Abstract
Wasps are a true model in studies on the origin and evolution of cooperative behavior and the mechanisms that help to stabilize sociality and resolve internal conflicts. Indeed, the wide variety of social organizations found in the group – ranging from solitary to highly social – provides unique opportunities to test how cooperation evolved and how conflicts are suppressed in societies with different degrees of social complexity. As the individuals in wasp colonies are not genetic clones of each other, inclusive fitness theory predicts that there should often be significant scope for conflicts between the queen and worker castes arising from specific genetic relatedness asymmetries within the colony. In many species, for example, the workers retain the ability to develop their ovaries and lay unfertilized male-destined eggs, thereby allowing them to challenge the reproductive monopoly of the queen. The amount of worker reproduction that is tolerated within the colony is a function of the genetic relatedness patterns within the colony and the costs and benefits of cheating, which under some circumstances can drive social enforcement mechanisms, whereby eggs laid by workers are selectively cannibalized or “policed” by the queen herself or by other workers. In some wasp species, such policing is so effective that workers are better off not trying to reproduce in the first place because nearly all the workers’ eggs would be policed anyway. The fact that policing can align the evolutionary interests of the queens and workers facilitates the evolution of queen signaling systems (i.e. queen pheromones, that act as honest signals for the presence of a healthy and fertile queen, resulting in workers refraining from reproducing in many social species). In many other species though, queen-worker conflict can be severe, with workers sometimes even engaging in matricide – killing their own mother queen to be able to reproduce without interference. This chapter provides an overview of both current and past research on reproductive conflicts within the Vespidae wasps and how their study has been instrumental in testing some key predictions of inclusive fitness theory.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
8.1 Inclusive Fitness Theory as a Basis to Understand Reproductive Conflicts
Wasps are a key model in the study of insect sociality . This is due to the fact that this group of insects presents a remarkable diversity of lifestyles – from solitary to truly social, “eusocial” species. Although the majority of species are solitary, the evolution of sociality in some lineages is thought to have greatly facilitated their ecological success (Wilson 1971; Grimaldi and Engel 2005). Wilson (1971) considered organisms eusocial when they possess three major characteristics: (1) a reproductive division of labor between a reproductive and nonreproductive caste, (2) overlap of generations, and (3) cooperative care of the young. Whether this definition should or should not include species with mostly behaviorally defined castes, such as most Polistinae and Stenogastrinae wasps, where workers are usually totipotent and retain the ability to mate, is a point of debate (Boomsma and Gawne 2018; Sumner et al. 2018). We will here follow the commonly adopted convention to refer to those as primitively eusocial and to the species with morphologically defined castes and an obligate worker caste, i.e., with workers that have lost the ability the mate, as highly eusocial (cf. Michener 1974; O’Donnell 1998; Wenseleers and van Zweden 2017). By this token, all of the Vespinae wasps are highly eusocial, whereas most of the Polistinae are primitively eusocial, with the possible exception of a few swarm-founding Polistinae, such as Agelaia, which can achieve colony sizes of over half a million individuals and which have quite pronounced caste dimorphism and have been suggested to possess an obligate worker caste (O’Donnell 1998; Wenseleers and van Zweden 2017; Piekarski et al. 2018). The Stenogastrinae hover wasps and some members of the Crabronid wasp Microstigmus are considered facultatively eusocial, given that many nests also just contain a single breeding female (Field 2008; Turillazzi 2012; Matthews 1991). Recent phylogenomic evidence indicates that the presence of preimaginally determined castes, which do not need to be reflected in overall morphology, was likely the ancestral condition for all of the Polistinae and Vespinae (Piekarski et al. 2018), implying that a subset of the daughters expressed a distinct, likely subfertile, phenotype at the very outset of eusociality. This has been explained on the basis that the queen-worker castes were likely built on preexisting ground plans and pathways in solitary ancestors, e.g., related to ancestral breeding cycles (Piekarski et al. 2018; Hunt and Amdam 2005; Hunt et al. 2007b). Recent phylogenetic data also show that social behavior in the Stenogastrinae evolved independently from that in the Polistinae and Vespinae (Piekarski et al. 2018).
The origin of a sterile worker caste that refrains from reproducing and instead helps to rear the queen’s offspring presents an important evolutionary paradox and an apparent contradiction with the expectation that organisms should be selected to maximize their personal reproduction (Ratnieks et al. 2011). Indeed, Darwin himself referred to this question as a “special difficulty” that seemed to jeopardize his theory of evolution via natural selection (Ratnieks et al. 2011). A formal resolution of this paradox only came with the development of Hamilton’s (1964) inclusive fitness theory, which showed that individuals can be selected to altruistically sacrifice their own reproduction if their altruism would be directed toward relatives, as these can indirectly help to propagate copies of the altruist’s own genes, thereby allowing the altruistic trait to persist in the population (Wenseleers et al. 2010; Gardner et al. 2011). In formulating this theory, Hamilton was heavily influenced by the social insects , and he noticed that insect sociality appeared to be unusually common among the haplodiploid Hymenoptera, where females (workers and queens) develop from fertilized diploid eggs and males are usually derived from unfertilized, haploid eggs. He pointed out that as a result of this genetic system, any female would genetically be more highly related to full sisters (relatedness coefficient r = 0.75) than to its own daughters (r = 0.5, Fig. 8.1) and suggested this could predispose them to evolve helping behavior and give up the option to mate and reproduce on their own (Hamilton 1964). However, later authors noted that his argument was not quite correct, since haplodiploidy also causes females to be less related to their full brothers (r = 0.25) than to their own sons (r = 0.5), resulting in an average relatedness to siblings (brothers and sisters) that is the same as that to own offspring, 0.5, if they produce both sexes in an equal ratio (Trivers and Hare 1976). Later analyses have therefore concluded that haplodiploidy can only slightly increase the likelihood for eusociality to evolve and only does so under very specific conditions, e.g., if the workers would be able to bias the sex ratio toward highly related sisters (Grafen 1986; Seger 1983; Quiñones and Pen 2017), if the workers would be able to combine the rearing of sisters with the production of their own male offspring (Craig 1979, Fig. 8.1), or if there is male-biased dispersal (Johnstone et al. 2011). Indeed, several recent studies have concluded that haplodiploidy as such was most likely not a strong driving force in the evolution of eusociality but that high relatedness, linked with lifetime monogamy, and a high benefit to cost ratio of helping surely were (Hughes et al. 2008; Davies and Gardner 2018; Boomsma 2009; Boomsma and Gawne 2018; Liao et al. 2015; Boomsma et al. 2011; Quiñones and Pen 2017).
Genetic relatedness coefficients in social Hymenoptera if the colony is headed by a single-mated (top) or double-mated queen (bottom). The relatedness coefficients r are the genetic relatedness values between a focal worker and individual sisters, brothers, sons, or nephews, whereas \( \overline{r} \) is the average genetic relatedness of a focal work to each of these classes of individuals. The different thorax colors represent distinct genetic backgrounds. In this diagram, the production of diploid (2n) females, derived from fertilized eggs, are indicated with black arrows, whereas the production of haploid (n) males, derived from unfertilized eggs, are indicated with gray arrows
Another important insight that came from Hamilton’s work was the realization that inclusive fitness theory would not just be able to explain under what conditions sociality would be able to evolve but that it also predicted the exact conditions under which social conflicts could be expected. Insect societies , including many social wasps, have proven an excellent test bed to test these theories, given that the genetic asymmetries within their colonies produce much scope for conflict, both among the queen and workers as well as among the workers themselves (reviewed in Ratnieks et al. 2006).
8.2 The Scope for Conflict in Insect Societies
Empirical study of conflict within insect societies began with Trivers and Hare (1976), who used inclusive fitness theory to show that in social Hymenoptera there is strong potential conflict between the mother queen and her daughter workers over the rearing of young males versus queens. Specifically, they noted that in colonies headed by a single once-mated queen, workers would on average be three times more related to sisters (r = 0.75) than to brothers (r = 0.25), whereas the queen would always be related equally to sons and daughters (r = 0.5). Hence, it was predicted that the queen and workers would have conflicting evolutionary optima, with the workers being selected to invest three times more in females than in males but the queen being favored to invest equally in both (Trivers and Hare 1976). Since then, researchers have documented many other areas where conflict can be expressed, including in the context of conflict over male parentage (Ratnieks 1988; Bourke and Franks 1995; Hammond and Keller 2004; Wenseleers et al. 2004b; Wenseleers and Ratnieks 2006a; Ratnieks and Wenseleers 2008; Bourke 1988), queen rearing (Keller 1997; Tarpy et al. 2004; Ratnieks et al. 2006), and caste fate (Bourke and Ratnieks 1999; Wenseleers et al. 2003; Wenseleers and Ratnieks 2004) or, in societies lacking morphologically distinct queens and workers, over breeding role and the choice between helping and nesting independently (Hart and Ratnieks 2005; Hart and Monnin 2006; Reeve 1991; Reeve and Keller 2001; Tibbetts 2007; Strassmann et al. 2002). Below we will review how some of these conflicts have been documented in wasps and how this has led to some of the best tests of inclusive fitness theory to date. We start with two conflicts that theoretically are predicted to occur but which in wasps have only received relatively limited study, namely, conflict over sex allocation and queen rearing, and then move on to review types of conflicts that have been investigated very extensively in wasps, namely, conflicts over breeding roles in primitively eusocial wasps and conflicts over male parentage. We then discuss how in some species these conflicts are resolved via policing mechanisms and chemical signaling systems, which likely evolved hand in hand, while in others queen-worker conflict remains unresolved, resulting, for example, in worker matricide, where the workers kill their own mother queen to eliminate all reproductive competition and be able to reproduce on their own.
8.3 Conflict over Sex Allocation
Sex ratio theory predicts that workers in social Hymenoptera should be selected to invest in queens versus males in proportion to the relatedness asymmetry to both sexes (Bourke and Franks 1995; Crozier and Pamilo 1996; Ratnieks et al. 2006). This relatedness asymmetry becomes smaller in species where queens mate with multiple males, where colonies are headed by multiple queens, or when the majority of the males in the colony are produced by the workers (Bourke and Franks 1995; Crozier and Pamilo 1996; Ratnieks et al. 2006). Hence, in these situations, the sex ratio favored by the workers becomes closer to the 1 to 1 investment sex ratio favored by the mother queen(s), resulting in reduced potential conflict. Theory also predicts that intraspecific variation in the kin structure of colonies is expected to favor facultative sex ratio biasing or “split sex ratios,” whereby workers should produce more queens in colonies with single-mated queens or low queen numbers, where relatedness asymmetry is high, and more males in colonies with multiple-mated queens or high queen numbers, where relatedness asymmetry is low (Boomsma and Grafen 1991; Ratnieks 1991).
