Abstract
Life is the outcome of a complex network of chemical reactions and molecular interactions that emerged on Earth once primitive chemical automata could self-assemble in such a way that enabled them to self-reproduce and evolve. Yet exactly how, where and when life first appeared on our planet remains unknown. In this chapter, we review the various lines of evidence from fossil and geochemical traces of early life preserved in the geological record, which provide fundamental, albeit often rudimentary, insight into early life. The oldest fossils record the nature of life more than half a billion years after it emerged on Earth and suggest that considerable metabolic diversity had already evolved by this time. Microfossils, microbial mats, stromatolites and organic and inorganic geochemical signatures have been interpreted as compelling evidence for Archean biodiversity. In spite of the chemical enigmas of the earliest life and its limited record, characterization of the various classes of biosignatures indicative of life in its geological context provides guidance as to the earliest co-evolution of the geosphere and biosphere.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
10.1 Introduction
Life emerged on Earth when parts of the chemical automata self-assembled to generate automata capable of self-reproduction and evolution. Occassional minor errors in the assembly of these molecules would have generated more efficient automata that became the dominant entities. With time, repetition of these processes ultimately led to the emergence of life as we know it. Like Luigi Pirandello in his “Six Characters in Search of an Author,” chemists remain in search of the ingredients, the environment, and the most plausible scenarios by which life occurred on Earth around four billion years ago. The lack of a prebiotic geochemical record on Earth eliminates the possibility of identifying the essential evidence required to prove the pathway by which these processes occurred. Likewise, the oldest fossil evidence for life is not necessarily representative of the oldest forms of life on Earth. The fossil evidence for ancient life nonetheless elucidates the variety of settings that hosted early life.
The propensity for life to occupy and colonize surfaces in every favorable niche, which is certain to have happened once it emerged on our planet, adds to the ambiguities faced by chemists who are now closer than ever to completing the puzzle of life’s origins. A key step in the puzzle will be reconciling the scenario by which the ingredients for life come together in a sufficiently robust form to have withstood the challenges it would have faced in nature, which would have been considerably different than habitats on Earth today (Martin et al. 2008; Westall and Brack 2018; Westall et al. 2018).
Given that the exact steps, pathways and environmental constraints that led to the emergence of life remain hypothetical, a chemist’s playground of opportunities remain constrained primarily by the fundamental laws of science and the limitations imposed by physical/geological settings, the latter of which certainly lies on a spectrum, from having been only slightly different geochemically from their modern analogue environments to being drastically different from any type of environment known to us (Westall et al. 2018; Cavalazzi et al. 2019), and therefore having become obsolete as Earth evolved through time. The vagaries of time are certain to have masked nearly any clue of the events that led to the emergence of life in our geological record. Hence we review the necessary steps required to assemble life as we know it, followed by an overview of the evidence for the earliest life on Earth with insights as to how it is recognized, given the possibility that early life could have emerged in environmental settings that are not significantly different from the types of habitats with which we are familiar, such as hydrothermal settings, which have existed throughout Earth history (Nisbet and Sleep 2001).
10.2 The Record of Early Life
Life as we know it emerged during the Hadean Eon (~4.5–4.0 Ga), on an Earth that was truly a “distant world” in terms of the planet’s evolutionary timeline (Sleep 2010). Although various niches on Earth could likely have supported life since the early Hadean (Nisbet and Sleep 2001; Dass et al. 2016; Westall et al. 2018; Sleep 2018), the earliest evidence––direct and indirect––for a biosphere on Earth is archived within the fossil record of prokaryotes in the subsequent Archean Eon (4.0–2.5 Ga). This situation presents a worst-case scenario for deducing the nature of the earliest life, since the oldest evidence for life on Earth records the nature of life more than half a billion years after its origin. In spite of this constraint, by scrutinizing the earliest fossil record it becomes possible to extrapolate back in time to different plausible environments that could have hosted the origins of life. Comparisons of the possible environments that could have hosted the earliest forms of life have recently been reviewed (Westall et al. 2018; Camprubí et al. 2019).
Much of what is known about ancient microorganisms on the early Earth stems from a small number of highly contested purported fossils that were collected from rather limited quantities of rock. Prior to two billion years ago (i.e., the advent of the Paleoproterozoic Turee Creek-, Gunflint- and Francevillian-type biotas), almost all puported microfossil morphologies were remarkably simple (filamentous, vibrioidal, coccoidal and spheroidal,though not lenticular). These are the same morphologies that represent the major cellular morphotypes found on Earth today. Some have argued that many of the most ancient “microfossils” arose from mineralization phenomena, such as the silica-carbonate (witherite) biomorphs, silica-metal oxide vesicles, and iron mineral chemical gardens (Garcia-Ruiz et al. 2003, 2020; Bizzarri et al. 2017; McMahon 2019), Such hypotheses underscore the need to determine whether such objects preserve multiple biosignatures, an attribute of bona fide (i.e., carbonaceous) microbial fossils (e.g., Cady 2002).
The earliest rock record is highly fragmentary and limited in extent, as dynamic tectonic processes throughout Earth history have inexorably reworked Hadean and Eoarchean strata. Most compelling evidence for early life (>3 Ga) comes from only two regions on Earth: the Barberton greenstone belt of southern Africa and the greenstone belts of the East Pilbara terrane, Western Australia. These tectonostratigraphic entities present sequences of volcanic and sedimentary rocks, with the latter, primarily in the form of cherts and sandstones, preserving the canonical traces of life. This record provides the evidence closest in time to the origins of life and, hence, naturally guides our expectations both of the early evolution of life and the organic (geo)chemical processes that led to the first life. Decoding the evidence for early life remains, thus, one of the greatest challenges facing modern geobiology.
In the sections that follow, we summarize the evidence for early life, propose areas for future research, and illustrate how the various evidence is used as a means to understand the nature of early life on Earth at microbially relevant spatial and temporal scales.
10.2.1 Indicators of Early Life
Biosignatures, the general term for evidence of biological activity archived in the rock record can be broadly classified into five groups:
-
Cellular remains: fossilized (carbonaceous) cells and the extracellular remains of microbial communities;
-
Microbialites: bio-sedimentary constructions, including small-to-large scale (millimeter- to meter-scale) structures such as stromatolites, thrombolites, microbially induced sedimentary structures (MISS), and microbially influenced rock fabrics, e.g., biolaminites and mineralized mats and biofilms;
-
Biochemical signals of the co-evolving geosphere and biosphere: biomarker molecules, isotope biogeochemistry/fractionations, elemental geochemistry/enrichments (passive or paleo-metallomic), and the (ultra)structural characteristics of carbonaceous and organic materials preserved in rocks;
-
Biominerals: biologically induced or mediated minerals;
-
Bioalteration: textures in rocks that indicate the former presence of microbial activity and include, but are not limited to, tubular alteration and corrosion textures in lava, ambient microbial inclusion trails, and microbial borings at the surfaces of minerals.
Any effort to make a case for biology in ancient rocks remains challenging for the simple reason that any one of the various types of microbial biosignatures (sans rare ancient bona fide microbial fossils (c.f. Cady 2002) and uniquely biological molecular biomarker suites (c.f. Summons et al. 2011) can potentially, in isolation, be accounted for by non-biological means (Brasier et al. 2005, 2015). The substantial burden of proof required to demonstrate the presence of early life in geological materials is thus contingent upon the presence of multiple correlated lines of evidence, each of which must be demonstrated with profound rigor after careful collection from geological deposits that represent an unequivocally habitable setting (Wacey 2009).
10.2.1.1 Cellular Remains
Establishing the biogenicity of ancient purported microfossils has led to widespread and often contentious discourse regarding the “characters” that must be exhibited to conclusively support a biological origin (e.g., Brasier et al. 2002; Schopf et al. 2002). As noted in the seminal work of Cloud (1976), “…a given microstructure can be considered demonstrably biogenic only if some of its representatives display a level of cellular, microstructural, or biogeochemical differentiation comparable with that of living organisms and implying a similarity of function and continuity of evolution between them.”
Subsequent refinements and restatements of Cloud’s criteria for establishing the biogenicity of ancient evidence for life resulted in new criteria for recognizing ancient microfossils and critical appraisals of proposed ancient microfossil assemblages from specific localities. Buick (1990), for example, challenged previous biological interpretations of non-carbonaceous “microbiota” from the ~3.5 Ga Dresser Formation at North Pole, Pilbara, and Brasier et al. (2002, 2005) and Wacey et al. (2016) demonstrated an abiogenic origin for the microfossil-like suite of objects that, at the time, was considered the oldest convincing evidence for ancient fossil remains (the ~3.46 Ga Apex chert, also Pilbara; Schopf and Packer 1987).
Since each ancient microfossil discovery reveals unique combinations of cellular morphotypes, palaeoenvironmental processes, taphonomic pathways, and modes of preservation, the traditional approach to microfossil acceptance, which focussed on descriptive and comparative assessments of the characteristics of objects of putative biogenic origins with objects of known biogenic origin, has more recently been followed with a more holistic contextual approach, which considers a combination of regional, local, and fossil-scale biogeochemical characteristics of putative microfossils as a means to demonstrate whether microfossil-like objects are not abiogenic (c.f., Tomescu et al. 2016).
This contextual, holistic approach is used in most recent assessments of microfossil biogenicity and places the burden of proof on falsifying alternative non-biological (null) hypotheses (Brasier et al. 2002, 2005), which is more resilient to inductive reasoning than the traditional approach noted above. A discussion of the oldest examples of key morphotypes of microbial body fossils illustrates why a contextual, rather than traditional, approach is necessary when evaluating the most convincing evidence for ancient life.
Filaments
Filamentous cell-like objects have been identified in multiple geological horizons dated to be between 3.0 and 3.5 Ga, including the 3.48 Ga Dresser Formation (Ueno et al. 2001a, b), the 3.45 Ga Hooggenoeg Cherts (Walsh 1992), the 3.416 Buck Reef Chert (Walsh and Lowe 1985; Walsh 1992), unspecified 3.3–3.4 Ga Kromberg Formation cherts (Muir and Grant 1976), the 3.24 Ga Kangaroo Caves Formation (Rasmussen 2000; Wacey 2009), the 3.2 Ga Dixon Island Formation (Kiyokawa et al. 2006, 2014), the 3.020 Ga Cleaverville Formation (Ueno et al. 2006a), and the 3.0 Ga Farrell Quartzite (Sugitani et al. 2007) (Fig. 10.1). In most cases, ancient filamentous structures are solid or hollow thread-like carbonaceous materials up to several micrometers in diameter and possibly tens or hundreds of micrometers in length, bearing morphological and dimensional similarity to microfossils from younger horizons, such as Gunflintia spp. from the 1.88 Ga Gunflint Formation. Exceptions to this rule are the pyritized filaments reported from the Kangaroo Caves Formation, an extensively remineralized volcanogenic massive sulfide deposit.
