Abstract
Approximately two-thirds of patients with focal epilepsy will not achieve a sustained seizure-free outcome. When two appropriate antiseizure medications (ASMs) fail to produce sustained seizure freedom, used at optimal doses over a reasonable period of time, further trials of antiseizure drugs are unlikely to be effective. Epilepsy surgery is a standard of care for selected patients who fail medical management. Suitable candidates are motivated, have a single localized seizure focus, develop seizures from an accessible area in the brain, and are disabling to the patient. The most common operation involves temporal lobe surgery in the form of anterior temporal lobectomy. However selective amygdalohippocampectomy and laser ablation are other alternative surgical techniques that are less invasive. When a high-resolution brain MRI identifies a structural lesion such as hippocampal sclerosis and the clinical history and electrophysiological testing are concordant, the outcome for seizure freedom following surgery is excellent. Furthermore, when all lines of evidence during “phase 1” presurgical evaluation are concordant (i.e., history and semiology, MRI, PET, video-EEG monitoring, neuropsychological testing), these candidates may “skip” implantation of invasive electrodes with intracranial EEG monitoring and proceed directly to surgery. Unfortunately, only a small percentage of potential surgical candidates are being referred to surgical epilepsy centers. Lengthy delays of 15–20 years are unfortunately common. Overall, 50–70% of patients become seizure-free of disabling seizures with limited morbidity postoperatively, and with lesional epilepsy 70–90% become seizure-free.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keyword
Case Presentation
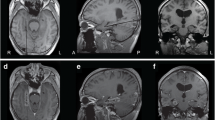
A 47-year-old right-handed Indian-American female was self-referred for uncontrolled seizures. She was healthy other than taking antiseizure medication (ASM) for epilepsy. When she was 2 years of age, she experienced a prolonged febrile convulsion lasting 15 min associated with a fever of 103°. At the time she was told “nothing was wrong” and that is was a benign occurrence. She developed normally throughout childhood with above average scholastic achievement. At 11 years of age, she developed her first afebrile seizure which recurred manifest as a staring spell that she referred to as a “petit mal” seizure . Her seizures involved a warning where she would experience an indescribable feeling just prior to a wide-eyed stare, manifest subtle lip smacking, and impaired responsiveness for 45 seconds in duration. Following this she would be sleepy with transient difficulty “getting the words out.” She failed five ASM with ongoing seizures and was maintained on lamotrigine and levetiracetam. Several seizures per month occurred with rare injury mostly involving lacerations, abrasions, and contusions of the head. She never experienced a “grand mal” seizure . Her neurological examination was normal. A high-resolution brain MRI with an epilepsy protocol demonstrated left mesial temporal sclerosis (Fig. 41.1a). EEG revealed left anterior temporal interictal epileptiform discharges (Fig. 41.1b ). A surgical evaluation was recommended to her by her neurologist after she fell down a flight of stairs. Subsequent evaluation included a FDG-PET scan of the brain that revealed hypometabolism of the left temporal lobe. She was admitted to the hospital’s epilepsy monitoring unit and underwent video-EEG monitoring . During this time frequent state-independent left anterior temporal epileptiform discharges were apparent, and three focal seizures typical of her outpatient events were recorded (Fig. 41.2). Neuropsychological testing revealed mild verbal memory deficit. A Wada test was performed. Injection of sodium brevital revealed 8/8 object recall and aphasia upon left hemispheric injection and 0/8 recall and dysarthria upon right injection. A left amygdalohippocampectomy was recommended; however, her son (an anesthesiologist) recommended against it. 10 years elapsed before surgery was performed. Following surgery, she became seizure-free for more than 2 years.
Clinical Questions
-
1.
What parts of the clinical history suggest focal seizures?
-
2.
What is the likelihood that further ASM will result in seizure freedom, and what are the reasons for drug resistance?
-
3.
Why is seizure monitoring needed when the MRI is abnormal?
-
4.
What further testing is required if surgery is to be pursued?
-
5.
What is the prognosis after surgery for seizures, and what about surgical consequences?
Diagnostic Discussion
-
1.
The diagnosis of epilepsy is suggested by the paroxysmal recurrent episodes of impaired consciousness, and a treatment algorithm is practical [1]. Many patients describe their seizures as “petit mal” seizures when they are nonconvulsive, though in 70% of adults, focal seizures rather than generalized seizures predominate. In two-thirds of these individuals, ASM will not result in sustained seizure control. Many adults with focal seizures experience a warning (aura), though it is the post-ictal state that is the characteristic feature of focal seizures to distinguish it from other events associated with transitory loss of consciousness including absence seizures or “petit mal.” If uncontrolled seizures are permitted to continue, a greater risk for morbidity with higher accident and injury rates, psychiatric and cognitive deterioration (especially memory), social isolation, stigmatization, and impaired self-esteem, and even mortality from sudden unexplained death accrues.