These sex ratio predictions have been tested most extensively in ants, and data from many species support the hypothesis that workers selfishly bias sex allocation in their own favor or facultatively bias sex allocation in response to the colony kin structure (reviewed in Bourke and Franks 1995; Crozier and Pamilo 1996; West 2009). For example, a meta-analysis comprising data from 22 species of ants, bees, and wasps provided strong evidence for facultative sex allocation biasing in response to variation in relatedness asymmetry (or mate or queen number, which directly correlates with relatedness asymmetry) (Meunier et al. 2008). Key supporting evidence in this analysis came from primitively eusocial wasps, as in both Polistes fuscatus (Noonan 1978) and in five species of swarm-founding epiponine wasps (Queller et al. 1993; Hastings et al. 1998), sex ratios were found to be split and follow variation in queen number, thereby suggesting worker control of sex allocation. In Polistes metricus, Metcalf (1980) also reported that sex ratios were split due to some colonies being orphaned, causing the workers in those colonies to rear their own males and the remaining colonies to be selected to compensate for this male bias by producing more female-biased sex ratios. A different outcome though was recorded in Polistes chinensis, where Tsuchida (2003) also recorded male-biased allocation in orphaned nests but found that workers in queenright nests did not compensate by increasing female investment but rather by engaging in high levels of worker reproduction in the presence of the mated foundress, causing over half of all the adult males to be the workers’ sons, whilst producing an investment sex ratio that was close to the queen’s optimum (Tsuchida et al. 2003; Wenseleers and Ratnieks 2006b). This shows how different types of social conflict can interact with each other and how the workers can sometimes win one type of conflict at the cost of losing another. Such outcomes as well as multiple stable queen-worker equilibria or ever continuing queen-worker arms races have been predicted by models that considered how multiple traits in queens and different types of social conflicts coevolve with each other (Helms 1999; Wenseleers et al. 2013). Many other factors though are suspected to influence optimal sex allocation in social insects (West 2009). Suzuki (1986), for example, reported investment sex ratios for 11 Polistini and 2 Ropalidiini paper wasps and found an approximately equal investment sex ratio in species that produce males and reproductive females simultaneously, implying queen-controlled sex allocation in those species but a female-biased investment sex ratio and worker control in both protandrous and protogynous species that produce either males or females first in the season. These data illustrate the importance of considering the full dynamics of the production schedule of both sexes, which in most models of social Hymenopteran sex allocation have so far been neglected (but see Avila et al. 2019 for a promising approach to do so).
Whether investment sex ratios across different Vespine wasps correlate with queen mating frequency and relatedness asymmetry remains largely unknown, as sex ratios are quite hard to measure due to the long period over which sexuals are produced and the fact that in Vespine wasps, two different sizes of males are produced, derived from sexual and worker comb (Bonckaert et al. 2011b). Johnson et al. (2009), however, provided tentative evidence that queen polyandry reduces queen-worker conflict over sex allocation in the North American yellowjacket Vespula maculifrons, as the predicted optimal investment sex ratio of the mother queens and the workers, given the observed queen mating frequency, was very close to each other, 0.5 and 0.56, and the observed investment sex ratio intermediate and not significantly different from either. Likewise, the numerical sex ratios reported for the polyandrous yellowjackets V. germanica and V. vulgaris in Edwards (1980) and Spradbery (1971) translate into investment sex ratio values of 0.63 and 0.56, which are again intermediate between the queen’s (0.5) and the workers’ optima (0.65 and 0.65, given observed queen mating frequencies, Bonckaert et al. 2007; Foster and Ratnieks 2001b). On the other hand, Archer (1998) observed that workers selectively eliminate some of the male larvae in V. vulgaris, thereby suggesting that even in these polyandrous species, workers do bias sex allocation in their own favor to a limited extent. Hence, observed levels of polyandry are not fully effective at eliminating all potential conflict in these yellowjackets. Detailed comparative studies of investment sex ratios in other Vespine wasps, such as in Dolichovespula and Vespa, as far as we are aware are not available but a priori would be expected to show stronger potential female bias under worker control due to the effective mating frequency in those genera being much lower than in the large-colony Vespula, often being close to one (Foster et al. 2001; Foster and Ratnieks 2001a; Hughes et al. 2008; van Zweden et al. 2014). This prediction remains to be tested. In D. arenaria, however, split sex ratios have been suggested to occur, even though two studies reported opposite effects, with Foster and Ratnieks (2001a) citing decreased female investment in function of paternity but Loope (2016) finding the opposite pattern. Hence, these studies require further replication.
8.4 Conflict over Queen Rearing
Another potential conflict that can occur in insect societies is when there are different degrees of relatedness between individual workers and different queens raised in the colony. In wasps, this can occur when queens are multiple mated, as is common in many Vespine wasps (Foster and Ratnieks 2001a; Hughes et al. 2008), or when the colony contains multiple mother queens or foundresses, which is common in many Polistinae and Stenogastrinae paper wasps (Turillazzi and West-Eberhard 1996; Ross and Matthews 1991; Hughes et al. 2008). In these cases, one would expect that workers could be selected to nepotistically aid only in the rearing of new queens to which they are most related, i.e., full-sister queens. In a swathe of species, from the honeybee to stingless bees, ants and Polistine, and Vespine wasps, such nepotism has been looked for, but the consensus is that it is quite rare in social insects overall (Keller 1997; Tarpy et al. 2004; Goodisman et al. 2007; Wenseleers 2007). In wasps, nepotism and within-colony kin discrimination was, for example, found to be absent in the multiqueen colonies of the swarm-founding wasp Parachartergus colobopterus (Strassmann et al. 1997) and in the polyandrous colonies of the yellowjacket Vespula maculifrons (Goodisman et al. 2007). It is thought that even though potential conflict over queen rearing exists, recognitional errors often make it too costly in practice to be evolutionarily favored (Keller 1997; Wenseleers 2007; van Zweden and d’Ettorre 2010). Indeed, cuticular hydrocarbons from Polistinae and Vespinae wasps failed to distinguish reliably among patrilines in the hornet Vespa crabro, even though they could distinguish among nestmate workers of different matrilines in the paper wasp Polistes dominulus (Dani et al. 2004). In line with this last finding, Leadbeater et al. (2014) reported a limited degree of kin-biased behavior in P. dominulus, thereby providing the first evidence for within-nest kin discrimination in a primitively eusocial wasp.
8.5 Conflict over Breeding Role in Species with Totipotent Females
In most Polistinae and Stenogastrinae wasps and in the Crabronid genus Microstigmus, queen-worker dimorphism is either weak or absent (Noll and Wenzel 2008; Turillazzi 2012; Matthews 1991), and all or most females are thought to be “totipotent,” i.e. being able to mate and produce both male and female offspring (Reeve et al. 1998; Tibbetts 2007; Strassmann et al. 2002). Totipotency provides significant scope for conflict over breeding role, as it allows any individual in the colony to replace the existing queen (Reeve 1991; Jandt et al. 2014; Hart and Monnin 2006; Hart and Ratnieks 2005), to become an additional queen (Strassmann et al. 2002), or, except in the swarm-founding Polistinae, to found a nest independently (Reeve and Keller 2001). The relatedness benefits for females to mate and breed are similar to those of becoming a morphologically specialized queen (Bourke and Ratnieks 1999; Wenseleers et al. 2003). That is, in the face of reproductive competition with other females, a female benefits if she can become the dominant breeder and produce own offspring (sons and daughters, r = 0.5) rather than a worker rearing the less-related offspring of other females (r < < 0.5). The cost of helping can be significantly reduced though if there are potential direct fitness benefits associated with delayed nest inheritance (Leadbeater et al. 2011) or if there are insurance-based advantages of helping (Reeve and Nonacs 1997; Field et al. 2000; Shreeves et al. 2003), e.g., so-called assured fitness returns resulting from the fact that when a helper dies, the offspring she contributed may be brought to maturity by surviving nestmates (Gadagkar 1990), or “bet hedging”-type benefits of helping (Kennedy et al. 2018). Indeed, in both Polistinae paper wasps and Stenogastrinae hover wasps, insurance-based advantages (Reeve and Nonacs 1997; Field et al. 2000; Shreeves et al. 2003; Field 2008) as well as advantages associated with delayed nest inheritance (Field et al. 1999; Field et al. 2006; Shreeves and Field 2002; Field 2008; Monnin et al. 2009; Leadbeater et al. 2011) have both been shown to be important in driving helping behavior. In the paper wasp Polistes dominula, for example, females that inherit the dominant position were shown to produce more offspring than solitary females (Leadbeater et al. 2011).
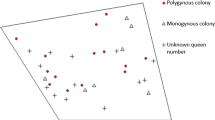
There is extensive evidence for conflict over breeding role in wasps with totipotent females. In Polistinae with independent nest founding, dominance interactions frequently determine who will take the dominant breeding spot (Fig. 8.2, Reeve 1991; Jandt et al. 2014), and subordinates sometimes challenge the dominant breeder to attempt to overthrow it (Hart and Monnin 2006; Hart and Ratnieks 2005; Jandt et al. 2014). Conflict over the decision to either help or reproduce is also apparent from the fact that some first-generation females, which normally act as workers, have been observed to leave the colony early to become a queen the next year (in Polistes annularis, Strassmann 1989; P. fuscatus, Reeve et al. 1998; and P. dominula, Tibbetts 2007). Similarly, first-generation females of P. exclamans may leave the nest to find satellite nests elsewhere the same season (in P. exclamans, Strassmann 1981). In swarm-founding Epiponini, conflict over breeding role (“caste fate conflict”) has been suggested to result in an excess of females mating to be readopted in the natal nest when colonies have few mother queens left (Henshaw et al. 2000; Queller et al. 1993; Strassmann et al. 2002). This excess is then reduced as a result of queen-queen competition or active elimination by workers (Strassmann et al. 2002; Platt et al. 2004), thereby driving the characteristic cycles in queen number that occur in this group (Noll and Wenzel 2008). Such queen overproduction only occurs in colonies with few mother queens, presumably because the workers as a collective then favor their production (Strassmann et al. 2002) and because the potential benefits for females to mate and reproduce are largest, as there would be few competing queens already present. It is possible that the high levels of queen production observed in Epiponine wasp colonies with few queens is therefore not just a consequence of split sex ratios as Queller et al. (1993) proposed but that caste fate conflict is an important additional driver of this pattern.