(a) Clumps of filaments from the Kromberg Formation; (b, c) Pyritised filaments from the Kangaroo Caves Formation, (c) shows 3D visualisation; (d) Carbonaceous filaments from the Dresser Formation; (e) Carbonaceous filaments from the Dixon Island Formation. (a) Modified from Walsh and Westall (2003); (b) Courtesy of Birger Rasmussen; (c) Modified from Wacey et al. (2014); (d) Modified from Ueno et al. (2001a); (e) Courtesy of Shoichi Kiyokawa
Once the syngenetic context, both regional and local, of ancient filamentous microfossil-like objects has been demonstrated, high-resolution (sub-micrometric) analytical techniques are required to distinguish their biogenic origin (c.f., Oehler and Cady 2014). For example, one of the traditional criteria for recognizing ancient filaments is the presence of microscopically visible septae that regularly subdivide elongate filaments (Buick 1990). However, a high-resolution study that re-examined the submicroscopic attributes of some iron oxide replaced Gunflintia spp. filaments (Maldanis et al. 2020) shows that septae can be lost during mineral replacement of filamentous cells. High-resolution analysis has also proven indispensible in demonstrating the non-biogenic origin of some ancient filamentous objects, as shown when pseudo-septate patterns observed in filament-like objects from the Apex chert were identified as having been produced from the condensation of carbonaceous material around phyllosilicate templates with no biological origin (Wacey et al. 2016).
Spheroids and Coccoids
Ancient spheroidal or coccoidal microfossil-like objects have been identified in the 3.45 Ga Hooggeoneg Formation (Walsh 1992; Glikson et al. 2008), the 3.446 Ga Kitty’s Gap Chert (Westall et al. 2006, 2011), the 3.35–3.43 Ga Strelley Pool Chert (Wacey et al. 2011; Sugitani et al. 2015), the 3.26 Ga Fig Tree Group (Knoll and Barghoorn 1977), and the 3.0 Ga Farrell Quartzite (Sugitani et al. 2007, 2009) (Fig. 10.2). Coccoidal objects exhibit substantial size variation. Although in early work, a statistically normal size distribution was considered a requirement for demonstrating the biogenic origin of a coccoid population (e.g., Schopf and Walter 1983), it is now known that a normal distribution of spherical objects can also arise naturally in inorganic particle populations (Buick 1990). Spheroidal microfossil-like objects also exhibit considerable morphological differentiation: within millimeters, hollow and solid forms, both solitary and grouped, have been identified (Sugitani et al. 2007, 2015). Compelling evidence for biogenesis has been found in certain populations of spheroidal microstructures: (i) both Knoll and Barghoorn (1977) and Westall et al. (2006, 2011) identified evidence for cell division; (ii) Walsh (1992) and Sugitani et al. (2007, 2015) showed that coccoidal structures can occur in pairs and groups consistent with biological cell distributions; Walsh (1992) and Sugitani et al. (2007) identified features consistent with the rupturing of cell walls; and Westall et al. (2006) noted deflated coccoid surface textures consistent with cell lysis. In this regard, coccoidal-shaped microfossils are the only ancient fossil morphotype that displays evidence for multiple stages of the cell cycle, though caution is warranted as some of the apparent “life-cycle stages” can also occur during the growth of microfossil-like minerals: silica microspheres and botryoidal oxides exhibit division-like phenomena, and many mineral species show self-organization into arrangements not dissimilar to the pairing and grouping shown by coccoidal microfossil-like objects. Unlike filaments, Early Archean coccoidal microfossils have not, at the time of writing, been subjected to high-resolution microanalysis, which illustrates a potentially impactful future research direction for which the three dimensional tomographic analyses of coccoidal Gunflint-type microfossils (Huroniospora spp. and Eosphaera) in the works of Wacey et al. (2013), Hickman-Lewis et al. (2017) and Maldanis et al. (2020) provide a benchmark.
(a) Ruptured spheroid from the Kromberg Formation; (b, c) Lenticular forms from the Kromberg Formation; (d) Spheroids from the Strelley Pool Formation; (e) Lenticular forms from the Farrell Quartzite; (f, g) Dividing spheroids from the Sheba Formation (arrows). (a, b) Modified from Walsh (1992); (c) Courtesy of Maud M. Walsh; (d) Courtesy of Wacey et al. (2011); (e) Courtesy of Kenichiro Sigitani; (f, g) Courtesy of Andrew H. Knoll
Lenticular
A third group of proposed microfossils are described as lenticular (earlier work used the term spindle-like); these are large, thick-walled structures with single or double internal cavities and diaphanous carbonaceous coatings. Lenticular microstructures have been identified in the Hooggenoeg Formation (Walsh 1992), the Strelley Pool Chert (Sugitani et al. 2015; Oehler et al. 2017), various Kromberg Formation cherts (Walsh 1992; Oehler et al. 2017; Alleon et al. 2018), and the Farrell Quartzite (Sugitani et al. 2007). They have been ascribed a multitude of biological functions, including the outer sheath of microbial colonies (Walsh 1992) and planktonic organisms (Sugitani et al. 2007, 2015; Oehler et al. 2017; Kozawa et al. 2019) (Fig. 10.2). Their occurrence in pairs and linked chains has been hypothesized to reflect cell division by binary fission (Sugitani et al. 2015); however, no detailed comparison with the few younger microstructures bearing similarity to lenticular microfossils has yet been undertaken and their origin remains open to discussion. One particular challenge with regard to lenticular microstructures is their highly variable morphology; in spite of the large volume of research that supports a biogenic origin for these microstructures, similar populations of objects, some of which are non-carbonaceous, have been found in association with microbial mats of the Middle Marker horizon (Hickman-Lewis et al. 2018).
Analytical Approaches to Archean Microfossils
It is clear that further high-resolution studies of Early Archean microfossils are required to evaluate their biogenicity, demonstrate their metabolic affinities, and explore their evolution through diagenesis. High-resolution approaches have been widely applied to younger Proterozoic microfossils to constrain life processes (e.g., Wacey et al. 2013) and diagenetic changes (e.g., Maldanis et al. 2020). Unfortunately, for the Archean, such studies are hindered by the scarcity of microfossils preserved in rocks of this age. Nonetheless, approaches such as transmission electron microscopy (De Gregorio and Sharp 2006), micro-scale X-ray computed tomography (X-ray μCT; Hickman-Lewis et al. 2017), hydrolysis–pyrolysis (Duda et al. 2018), synchrotron X-ray fluorescence (Sforna et al. 2017) and micro-scale particle-induced X-ray emission (μPIXE; Hickman-Lewis et al. 2020b), have been used to study Archean microfossils and organic materials with promising results. One enduring challenge is that microfossils are rarely preserved within Archean stromatolites and microbial mats (Schopf et al. 2007), and this lack of spatial association leads to reciprocal challenges in proving the biogenicity of both types of traces of life: how can one be assured of the biogenic origins of a stromatolite when no traces of its architects are preserved? Furthermore, many microfossil-oriented studies fail to properly account for the chemistry of the depositional environment at local and regional scales. Taking into account the combination of stratigraphy, sedimentology, and layer-specific geochemistry (both mineralogy and evidence of aqueous alteration) can provide key constraints regarding the conditions of deposition relevant to microbial growth and thus the types of organisms that could plausibly have been present (Kiyokawa et al. 2006; Sugitani et al. 2015; Hickman-Lewis et al. 2020a).
10.2.1.2 Microbialites
Interpretation of the biogenicity of ancient microbialites suffers from similar shortcomings to ancient microfossils, since several macroscopic biological fabrics can be reproduced through abiotic processes (Lowe 1994; McLoughlin et al. 2008; Hickman-Lewis et al. 2018, 2019). Suites of biogenicity criteria have been devised to critically assess the origins of microbial mat fabrics (Hickman-Lewis et al. 2018) and MISS (Noffke 2010), taking into account both the macroscopic and microscopic morphologies of the structures and their relationships with the surrounding stratigraphy. When using these nested microbial–environmental biogenicity criteria, the biogenic origin of fossilized microbial mats, mat relicts, and MISS can be concluded beyond any reasonable doubt. Although the individual constituent parts of microbial mats—filamentous microstructures, carbonaceous aggregates, entrained mineral particles, and laminar macromorphologies, among others—can be accounted for individually by abiotic or hydrodynamic mechanisms, their occurrence in a unified structure cannot. As a consequence of this multi-scale, multi-dimensional approach, fossilized microbial mats, biofilms, and MISS are perhaps the most uncontroversial fossils in deep time and have been identified widely throughout Paleoarchean and Mesoarchean successions, including the Dresser Formation (Noffke et al. 2013), the Middle Marker horizon (Hickman-Lewis et al. 2018, 2019), the Apex Basalt (Hickman-Lewis et al. 2016), the Hooggenoeg Formation (Walsh 1992; Walsh and Lowe 1999) the Buck Reef Chert (Walsh 1992; Walsh and Lowe 1999; Tice and Lowe 2004, 2006a, b; Tice 2009), the Footbridge Chert (Hickman-Lewis et al. 2020a), the Josefsdal Chert (Westall et al. 2006, 2011, 2015), the Mendon Formation (Trower and Lowe 2016), the Moodies Group (Noffke et al. 2006; Heubeck 2009; Homann et al. 2015, 2018) and the Dixon Island Formation (Kiyokawa et al. 2006) (Fig. 10.3).
(a, b) Microbial mats from the Middle Marker horizon, (b) shows high-resolution X-ray μCT scan of mats exhibiting micro-tufts (arrow); (c) microbial mats from the Buck Reef Chert; (d) Microbial mats from the Moodies Group. (a, b) Credit: Keyron Hickman-Lewis; (c) Modified from Tice (2009); (d) Modified from Homann et al. (2015)
Fossilized microbial mats and biofilms comprise fine, undulating or crinkly carbonaceous laminations, each level of which represents microbial communities that colonized the sediment surface; non-isopachous laminae (i.e., growth with a low degree of inheritance from underlying laminations) exhibit anastomosis behavior that is incompatible with a sedimentary or diagenetic precursor, but rather evidences trophic or ecophysiological behavior (Hickman-Lewis et al. 2016, 2018). Non-isopachous morphologies on multiple scales are taken to indicate growth according to trophic requirements; for example, photosynthetic microbial mats grow more rapidly at crests, in part due to exposure to sunlight, while permanently submerged benthic mats can form crested or tufted morphologies due to emergence from the laminar bottom flow into hydrodynamically well-mixed, nutrient-rich waters above, or to avoid chemical stresses that result from concentrations of heavy metals arising from EPS (extracellular polymeric substances) detoxification mechanisms. At the sub-millimeter scale, early Archean fossilized microbial mats alternate with pure silica or granular layers and lenses and are laterally discontinuous at micrometer-scales, though broadly continuous at the centimeter-scale (Walsh 1992; Noffke et al. 2013); this denotes their original filament-like morphology, for which limited evidence has been observed in three dimensions (Hickman-Lewis et al. 2017). Key is the entrainment and orientation of sediment particles due to trapping and binding processes by EPS (Noffke 2010; Noffke et al. 2013); preferentially oriented sediment particles are among the key MISS attributes that provide evidence for biological action. The in vivo plasticity of many microbial mats is also denoted by torn and reworked cohesive mat fragments or tangles of filament-like objects that are deposited within the immediately adjacent sediment (Tice and Lowe 2004; Kiyokawa et al. 2006).
