Abstract
Background: Standard semen evaluation according to WHO thresholds (2010) is still insufficient to disclose individuals with a diagnosis of idiopathic infertility. Sperm DNA fragmentation is considered a possible valuable marker, and flow-cytometry-based Sperm Chromatin Structure Assay (SCSA) analysis has been used. However, the method is cumbersome and time-consuming; hardly applicable in a clinical setting where split ejaculates are used for diagnostics and intervention. The present study explores an alternative method using conventional bright-field microscopy (HalospermG2, Halothech, Madrid, Spain) with Sperm DNA fragmentation (SDF) as end values. Analyses were done on split ejaculates (F1, sperm-rich fraction, mainly prostate-dominated versus F2, sperm-poor fraction, mainly seminal vesicle-dominated) of documented fertile donors and partners in a couple seeking for ART-intervention with a clinical diagnosis of idiopathic infertility (patients).
Access provided by Autonomous University of Puebla. Download conference paper PDF
Similar content being viewed by others
Background: Standard semen evaluation according to WHO thresholds (2010) is still insufficient to disclose individuals with a diagnosis of idiopathic infertility. Sperm DNA fragmentation is considered a possible valuable marker, and flow-cytometry-based Sperm Chromatin Structure Assay (SCSA) analysis has been used. However, the method is cumbersome and time-consuming; hardly applicable in a clinical setting where split ejaculates are used for diagnostics and intervention. The present study explores an alternative method using conventional bright-field microscopy (HalospermG2, Halothech, Madrid, Spain) with Sperm DNA fragmentation (SDF) as end values. Analyses were done on split ejaculates (F1, sperm-rich fraction, mainly prostate-dominated versus F2, sperm-poor fraction, mainly seminal vesicle-dominated) of documented fertile donors and partners in a couple seeking for ART-intervention with a clinical diagnosis of idiopathic infertility (patients).
Main Question: Can DNA-fragmentation analysis by HalospermG2 be applied in a clinical setting for an idiopathic infertility survey?
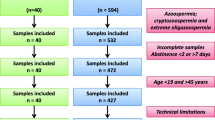
Experimental Design: Consensual fertile semen donors (n = 14) and patients (n = 6) attending clinical setting (Reproduction Medicine Centre (RMC), Linköping, Sweden). Ejaculates were collected by masturbation in two consecutive fractions (sperm-rich F1 and sperm-poor F2). Samples were evaluated for volume, sperm concentration (manual counting), and motility (subjective) following WHO-criteria and also for proportions of DNA fragmentation (SDF) using a commercially available kit (HalospermG2; Halotech DNA, Madrid, Spain). Statistical analysis was performed in the R statistical environment (ISBN 3-900051-07-0). Data are expressed as means ± SD, (range).
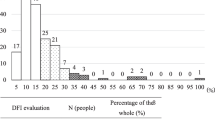
Main Results: The restricted number of individuals hereby explored yield no differences between donors (SDF (%): F1, 19.8 ± 9.4 (7.87–42.15); F2, 23.2 ± 13.4 (5.95–50.68)) (means ± SD, ranges between brackets) and the patients (SDF (%): F1, 17.7 ± 6.6 (9.66–28.98); F2, 20.8 ± 10.9 (10.60–43.18)), which was extensive to the fraction where spermatozoa were fortuitously recovered from. However, there was a wider range for F1 than for F2.
Conclusions: DNA fragmentation analysis using the HalospermG2 kit can be easily performed in an ART setting. HalospermG2 calculates a global ratio from the fragmented-degraded sperm divided by the total of spermatozoa counted. The limited number of individuals diagnosed with idiopathic infertility makes us refrain from other conclusions, but values are similar to data emanated from SCSA (Sperm Chromatin Stability Analysis) using flow cytometry on spermatozoa from the same individuals (Alvarez-Rodriguez et al. 2016), considering SCSA has an inbuilt threshold in the resulting data (a DNA-fragmentation index (DFI) lower than 25% is considered normal).
Reference
Alvarez-Rodriguez M, Liffner S, Vicente-Carrillo A, Hammar M, Rodriguez-Martinez H (2016) Sperm chromatin integrity (SCSA analysis) in specific ejaculate fractions can improve semen evaluation under clinical settings. Annual meeting ESHRE, Helsinki, Finland, 2016. Human Reproduction 31 S1 i163-i164
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 Springer Nature Switzerland AG
About this paper
Cite this paper
Alvarez-Rodríguez, M., Pehrson, I., Liffner, S., Hammar, M., Rodríguez-Martinez, H. (2021). Sperm DNA Fragmentation in Human Split Ejaculates. In: Björndahl, L., Flanagan, J., Holmberg, R., Kvist, U. (eds) XIIIth International Symposium on Spermatology. Springer, Cham. https://doi.org/10.1007/978-3-030-66292-9_12
Download citation
DOI: https://doi.org/10.1007/978-3-030-66292-9_12
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-66291-2
Online ISBN: 978-3-030-66292-9
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)