Several mechanisms have been suggested by which overt conflict over breeding role could be avoided in primitively eusocial wasps. In North American populations of Polistes dominula and P. exclamans paper wasps, signals of dominance and fighting ability, so-called badges of status, have been suggested to be encoded by specific patterns on the foundresses’ clypeus, which correlate with body size (Tibbetts and Dale 2004; Tibbetts and Sheehan 2011). In this way, conventional settlements can resolve conflict and prevent overt aggression. Furthermore, individuals that were painted to emulate the wasps’ having a higher rank than their true rank were found to be punished by nestmates, thereby helping the signaling system to remain honest (Tibbetts and Dale 2004). Clypeal status badges, however, were not found to be used in European populations of P. dominula (Green and Field 2011; Green et al. 2013), while clypeal patterning was shown to be correlative with fertility in the Brazilian paper wasp P. satan (Tannure-Nascimento et al. 2008). Another potential mechanism that the dominant breeder could use to reduce the chances that it’s offspring would reproduce would be to provide them with limited food to keep them smaller and less fecund. In neither paper wasps nor hover wasps, however, this subfertility hypothesis has so far received any empirical support (Field and Foster 1999; Couchoux and Field 2019). Some models have also suggested that social contracts could help to reduce conflict among cofounding females in primitively eusocial wasps (Reeve and Keller 2001; Nonacs and Hager 2011). In so-called transactional skew models, dominants were argued to offer a sufficiently large reproductive share to the subordinate, lest the subordinate would leave to found her own nest (Reeve and Keller 2001; Nonacs and Hager 2011). Although initial data from some Polistes paper wasps appeared to support social contract-type transactional skew models (e.g., Reeve et al. 2000; Tibbetts and Reeve 2000, reviewed in Nonacs and Hager 2011), many later studies have supported other models, such as ones where reproductive shares arise from selfish competition among individuals (so-called “tug of war” models) (Nonacs and Hager 2011) or follow biological market theory, in which dominants will compete for the helpers and pay a price for it, sharing part of reproduction with the subordinates, and subordinates choose who they will help depending on the options they are offered (Grinsted and Field 2017a, b). Over the past decade, the exact support for different classes of reproductive skew models has become quite contentious, however, and many of the formulated models have become quite hard to test due to the difficulty of measuring the underlying model parameters (Nonacs and Hager 2011). Finally, in some species of independently founding and swarm-founding wasps, a transition from aggressive regulation of reproduction via dominance interactions to one based on chemical signaling has been suggested (e.g., Mitra et al. 2011; Mitra and Gadagkar 2011; Dapporto et al. 2007b, reviewed in Jeanne 2020 this book). However, this would only be expected to be evolutionarily stable in those species where the queen and workers’ interests are strongly aligned (Oi et al. 2015b).
8.6 Conflict over Male Parentage
A final area in which significant scope for conflict is expected in wasp societies is in the context of male parentage (Trivers and Hare 1976; Bourke 1988; Wenseleers and Ratnieks 2006a). Conflict over male parentage has been intensively studied across many ants, bees, and wasps and has yielded some of the best support for inclusive fitness theory to date (Wenseleers et al. 2004b; Wenseleers and Ratnieks 2006a, b; Ratnieks and Wenseleers 2008). The scope for conflict over male parentage stems from the fact that workers, though unable to mate, usually still possess functional ovaries and are therefore capable of laying unfertilized, male eggs (Wenseleers and Ratnieks 2006a). However, since both the queen and each of the workers are genetically most related to their own sons (r = 0.5), they are in conflict over who should produce the males in the colony. This can favor the queen to try to stop the workers from reproducing via aggression or egg eating. This process is known as “queen policing” and appears particularly common and effective in species with small colonies, including small-colony Polistine and Vespine wasps (e.g., Polistes chinensis and P. dominulus, Saigo and Tsuchida 2004; Liebig et al. 2005; Dolichovespula sylvestris, Wenseleers et al. 2005b; D. norwegica, Bonckaert et al. 2011a; Vespula rufa, Wenseleers et al. 2005a), as well as in Halictid bees and bumble bees (Ratnieks et al. 2006; Wenseleers and Ratnieks 2006a; Zanette et al. 2012).
Conflict over male parentage not only occurs between the queen and workers but also among the different workers themselves. Individual workers, for example, may benefit from producing highly related sons, but the workers as a collective may rather prefer that the queen would produce all the male offspring. This conflict is most pronounced in species where colonies are headed by multiple-mated queens or by multiple related queens, as in both cases the workers can end up being more related to the sons of the queen than to the sons of other workers (Starr 1984; Ratnieks 1988; Pamilo 1991) (Fig. 8.1). In these situations, nonreproductive workers have been shown to benefit from trying to suppress the reproduction of other workers via targeted aggression or the selective cannibalism of worker-laid eggs, a process that was dubbed “worker policing” (Ratnieks 1988; Ratnieks and Visscher 1989). In line with prediction, worker policing was first documented to occur in the multiple-mated honeybees (Ratnieks and Visscher 1989; Ratnieks et al. 2006) but has since been discovered to occur also in several social wasps, including in the multiple-mated large-colony yellowjackets Vespula vulgaris and Vespula germanica (Foster and Ratnieks 2001b; Bonckaert et al. 2007, Fig. 8.3) as well as in the predominantly single-mated European hornet Vespa crabro (Foster et al. 2002) and both monandrous and polyandrous ant species (Wenseleers and Ratnieks 2006a; Ratnieks et al. 2006).
Three types of policing regulate conflict over male parentage in social wasps. a In species where colonies are headed by a multiple-mated queen, as in the German wasp Vespula germanica (pictured) and the common wasp Vespula vulgaris, regular “worker policing” occurs (Foster and Ratnieks 2001b; Bonckaert et al. 2007). As in the honeybee (Ratnieks and Visscher 1989), such policing involves nonreproductive worker selectively detecting and removing eggs laid by “cheater” workers that try to reproduce inside the colony. By contrast, in species where colonies are normally headed by a single-mated queen, such as in the tree wasp Dolichovespula sylvestris (pictured in the video stills in panels b to g) (Wenseleers et al. 2005b), the Norwegian wasp Dolichovespula norwegica (Bonckaert et al. 2011a), and the small-colony yellowjacket Vespula rufa (Wenseleers et al. 2005a), it is usually either the queen herself that polices worker-laid eggs (“queen policing,” panels b to d show a worker laying an egg, the queen then removing it, after which she deposits her own egg in the same cell) or reproductive workers that selectively destroy the eggs laid by other competing workers (“selfish policing,” panels e to g show a worker laying an egg, after which another worker destroys the egg to then lay her own egg). Photos by Tom Wenseleers
The fact that worker policing is not purely restricted to species with multiple-mated queens suggests that the relatedness difference between queen’s sons versus workers’ sons is not the only evolutionary driver of this behavior. Indeed, several studies have pointed out that worker policing likely also has other benefits, e.g., linked to the fact that workers may kill worker-laid eggs as part of a strategy to cause a female-biased sex allocation ratio (Foster and Ratnieks 2001c), that workers may aggress egg-laying workers to increase their work rates and improve colony productivity (Ratnieks 1988; Wenseleers and Ratnieks 2006a), or that the policing may in fact be carried out by reproductive workers, who may gain direct fitness benefits from eliminating the eggs laid by other workers (“selfish policing”, (Wenseleers et al. 2005b; Bonckaert et al. 2011a; Zanette et al. 2012). In a large meta-analysis of the male parentage in over 100 ants, bees, and wasps, however, colony kin structure did significantly correlate with the percentage of adult males that were workers’ sons in the direction predicted by inclusive fitness theory (Wenseleers and Ratnieks 2006a, Fig. 8.4a). In wasps, for example, it was the case that in species where colonies were headed by a multiple-mated queen, as in large-colony yellowjackets (Vespula spp.) (Bonckaert et al., 2007; Foster and Ratnieks 2001b; Ross and Matthews 1991), or by multiple related queens, as in the swarm-founding Polistinae (Epiponinae) (Hastings et al. 1998; Henshaw et al. 2002; Henshaw et al. 2000), only few or none of the adult males were workers’ sons, suggesting the presence of highly effective worker policing (Fig. 8.4a). By contrast, in species where colonies were usually headed by a single, once-mated queen, and where workers were collectively most related to the sons of other workers, a significant fraction of the adult males could be produced by the workers (Wenseleers and Ratnieks 2006a, Fig. 8.4a). In the Asian paper wasp Polistes chinensis , for example, genotyping data showed that over half of all adult males were worker produced (Tsuchida et al. 2003). In addition, in a British population of the Saxon wasp Dolichovespula saxonica , patterns of male parentage suggested that workers show a facultative expression of worker policing, with workers only policing the eggs laid by other workers in colonies with a multiple-mated queen, but not in those with a single-mated one (Foster and Ratnieks 2000). Such facultative worker policing, however, did not appear universal and was not found in a Danish population of the Saxon wasp (Bonckaert et al. 2011b).
Worker reproduction in wasp societies in function of relatedness (a) and policing (b). (a) Patterns of male parentage support the relatedness theory for the evolution of worker policing. In wasps, the percentage of adult males that are workers’ sons, derived, e.g., from genotyping data, correlates with the relatedness difference of the workers to other workers’ vs. the queen’s sons. This is in line with the prediction that in species where colonies are headed by a multiple-mated queen or by multiple related queens, workers should be selected to suppress the reproduction of other workers due to the fact that they would then be more related to the sons of the queen than to Fig. 8.4 (continued) the sons of other workers (Ratnieks 1988; Pamilo 1991) (Data from Wenseleers and Ratnieks 2006a). (a) More effective worker policing favors greater cooperation. Across nine species of wasps, the percentage of workers that attempt to reproduce inside the colony is a strongly decreasing function of how effectively worker-laid eggs are policed by nestmates, as this disincentivizes them to even try to reproduce (Wenseleers et al. 2004a, b). Species with queen polyandry (orange), where nonreproductive workers police eggs laid by other workers, have more effective worker policing and have fewer workers attempting to reproduce inside the colony than species with low queen paternity (blue), where it is mainly the queen and reproductive workers that remove eggs laid by other workers (“queen policing” and “selfish policing”) (Data from Wenseleers and Ratnieks 2006b). Species abbreviations: Panel a: Crabronidae – Mc, Microstigmus comes; Epiponini – Pt, Polybioides tabidus; Bm, Brachygastra mellifica; Pt, Parachartergus colobopterus; Polistini – Pb, Polistes bellicosus; Pc, P. chinensis; Pd, P. dorsalis; Pf, P. fuscatus variatus; Pg, P. gallicus; Pm, P. metricus; Vespinae – Da, Dolichovespula arenaria; Dm, D. maculata; Dmed, D. media; Dn, D. norwegica; Ds, D. saxonica; Dsy, D. sylvestris; Vcf, Vespa crabro flavofasciata; Vcg, V. crabro gribodi; Vd, V. ducalis; Vman, V. mandarinia; Vg, Vespula germanica; Vmac, V. maculifrons; Vr, V. rufa; Vs, V. squamosa; Vv, V. vulgaris. Panel b: Pc, Polistes chinensis; Vr, Vespula rufa; Dmed, Dolichovespula media; Dn, D. norwegica; Ds, D. saxonica; Dsy, D. sylvestris; Vc, Vespa crabro; Vg, Vespula germanica; Vv, V. vulgaris
In a number of Vespine wasps, several of the aforementioned forms of policing have been shown to co-occur within the same species. For example, in the tree wasp Dolichovespula sylvestris, the Norwegian wasp D. norwegica, and the small-colony yellowjacket Vespula rufa, eggs laid by rare cheater workers were found to be policed both by the queen (queen policing) and by competing reproductive workers (selfish policing) (Wenseleers et al. 2005b; Bonckaert et al. 2011a; Wenseleers et al. 2005a, Fig. 8.3). Interestingly, selfish worker policing , though driven purely by direct reproductive competition, was exclusively targeted toward the eggs laid by other workers, as the workers spared the eggs laid by the queen (Wenseleers et al. 2005b; Bonckaert et al. 2011a). Presumably, this is linked to the fact that if the reproductive workers would remove the eggs laid by the queen, it would become too costly from a colony-level perspective; as such, reproductive competition would then severely compromise the successful rearing of new brood in the colony, which would entail large indirect fitness costs.