Stromatolites are the most iconic microbial fossils and spectacular living examples are directly comparable to ancient forms in many respects, which provides an opportunity to elucidate the nature and role of biological and environmental processes that characterize them (Reid et al. 2003; Riding 2011). The oldest reported stromatolites are found in the Dresser Formation (Walter et al. 1980; Van Kranendonk 2006; Djokic et al. 2017), the Strelley Pool Chert (Hofmann et al. 1999; Allwood et al. 2006, 2009, 2010), and the Mendon Formation (Byerly et al. 1986; Byerly and Palmer 1991). The stromatolites in the Dresser Formation include the domical–conical forms and a diverse assemblage of morphotypes in the Strelley Pool Chert, which include complex conical Conophyton-type, encrusting, cuspate, and “egg carton” forms. Geochemical work has shown that the predominance of these forms varies predictably with sedimentary depositional facies (Allwood et al. 2009).
Though controversial, older (~3.7 Ga) stromatolite forms are proposed by Nutman et al. (2016), who argue that ancient laminated conical structures found in the Isua region of Greenland are poorly preserved stromatolites. That work has been challenged by Allwood et al. (2018), whose subsequent analysis of photographs of multiple faces of rock samples and X-ray fluorescence elemental mapping, combined with structural analysis of the outcrop, showed that the structures possessed neither internal lamination nor elemental distributions that could be argued to result from microbial processes. Allwood et al. (2018) found that the putative stromatolites were characterized by ridge-like rather than conical/domical morphologies and argue that the structures are best interpreted as deformation features, with ridge-like morphologies that reflect the applied stress field. In their follow-on study, which addresses additional attributes and geochemical signatures associated with the purported stromatolites, Nutman et al. (2019) claim that the preponderance of evidence remains consistent with a biogenic origin.
Establishing the biogenicity of stromatolites presents formidable challenges since they reflect a complex interplay of multiple processes: biomass growth; microbial community composition; sedimentation rate and character; surface normal growth driven by nutrient diffusion, mineralization, and biophysical constraints; the production and properties of EPS, environmental hydrodynamic processes, and stochastic events (Grotzinger and Knoll 1999; Bosak et al. 2013; Hickman-Lewis et al. 2019). Furthermore, as noted previously, stromatolites rarely include direct evidence and/or expressions of the microorganisms that were involved in their construction. Stromatolite geobiology has thus expanded to include mathematical predictive frameworks founded in the observation that stromatolite morphologies can be partly or fully accounted for by a combination of Kardar-Parisi-Zhang (KPZ) dynamics, diffusion-limited aggregation, and stochastic growth (Grotzinger and Rothman 1996; Batchelor et al. 2002, 2003; Dupraz et al. 2006; Petroff et al. 2010). The application of such models, however, generates considerable controversy since it remains unclear how best to set the initial conditions of stromatolite growth, incorporate fluctuating palaeoenvironmental parameters, and incorporate stochastic events that may drive the formation of unique morphologies and variants. Many growth models are explicitly deterministic and include no randomness, producing results that depend directly upon the initial settings; this is inconsistent with natural microbialite growth, especially in ecosystem settings where both intrinsic and extrinsic variables fluctuate episodically to produce macroscale and microscale peculiarities (Dupraz et al. 2006; Cuerno et al. 2012). More sophisticated models, while needed, are generally computationally intensive.
Lowe (1994) and Grotzinger and Rothman (1996), among others, argued that simple stromatolite forms, such as domes and cones, can be replicated through abiotic processes. Yet the extraordinary diversity of stromatolite forms with plausibly similar biological genesis––greatest in the Proterozoic, though not insubstantial in the Archean––cannot be explained by abiotic models alone. Indeed, it is difficult even to model conical Conophyton-like forms without considering inherent biological attributes (Batchelor et al. 2003; Petroff et al. 2010). It has been suggested more recently that “true” biological signatures in stromatolites may be indicated in macromorphologies that do not conform to certain model expectations (e.g., overhangs; Cuerno et al. 2012) or micromorphologies that can be reproduced through geomicrobiological experimentation (e.g., fabric orientation and density differences within microbial pinnacles; Hickman-Lewis et al. 2019). Observations of modern stromatolites show that such complexity arises from the interaction of the stromatolite-building biota with paleoenvironmental parameters (Reid et al. 2003) (Fig. 10.4).
(a) Conical stromatolites from the Strelley Pool Formation; (b) High-resolution X-ray μCT scan of mineralised stromatolites from the Dresser Formation showing 3D variations in mineralogy and lamination micromorphology (red = iron oxide, yellow = silica, white = baryte, blue = void space); (c) Stromatolites from the Mendon Formation. (a) Courtesy of S.M. Awramik; (b) Credit: Keyron Hickman-Lewis; (c) Modified from Byerly and Palmer (1991)
10.2.1.3 Biogeochemical Signals of the Co-evolving Geosphere and Biosphere
Biomarker Molecules
Chemical fossils include lipid biomarker molecules that can only be derived directly from biological processes (Summons et al. 2011). Regrettably, the biomarker record is plagued with uncertainty due to likely/demonstrable contamination (French et al. 2015) and the inability to consider an individual biomarker compound in isolation as a reliable indicator of Archean metabolism. In the absence of a suite of biomarkers, isotope geochemistry and trace and rare earth element compositions are used to decode aspects of co-evolving ecosystems and environments. Recently, Fourier Transform Infra-Red (FTIR) spectroscopy has been applied to Archean carbonaceous materials as an alternative means of decoding primary aliphatic biochemistry, being sensitive to organic materials but less susceptible to contamination. FTIR investigations illustrate that microfossils and microbial mats comprise organic residues from both Bacteria and Archaea based on the exceptional preservation of relict membrane proteins with diverse aliphatic compositions, e.g., the relative abundances of CH2 and CH3 moieties (Igisu et al. 2018; Hickman-Lewis et al. 2020c).
Isotope Biogeochemistry
Isotope geochemistry is a powerful approach toward both paleoenvironmental and ecological reconstructions in deep time. Traditional and non-traditional stable isotope approaches using Li, C, N, O, Mg, Si, S, K, V, Cr, Fe, Ni, Cu, Se, Mo, Te, and Tl, among other elements, have been used throughout the Archean geological record to unveil a multitude of biogeochemical and paleoenvironmental parameters (see a comprehensive review by Teng et al. 2017). Most commonly, carbonaceous microfossils are routinely analyzed for their δ13C composition, since metabolic processes typically yield products with negative δ13C (Schidlowski 2001). Since the abiotic synthesis of carbonaceous materials can produce similarly wide-ranging δ13C values (McCollum and Seewald 2006), the argument that negative δ13C is solely indicative of a biogenic precursor is far from infallible; a more reasonable biogenic signature is a restricted range of δ13C. Coupled isotopic records, such as the δ13C records of concomitant organic carbon and associated carbonates, can also chart local biogeochemical events (e.g., the pairing of fluctuations in organic and inorganic δ13C that correspond to terrestrial water influxes into marine environments). Strongly negative C isotope signals (δ13C) associated with Archean organic material, both body fossils and disseminated carbonaceous materials, include δ13C values of −28.2‰ in >3.7 Ga graphite from Labrador, Canada (Tashiro et al. 2017), −30‰ to −19‰ in >3.7 Ga graphite from Isua (Rosing 1999; Grassineau et al. 2006), as low as −56‰ to −32‰ in methane inclusions from the Dresser Formation (Ueno et al. 2001a, b, 2006b), and −40 to −10‰ in cherts of the Barberton greenstone belt (Grassineau et al. 2006; van Zuilen et al. 2007; Hickman-Lewis et al. 2020b).
An important consideration is the fact that negative C isotope fractionations (between ca. –40‰ and 0‰) are compatible with multiple biological pathways, hence in the absence of strongly grouped δ13C measurements associated with a single microstructure or a cellular morphotype, it can be difficult or impossible to detect specific metabolisms. Exceptions include a strongly negative δ13C signal considered incompatible with photosynthesis though consistent with methanogenesis. Less well-constrained distributions of δ13C associated with different fossil morphotypes could indicate either a mixed non-syntrophic microbial consortium or the influence of non-biological carbon, for example, from FTT processes, which is not expected to conform to a restricted range of values. In such cases, additional evidence is necessitated. As examples, van Zuilen et al. (2007) combined C isotopes with Raman and H/C measurements, Grassineau et al. (2006) used both C and S isotopes from the same horizons, and Hickman-Lewis et al. (2020b) combined C isotopes with Raman, TEM and μPIXE spatially quantified trace element mapping; in all cases, the carbonaceous materials studied were found to be most plausibly of biogenic origin.
Ambiguity in the interpretation of organic δ13C in ancient microfossils can also be resolved by correlating δ13C measurements with δ34S, through which the contributions of sulfur-metabolizing pathways can be incorporated into a more comprehensive understanding of the dominant biogeochemical processes (Wacey et al. 2011; Bontognali et al. 2012). Wacey et al. (2011) identified cell-like structures in the Strelley Pool Formation associated with pyrite crystals with Δ33S values between −1.65 and +1.43‰ and δ34S values from −12 to +6‰ (normalized to the Vienna Canyon Diablo Troilite). These values, when considered alongside strongly negative δ13C (−33 to −46‰) in the same structures, indicate sulfate-reduction and sulfur-disproportionation metabolic pathways. Bontognali et al. (2012), who also analyzed carbonaceous materials from Strelley Pool, measured large δ34S fractionations at small spatial scales, including values below −15‰, and positive Δ33S values which, taken together, suggest the sulfurization of organic matter by respiratory sulfur metabolisms alongside elemental sulfur disproportionation. On regional and global scales, the change from dominantly mass-independent to mass-dependent sulfur fractionation during the Archean has been interpreted to reflect a global “chemical shift” from anoxic to oxic environments.