-
2.
Approximately one-third of patients with focal seizures and 15–20% with generalized epilepsy will remain refractory to ASM despite different treatment options [2]. After the failure of two appropriate ASMs given for an adequate duration, at an effective dose, there is less than a 5–10% likelihood that further ASM changes will result in seizure freedom. It is important to exclude pseudo-resistance as the reason for drug failure. An incorrect diagnosis may result in ongoing seizures because treatment of a nonepileptic seizure mimic is unlikely to respond to ASM. Similarly, treatment with an incorrect ASM choice targeting the wrong seizure or epilepsy type or too low a dose of ASM will result in apparent drug failure. Genetic generalized epilepsies for example may be aggravated by narrow spectrum ASM such as carbamazepine or phenytoin, and the use of ethosuximide to treat “petit mal” seizures will be ineffective in patients with focal seizures. Patient-related issues are yet another reason for poor results. When noncompliance or an adverse lifestyle is encountered, the ASM may be the correct choice, though efficacy may be compromised due to reasons such as subtherapeutic use from non-compliance or drug and alcohol abuse. Epilepsy surgery is a standard of care [3] and remains a cornerstone of therapy due to efficacy proven in randomized controlled trials as a more effective treatment compared to continued medial therapy when seizures continue despite ASM [4, 5].
-
3.
When the MRI and interictal EEG are concordant, the likelihood of a correct localization is approximately 80%. The demonstration of a “lesion” (mesial temporal sclerosis on MRI in our patient) has the best predictive value as a localizing feature and as a favorable prognosticator for a successful outcome following epilepsy surgery [6,7,8]. Ictal EEG recordings are recommended to confirm the diagnosis of epilepsy. Approximately 20–30% of patients admitted for epilepsy monitoring will not have epilepsy. The majority of them have psychogenic nonepileptic attacks (PNEA) . Even in patients with epileptic seizures (ES), about 10–15% may exhibit both ES and PNES. Excluding incorrect ASM choices will be made possible by accurately classifying the seizures correctly when they are captured during video-EEG monitoring. Recording EEG during seizures will also identify a single semiology and ictal EEG pattern that morphologically is able to support a diagnosis of unifocal epilepsy. Excluding more than one source for generating recurrent seizures may be difficult based solely upon semiology. An example of the latter situation may be seen in patients with bitemporal epilepsy where staring episodes may be caused by focal seizures arising from each hemisphere independently.
-
4.
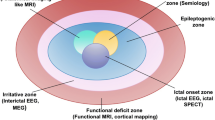
When all aspects of a “phase 1” evaluation (non-invaive) are concordant (i.e., history and semiology, MRI, PET, video-EEG monitoring , neuropsychological testing ), these candidates may “skip” and proceed directly to surgery without the need to undergo further invasive EEG monitoring with intracranial electrodes for further seizure localization. In our patient, Wada testing was used to firmly localize language function and predict memory function after surgery which was robust, demonstrating a significant difference in participation between the hemispheres to predict a favorable outcome with respect to working memory in addition to anticipating seizure freedom. Functional MRI has been used to identify atypical areas subserving language and is increasingly being used in place of Wada testing as a noninvasive alternative. All of the results of the presurgical evaluation are favorable in our patient and provide localizing information to identify impaired hippocampal function within a limbic neural network. Each of the classic aspects of the presurgical evaluation strongly suggest a favorable outcome with respect to eliminating seizures through surgery.
-
5.
This patient illustrates the most common surgically remediable syndrome of drug-resistant temporal lobe epilepsy (TLE) . The presurgical evaluation above involving MRI, PET, and interictal and ictal EEG demonstrate concordance to support unifocal localization. Unfortunately, only a small percentage of potential surgical candidates are being referred to surgical epilepsy centers. Lengthy delays of 15–20 years are unfortunately common. However, 50–90% of patients become seizure-free with limited morbidity postoperatively. Laser interstitial thermal therapy is another option that potentially has less cognitive consequences albeit with a slightly reduced likelihood of resultant seizure-free outcome [9, 10]. Overall, the most favorable predictor to obtain a seizure-free outcome following surgery or ablation exists when a lesion is present on neuroimaging. It has a high rate of success especially if it is due to hippocampal sclerosis as in our patient. Complications are related to the craniotomy and to the site and the extent of resected tissue. Nobody “wants” surgery, but it is important to present surgery as an option in a realistic and objective fashion. After declining epilepsy surgery for years despite experiencing uncontrolled seizures, our patient underwent successful surgery despite the urging of her family to the contrary. Early surgery has proven to be 15–21% more effective than delayed surgery [11]. She had no complications and became seizure-free as expected. She later wished she would have undergone surgery 10 years earlier and not listened to her son who advised against it.