Lastly, potential conflict over male parentage could also be caused by intraspecific worker parasitism, whereby workers could try to deposit male eggs in other colonies to have them reared by nonrelatives. Such “egg dumping” or intraspecific worker parasitism has been well-documented in bees (Beekman and Oldroyd 2008), and several studies have also tried to document it in primitively and highly eusocial wasps (Sumner et al. 2007; Oliveira et al. 2016). In the common wasp Vespula vulgaris , it was found that workers from queenright colonies left the colony at a higher rate than those in queenless colonies, presumably to evade worker policing in their natal colony, and that drifted workers activated their ovaries at ca. five times the rate than natal workers (Oliveira et al. 2016). RFID tracking technology also demonstrated very high rates of drifting in the paper wasp Polistes canadensis, even though rates of worker ovary activation were not higher among the drifters than among the natal workers (Sumner et al. 2007).
8.7 Resolution of Conflict over Male Parentage via Policing
In general terms, all the forms of social policing described above can be regarded as mechanisms that should reduce the incentive for individuals to act selfishly and in the long term should benefit the functional cohesion of the group (Ratnieks 1988; Leigh 1977; Frank 2003; Agren et al. 2019). Hence, over evolutionary time, the mere presence of these forms of policing would be expected to increase the level of cooperation and reduce the percentage of workers in the colony that try to cheat by laying their own eggs (Wenseleers et al. 2004a, b; Ratnieks and Wenseleers 2008; Agren et al. 2019). A comparative study that looked at the relationship between policing and the percentage of workers that attempted to reproduce inside the colony supported this prediction (Wenseleers and Ratnieks 2006b). In particular, across nine species of wasps, the percentage of workers with active ovaries was found to be a strongly decreasing function of how effectively worker-laid eggs were policed by nestmates (Wenseleers and Ratnieks 2006b), as this disincentivizes them to even try to reproduce (Wenseleers et al. 2004a, Fig. 8.4b, b). In species where queens are predominantly single-mated, such as in Dolichovespula spp. and in the small-colony yellowjacket Vespula rufa, worker-laid eggs are policed mainly by the queen or by other egg-laying workers (selfish policing). Yet, these forms of policing are not 100% effective (Wenseleers et al. 2005a, b; Bonckaert et al. 2011a), implying that workers in these species can still have a significant incentive to reproduce inside the colony (Wenseleers et al. 2004b), often causing a significant fraction of the workers to activate their ovaries (Fig. 8.4b). By contrast, in two polyandrous large-colony yellowjackets, Vespula germanica and V. vulgaris, nearly all worker-laid eggs are removed by worker policing (Foster and Ratnieks 2001b; Bonckaert et al. 2007). Hence, in those species workers have almost no incentive to even try to reproduce in presence of the queen, causing only few to activate their ovaries (Foster and Ratnieks 2001b; Bonckaert et al. 2007, Fig. 8.4b). These data are interesting in that they document a form of “enforced altruism,” whereby workers refrain from reproducing because it is made unprofitable by the presence of policing (Wenseleers and Ratnieks 2006b; Ratnieks and Wenseleers 2008). Paradoxically, this also causes levels of cheating to be the lowest and the level of cooperation to be the highest in the species with the lowest genetic relatedness, i.e., in colonies of the polyandrous yellowjackets V. germanica and V. vulgaris, because low relatedness selects more strongly for worker policing (Ratnieks 1988). This pattern, however, flips when the same nine wasp species were studied in the absence of the queen, as worker reproduction is then limited merely by genetic relatedness and the indirect fitness costs of having an excess of workers reproduce inside the colony (Wenseleers et al. 2004b). Hence, in that situation, a negative relationship was observed between the percentage of reproductive workers in the colony and genetic relatedness (Wenseleers and Ratnieks 2006b; Ratnieks and Wenseleers 2008). In yellowjackets, the breakdown of cooperation seen in queenless colonies is particularly apparent. In the common wasp Vespula vulgaris, for example, only around 1% of the workers try to reproduce in the presence of the queen, but this increases to 30% following the loss of the mother queen (Wenseleers and Ratnieks 2006b). This is far more than what is required for the successful reproduction of the colony, as shown by the fact that in queenless colonies, the workers deposit multiple eggs in the cells of their combs (Fig. 8.5). Such conflicts, where individual selfishness causes a cost to all, are known as “tragedies of the commons” and have also been documented in the context of conflict over caste fate (Wenseleers and Ratnieks 2004).
Cooperation breaks down in queenless wasp colonies. (a) In queenright colonies of most social wasp species, such as in the common wasp Vespula vulgaris (pictured), the queen typically deposits just a single egg per cell, in line with the needs of the colony. (b) However, following the death of the mother queen, cooperation breaks down, and far more workers start reproducing than is necessary for the functioning of the colony, which is apparent from the fact that the workers then start depositing multiple eggs per cell. (Photos by Cintia Akemi Oi)
8.8 Queen Pheromones Can Regulate Conflict over Male Parentage
Given the widely divergent reproductive interests of workers in colonies with or without a queen (Ratnieks and Wenseleers 2008), it is imperative that the workers can unequivocally assess the queen’s presence in order to optimally modulate their reproduction and behavior. In species with small colonies, individuals are in close contact with one another, and the queen alone is able to control the workers’ reproduction, usually by being aggressive toward them or by policing worker-laid eggs (Ratnieks and Wenseleers 2005). This is the case, for example, in Polistes wasps and permanently queenless ant species where the dominant reproductive individual limits mating opportunities and reproduction of subordinates via targeted aggression (Reeve 1991; Monnin and Peeters 1998; Peeters 1993; Liebig et al. 2005; Saigo and Tsuchida 2004). Nevertheless, when the number of individuals reaches a certain threshold, the queen is no longer able to effectively keep workers from reproducing merely via the use of aggression (Keller and Nonacs 1993; Ratnieks and Reeve 1992). In such cases, the workers assess whether or not the queen is present based on specific chemicals that she emits, which signal her presence and fertility status. When workers respond to such signals by inhibiting their ovary activation, such chemicals are referred to as queen signals or queen pheromones (Howard and Blomquist 2005; Holman et al. 2010, 2013; Van Oystaeyen et al. 2014; Monnin 2006; Liebig 2010; Peeters and Liebig 2009; Holman 2010; Liebig et al. 2000; D’Ettorre et al. 2004; Oliveira et al. 2015; Fig. 8.5). The first queen pheromone that was identified in social insects was in the well-studied honeybee Apis mellifera (Butler 1959; Wossler and Crewe 1999; Hoover et al. 2003). In this species, a honeybee-specific blend of pheromones derived from the queen’s mandibular gland (QMP) , containing the keto acid 9-oxo-decenoic acid as major compound, was shown to inhibit worker reproduction (Butler 1959; Hoover et al. 2003). Recent work, however, has shown that specific cuticular wax compounds act as more conserved queen pheromones across several groups of Hymenoptera, including wasps, ants, bumblebees, and the honeybee (Van Oystaeyen et al. 2014, reviewed in Oi et al. 2015b; Princen et al. 2019a). The fact that structurally related and sometimes even identical hydrocarbon compounds were found to suppress worker reproduction across these different independently evolved lineages of social insects implies very strong evolutionary conservation and suggested that these signals likely evolved from preexisting fertility-linked compounds that were already present in the common solitary ancestors of all extant social Hymenoptera (Van Oystaeyen et al. 2014; Oi et al. 2015b).
In wasps, nearly all fertility or queen-characteristic compounds have been shown to consist of linear or branched saturated alkanes or alkenes (Table 8.1). The only possible exception is provided by the Stenogastrinae wasp Liostenogaster flavolineata, where egg-laying females also upregulate the production of icosanol (Table 8.1). An actual queen signaling function of these compounds, however, has been demonstrated via bioassays with pure compounds in only two Vespine wasps – the common wasp Vespula vulgaris (Van Oystaeyen et al. 2014, Fig. 8.6) and the Saxon wasp Dolichovespula saxonica (Oi et al. 2016) – where a partly overlapping set of hydrocarbons was found to suppress worker reproduction (Van Oystaeyen et al. 2014; Oi et al. 2016). In the common wasp, it was further shown that one of the worker reproduction-inhibiting queen pheromones, 3-methylnonacosane, also acted as a queen egg marking pheromone that helps workers to recognize queen-laid eggs from worker-laid eggs and selectively police worker-laid eggs (Fig. 8.6, Oi et al. 2015a). Such queen egg marking signals had long been proposed to exist (Endler et al. 2004; Ratnieks 1995; Oldroyd et al. 2002; Martin et al. 2004), but previous work had not succeeded in identifying the active compounds in any species (Martin et al. 2005).
The dual role of queen pheromones in regulating sociality in the common wasp. In the common wasp Vespula vulgaris, specific hydrocarbons that are abundant on the queen’s cuticle and eggs, as established by gas chromatography-mass spectrometry analysis (a), appear to have a dual role in regulating the social order in the colony (b), with heptacosane (n-C27), nonacosane (n-C29), and 3-methyl nonacosane (3-MeC29) acting as a worker reproduction inhibiting queen pheromone (b, left) (Van Oystaeyen et al. 2014) and also acting as a queen egg marking signal that helps workers to recognize queen-laid eggs from worker-laid eggs and selectively destroy or “police” worker-laid eggs (b, right) (Oi et al. 2015a). This was shown by the fact that administering these pheromones to queenless worker groups and applying them onto the surface of worker-laid eggs reduce the odds of the workers to activate their ovaries or reduce the odds of worker-laid eggs to be policed compared to in a solvent-only control treatment (*** = p < 0.001). (Modified based on data in Van Oystaeyen et al. (2014) and Oi et al. (2015a))
At an ultimate level, there is an ongoing debate over whether queen pheromones could ever “chemically castrate” the workers (the so-called “queen control” hypothesis ) or if workers merely respond to the queen pheromones in their own best interests (the “honest signaling” hypothesis ) (Keller and Nonacs 1993; Grüter and Keller 2016; Oi et al. 2015b). Evidence to date seems to give more support to the latter hypothesis, with workers merely modulating their reproductive physiology and behavior in accordance with their own evolutionary interests (Oi et al. 2015b). First, queen pheromones appear highly conserved across species, and this goes against the rapid evolution of the bioactive queen substances expected under the queen control hypothesis, where workers would be expected to rapidly evolve resistance to the chemicals that the queen emits (Heinze and d’Ettorre 2009; Van Oystaeyen et al. 2014). Further in line with honest signaling hypothesis is that queen pheromones in several species of ants, bees, and wasps have been shown to be positively correlated with fecundity (Liebig 2010; Peeters and Liebig 2009; Monnin 2006). This implies they are indeed honest signals of fertility. In some species, such as in the common wasp Vespula vulgaris, signal honesty has been shown to be maintained partly by the fact that queen fecundity and the production of specific hydrocarbon queen signals are under identical endocrine control (“hormonal pleiotropy,” Oliveira et al. 2017). Finally, the fact that worker reproduction was found to be modulated by the presence or absence of the queen in exact accordance with inclusive fitness theory and was found to be affected by the colony kin structure in the predicted direction shows that workers respect the queen’s signals only to the extent that it serves the workers’ own inclusive fitness interests (e.g. Wenseleers and Ratnieks 2006b; van Zweden et al. 2014; Wenseleers et al. 2013; Ratnieks and Wenseleers 2008; Oi et al. 2015b).