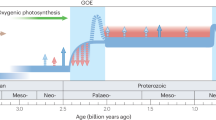
Isotope records are particularly powerful for understanding changing paleo-environments, the most pronounced expression of which are the major environmental changes associated with episodes of oxygenation, such as the Great Oxygenation Event (GOE), since such shifts reflect and would have impacted the composition and activities of major biological communities. The GOE and numerous “whiffs” of oxygen––for up to several hundred million years prior––have been studied via multiple isotope chemostratigraphy approaches that include, though are not limited to, coupled C-S (Izon et al. 2017), S (Philippot et al. 2018), Fe (Oonk et al. 2016), Cu (Chi Fru et al. 2016), Mo (Duan et al. 2010) and U (Kendall et al. 2013). Although isotope chemostratigraphy has seen limited application to Early Archean rocks, isotope systematics have nonetheless been used to infer the characteristics of habitable environments on the early Earth. Particularly informative isotopic data include a detailed δ18C record from Paleoarchean–Neoarchean cherts that demonstrate that the early oceans were warm (Knauth 2005; Hren et al. 2009; Tartèse et al. 2017) and δ30Si isotopes from predominantly South African cherts that indicate shared roles of Si-saturated seawater and hydrothermal fluids in the silicification of sediments (van den Boorn et al. 2007, 2010; Abraham et al. 2011). Deviations of around 2‰ in δ30Si between sedimentary laminated and massive chemical cherts indicate that, while Si-saturated seawater was the principal agent of silicification in laminated cherts, silicification of massive cherts was due to the mixing of hydrothermal fluids and seawater. Combining O and Si isotopes could place distinct temperature-redox constraints on the earliest habitable environments and facilitate determination of the degree of thermophilic adaptation that would be required by microbial inhabitants. O isotopes indicate that the temperatures of the oceans have gradually decreased over geological time, with temperatures of 40–90 °C being argued for the depositional fluid of Archean cherts, consistent with the hypothesis of thermophilic life occupying the early oceans (Knauth 2005).
Elemental Biogeochemistry
A third branch of biochemical signal includes the various types of elemental enrichments associated with fossil and extracellular fossil remains, a phenomenon that could have been concentrated either passively (Allwood et al. 2010; Hickman-Lewis et al. 2019, 2020b) or actively by life (Fraústo da Silva and Williams 2001; Edwards et al. 2014; Hickman-Lewis et al. 2019, 2020b). Deciphering elemental enrichments in paleoenvironments and fossilized biomass is contingent upon constraining the biogeochemistry of individual elements. Paleoenvironmental reconstruction in ancient geological units primarily utilizes trace and rare earth elements plus yttrium (REE + Y) chemistry, the composition of which exhibits predictable aqueous complexation behavior throughout geological time (Bau and Dulski 1996; Kamber and Webb 2001; Shields and Webb 2004; Gourcerol et al. 2015). Early Archean fossiliferous units typically exhibit normalized REE + Y compositions (i.e., patterns of elemental enrichment and depletion) that indicate influences from seawater and hot hydrothermal fluids (Danielson et al. 1992; Van Kranendonk et al. 2003; Allwood et al. 2010), though most fossiliferous cherts from the Barberton greenstone belt exhibit an additional major component from river-derived inputs (Hickman-Lewis et al. 2020a). In brief, widespread phototrophic microbial communities on the early Earth flourished under disequilibrium conditions at the confluence of marine, terrigenous, and hydrothermal activity (Hickman-Lewis et al. 2020a), whereas ecosystems dominated by chemosynthesis appear to have been concentrated around zones of elevated hydrothermal activity, sustained by the greater magnitude of the positive Eu anomaly (Westall et al. 2015). It has been suggested that some stromatolites exhibit REE + Y enrichment that could function as a biogenicity indicator (Allwood et al. 2010). While this correlation is consistent with known mechanisms of REE-ligand complexation within biomass, particularly in the case of the middle and heavy REEs (Takahashi et al. 2005; Censi et al. 2013), the precise means of REE + Y uptake by living microbial biofilms and bioconstructions remains poorly constrained (Johannesson et al. 2013).
Perhaps a more promising avenue toward an understanding of bio-indicative elemental enrichments are specific elemental distributions that result from uptake by the cell or extracellular polymers. The metallome, first outlined in detail by Williams (2001) and Fraústo da Silva and Williams (2001), was defined to reflect the chemical selection of elements by the cell according to its biochemical requirements. The potential then exists for such microbial communities to preserve “fingerprints” of elemental enrichments that could distinguish them from other communities, as demonstrated by the fact that the elemental compositions of extant organisms vary according to metabolism. For example, methanogens require higher concentrations (or fractional contributions to their metallome) of V and Ni than other groups (Zerkle et al. 2005; Cameron et al. 2012).
The metallome has been considered the inorganic complement to the proteome and genome and, thus, a possible inherent expression of cell biochemistry. The potential for this biosignature to endure geological processing is promising, and it has recently been suggested that diagnostic fragments of the metallome can be preserved even in Earth’s oldest fossil record, revealing the metabolisms of the original biomass, for example, enrichments of Fe, Ni, Co, and V in carbonaceous materials, consistent with a putatively lithotrophic consortium cycling methane and nitrogen (Hickman-Lewis et al. 2020b). Although primary elemental concentrations remain associated with biomass through early diagenesis (Edwards et al. 2014), the possibility of their preservation into deep time remains to be stringently tested. In this regard, elemental enrichments in carbonaceous materials that have undergone rapid preservation should be considered when the origin of the carbonaceous material is likely to be biogenic, as such a biosignature can be used to prove biogenicity and identify metabolism in the absence of obvious cellular preservation (Hickman-Lewis et al. 2020b).
10.2.1.4 Biominerals
Many organisms produce mineral phases as a metabolic by-product. Biomineralization, both extracellular and intracellular, is a widespread phenomenon in stromatolite-building communities, for example, cyanobacteria (Benzerara et al. 2006, 2014) and anoxygenic photosynthesizers (Bosak et al. 2007; Bundeleva et al. 2012). The formation of minerals either induced or mediated by microbes, particularly cyanobacteria, is often factored into morphogenetic models of stromatolites as a dominant growth vector (e.g., Grotzinger and Knoll 1999; Petroff et al. 2010). Microscopic pyrite heavily depleted in 36S is commonly associated with microbial sulfate-reducers (Ueno et al. 2008; Wacey et al. 2011; Marin-Carbonne et al. 2018); though pyrite can also be a by-product of the anaerobic oxidation of methane (AOM; Zhang et al. 2014). Among other mechanisms of biomineralization, oriented microcrystals of magnetite formed by magnetotactic bacteria is another potential biosignature that could date back to the Archean, since the gene cluster responsible for the biomineralization of magnetosomes is thought to have originated between 2.7 and 3.6 Ga (Lin et al. 2017). A review of biomineralization and its application to the fossil record is given by Benzerara et al. (2011).
10.2.1.5 Bioalteration
The fifth group of biosignatures is bioalteration, indirect evidence for life manifested as traces of its influence upon the substrate, that is, ichnofossils. In the context of early life, two main groups of bio-induced alteration of the rock are discussed: in vivo microbial corrosion of rocks, usually basalts and hyaloclastites, by endolithic microbes (forming tubules), and diagenetic or post-diagenetic migration of fabric elements due to the alteration (degradation and degassing) of carbonaceous materials (forming ambient inclusion trails). Features that resemble microbial corrosion of basalts have been identified in pillow lavas as old as 3.47 Ga (Furnes et al. 2004, 2007; Banerjee et al. 2006); although their morphology and petrological setting strongly resemble those of modern microbial corrosion textures in ocean crust, subsequent studies have suggested that the fossilized forms may result from post-diagenetic metamorphic processes (Grosch and McLoughlin 2014). Since the formation processes of even the modern examples remain poorly constrained (Staudigel et al. 2015), the debate concerning the origins of such ancient structures remains unresolved. Recently, however, Wacey et al. (2018) identified the co-occurrence of Ti and C in modern microbial corrosion tubules, directly echoing the concentrations of carbonaceous materials in titanite-remineralized tubules from the Archean, which suggests that aspects of the complex formation and mineralization processes outlined for the genesis of these structures in Furnes et al. (2007) are correct.
Ambient inclusion trails are another type of tubular alteration structure, usually with a single microscopic (1–15 μm) pyrite grain at their terminus, that occur adjacent to loci of decaying organic matter. They have been identified throughout the geological record and are attributed to the effects of thermal decomposition of residual organic material that produces CO2, and potentially CH4, as propulsive gases that drive the crystal through a microquartz matrix that is simultaneously locally dissolved by organic acids (Wacey et al. 2008a, b, 2016; Olempska and Wacey 2016). The correlation of C and N within ambient inclusion trails has been used to demonstrate a biological role in their formation (Wacey et al. 2008a, b); nonetheless, a range of abiotic means for the formation of ambient inclusion trails have also been demonstrated, for example matrix dissolution due to the influence of clay minerals (Wacey et al. 2016). Such features may thus occur in the absence of biogenic carbonaceous materials.
10.3 Relevance of the Palaeobiological Record for Life’s Origin
The ancient geobiological record provides insights into the types of ecosystems early life occupied, how it behaved, and how different populations interacted with one another during that time. Microbial biosignatures preserved in the ancient fossil record have revealed that early life has retained a remarkable degree of morphological conservatism relative to extant life on multiple spatial scales. Cellular morphotypes of early bacteria are similar to those of extant life today, with the exception of lenticular microfossils that are unique to the ancient fossil record. The relatively early diversification of life that colonized available surfaces led to distinct influences of microbial life on sediment fabrics and microbialite structures. The evolution of different metabolic stratigies, evidenced in the isotopic record, enabled early life to occupy a wide range of environments, which for shallow water environments required evolutionary adaptations so photosynthetic microbial populations could shield themselves from the deleterious effects of UV.
Though the exact nature of the earliest form of life is unknown, several lines of evidence suggest it was thermophilic. Hydrothermal systems provide environments where fluids enriched in reduced inorganic chemical species generate proton gradients and redox disequilibria that could support early life metabolisms (Branscomb and Russell 2013). An analysis of metal utilization by microbes throughout early Earth history illustrates that the earliest metabolisms were likely characterized by metal dependencies similar to those of thermophilic extremophiles (Moore et al. 2017). Almost all paleoenvironmental reconstructions of early life habitats invoked some degree of necessity for thermophily, usually as a result of the influence of volcanism and hydrothermal fluids (Danielson et al. 1992; Nisbet and Sleep 2001; Van Kranendonk et al. 2003; Allwood et al. 2010; Sugahara et al. 2010; Hickman-Lewis et al. 2018, 2020a).
The timing of life’s origins is unknown and not recorded in the geological record. Though molecular clocks have been developed to extrapolate the timing of key events during the evolution of life, none have yet been calibrated with the fossil evidence preserved in either the Barberton greenstone belt or the numerous greenstone belts of the Zimbabwe craton. As such, the current generation of phylogenetic trees could be improved with a comprehensive interpretation of the evidence preserved in the oldest fossil record.