Pearls of Wisdom
-
1.
Epilepsy surgery is a standard of care and should be considered early when a patient with focal seizures has proven to be drug-resistant to ASM.
-
2.
A lesion on neuroimaging is the best predictor for localizing seizure onset and for prognosticating a seizure-free outcome following epilepsy surgery or stereotactic laser interstitial thermal ablation.
-
3.
Video-EEG monitoring is essential to perform before epilepsy surgery. It will verify the diagnosis of epilepsy and exclude the possibility of other seizure mimics as the reason for drug resistance. In addition, it can provide localizing information about the site of seizure onset by demonstrating electrophysiological information that is concordant with the other “phase 1” evaluations to allow the patient to “skip” invasive monitoring and proceed directly to surgical therapy when concordance is identified.
-
4.
Temporal lobe epilepsy is the most common epilepsy surgery performed. TLE is often due to hippocampal sclerosis and the most common adult epilepsy syndrome that is amenable to surgery.
-
5.
Overall, about 70% of patients are seizure-free after surgery, and an additional 20% have a significant reduction in their seizures. Though the ideal surgical candidate has the best predictability for a seizure-free outcome, epilepsy surgery is more likely to result in seizure freedom when patients have failed >2 appropriate trials of ASM.
References
Jobst BC. Treatment algorithms in refractory partial epilepsy. Epilepsia. 2009;50(Suppl 8):51–6.
Engel J Jr. What can we do for people with drug-resistant epilepsy? The 2016 Wartenberg lecture. Neurology. 2016;87:2483–9.
Wiebe S, Blume WT, Girvin JP, Eliasziw M. A randomized, controlled trial of surgery for temporal-lobe epilepsy. N Engl J Med. 2001;345(5):311–8.
Engel J Jr, McDermott MP, Wiebe S, Langfitt JT, Stern JM, Dewar S, Sperling MR, Gardiner I, Erba G, Fried I, Jacobs M, Vinters HV, Mintzer S, Kieburtz K. Early surgical therapy for drug-resistant temporal lobe epilepsy: a randomized trial. JAMA. 2012;307:922–30.
Engel J Jr, Wiebe S, French J, et al. Practice parameter: temporal lobe and localized neocortical resections for epilepsy: report of the quality standards subcommittee of the American Academy of Neurology, in association with the American Epilepsy Society and the American Association of Neurological Surgeons. Neurology. 2003;60(4):538–47.
Spencer SS, Berg AT, Vickrey BG, et al. Initial outcomes in the Multicenter study of epilepsy surgery. Neurology. 2003;61(12):1680–5.
Englot DJ, Chang EF. Rates and predictors of seizure freedom in resective epilepsy surgery: an update. Neurosurg Rev. 2014;37:389–404.
Capraz IY, Kurt G, Akdemir O, Hirfanoglu T, Oner Y, Sengezer T, Kapucu LO, Serdaroglu A, Bilir E. Surgical outcome in patients with MRI-negative, PET-positive temporal lobe epilepsy. Seizure. 2015;29:63–8.
Grewal SS, Tatum WO. Laser thermal ablation in epilepsy. Expert Rev Neurother. 2019;6:1–8.
Drane DL, Loring DW, Voets NL, Price M, Ojemann JG, Willie JT, Saindane AM, Phatak V, Ivanisevic M, Millis S, Helmers SL, Miller JW, Meador KJ, Gross RE. Better object recognition and naming outcome with MRI-guided stereotactic laser amygdalohippocampotomy for temporal lobe epilepsy. Epilepsia. 2015;56:101–13.
Bjellvi J, Olsson I, Malmgren K, Ramsay KW. Epilepsy duration and seizure outcome in epilepsy surgery. Neurology. 2019;93(2):e159. https://doi.org/10.1212/WNL.0000000000007753.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Tatum, W.O., Grewal, S.S. (2021). Surgical Candidate (Skip). In: Tatum, W.O., Sirven, J.I., Cascino, G.D. (eds) Epilepsy Case Studies. Springer, Cham. https://doi.org/10.1007/978-3-030-59078-9_41
Download citation
DOI: https://doi.org/10.1007/978-3-030-59078-9_41
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-59077-2
Online ISBN: 978-3-030-59078-9
eBook Packages: MedicineMedicine (R0)