Exactly how the hydrocarbon queen signaling system evolved from preexisting pathways in solitary ancestors is still not known with certainty, but it has been suggested that the queen signals could be derived from one of several preexisting pathways or unrelated signaling systems, e.g., they could (1) initially have been mere by-products of ovary activation, (2) be derived from ancestral oviposition-deterrent pheromones, or (3) be derived from ancestral sex pheromones (Oi et al. 2015b; Van Oystaeyen et al. 2014; Oliveira et al. 2015). In support of the by-product hypothesis, it has been shown that in both solitary and social species, the activation of the ovaries causes distinct changes in the cuticular hydrocarbon profiles of individuals (Blomquist and Bagnères 2010; Liebig 2010; Peeters and Liebig 2009; Holman 2012). In addition, in the primitively eusocial paper wasp Polistes satan, it was shown that dominant breeders overproduce particular hydrocarbons but that in bioassays these compounds did not inhibit worker reproduction (Oi et al. 2019). This supports the hypothesis that queen signals in highly eusocial species were derived from fertility cues that initially lacked any signaling function. Queen pheromones could also have been co-opted from oviposition-deterrent pheromones. Such pheromones are known in several solitary wasp species, where they mark the oviposition site with chemicals that signal to other females that the particular site is already in use (e.g., Stelinski et al. 2007; Anderson et al. 2002). Although these pheromones are directed to inhibiting other females from egg-laying, the lack of evidence for such pheromones from other groups of insects with a different social organization weakens this hypothesis (Oi et al. 2015b). Finally, queen pheromones could have been derived from sex pheromones and be initially directed at the male sex. In some bees, for example, the exact same compounds were shown to act both as a sex pheromone and as a queen pheromone or fertility-linked cue, with honeybee QMP being the best-known example (Oliveira et al. 2015). In honeybees, there is also some evidence that the queen-worker signaling system may have evolved by exploiting preexisting receiver physiology, since QMP has been found to also inhibit reproduction in the distantly related fruit fly and in bumblebees (Princen et al. 2019b), but in wasps there is so far no evidence to also support such an evolutionary scenario.
8.9 Overt Conflict over Male Parentage: Worker Matricide
While the sections above illustrate clear cases where conflict over male parentage is effectively resolved via the presence of policing mechanisms and queen signaling systems that coevolved with them, there are also clear examples in wasps where conflict over male parentage remains unresolved. A spectacular example is provided by worker matricide, where workers kill their own mother queen to be able to reproduce without interference, which has been documented not only in wasps but also in some ants and bumblebees (Ratnieks 1988; Bourke 1994; Foster and Ratnieks 2001a). Various direct observations of worker matricide exist from several Vespine wasps, including the European hornet Vespa crabro and several Dolichovespula species (Bourke 1994; Foster and Ratnieks 2001a; Loope 2015). The exact timing and conditions under which such matricide should be favored can be calculated from inclusive fitness theory (Bourke 1994; Almond et al. 2019). In particular, theory predicts that worker matricide should be favored most strongly in species with low queen paternity, where queen-worker conflict over male parentage is strongest and the workers are collectively mostly related to the sons of other workers (Ratnieks 1988; Bourke 1994). In line with this prediction, workers in the Vespine wasp Dolichovespula arenaria were found to kill their mother queen preferentially in colonies headed by a singly mated queen, that is, in colonies in which workers stood to gain the greatest fitness (Loope 2015). It was also shown that this was costly, as matricide resulted in the production of fewer queens (Loope 2016). Furthermore, in a comparative analysis of nine Vespine wasps, Foster et al. (2001) reported colonies to be queenless more frequently in species with low queen paternity, in accordance with worker matricide there being more common . Together, these studies again provide strong support for inclusive fitness theory.
8.10 Outstanding Questions
As we have shown, studies of reproductive conflicts in social wasps have yielded some remarkable insight into the basic structure of social conflicts and how they can be resolved. At the same time, several questions remain. In the areas of the study of social conflicts in wasp societies, the determinants of variation in investment sex ratio and the amount of overt conflict over male parentage in independently founding paper wasps and some monandrous Vespine wasps remain elusive (Foster et al. 2000; Foster and Ratnieks 2001a; Hammond and Keller 2004; Wenseleers and Ratnieks 2006a; Suzuki 1986; Tsuchida et al. 2003). To obtain a good match between empirically observed patterns and empirical data, it is likely that we will need new theoretical models that better take into account the dynamics of colony growth and sex allocation as well as the way in which different types of social conflict and traits in queens and workers coevolve with each other. At present, such theory still needs further development and testing as well as tailoring to the specific biology of particular species (e.g. Foster and Ratnieks 2001c; Wenseleers et al. 2013; Avila et al. 2019). The fact that model predictions have been shown to partly depend on the underlying genetic architecture of the traits may complicate such an endeavor (Olejarz et al. 2015, 2016; Davies and Gardner 2018). Another area that will be very rewarding to study further is the actual origin of sociality, which over the last decade has again become center stage. Indeed, fully understanding the factors that drove the major evolutionary transition to a eusocial lifestyle is surely one of the most important outstanding questions in evolutionary biology today. Social wasps are an excellent model system to study this question (Taylor et al. 2018). In fact, at an empirical level, research on primitively and facultatively eusocial wasps has given us some fantastic insights already, e.g., documenting direct fitness benefits of helping (Field et al. 2000, 2006, 2008), which has been matched by similar findings in other taxa (Korb and Heinze 2016; Downing et al. 2016), and showing how the evolution of helping behavior is subject to biological market theory (Grinsted and Field 2017b). At a theoretical level, social evolution theory and bet hedging theory have recently been combined in a unified framework (Kennedy et al. 2018), and the various preadaptations that can favor the evolutionary transition to eusociality have been subjected to systematic study, both from a proximate angle (Hunt 2012) and an ultimate perspective (Quiñones and Pen 2017). The detailed testing of some of these new models using comparative studies will remain an important task for the future. Lastly, much is yet to be unraveled about the genomic basis of social traits in wasps (Taylor et al. 2018). With many full genomes of species of key phylogenetic placement and contrasting social structure expected to become available in the near future (Taylor et al. 2018), the study of social wasps are expected to provide key insight in the genomic basis of a diverse set of traits, ranging from group living and altruistic behavior to reproductive plasticity, preimaginal caste differentiation, swarm founding, and queen-worker conflict over male parentage (Jandt and Toth 2015; Patalano et al. 2015; Taylor et al. 2018). Key questions that such data will be able to address is the extent to which sociality was built on novel genetic innovations (Ferreira et al. 2013) as opposed to the mere repurposing of existing pathways (Hunt and Amdam 2005; Hunt et al. 2007a) and how primitive and advanced eusocial systems, including queen-worker signaling systems and various conflict-resolving mechanisms, may have been built from preexisting traits in solitary ancestors (Johnson and Linksvayer 2010; Rehan and Toth 2015; Toth and Rehan 2017).
8.11 Concluding Remarks
Our aim in this chapter was to provide an overview on the current understanding of the causes and consequences of reproductive conflicts and conflict resolution mechanisms in social wasps and to present some new insights and perspectives for future research directions. Despite being among the most interesting organisms to study questions with respect to the origin and maintenance of sociality, we feel that wasps are still understudied compared to other social insect groups. We hope this chapter will incentivize students and young researchers to be as excited as we are to study and understand more about this fascinating group.
References
Agren JA, Davies NG, Foster KR (2019) Enforcement is central to the evolution of cooperation. Nat Ecol Evol 3:1018–1029. https://doi.org/10.1038/s41559-019-0907-1
Almond EJ, Huggins TJ, Crowther LP, Parker JD, Bourke AFG (2019) Queen longevity and fecundity affect conflict with workers over resource inheritance in a social insect. Am Nat 193:256–266. https://doi.org/10.1086/701299
Anderson P, Hilker M, Meiners T (2002) Oviposition pheromones in herbivorous and carnivorous insects. In: Chemoecology of insect eggs and egg deposition. Blackwell, Oxford, Berlin, pp 235–263
Archer M (1998) Worker versus sexual, and sex ratio investments in the social wasp Vespula vulgaris (L.) (Hymenoptera: Vespinae) in England. J Hymenopt Res 7:257–267
Avila P, Fromhage L, Lehmann L (2019) Sex-allocation conflict and sexual selection throughout the lifespan of eusocial colonies. Evolution 73:1116–1132
Beekman M, Oldroyd BP (2008) When workers disunite: Intraspecific parasitism by eusocial bees. Annu Rev Entomol 53:19–37. https://doi.org/10.1146/annurev.ento.53.103106.093515
Bhadra A, Mitra A, Deshpande SA, Chandrasekhar K, Naik DG, Hefetz A, Gadagkar R (2010) Regulation of reproduction in the primitively eusocial wasp Ropalidia marginata: on the trail of the queen pheromone. J Chem Ecol 36:424–431