In summary, and for the purposes of moving closer to unravelling life’s origins, “characters” shared by the ancient geological settings that hosted the earliest evidence for life have recorded key aspects of the environmental components of the prebiotic Earth. Water, higher temperatures, (bio)polymer compositions, and the ability to colonize surfaces are examples of these “characters.” The progressive complexification of life throughout the Archean and Proterozoic reciprocally informs us of the types of life and settings that are not thought to be relevant to the origins of life, such as oxygenic metabolisms, which certainly post-date anaerobic, anoxygenic cellular machinery. The interested reader is referred to the work of Westall et al. (2018) and Camprubí et al. (2019) for additional discussion of the environmental parameters relevant to the emergence of life on Earth.
References
Abraham, K., Hofmann, A., Foley, S.F., Cardinal, D., Harris, C., Barth, M.G., André, L.: Coupled silicon–oxygen isotope fractionation traces Archaean silicification. Earth Planet. Sci. Lett. 301, 222–230 (2011)
Alleon, J., Bernard, S., Le Guillou, C., Beyssac, O., Sugitani, K., Robert, F.: Chemical nature of the 3.4 Ga Strelley Pool microfossils. Geochem. Perspect. Lett. 7, 37–42 (2018)
Allwood, A.C., Walter, M.R., Kamber, B.S., Marshall, C.P., Burch, I.W.: Stromatolite reef from the early Archean era of Australia. Nature. 441, 713–718 (2006)
Allwood, A.C., Grotzinger, J.P., Knoll, A.H., Burch, I.W., Anderson, M.S., Coleman, M.L., Kanik, I.: Controls on development and diversity of Archean stromatolites. Proc. Natl. Acad. Sci. USA. 106, 9548–9555 (2009)
Allwood, A.C., Kamber, B.S., Walter, M.R., Burch, I.W., Kanik, I.: Trace element record depositional history of an Early Archean stromatolitic carbonate platform. Chem. Geol. 270, 148–163 (2010)
Allwood, A.C., Rosing, M.T., Flannery, D.T., Hurowitz, J.A.: Reassessing evidence of life in 3,700-million-year-old rocks of Greenland. Nature 563(7730), 241–244 (2018)
Banerjee, N.R., Furnes, H., Muehlenbachs, K., Staudigel, H., de Wit, M.: Preservation of ~3.4–3.5 Ga microbial biomarkers in pillow lavas and hyaloclastites from the Barberton Greenstone Belt, South Africa. Earth Planet. Sci. Lett. 241, 707–722 (2006)
Batchelor, M.T., Burne, R.V., Henry, B.I., Watt, S.D.: Deterministic KPZ model for stromatolite laminae. Phys. A. 282, 123–136 (2002)
Batchelor, M.T., Burne, R.V., Henry, B.I., Watt, S.D.: Mathematical and image analysis of stromatolite morphogenesis. Math. Geol. 35, 789–803 (2003)
Bau, M., Dulski, P.: Distribution of yttrium and rare-earth elements in the Penge and Kuruman iron- formations, Transvaal Supergroup, South Africa. Precambrian Res. 79, 37–55 (1996)
Benzerara, K., Menguy, N., López-García, P., Yoon, T.-H., Kazmierczak, J., Tyliszczak, T., Guyot, F., Brown Jr., G.E.: Nanoscale detection of organic signatures in carbonate microbialites. Proc. Natl. Acad. Sci. USA. 103(25), 9440–9445 (2006)
Benzerara, K., Miot, J., Morin, G., Ona-Nguema, G., Skouri-Pane, F., Ferard, C.: Significance, mechanisms and environmental implications of microbial biomineralization. Compt. Rendus Geosci. 343(2–3), 160–167 (2011)
Benzerara, K., Skouri-Panet, F., Li, J., Férard, C., Gugger, M., Laurent, T., Couradeau, E., Ragon, M., Cosmidis, J., Menguy, N., Margaret-Oliver, I., Tavera, R., López-García, P., Moreira, D.: Intracellular Ca-carbonate biomineralization is widespread in cyanobacteria. Proc. Natl. Acad. Sci. USA. 111(30), 10933–10938 (2014)
Bizzarri, B.M., Botta, L., Pérez-Valverde, M.I., Saladino, R., Di Mauro, E., Garcia-Ruiz, J.M.: Silica metal oxide vesicles catalyze comprehensive prebiotic chemistry. Chem. Eur. J. 24, 8126–8132 (2017)
Bontognali, T.R.R., Session, A.L., Allwood, A.C., Fischer, W.W., Grotzinger, J.P., Summons, R.E., Eiler, J.M.: Sulfur isotopes of organic matter preserved in 3.45-billion-year-old stromatolites reveal microbial metabolism. Proc. Natl. Acad. Sci. USA. 109, 15146–15151 (2012)
Bosak, T., Greene, S.E., Newman, D.K.: A likely role for anoxygenic photosynthetic microbes in the formation of ancient stromatolites. Geobiology. 5(2), 119–126 (2007)
Bosak, T., Knoll, A.H., Petroff, A.P.: The meaning of stromatolites. Annu. Rev. Earth Planet. Sci. 41, 21–44 (2013)
Branscomb, E., Russell, J.M.: Turnstiles and bifurcators: The disequilibrium converting engines that put metabolism on the road. Biochim. Biophys. Acta 1827(2), 62–78 (2013)
Brasier, M.D., Green, O.R., Jephcoat, A.P., Kleppe, A.K., Van Kranendonk, M.J., Lindsay, J.F., Steele, A., Grassineau, N.V.: Questioning the evidence for Earth’s oldest fossils. Nature. 416, 78–81 (2002)
Brasier, M.D., Green, O.R., Lindsay, J.F., McLoughlin, N., Steele, A., Stoakes, C.: Critical testing of Earth’s oldest putative fossil assemblage from the 3.5 Ga Apex chert, Chinaman Creek Western Australia. Precambrian Res. 140, 55–102 (2005)
Brasier, M.D., Antcliffe, J., Saunders, M., Wacey, D.: Earth’s earliest fossils (3.5-1.9 Ga): changing the picture with new approaches and new discoveries. Proc. Natl. Acad. Sci. 112, 4859–4864 (2015)
Buick, R.: Microfossil recognition in Archaean rocks: an appraisal of spheroids and filaments from 3500 M.Y. old chert-barite at North Pole, Western Australia. PALAIOS. 5, 441–459 (1990)
Bundeleva, I.A., Shirokova, L.S., Bénézeth, P., Pokrovsky, O.S., Kompantseva, E.I., Balor, S.: Calcium carbonate precipitation by anoxygenic phototrophic bacteria. Chem. Geol. 291, 116–131 (2012)
Byerly, G.R., Palmer, M.R.: Tourmaline mineralization in the Barberton greenstone belt, South Africa: early Archean metasomatism by evaporite-derived boron. Contrib. Mineral. Petrol. 107, 387–402 (1991)
Byerly, G.R., Lowe, D.R., Walsh, M.M.: Stromatolites from the 3300-3500-Myr Swaziland Supergroup, Barberton Mountain Land, South Africa. Nature. 319, 489–491 (1986)
Cady, S.L.: Formation and preservation of bona fide microfossils. In: Signs of Life: A Report Based on the April, 2000 Workshop on Life-Detection Techniques, pp. 149–155. NRC-NAP, Washington, D.C. (2002)
Cameron, V., House CH, Brantley, S.L.: A first analysis of metallome biosignatures of hyperthermophilic archaea. Archaea. (2012). https://doi.org/10.1155/2012/789278
Camprubí, E., de Leeuw, J.W., House CH, Raulin, F., Russell, M.J., Spang, A., Tirumalai, M.R., Westall, F.: The emergence of life. Space Sci. Rev. 215, 56 (2019)
Cavalazzi, B., Barbieri, R., Gomez, F., Capaccioni, B., Olsson-Francis, K., Pondrelli, M., Rossi, A.P., Hickman-Lewis, K., Agangi, A., Gasparotto, G., Glamoclija, M., Ori, G.G., Rodriguez, N., Hagos M.: The Dallol Geothermal Area, Northern Afar (Ethiopia)–An Exceptional Planetary Field Analog on Earth. Astrobiology 19, 553–578 (2019)
Censi, P., Cangemi, M., Brusca, L., Madonia, P., Saiano, F., Zuddas, P.: The behavior of rare-earth elements, Zr and Hf during biologically-mediated deposition of silica-stromatolites and carbonate-rich microbial mats. Gondwana Res. 27, 209–215 (2013)
Chi Fru, E., Rodríguez, N.P., Partin, C.A., Lalone, S.V., Andersson, P., Weiss, D.J., El Albani, A., Rodushkin, I., Konhauser, K.O.: Cu isotopes in marine black shales record the Great Oxidation Event. Proc. Natl. Acad. Sci. USA. 113(18), 4941–4946 (2016)
Cloud, P.: Beginnings of biospheric evolution and their biogeochemical consequences. Paleobiology. 2(4), 351–387 (1976)
Cuerno, R., Escudero, C., García-Ruiz, J.-M., Herrero, M.A.: Pattern formation in stromatolites: Insights from mathematical modelling. J. R. Soc. Interface. 9, 1051–1062 (2012)
Danielson, A., Möller, P., Dulski, P.: The europium anomalies in banded iron formations and the thermal history of the oceanic crust. Chem. Geol. 97, 89–100 (1992)
Dass, A.V., Hickman-Lewis, K., Brack, A., Kee, T.P., Westall, F.: Stochastic prebiotic chemistry within realistic prebiotic geological systems. Chem. Select. 1(15), 4906–4926 (2016)
De Gregorio, B.T., Sharp, T.G.: Determining the biogenicity of microfossils in the apex chert, Western Australia, using Transmission Electron Microscopy. Lunar and Planetary Science XXXIV Houston (2003)
Djokic, T., Van Kranendonk, M.J., Campbell, K.A., Walter, M.R., Ward, C.R.: Earliest signs of life on land preserved in ca. 3.5 Ga hot spring deposits. Nat. Commun. 8 (2017). https://doi.org/10.1038/ncomms15263
Duan, Y., Anbar, A.D., Arnold, G.L., Lyons, T.W., Gordon, G.W., Kendall, B.: Molybdenum isotope evidence for mild environmental oxygenation before the great oxidation event. Geochim. Cosmochim. Acta. 74(23), 6655–6668 (2010)
Duda, J.-P., Thiel, V., Bauersachs, T., Mißbach, H., Reinhardt, M., Schäfer, N., Van Kranendonk, M.J., Reitner, J.: Ideas and perspectives: hydrothermally driven redistribution and sequestration of early Archaean biomass - the hydrothermal pump hypothesis. Biogeosciences 15, 1535–1548 (2018)