Blomquist GJ, Bagnères A-G (2010) Insect Hydrocarbons Biology, Biochemistry, and Chemical Ecology. Cambridge University Press, Cambridge
Bonavita-Cougourdan A, Theraulaz G, Bagnères AG, Roux M, Pratte M, Provost E, Clément JL (1991) Cuticular hydrocarbons, social organization and ovarian development in a polistine wasp: Polistes dominulus Christ. Comp Biochem Physiol B 100:667–680
Bonckaert W, Vuerinckx K, Billen J, Hammond RL, Keller L, Wenseleers T (2007) Worker policing in the German wasp Vespula germanica. Behav Ecol 19:272–278
Bonckaert W, Tofilski A, Nascimento FS, Billen J, Ratnieks FL, Wenseleers T (2011a) Co-occurrence of three types of egg policing in the Norwegian wasp Dolichovespula norwegica. Behav Ecol Sociobiol 65:633–640
Bonckaert W, van Zweden JS, d’Ettorre P, Billen J, Wenseleers T (2011b) Colony stage and not facultative policing explains pattern of worker reproduction in the Saxon wasp. Mol Ecol 20:3455–3468. https://doi.org/10.1111/j.1365-294X.2011.05200.x
Bonckaert W, Drijfhout FP, d’Ettorre P, Billen J, Wenseleers T (2012) Hydrocarbon signatures of egg maternity, caste membership and reproductive status in the common wasp. J Chem Ecol 38:42–51
Boomsma JJ (2009) Lifetime monogamy and the evolution of eusociality. Philos Trans R Soc B 364:3191–3207
Boomsma JJ, Gawne R (2018) Superorganismality and caste differentiation as points of no return: how the major evolutionary transitions were lost in translation. Biol Rev 93:28–54
Boomsma J, Grafen A (1991) Colony-level sex ratio selection in the eusocial Hymenoptera. J Evol Biol 4:383–407
Boomsma JJ, Beekman M, Cornwallis CK, Griffin AS, Holman L, Hughes WO, Keller L, Oldroyd BP, Ratnieks FL (2011) Only full-sibling families evolved eusociality. Nature 471E4–E5; author reply E9–10. https://doi.org/10.1038/nature09832
Bourke AF (1988) Worker reproduction in the higher eusocial Hymenoptera. Q Rev Biol 63:291–311
Bourke AFG (1994) Worker Matricide in Social Bees and Wasps. J Theor Biol 167:283–292. https://doi.org/10.1006/jtbi.1994.1070
Bourke AF, Franks NR (1995) Social evolution in ants. Princeton University Press, Princeton
Bourke AF, Ratnieks FL (1999) Kin conflict over caste determination in social Hymenoptera. Behav Ecol Sociobiol 46:287–297
Butler CG (1959) The source of the substance produced by a queen honeybee (Apis Mellifera L.) which inhibits development of the ovaries of the worker of her colony. Proc R Entomol Soc Lond Ser A 34:137–138. https://doi.org/10.1111/j.1365-3032.1959.tb00249.x
Butts DP, Espelie KE, Hermann HR (1991) Cuticular hydrocarbons of 4 species of social wasps in the subfamily Vespinae - Vespa crabro L, Dolichovespula maculata (L), Vespula squamosa (Drury), and Vespula maculifrons (Buysson). Comp Biochem Physiol B 99:87–91
Butts DP, Camann MA, Espelie KE (1995) Workers and queens of the European hornet Vespa crabro L. have colony-specific cuticular hydrocarbon profiles (Hymenoptera, Vespidae). Insect Soc 42:45–55
Couchoux C, Field J (2019) Parental manipulation of offspring size in social groups: a test using paper wasps. Behav Ecol Sociobiol 73:36
Craig R (1979) Parental manipulation, kin selection, and the evolution of altruism. Evolution 33:319–334
Crozier RH, Pamilo P (1996) Evolution of social insect colonies: sex allocation and kin selection. Ecology and evolution. Oxford University Press, Oxford
D’Ettorre P, Heinze J, Schulz C, Francke W, Ayasse M (2004) Does she smell like a queen? Chemoreception of a cuticular hydrocarbon signal in the ant Pachycondyla inversa. J Exp Biol 207:1085–1091. https://doi.org/10.1242/jeb.00865
Dani FR, Foster KR, Zacchi F, Seppä P, Massolo A, Carelli A, Arévalo E, Queller DC, Strassmann JE, Turillazzi S (2004) Can cuticular lipids provide sufficient information for within–colony nepotism in wasps? Proc R Soc Lond Ser B 271:745–753
Dapporto L, Romana Dani F, Turillazzi S (2007a) Social dominance molds cuticular and egg chemical blends in a paper wasp. Curr Biol 17:R504–R505
Dapporto L, Santini A, Dani FR, Turillazzi S (2007b) Workers of a Polistes paper wasp detect the presence of their queen by chemical cues. Chem Senses 32:795
Davies NG, Gardner A (2018) Monogamy promotes altruistic sterility in insect societies. R Soc Open Sci 5:172190
Downing P, Cornwallis C, Griffin A (2016) How to make a sterile helper. BioEssays 39:1600136
Edwards R (1980) Social wasps. Their biology and control. Rentokil Ltd., East Grinstead
Endler A, Liebig J, Schmitt T, Parker JE, Jones GR, Schreier P, Hölldobler B (2004) Surface hydrocarbons of queen eggs regulate worker reproduction in a social insect. Proc Natl Acad Sci U S A 101:2945–2950
Ferreira PG, Patalano S, Chauhan R, Ffrench-Constant R, Gabaldon T, Guigo R, Sumner S (2013) Transcriptome analyses of primitively eusocial wasps reveal novel insights into the evolution of sociality and the origin of alternative phenotypes. Genome Biol 14:R20. https://doi.org/10.1186/gb-2013-14-2-r20
Field J (2008) The ecology and evolution of helping in hover wasps (Hymenoptera: Stenogastrinae). In: Ecology of social evolution. Springer, Heidelberg, pp 85–107
Field J, Foster W (1999) Helping behaviour in facultatively eusocial hover wasps: an experimental test of the subfertility hypothesis. Anim Behav 57:633–636
Field J, Shreeves G, Sumner S (1999) Group size, queuing and helping decisions in facultatively eusocial hover wasps. Behav Ecol Sociobiol 45:378–385
Field J, Shreeves G, Sumner S, Casiraghi M (2000) Insurance-based advantage to helpers in a tropical hover wasp. Nature 404:869–871. https://doi.org/10.1038/35009097
Field J, Cronin A, Bridge C (2006) Future fitness and helping in social queues. Nature 441:214–217. https://doi.org/10.1038/nature04560
Foster K, Ratnieks F (2000) Facultative worker policing in a wasp. Nature 407:692–693. https://doi.org/10.1038/35037665
Foster KR, Ratnieks FL (2001a) Paternity, reproduction and conflict in vespine wasps: a model system for testing kin selection predictions. Behav Ecol Sociobiol 50:1–8
Foster KR, Ratnieks FLW (2001b) Convergent evolution of worker policing by egg eating in the honeybee and common wasp. Proc R Soc Lond Ser B 268:169–174
Foster KR, Ratnieks FLW (2001c) The effect of sex-allocation biasing on the evolution of worker policing in hymenopteran societies. Am Nat 158:615–623. https://doi.org/10.1086/323588
Foster KR, Ratnieks FL, Raybould AF (2000) Do hornets have zombie workers? Mol Ecol 9:735–742
Foster KR, Ratnieks FL, Gyllenstrand N, Thorén PA (2001) Colony kin structure and male production in Dolichovespula wasps. Mol Ecol 10:1003–1010
Foster KR, Gulliver J, Ratnieks FL (2002) Worker policing in the European hornet Vespa crabro. Insect Soc 49:41–44
Frank SA (2003) Repression of competition and the evolution of cooperation. Evolution 57:693–705
Gadagkar R (1990) Evolution of eusociality: the advantage of assured fitness returns. Philos Trans R Soc Lond Ser B 329:17–25. https://doi.org/10.1098/rstb.1990.0146
Gardner A, West SA, Wild G (2011) The genetical theory of kin selection. J Evol Biol 24:1020–1043
Goodisman MA, Kovacs JL, Hoffman EA (2007) Lack of conflict during queen production in the social wasp Vespula maculifrons. Mol Ecol 16:2589–2595
Grafen A (1986) Split sex ratios and the evolutionary origins of eusociality. J Theor Biol 122:95–121
Green JP, Field J (2011) Interpopulation variation in status signalling in the paper wasp Polistes dominulus. Anim Behav 81:205–209
Green JP, Leadbeater E, Carruthers JM, Rosser NS, Lucas ER, Field J (2013) Clypeal patterning in the paper wasp Polistes dominulus: no evidence of adaptive value in the wild. Behav Ecol 24:623–633
Grimaldi D, Engel M (2005) Evolution of the Insects. Cambridge University Press, Cambridge
Grinsted L, Field J (2017a) Biological markets in cooperative breeders: quantifying outside options. Proc R Soc B 284:20170904
Grinsted L, Field J (2017b) Market forces influence helping behaviour in cooperatively breeding paper wasps. Nat Commun 8:13750. https://doi.org/10.1038/ncomms13750
Grüter C, Keller L (2016) Inter-caste communication in social insects. Curr Opin Neurobiol 38:6–11
Hamilton WD (1964) The genetical evolution of social behaviour. I & II. J Theor Biol 7:1–52
Hammond RL, Keller L (2004) Conflict over male parentage in social insects. PLoS Biol 2:e248
Hart A, Monnin T (2006) Conflict over the timing of breeder replacement in vertebrate and invertebrate societies. Insect Soc 53:375–389
Hart AG, Ratnieks F (2005) Crossing the taxonomic divide: conflict and its resolution in societies of reproductively totipotent individuals. J Evol Biol 18:383–395
Hastings MD, Queller DC, Eischen F, Strassmann JE (1998) Kin selection, relatedness, and worker control of reproduction in a large-colony epiponine wasp, Brachygastra mellifica. Behav Ecol 9:573–581
Heinze J, d’Ettorre P (2009) Honest and dishonest communication in social Hymenoptera. J Exp Biol 212:1775–1779
Helms KR (1999) Colony sex ratios, conflict between queens and workers, and apparent queen control in the ant Pheidole desertorum. Evolution 53:1470–1478
Henshaw MT, Strassmann JE, Queller DC (2000) The independent origin of a queen number bottleneck that promotes cooperation in the African swarm-founding wasp, Polybioides tabidus. Behav Ecol Sociobiol 48:478–483
Henshaw MT, Queller DC, Strassman JE (2002). Control of male production in the swarm-founding wasp, Polybioides tabidus. J Evol Biol 15:262–268 https://doi.org/10.1046/j.1420-9101.2002.00392.x
Holman L (2010) Queen pheromones: The chemical crown governing insect social life. Commun Integr Biol 3:558–560. https://doi.org/10.4161/cib.3.6.12976
Holman L (2012) Costs and constraints conspire to produce honest signaling: insights from an ant queen pheromone. Evolution 66:2094–2105
Holman L, Jorgensen CG, Nielsen J, d’Ettorre P (2010) Identification of an ant queen pheromone regulating worker sterility. Proc R Soc Lond Ser B 277:3793–3800. https://doi.org/10.1098/rspb.2010.0984
Holman L, Lanfear R, d’Ettorre P (2013) The evolution of queen pheromones in the ant genus Lasius. J Evol Biol 26:1549–1558. https://doi.org/10.1111/jeb.12162
Hoover SR, Keeling C, Winston M, Slessor K (2003) The effect of queen pheromones on worker honey bee ovary development. Naturwissenschaften 90:477–480. https://doi.org/10.1007/s00114-003-0462-z