Dupraz, C., Pattisina, R., Verrecchia, E.P.: Translation of energy into morphology: simulation of stromatolite morphospace using a stocastic model. Sedimentary Geology 185(3-4), 185–203 (2006)
Edwards, N.P., Manning, P.L., Bergmann, U., Larson, P.L., van Dongen, B.E., Sellers, W.I., Webb, S.M., Sokaras, D., Alonso-Mori, R., Ignatyev, K., Barden, H.E., van Veelen, A., Anné, J., Egerton, V.M., Wogelius, R.A.: Leaf metallome preserved over 50 million years. Metallomics. 6, 774–782 (2014)
Fraústo da Silva, J.J.R., Williams, R.J.P.: The Biological Chemistry of the Elements, p. 600. Oxford University Press, Oxford (2001)
French, K.L., Hallmann, C., Hope, J.M., Schoon, P.L., Zumberge, J.A., Hoshino, Y., Peters, C.A., George, S.C., Love, G.D., Brocks, J.J., Buick, R., Summons, R.E.: Reappraisal of hydrocarbon biomarkers in Archean rocks. Proc. Natl. Acad. Sci. USA. 112(19), 5915–5920 (2015)
Furnes, H., Banerjee, N.R., Muehlenbachs, K., Staudigel, H., de Wit, M.: Early life recorded in Archean pillow lavas. Science. 304, 578–581 (2004)
Furnes, H., Banerjee, N.R., Staudigel, H., Muehlenbachs, K., McLoughlin, N., de Wit, M., Van Kranendonk, M.: Comparing petrographic signatures of bioalteration in recent to Mesoarchean pillow lavas: tracing subsurface life in oceanic igneous rocks. Precambrian Res. 158, 156–176 (2007)
Garcia-Ruiz, J.M., Hyde, S.T., Carnerup, A.M., Christy, A.G., Van Kranendonk, M.J., Welham, N.J.: Self-assembled silica-carbonate structures and detection of ancient microfossils. Science. 302(5648), 1194–1197 (2003)
Garcia-Ruiz, J.M., van Zuilen, M.A., Bach, W.: Mineral self-organization on a lifeless planet. Phys Life Rev. (2020)
Glikson, M., Duck, L.J., Golding, S.D., Hofmann, A., Bolhar, R., Webb, R., Baiano, J.C.F., Sly, L.I.: Microbial remains in some earliest Earth rocks: comparison with a potential modern analogue. Precambrian Res. 164, 187–200 (2008)
Gourcerol, B., Thurston, P.C., Kontak, D.J., Côté-Mantha, O.: Interpretations and implications of LA ICP-MS analysis of chert for the origin of geochemical signatures in banded iron formations (BIFs) from the Meadowbank gold deposit, Western Churchill Province, Nunavut. Chem. Geol. 410, 89–107 (2015)
Grassineau, N.F., Abell, P., Appel, P.W.U., Lowry, D., Nisbet, E.G.: Early life signatures in sulfur and carbon isotopes from Isua, Barberton, Wabigoon (Steep Rock), and Belingwe Greenstone Belts (3.8 to 2.7 Ga). In: Kesler, S.E., Ohmoto, H. (eds.) Evolution of Early Earth’s Atmosphere, Hydrosphere, and Biosphere––Constraints from Ore Deposits: GSA Memoir, vol. 198, pp. 33–52. Sage, New York (2006)
Grosch, E.G., McLoughlin, N.: Reassessing the biogenicity of Earth’s oldest trace fossil with implications for biosignatures in the search for early life. Proc. Natl. Acad. Sci. USA. 111, 8380–8385 (2014)
Grotzinger, J.P., Knoll, A.H.: Stromatolites in precambrian carbonates: evolutionary mileposts or environmental dipsticks? Annu. Rev. Earth Planet. Sci. 27, 313–358 (1999)
Grotzinger, J.P., Rothman, D.R.: An abiotic model for stromatolite morphogenesis. Nature. 383, 423–425 (1996)
Heubeck, C.: An early ecosystem of Archean tidal microbial mats (Moodies Group, South Africa, ca. 3.2 Ga). Geology. 37, 931–934 (2009)
Hickman-Lewis, K., Garwood, R.J., Brasier, M.D., Goral, T., Jiang, H., McLoughlin, N., Wacey, D.: Carbonaceous microstructures of the 3.46 Ga stratiform ‘Apex chert’, Chinaman Creek locality, Pilbara, Western Australia. Precambrian Res. 278, 161–178 (2016)
Hickman-Lewis, K., Garwood, R.J., Withers, P.J., Wacey, D.: X-ray microtomography as a tool for investigating the petrological context of Precambrian cellular remains. In: Brasier, A.T., McIlroy, D., McLoughlin, N. (eds.) Earth System Evolution and Early Life: A Celebration of the Work of Martin Brasier, vol. 448, pp. 33–56. Geological Society of London Special Publication, London (2017)
Hickman-Lewis, K., Cavalazzi, B., Foucher, F., Westall, F.: Most ancient evidence for life in the Barberton Greenstone Belt: microbial mats and biofabrics of the ~3.47 Ga Middle Marker Horizon. Precambrian Res. 312, 45–67 (2018)
Hickman-Lewis, K., Gautret, P., Arbaret, L., Sorieul, S., De Wit, R., Foucher, F., Cavalazzi, B., Westall, F.: Mechanistic morphogenesis of organo-sedimentary structures growing under geochemically stressed conditions: Keystone to the interpretation of some Archaean stromatolites? Geosciences. 9, 359 (2019)
Hickman-Lewis, K., Gourcerol, B., Westall, F., Manzini, D., Cavalazzi, B.: Reconstructing Palaeoarchaean microbial biomes flourishing in the presence of emergent landmasses using trace and rare earth element systematics. Precambrian Res. (2020c). https://doi.org/10.1016/j.precamres.2020.105689
Hickman-Lewis, K., Cavalazzi, B., Sorieul, S., Gautret, P., Foucher, F., Whitehouse, M.J., Jeon, H., Cockell, C.S., Georgelin, T., Westall, F.: Metallomics in deep time and the influence of ocean chemistry on the metabolic landscapes of Earth’s earliest ecosystems. Sci. Rep. 10(1) (2020b). https://doi.org/10.1038/s41598-020-61774-w
Hickman-Lewis, K., Westall, F., Cavalazzi, B.: Diverse communities of bacteria and archaea flourished in Palaeoarchaean (3.5-3.3 Ga) microbial mats. Palaeontology. (2020a). https://doi.org/10.1111/pala.12504
Hofmann, H.J., Grey, K., Hickman, A.H., Thorpe, R.I.: Origin of 3.45 Ga coniform stromatolites in Warrawoona Group, Western Australia. GSA Bull. 111, 1256–1262 (1999)
Homann, M., Heubeck, C., Airo, A., Tice, M.M.: Morphological adaptations of 3.22 Ga-old tufted microbial mats to Archean coastal habitats (Moodies Group, Barberton Greenstone Belt, South Africa). Precambrian Res. 266, 47–64 (2015)
Homann, M., Sansjofre, P., van Zuilen, M., Heubeck, C., Gong, J., Killingsworth, B., Foster, I.S., Airo, A., Van Kranendonk, M.J., Ader, M., Lalonde, S.V.: Microbial life and biogeochemical cycling on land 3,220 million years ago. Nat. Geosci. 11, 665–671 (2018)
Hren, M.T., Tice, M.M., Chamberlain, C.P.: Oxygen and hydrogen isotope evidence for a temperate climate 3.42 billion years ago. Nature. 205, 205–208 (2009)
Igisu, M., Ueno, Y., Takai, K.: FTIR microspectroscopy of carbonaceous matter in ~3.5 Ga seafloor hydrothermal deposits in the North Pole area, Western Australia. Prog. Earth Planet. Sci. 5 (2018). https://doi.org/10.1186/s40645-018-0242-1
Izon, G., Zerkle, A.L., Williford, K.H., Farquhar, J., Poulton, S.W., Claire, M.W.: Biological regulation of atmospheric chemistry en route to planetary oxygenation. Proc. Natl. Acad. Sci. USA. 114(13), E2571–E2579 (2017)
Johannesson, K.H., Telfeyan, K., Chevis, D.A., Rosenheim, B.E., Leybourne, M.I.: Rare earth elements in stromatolites–1. Evidence that modern terrestrial stromatolites fractionate rare earth elements during incorporation from ambient waters. In: Dilek, Y., Furnes, H. (eds.) Evolution of Archean Crust and Early Life. Springer, Dordrecht (2013). https://doi.org/10.1007/978-94-007-7615-9_14
Kamber, B.S., Webb, G.E.: The geochemistry of late Archean microbial carbonate: implications for ocean chemistry and continental erosion history. Geochim. Cosmochim. Acta. 65, 2509–2525 (2001)
Kendall, B., Brennecka, G.A., Weyer, S., Anbar, A.D.: Uranium isotope fractionation suggests oxidative uranium mobilization at 2.50 Ga. Chem. Geol. 362, 105–114 (2013)
Kiyokawa, S., Ito, T., Ikehara, M., Kitajima: Middle Archean volcano-hydrothermal sequience: Bacterial microfossil-bearing 3.2 Ga Dixon Island Formation, coastal Pilbara terrane, Australia. GSA Bull. 118(1–2), 3–22 (2006)
Kiyokawa, S., Koge, S., Ito, T., Ikehara, M.: An ocean-floor carbonaceous sedimentary sequence in the 3.2-Ga Dixon Island Formation, coastal Pilbara terrane, Western Australia. Precambrian Res. 255(1), 124–143 (2014)
Knauth, L.P.: Temperature and salinity history of the Precambrian ocean: implications for the course of microbial evolution. Palaeogeogr. Palaeoclimatol. Palaeoecol. 219, 53–69 (2005)
Knoll, A.H., Barghoorn, E.S.: Archean microfossils showing cell division from the Swaziland system of South Africa. Science. 198, 396–398 (1977)
Kozawa, T., Sugitani, K., Oehler, D.Z., House CH, Saito, I., Watanabe, T., Gotoh, T.: Early Archean plaktonic mode of life: implications from fluid dynamics of lenticular microfossils. Geobiology. 17(2), 113–126 (2019)
Lin, W., Paterson, G.A., Zhu, Q., Wang, Y., Kopylova, E., Li, Y., Knight, R., Bazylinski, D.A., Zhu, R., Kirschvink, J.L., Pan, Y.: Origin of microbial biomineralization and magnetotaxis during the Archean. Proc. Natl. Acad. Sci. USA. 114(9), 2171–2176 (2017)
Lowe, D.R.: Abiological origin of described stromatolites older than 3.2 Ga. Geology. 22, 387–390 (1994)