Howard RW, Blomquist GJ (2005) Ecological, behavioral, and biochemical aspects of insect hydrocarbons. Annu Rev Entomol 50:371–393. https://doi.org/10.1146/annurev.ento.50.071803.130359
Hughes WO, Oldroyd BP, Beekman M, Ratnieks FL (2008) Ancestral monogamy shows kin selection is key to the evolution of eusociality. Science 320:1213–1216
Hunt JH (2012) A conceptual model for the origin of worker behaviour and adaptation of eusociality. J Evol Biol 25:1–19
Hunt JH, Amdam GV (2005) Bivoltinism as an antecedent to eusociality in the paper wasp genus Polistes. Science 308:264–267
Hunt JH, Kensinger BJ, Kossuth JA, Henshaw MT, Norberg K, Wolschin F, Amdam GV (2007a) A diapause pathway underlies the gyne phenotype in Polistes wasps, revealing an evolutionary route to caste-containing insect societies. Proc Natl Acad Sci 104:14020–14025
Jandt JM, Toth AL (2015) Physiological and genomic mechanisms of social organization in wasps (Family: Vespidae). In: Advances in insect physiology, vol 48. London, Academic Press, pp 95–130
Jandt J, Tibbetts E, Toth A (2014) Polistes paper wasps: a model genus for the study of social dominance hierarchies. Insect Soc 61:11–27
Johnson BR, Linksvayer TA (2010) Deconstructing the superorganism: social physiology, groundplans, and sociogenomics. Q Rev Biol 85:57–79
Johnson EL, Cunningham TW, Marriner SM, Kovacs JL, Hunt BG, Bhakta DB, Goodisman MA (2009) Resource allocation in a social wasp: effects of breeding system and life cycle on reproductive decisions. Mol Ecol 18:2908–2920
Johnstone RA, Cant MA, Field J (2011) Sex-biased dispersal, haplodiploidy and the evolution of helping in social insects. Proc R Soc B 279:787–793
Keller L (1997) Indiscriminate altruism: unduly nice parents and siblings. Trends Ecol Evol 12:99–103
Keller L, Nonacs P (1993) The role of queen pheromones in social insects: queen control or queen signal? Anim Behav 45:787–794. https://doi.org/10.1006/anbe.1993.1092
Kelstrup HC, Hartfelder K, Nascimento FS, Riddiford LM (2014a) Reproductive status, endocrine physiology and chemical signaling in the Neotropical, swarm-founding eusocial wasp Polybia micans. J Exp Biol 217:2399–2410
Kelstrup HC, Hartfelder K, Nascimento FS, Riddiford LM (2014b) The role of juvenile hormone in dominance behavior, reproduction and cuticular pheromone signaling in the caste-flexible epiponine wasp, Synoeca surinama. Front Zool 11:78
Kelstrup HC, Hartfelder K, Esterhuizen N, Wossler TC (2017) Juvenile hormone titers, ovarian status and epicuticular hydrocarbons in gynes and workers of the paper wasp Belonogaster longitarsus. J Insect Physiol 98:83–92
Kennedy P, Higginson AD, Radford AN, Sumner S (2018) Altruism in a volatile world. Nature 555:359
Korb J, Heinze J (2016) Major Hurdles for the Evolution of Sociality. Annu Rev Entomol 61:297–316. https://doi.org/10.1146/annurev-ento-010715-023711
Layton JM, Camann MA, Espelie KE (1994) Cuticular lipid profiles of queens, workers, and males of social wasp Polistes metricus Say are colony specific. J Chem Ecol 20:2307–2321
Leadbeater E, Carruthers JM, Green JP, Rosser NS, Field J (2011) Nest inheritance is the missing source of direct fitness in a primitively eusocial insect. Science 333:874–876. https://doi.org/10.1126/science.1205140
Leadbeater E, Dapporto L, Turillazzi S, Field J (2014) Available kin recognition cues may explain why wasp behavior reflects relatedness to nest mates. Behav Ecol 25:344–351. https://doi.org/10.1093/beheco/art113
Leigh EG (1977) How does selection reconcile individual advantage with the good of the group? Proc Natl Acad Sci U S A 74:4542–4546
Liao X, Rong S, Queller DC (2015) Relatedness, conflict, and the evolution of eusociality. Plos Biol 13:e1002098
Liebig J (2010) Hydrocarbon profiles indicate fertility and dominancy status in ant, bee, and wasp colonies. In: Insect hydrocarbons: biology biochemistry and chemical ecology. Cambridge University Press, Cambridge, pp 254–281
Liebig J, Peeters C, Oldham NJ, Markstädter C, Hölldobler B (2000) Are variations in cuticular hydrocarbons of queens and workers a reliable signal of fertility in the ant Harpegnathos saltator? Proc Natl Acad Sci USA 97:4124–4131. https://doi.org/10.1073/pnas.97.8.4124
Liebig J, Monnin T, Turillazzi S (2005) Direct assessment of queen quality and lack of worker suppression in a paper wasp. Proc R Soc Lond Ser B 272:1339–1344
Loope KJ (2015) Queen killing is linked to high worker-worker relatedness in a social wasp. Curr Biol 25:2976–2979
Loope KJ (2016) Matricide and queen sex allocation in a yellowjacket wasp. Sci Nat 103:57
Martin SJ, Châline N, Oldroyd BP, Jones GR, Ratnieks FL (2004) Egg marking pheromones of anarchistic worker honeybees (Apis mellifera). Behav Ecol 15:839–844
Martin SJ, Châline NG, Ratnieks FLW, Jones GR (2005) Searching for the egg-marking signal in honeybees. J Negat Reults 2:1–9
Matthews R (1991) Evolution of social behavior in Sphecid wasps. In: Ross KG, Matthews RW (eds) The social biology of wasps. Cornell University Press, Ithaca, pp 570–602
Metcalf RA (1980) Sex ratios, parent-offspring conflict, and local competition for mates in the social wasps Polistes metricus and Polistes variatus. Am Nat 116:642–654
Meunier J, West SA, Chapuisat M (2008) Split sex ratios in the social Hymenoptera: a meta-analysis. Behav Ecol 19:382–390
Michener CD (1974) The social behavior of the bees: a comparative study, vol 73. Harvard University Press, Cambridge, MA
Mitra A, Gadagkar R (2011) Can Dufour’s gland compounds honestly signal fertility in the primitively eusocial wasp Ropalidia marginata? Naturwissenschaften 98:157–161
Mitra A, Saha P, Chaoulideer ME, Bhadra A, Gadagkar R (2011) Chemical communication in Ropalidia marginata: Dufour’s gland contains queen signal that is perceived across colonies and does not contain colony signal. J Insect Physiol 57:280–284
Monnin T (2006) Chemical recognition of reproductive status in social insects. Ann Zool Fenn 43:515–530
Monnin T, Peeters C (1998) Monogyny and regulation of worker mating in the queenless ant Dinoponera quadriceps. Anim Behav 55:299–306
Monnin T, Cini A, Lecat V, Fédérici P, Doums C (2009) No actual conflict over colony inheritance despite high potential conflict in the social wasp Polistes dominulus. Proc R Soc B 276:1593–1601
Noll FB, Wenzel JW (2008) Caste in the swarming wasps: ‘queenless’ societies in highly social insects. Biol J Linn Soc 93:509–522
Nonacs P, Hager R (2011) The past, present and future of reproductive skew theory and experiments. Biol Rev 86:271–298
Noonan KM (1978) Sex Ratio of Parental Investment in Colonies of the Social Wasp Polistes fuscatus. Science 199:1354–1356. https://doi.org/10.1126/science.199.4335.1354
O’Donnell S (1998) Reproductive caste determination in eusocial wasps (Hymenoptera: Vespidae). Annu Rev Entomol 43:323–346. https://doi.org/10.1146/annurev.ento.43.1.323
Oi CA, Van Oystaeyen A, Oliveira RC, Millar JG, Verstrepen KJ, van Zweden JS, Wenseleers T (2015a) Dual effect of wasp queen pheromone in regulating insect sociality. Curr Biol 25:1638–1640
Oi CA, van Zweden JS, Oliveira RC, Van Oystaeyen A, Nascimento FS, Wenseleers T (2015b) The origin and evolution of social insect queen pheromones: novel hypotheses and outstanding problems. BioEssays 37:808–821
Oi CA, Millar JG, van Zweden JS, Wenseleers T (2016) Conservation of queen pheromones across two species of vespine wasps. J Chem Ecol 42:1175–1180
Oi CA, Oliveira RC, van Zweden JS, Mateus S, Millar JG, Nascimento FS, Wenseleers T (2019) Do primitively eusocial wasps use queen pheromones to regulate reproduction? A case study of the paper wasp Polistes satan. Front Ecol Evol 7:199
Oldroyd BP, Ratnieks FL, Wossler T (2002) Egg-marking pheromones in honey-bees Apis mellifera. Behav Ecol Sociobiol 51:590–591
Olejarz JW, Allen B, Veller C, Nowak MA (2015) The evolution of non-reproductive workers in insect colonies with haplodiploid genetics. Elife 4:e08918
Olejarz JW, Allen B, Veller C, Gadagkar R, Nowak MA (2016) Evolution of worker policing. J Theor Biol 399:103–116
Oliveira RC, Oi CA, do Nascimento MM, Vollet-Neto A, Alves DA, Campos MC, Nascimento F, Wenseleers T (2015) The origin and evolution of queen and fertility signals in Corbiculate bees. BMC Evol Biol 15:254
Oliveira RC, Oi CA, Vollet-Neto A, Wenseleers T (2016) Intraspecific worker parasitism in the common wasp, Vespula vulgaris. Anim Behav 113:79–85. https://doi.org/10.1016/j.anbehav.2015.12.025
Oliveira RC, Vollet-Neto A, Oi CA, van Zweden JS, Nascimento F, Brent CS, Wenseleers T (2017) Hormonal pleiotropy helps maintain queen signal honesty in a highly eusocial wasp. Sci Rep 7:1654
Pamilo P (1991) Evolution of colony characteristics in social insects. II. Number of reproductive individuals. Am Nat 138:412–433
Patalano S, Vlasova A, Wyatt C, Ewels P, Camara F, Ferreirab PG, Asher CL, Jurkowski TP, Segonds-Pichon A, Bachman M, Gonzalez-Navarrete I, Minoche AE, Krueger F, Lowy E, Marcet-Houben M, Rodriguez-Ales JL, Nascimento FS, Balasubramanian S, Gabaldon T, Tarver JE, Andrews S, Himmelbauer H, Hughes WOH, Guigo R, Reik W, Sumner S (2015) Molecular signatures of plastic phenotypes in two eusocial insect species with simple societies. Proc Natl Acad Sci U S A 112:13970–13975. https://doi.org/10.1073/pnas.1515937112
Peeters C (1993) Monogyny and polygyny in ponerine ants with or without queens. In: Keller L (ed) Queen Number and Sociality in Insects. Oxford University Press, Oxford, pp 234–261
Peeters C, Liebig J (2009) Fertility signaling as a general mechanism of regulating reproductive division of labor in ants. In: Fewell J, Gadau J (eds) Organization of insect societies: from genome to socio-complexity. Harvard University Press, Cambridge, MA, pp 220–242
Piekarski PK, Carpenter JM, Lemmon AR, Moriarty Lemmon E, Sharanowski BJ (2018) Phylogenomic evidence overturns current conceptions of social evolution in wasps (Vespidae). Mol Biol Evol 35:2097–2109
Platt TG, Queller DC, Strassmann JE (2004) Aggression and worker control of caste fate in a multiple-queen wasp, Parachartergus colobopterus. Anim Behav 67:1–10
Princen SA, Oliveira RC, Ernst UR, Millar JG, van Zweden JS, Wenseleers T (2019a) Honeybees possess a structurally diverse and functionally redundant set of queen pheromones. Proc R Soc B 286:20190517. https://doi.org/10.1098/rspb.2019.0517