Maldanis, L., Hickman-Lewis, K., Verezhak, M., Gueriau, P., Guizar-Sicairos, M., Jaqueto, P., Trinidade, R.I.F., Rossi, A.L., Berenguer, F., Westall, F., Bertrand, L., Galante, D.: Nanoscale 3D quantitative imaging of 1.88 Ga Gunflint microfossils reveals novel insights into taphonomic and biogenic characters. Sci. Rep. 10, 8163 (2020)
Marin-Carbonne, J., Remusat, L., Sforna, M.C., Thomazo, C., Cartigny, P., Philippot, P.: Sulfur isotope’s signal of nanopyrites enclosed in 2.7 Ga stromatolitic organic remains reveal microbial sufate reduction. Geobiology. 16(2), 121–138 (2018)
Martin, W., Baross, J., Kelley, D., Russell, M.J.: Hydrothermal vents and the origin of life. Nat. Rev. Microbiol. 6, 805–814 (2008)
McCollum, T.M., Seewald, J.S.: Carbon isotope composition of organic compounds produced by abiotic synthesis under hydrothermal conditions. Earth Planet. Sci. Lett. 243, 74–84 (2006)
McLoughlin, N., Wilson, L.A., Brasier, M.D.: Growth of synthetic stromatolites and wrinkle structures in the absence of microbes – implications for the early fossil record. Geobiology. 6, 95–105 (2008)
McMahon, S.: Earth’s earliest and deepest purported fossils may be iron-mineralized chemical gardens. Proc. R. Soc. B. 286, 20192410 (2019)
Moore, E.K., Jelen, B.I., Giovannelli, D., Raanan, H., Falkowski, P.G.: Metal availability and the expanding network of microbial metabolisms in the Archaean eon. Nat. Geosci. 10, 629–636 (2017)
Muir, M.D., Grant, P.R.: Micropalaeontological evidence from the Onverwacht Group, South Africa. In: Windley, B.F. (ed.) The Early History of the Earth, pp. 595–608. Wiley/Interscience, London (1976)
Nisbet, E.G., Sleep, N.H.: The habitat and nature of early life. Nature. 409, 1083–1091 (2001)
Noffke, N.: Microbial Mats in Sandy Deposits from the Archean Era to Today, 175 p. Springer, New York (2010)
Noffke, N., Eriksson, K.A., Hazen, R.M., Simpson, E.L.: A new window into Archean life: microbial mats in Earth’s oldest siliciclastic tidal deposits (3.2 Ga Moodies Group, South Africa). Geology. 34, 253–256 (2006)
Noffke, N., Christian, D., Wacey, D., Hazen, R.M.: Microbially induced sedimentary structures recording an ancient ecosystem in the ca. 3.48 billion-year-old Dresser Formation, Pilbara, Western Australia. Astrobiology. 13, 1103–1124 (2013)
Nutman, A.P., Bennett, V.C., Friend, C.R., Van Kranendonk, M.J., Chivas, A.R.: Rapid emergence of life shown by discovery of 3,700-million-year-old microbial structures. Nature. 537, 535–538 (2016)
Nutman, A.P., Bennett, V.C., Friend, C.R.L., Van Kranendonk, M.J., Rothacker, L., Chivas, A.R.: Cross-examining earth’s oldest stromatolites: seeing through the effects of heterogeneous deformation, metamorphoism and metasomatism affecting Isua (Greenland) ~3700 Ma sedimentary rocks. Precambrian Res. 331 (2019). https://doi.org/10.1016/j.precamres.2019.105347
Oehler, D., Cady, S.L.: Biogenicity and syngeneity of organic matter in ancient sediments: recent advances in the search for evidence of past life. Challenges. 5(2), 260–283 (2014)
Oehler, D.Z., Walsh, M.M., Sugitani, K., Liu, M.-C., House, C.H.: Large and robust lenticular microorganisms on the young Earth. Precambrian Res. 296, 112–119 (2017)
Olempska, E., Wacey, D.: Ambient inclusion trails in Palaeozoic crustaceans (Phosphatocopina and Ostracoda). Palaeogeogr. Palaeoclimatol. Palaeoecol. 441(4), 949–958 (2016)
Oonk, P., Tsikos, H., Mason, P., Henkel, S., Staubwasser, M., Williams, H.: Species-specific Fe-isotopes on Palaeoproterozoic BIF and their implications. In: 35th International Geological Congress, Cape Town, South Africa, 27 August 2016-4 September 2016 (2016)
Petroff, A.P., Sim, M.S., Maslov, A., Krupenin, M., Rothman, D.H., Bosak, T.: Biophysical basis for the geometry of conical stromatolites. Proc. Natl. Acad. Sci. USA. 107, 9956–9961 (2010)
Philippot, P., Ávila, J.N., Killingsworth, B.A., Tessalina, S., Baton, F., Caquineau, T., Muller, E., Pecoits, E., Cartigny, P., Lalonde, S.F., Ireland, T.R., Thomazo, C., vanKranendonk, M.J., Busigny, V.: Globally asynchronous sulphur isotope signatls require re-definition of the great oxidation event. Nat. Commun. 9 (2018). https://doi.org/10.1038/s41467-018-04621-x
Rasmussen, B.: Filamentous microfossils in a 3,235-million-year-old volcanogenic massive sulphide deposit. Nature. 405, 676–679 (2000)
Reid, R.P., James, N.P., Macintyre, I.G., Dupraz, C.P., Burne, R.V.: Shark Bay stromatolites: microfabrics and reinterpretations of origins. Facies. 9, 243–270 (2003)
Riding, R.: The nature of stromatolites: 3,500 million year history and a century of research. In: Reitner, J., Quéric, N.-V., Arp, G. (eds.) Advances in Stromatolite Geobiology, pp. 29–74. Springer, Berlin, Heidelberg (2011)
Rosing, M.T.: 13C-depleted carbon microparticles in >3700-Ma sea-floor sedimentary rocks from West Greenland. Science. 283, 674–676 (1999)
Schidlowski, M.: Carbon isotopes as biogeochemical recorders of life over 3.8 Ga of Earth history: evolution of a concept. Precambrian Res. 106(1–2), 117–134 (2001)
Schopf, J.W., Packer, B.M.: Early Archean (3.3-billion to 3.5-billion-year-old) microfossils from Warrawoona Group, Australia. Science. 237(4810), 70–73 (1987)
Schopf, J.W., Walter, M.R.: Archean microfossils: new evidence of ancient microbes. In: Schopf, J.W. (ed.) Earth’s Earliest Biosphere: Its Origin and Evolution, pp. 214–239. Princeton University Press, Princeton, NJ (1983)
Schopf, J.W., Kudryavtsev, A.B., Agresti, D.G., Wdowiak, T.J., Czaja, A.D.: Laser-Raman imagery of Earth’s earliest fossils. Nature. 416, 73–76 (2002)
Schopf, J.W., Kudryavtsev, A.B., Dzaja, A.D., Tripathi, A.B.: Evidence of archean life: stromatolites and microfossils. Precambrian Res. 158, 141–155 (2007)
Sforna, M.C., Daye, M., Philippot, P., Somogyi, A., van Zuilen, M.A., Medjoubi, K., Gérard, E., Jamme, F., Dupraz, C., Braissant, O., Glunk, G., Visscher, P.T.: Patterns of metal distribution in hypersaline microbialites during early diagenesis: Implications for the fossil record. Geobiology 15(2), 259–279 (2017)
Shields, G., Webb, G.: Has the REE composition of seawater changed over geological time? Chem. Geol. 204, 103–107 (2004)
Sleep, N.H.: The Hadean-Archaean environment. Cold Spring Harb. Perspect. Biol. 2, a002527 (2010)
Sleep, N.H.: Planetary interior-atmosphere interaction and habitability. In: Deeg, H.J., Belmonte, J.A. (eds.) Handbook of Exoplanets, pp. 1–22. Springer, Berlin (2018)
Staudigel, H., Furnes, H., de Wit, M.: Paleoarchean trace fossils in altered volcanic glass. Proc. Natl. Acad. Sci. USA. 112, 6892–6897 (2015)
Sugahara, H., Sugitani, K., Mimura, K., Yamashita, F., Yamamoto, K.: A systematic rare-earth elements and yttrium study of Archean cherts at the Mount Goldsworthy greenstone belt in the Pilbara Craton: Implications for the origin of microfossil-bearing black cherts. Precambrian Res. 177, 73–87 (2010)
Sugitani, K., Grey, K., Allwood, A., Nagaoka, T., Mimura, K., Minami, M., Marshall, C.P., Van Kranendonk, M.J., Walter, M.R.: Diverse microstructures from Archean chert from the Mount Goldsworthy-Mount Grant area, Pilbara Craton, Western Australia: microfossils, dubiofossils, or pseudofossils? Precambrian Res. 158, 228–262 (2007)
Sugitani, K., Grey, K., Nagaoka, T., Mimura, K., Walter, M.R.: Taxonomy and biogenicity of Archaean spheroidal microfossils (ca. 3.0 Ga) from the Mount Goldsworthy-Mount Grant area in the northeastern Pilbara Craton, Western Australia: Precambrian Res. 173, 50–59 (2009)
Sugitani, K., Mimura, K., Takeuchi, M., Yamaguchi, T., Suzuki, K., Senda, R., Asahara, Y., Wallis, S., Van Kranendonk, M.J.: A Paleoarchean coastal hydrothermal field inhabited by diverse microbial communities: the Strelley Pool Formation, Pilbara Craton, Western Australia. Geobiology. 13, 522–545 (2015)
Summons, R.E., Amend, J.P., Bish, D., Buick, R., Cody, G.D., Des Marais, D.J., Dromart, G., Eigenbrode, J.L., Knoll, A.H., Sumner, D.Y.: Preservation of martian organic and environmental records: Final report of the biosignature working group. Astrobiology. 11(2), 157–181 (2011)
Takahashi, Y., Châtellier, X., Hattori, K.H., Kato, K., Fortin, D.: Adsorption of rare earth elements onto bacterial cell walls and its implication for REE sorption onto natural microbial mat. Chem. Geol. 219, 53–67 (2005)
Tartèse, R., Chaussidon, M., Gurenko, A., Delarue, F., Robert, F.: Warm Archean oceans reconstructed from oxygen isotope composition of early-life remnants. Geochem. Perspect. Lett. 3, 55–65 (2017)
Tashiro, T., Ishida, A., Hori, M., Igisu, M., Koike, M., Mejean, P., Takahata, N., Sano, Y., Komiya, T.: Early trace of life from 3.95 Ga sedimentary rocks in Labrador, Canada. Nature. 549, 516–518 (2017)
Teng, F.-Z., Dauphas, N., Watkins, J.M.: Non-traditional stable isotopes: retrospective and prospective. Rev. Mineral. Geochem. 82, 1–26 (2017)
Tice, M.M.: Environmental controls on photosynthetic microbial mat distribution on and morphogenesis on a 3.42 Ga clastic-starved platform. Astrobiology. 9, 989–1000 (2009)
Tice, M.M., Lowe, D.R.: Photosynthetic microbial mats in the 3416-Myr-old ocean. Nature. 431, 549–552 (2004)
Tice, M.M., Lowe, D.R.: Hydrogen-based carbon fixation in the earliest known photosynthetic organisms. Geology. 34, 37–40 (2006a)
Tice, M.M., Lowe, D.R.: The origin of carbonaceous matter in pre-3.0 Ga greenstone terrains: a review and new evidence from the 3.42 Ga Buck Reef Chert. Earth Sci. Rev. 76, 259–300 (2006b)
Tomescu, A.M.F., Klymiuk, A.A., Matsunaga, K.K.S., Bippus, A.C., Shelton, G.W.K.: Microbes and the fossil record. In: Hurst, C.J. (ed.) Advances in Environmental Microbiology. Their World: A Diversity of Microbial Environments, pp. 69–169. Springer, Heidelberg (2016)
Trower, E.J., Lowe, D.R.: Sedimentology of the ~3.3 Ga upper Mendon Formation, Barberton Greenstone Belt, South Africa. Precambrian Res. 281, 473–494 (2016)
Ueno, Y., Isozaki, Y., Yurimoto, H., Maruyama, S.: Carbon isotopic signatures of individual Archean microfossils (?) from Western Australia. Int. Geol. Rev. 43(3), 196–212 (2001a)
Ueno, Y., Maruyama, S., Isozaki, Y., Yurimoto, H.: Early Archean (ca. 3.5 Ga) microfossils and 13C- depleted carbonaceous matter in the North Pole area, Western Australia: field occurrence and geochemistry. In: Nakashima, S., Maruyama, S., Brack, A., Windley, B.F. (eds.) Geochemistry and the Origin of Life, pp. 201–236. Universal Academy Press, Tokyo (2001b)
Ueno, Y., Isozaki, Y., McNamara, K.J.: Coccoid-like microstructures in a 3.0 Ga chert from Western Australia. Int. Geol. Rev. 48(1), 78–88 (2006a)
Ueno, Y., Yamada, K., Yoshida, N., Maruyama, S., Isozaki, Y.: Evidence from fluid inclusions for microbial methanogenesis in the early Archaean era. Nature. 440, 516–519 (2006b)
Ueno, Y., Ono, S., Rumble, D., Maruyama, S.: Quadruple sulfur isotope analysis of ca. 3.5 Ga Dresser Formation: new evidence for microbial sulfate reduction in the early Archaen. Geochim. Cosmochim. Acta. 72(23), 5675–5691 (2008)
van den Boorn, S.H.J.M., van Bergen, M., Nijman, W., Vroon, P.: Dual role of seawater and hydrothermal fluids in Early Archean chert formation: evidence from silicon isotopes. Geology. 35, 939–942 (2007)
van den Boorn, S.H.J.M., van Bergen, M.J., Vroon, P.Z., de Vries, S.T., Nijman, W.: Silicon isotope and trace element constraints on the origin of ~3.5 Ga cherts: implications for early Archaean marine environments. Geochim. Cosmochim. Acta. 74, 1077–1103 (2010)
Van Kranendonk, M.J.: Volcanic degassing, hydrothermal circulation and the flourishing of early life on Earth: new evidence from the Warrawoona Group, Pilbara Craton, Western Australia. Earth Sci. Rev. 74, 197–240 (2006)
Van Kranendonk, M.J., Webb, G.E., Kamber, B.S.: Geological and trace element evidence for a marine sedimentary environment of deposition and biogenicity of 3.45 Ga stromatolitic carbonates in the Pilbara Craton, and support for a reducing Archaean ocean. Geobiology. 1(2), 91–108 (2003)
Van Zuilen, M.A., Chaussidon, M., Rollion-Bard, C., Marty, B.: Carbonaceous cherts of the Barberton Greenstone Belt, South Africa: Isotopic, chemical and structural characteristics of individual microstructures. Geochim. Cosmochim. Acta. 71(3), 655–669 (2007)
Wacey, D.: ~3,240 Ma, Kangaroo Caves formation, East Pilbara, Western Australia. In: Wacey, D. (ed.) Early Life on Earth, pp. 221–227. Springer, Dordrecht (2009)
Wacey, D., Kilburn, M., Stoakes, C., Aggleton, H., Brasier, M.: Ambient inclusion trails: their recognition, age, range, and applicability to early life on Earth. In: Dikel, Y., Furnes, H., Muehlenbachs, K. (eds.) Links Between Geological Processes, Microbial Activities and Evolution of Life, vol. 4, pp. 113–134. Springer, Dordrecht (2008b)
Wacey, D., Kilburn, M.R., McLoughlin, N., Parnell, J., Stoakes, C.A., Grovenor, C.R.M., Brasier, M.D.: Use of NanoSIMS in the search for early life on Earth: ambient inclusion trails in a c. 3400 Ma sandstone. J. Geol. Soc. 165, 43–53 (2008a)
Wacey, D., Kilburn, M.R., Saunders, M., Cliff, J., Brasier, M.D.: Microfossils of sulphur-metabolizing cells in 3.4-billion-year-old rocks of Western Australia. Nat. Geosci. 4, 698–702 (2011)
Wacey, D., McLouglin, N., Kilburn, M.R., Saunders, M., Cliff, J.B., Kong, C., Barley, M.E., Brasier, M.D.: Nanoscale analysis of pyritized microfossils reveals differential heterotrophic consumption in the ∼1.9-Ga Gunflint chert. Proc. Natl. Acad. Sci. USA. 110, 8020–8024 (2013)
Wacey, D., Saunders, M., Cliff, J., Kilburn, M.R., Kong, C., Barley, M.E., Brasier, M.D.: Geochemistry and nano-structure of a putative ~3240 million-year-old black smoker biota, Sulphur Springs Group, Western Australia. Precambrian Res. 249, 1–12 (2014)
Wacey, D., Saunders, M., Kong, C., Brasier, A.T., Brasier, M.D.: 3.46 Ga Apex chert ‘microfossils’ reinterpreted as mineral artefacts produced during phyllosilicate exfoliation. Gondwana Res. 36, 296–313 (2016)
Wacey, D., Saunders, M., Kong, C.: Remarkably preserved tephra from the 3430 Ma Strelley Pool Formation, Western Australia: Implications for the interpretation of Precambrian microfossils. Earth Planet. Sci. Lett. 487, 33–43 (2018)
Walsh, M.M.: Microfossils and possible microfossils from the early Archean Onverwacht group, Barberton Mountain Land, South Africa. Precambrian Res. 54, 271–293 (1992)
Walsh, M.M., Lowe, D.R.: Filamentous microfossils from the 3,500-Myr-old Onverwacht Group, Barberton Mountain Land, South Africa. Nature. 314, 530–532 (1985)
Walsh, M.M., Lowe, D.R.: Modes of accumulation of carbonaceous matter in the early Archaean: a petrographic and geochemical study of the carbonaceous cherts of the Swaziland Supergroup. In: Lowe, D.R., Byerly, G.R. (eds.) Geologic Evolution of the Barberton Greenstone Belt, South Africa, vol. 329, pp. 115–132. Geological Society of America, Boulder (1999)
Walsh, M.M., Westall, F.: Archean biofilms preserved in the Swaziland Supergroup, South Africa. In: Krumbein, W.E., Paterson, D.M., Zavarzin, G.A. (eds.) Fossil and Recent Biofilms, pp. 307–316. Kluwer, Dordrecht (2003)
Walter, M.R., Buick, R., Dunlop, J.: Stromatolites, 3400–3500 Myr old from the North Pole area, Western Australia. Nature. 284, 443–445 (1980)
Westall, F., Brack, A.: The importance of water for life. Space Sci. Rev. 214, 50 (2018)
Westall, F., de Vries, S.T., Nijman, W., Rouchon, V., Orberger, B., Pearson, V., Watson, J., Verchovsky, A., Wright, I., Rouzaud, J.-N., Marchesini, D., Anne, S.: The 3.466Ga Kitty’s Gap chert, an Early Archean microbial ecosystem. In: Reimold, W.U., Gibson, R.L. (eds.) Processes on the Early Earth, vol. 405, pp. 105–131. Geological Society of America, Boulder (2006)
Westall, F., Foucher, F., Cavalazzi, B., de Vries, S., Nijman, W., Pearson, V., Watson, J., Verchovsky, A., Wright, I., Rouzaud, J.-N., Marchesini, D., Anne, S.: Volcaniclastic habitats for early life on Earth and Mars: a case study from c.3.5 Ga-old rocks from the Pilbara, Australia. Planet. Space Sci. 59, 1093–1106 (2011)
Westall, F., Campbell, K.A., Bréhéret, F.G., Foucher, F., Gautret, P., Hubert, A., Sorieul, S., Grassineau, N., Guido, D.M.: Complex microbe-sediment systems are ancient (3.33 Ga) and flourished in a hydrothermal context. Geology. 43, 615–618 (2015)
Westall, F., Hickman-Lewis, K., Hinman, N., Gautret, P., Campbell, K.A., Bréhéret, J.G., Foucher, F., Hubert, A., Sorieul, S., Dass, A.V., Kee, T.P., Georgelin, T., Brack, A.: A hydrothermal-sedimentary context for the origin of life. Astrobiology. 18(3), 259–293 (2018)
Williams, R.J.P.: Chemical selection of elements by cells. Coord. Chem. Rev. 216–217, 583–595 (2001)
Zerkle, A.L., House, C.H., Brantley, S.L.: Biogeochemical signatures through time as inferred from whole microbial genomes. Am. J. Sci. 305, 467–502 (2005)
Zhang, M., Konishi, H., Xu, H., Sun, X., Lu, H., Wu, D., Wu, N.: Morphology and formation mechanism of pyrite induced by the anaerobic oxidation of methane from the continental slope of the NE South China Sea. J. Asian Earth Sci. 92, 293–301 (2014)
Acknowledgements
SLC thanks the NASA Astrobiology Institute (NASA CAN7 Award# 16BB06I to the SETI Institute) and the PNNL EMSL user facility (DoE BER program).
Author information
Authors and Affiliations
Corresponding authors
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Cavalazzi, B., Hickman-Lewis, K., Brack, A., Cady, S.L. (2021). Earliest Traces of Life as a Window on Life’s Origins. In: Neubeck, A., McMahon, S. (eds) Prebiotic Chemistry and the Origin of Life. Advances in Astrobiology and Biogeophysics. Springer, Cham. https://doi.org/10.1007/978-3-030-81039-9_10
Download citation
DOI: https://doi.org/10.1007/978-3-030-81039-9_10
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-81038-2
Online ISBN: 978-3-030-81039-9
eBook Packages: Physics and AstronomyPhysics and Astronomy (R0)