Princen SA, Van Oystaeyen A, Petit C, van Zweden JS, Wenseleers T (2019b) Cross-activity of honeybee queen mandibular pheromone in bumblebees provides evidence for sensory exploitation. Behav Ecol 31:303–310. https://doi.org/10.1093/beheco/arz191
Queller DC, Strassmann JE, Solís CR, Hughes CR, DeLoach DM (1993) A selfish strategy of social insect workers that promotes social cohesion. Nature 365:639
Quiñones AE, Pen I (2017) A unified model of Hymenopteran preadaptations that trigger the evolutionary transition to eusociality. Nat Commun 8:15920
Ratnieks FLW (1988) Reproductive harmony via mutual policing by workers in eusocial Hymenoptera. Am Nat 132:217–236. https://doi.org/10.1086/284846
Ratnieks FL (1991) Facultative sex allocation biasing by workers in social Hymenoptera. Evolution 45:281–292
Ratnieks FL (1995) Evidence for a queen-produced egg-marking pheromone and its use in worker policing in the honey bee. J Apic Res 34:31–37
Ratnieks FLW, Reeve HK (1992) Conflict in single-queen hymenopteran societies: the structure of conflict and processes that reduce conflict in advanced eusocial species. J Theor Biol 158:33–65
Ratnieks FLW, Visscher PK (1989) Worker policing in the honeybee. Nature 342:796–797
Ratnieks FLW, Wenseleers T (2005) Policing Insect Societies. Science 307:54–56. https://doi.org/10.1126/science.1106934
Ratnieks FLW, Wenseleers T (2008) Altruism in insect societies and beyond: voluntary or enforced? Trends Ecol Evol 23:45–52. https://doi.org/10.1016/j.tree.2007.09.013
Ratnieks FLW, Foster KR, Wenseleers T (2006) Conflict resolution in insect societies. Annu Rev Entomol 51:581–608. https://doi.org/10.1146/annurev.ento.51.110104.151003
Ratnieks FL, Foster KR, Wenseleers T (2011) Darwin’s special difficulty: the evolution of “neuter insects” and current theory. Behav Ecol Sociobiol 65:481–492
Reeve HK (1991) Polistes. In: Ross KG, Matthews RW (eds) The social biology of wasps. Cornell University Press, Ithaca
Reeve HK, Keller L (2001) Tests of reproductive-skew models in social insects. Annu Rev Entomol 46:347–385. https://doi.org/10.1146/annurev.ento.46.1.347
Reeve HK, Nonacs P (1997) Within-group aggression and the value of group members: theory and a field test with social wasps. Behav Ecol 8:75–82
Reeve HK, Peters JM, Nonacs P, Starks PT (1998) Dispersal of first “workers” in social wasps: causes and implications of an alternative reproductive strategy. Proc Natl Acad Sci 95:13737–13742
Reeve HK, Starks PT, Peters JM, Nonacs P (2000) Genetic support for the evolutionary theory of reproductive transactions in social wasps. Proc R Soc Lond Ser B 267:75–79
Rehan SM, Toth AL (2015) Climbing the social ladder: the molecular evolution of sociality. Trends Ecol Evol 30:426–433. https://doi.org/10.1016/j.tree.2015.05.004
Ross KG, Matthews RW (eds) (1991) The social biology of wasps. Cornell University Press, Ithaca
Saigo T, Tsuchida K (2004) Queen and worker policing in monogynous and monandrous colonies of a primitively eusocial wasp. Proc R Soc Lond Ser B 271:S509–S512
Seger J (1983) Partial bivoltinism may cause alternating sex-ratio biases that favour eusociality. Nature 301:59
Shreeves G, Field J (2002) Group size and direct fitness in social queues. Am Nat 159:81–95. https://doi.org/10.1086/324125
Shreeves G, Cant MA, Bolton A, Field J (2003) Insurance–based advantages for subordinate co–foundresses in a temperate paper wasp. Proc R Soc Lond Ser B 270:1617–1622. https://doi.org/10.1098/rspb.2003.2409
Sledge MF, Boscaro F, Turillazzi S (2001) Cuticular hydrocarbons and reproductive status in the social wasp Polistes dominulus. Behav Ecol Sociobiol 49:401–409
Spradbery J (1971) Seasonal changes in the population structure of wasp colonies (Hymenoptera: Vespidae). J Anim Ecol 40:501–523
Starr CK (1984) Sperm competition, kinship, and sociality in the aculeate Hymenoptera. Sperm Compet Evol Anim Mat Syst 428:459
Stelinski LL, Oakleaf R, Rodriguez-Saona C (2007) Oviposition-deterring pheromone deposited on blueberry fruit by the parasitic wasp, Diachasma alloeum. Behaviour 144:429–445. https://doi.org/10.1163/156853907780755996
Strassmann JE (1981) Evolutionary implications of early male and satellite nest production in Polistes exclamans colony cycles. Behav Ecol Sociobiol 8:55–64
Strassmann JE (1989) Early termination of brood rearing in the social wasp, Polistes annularis (Hymenoptera: Vespidae). J Kansas Entomol Soc 62:353–362
Strassmann JE, Klingler CJ, Arevalo E, Zacchi F, Husain A, Williams J, Seppä P, Queller DC (1997) Absence of within–colony kin discrimination in behavioural interactions of swarm–founding wasps. Proc R Soc Lond Ser B 264:1565–1570
Strassmann JE, Sullender BW, Queller DC (2002) Caste totipotency and conflict in a large–colony social insect. Proc R Soc Lond Ser B 269:263–270
Sumner S, Lucas E, Barker J, Isaac N (2007) Radio-tagging technology reveals extreme nest-drifting behavior in a eusocial insect. Curr Biol 17:140–145. https://doi.org/10.1016/j.cub.2006.11.064
Sumner S, Bell E, Taylor D (2018) A molecular concept of caste in insect societies. Curr Opin Insect Sci 25:42–50
Suzuki T (1986) Production schedules of males and reproductive females, investment sex ratios, and worker-queen conflict in paper wasps. Am Nat 128:366–378
Tannure-Nascimento IC, Nascimento FS, Zucchi R (2008) The look of royalty: visual and odour signals of reproductive status in a paper wasp. Proc R Soc Lond Ser B 275:2555
Tarpy DR, Gilley DC, Seeley TD (2004) Levels of selection in a social insect: a review of conflict and cooperation during honey bee (Apis mellifera) queen replacement. Behav Ecol Sociobiol 55:513–523
Taylor D, Bentley MA, Sumner S (2018) Social wasps as models to study the major evolutionary transition to superorganismality. Curr Opin Insect Sci 28:26–32
Tibbetts EA (2007) Dispersal decisions and predispersal behavior in Polistes paper wasp ‘workers’. Behav Ecol Sociobiol 61:1877–1883
Tibbetts EA, Dale J (2004) A socially enforced signal of quality in a paper wasp. Nature 432:218–222. https://doi.org/10.1038/nature02949
Tibbetts EA, Reeve HK (2000) Aggression and resource sharing among foundresses in the social wasp Polistes dominulus: testing transactional theories of conflict. Behav Ecol Sociobiol 48:344–352
Tibbetts EA, Sheehan MJ (2011) Facial patterns are a conventional signal of agonistic ability in Polistes exclamans paper wasps. Ethology 117:1138–1146
Toth AL, Rehan SM (2017) Molecular Evolution of Insect Sociality: An Eco-Evo-Devo Perspective. Annu Rev Entomol 62:419–442. https://doi.org/10.1146/annurev-ento-031616-035601
Trivers RL, Hare H (1976) Haploidploidy and the evolution of the social insect. Science 191:249–263. https://doi.org/10.1126/science.1108197
Tsuchida K, Saigo T, Nagata N, Tsujita S, Takeuchi K, Miyano S (2003) Queen-worker conflicts over male production and sex allocation in a primitively eusocial wasp. Evolution 57:2365–2373
Turillazzi S (2012) The evolution of social behaviour in insects and the hover wasps. In: The biology of hover wasps. Springer, New York, pp 233–263
Turillazzi S, West-Eberhard MJ (1996) Natural history and evolution of paper-wasps, vol 400. Oxford University Press, Oxford
Turillazzi S, Sledge MF, Dapporto L, Landi M, Fanelli D, Fondelli L, Zanetti P, Dani FR (2004) Epicuticular lipids and fertility in primitively social wasps (Hymenoptera Stenogastrinae). Physiol Entomol 29:464–471
Van Oystaeyen A, Oliveira RC, Holman L, van Zweden JS, Romero C, Oi CA, d’Ettorre P, Khalesi M, Billen J, Wackers F, Millar JG, Wenseleers T (2014) Conserved class of queen pheromones stops social insect workers from reproducing. Science 343:287–290. https://doi.org/10.1126/science.1244899
van Zweden JS, d’Ettorre P (2010) Nestmate recognition in social insects and the role of hydrocarbons. Insect hydrocarb: Biol, Biochem Chem Ecol 11:222–243
van Zweden JS, Bonckaert W, Wenseleers T, d’Ettorre P (2014) Queen Signaling in Social Wasps. Evolution 68:976–986. https://doi.org/10.1111/Evo.12314
Wenseleers T (2007) Nepotism absent in insect societies—or is it? Mol Ecol 16:3063–3065
Wenseleers T, Ratnieks FL (2004) Tragedy of the commons in Melipona bees. Proc R Soc Lond Ser B 271:S310–S312
Wenseleers T, Ratnieks FLW (2006a) Comparative analysis of worker reproduction and policing in eusocial Hymenoptera supports relatedness theory. Am Nat 168:E163–E179. https://doi.org/10.1086/508619
Wenseleers T, Ratnieks FLW (2006b) Enforced altruism in insect societies. Nature 444:50–50. https://doi.org/10.1038/444050a
Wenseleers T, van Zweden JS (2017) Sensory and cognitive adaptations to social living in insect societies. Proc Natl Acad Sci U S A 114:6424–6426
Wenseleers T, Ratnieks FL, Billen J (2003) Caste fate conflict in swarm-founding social Hymenoptera: an inclusive fitness analysis. J Evol Biol 16:647–658
Wenseleers T, Hart AG, Ratnieks FLW (2004a) When resistance is useless: Policing and the evolution of reproductive acquiescence in insect societies. Am Nat 164:E154–E167. https://doi.org/10.1086/425223
Wenseleers T, Helanterä H, Hart A, Ratnieks FL (2004b) Worker reproduction and policing in insect societies: an ESS analysis. J Evol Biol 17:1035–1047
Wenseleers T, Badcock N, Erven K, Tofilski A, Nascimento F, Hart A, Burke T, Archer M, Ratnieks F (2005a) A test of worker policing theory in an advanced eusocial wasp, Vespula rufa. Evolution 59:1306–1314
Wenseleers T, Tofilski A, Ratnieks FL (2005b) Queen and worker policing in the tree wasp Dolichovespula sylvestris. Behav Ecol Sociobiol 58:80–86
Wenseleers T, Gardner A, Foster KR (2010) Social evolution theory: a review of methods and approaches. In: Szekely T, Moore AJ, Komdeur J (eds) Social behaviour: genes, ecology and evolution. Cambridge University Press, Cambridge, pp 132–158
Wenseleers T, Helantera H, Alves DA, Duenez-Guzman E, Pamilo P (2013) Towards greater realism in inclusive fitness models: the case of worker reproduction in insect societies. Biol Lett 9:20130334. https://doi.org/10.1098/Rsbl.2013.0334
West S (2009) Sex allocation, vol 48. Princeton University Press, Princeton
Wilson EO (1971) The insect societies. Harvard University Press, Cambridge, MA
Wossler TC, Crewe RM (1999) Honeybee queen tergal gland secretion affects ovarian development in caged workers. Apidologie 30:311–320. https://doi.org/10.1051/apido:19990407
Zanette LR, Miller SD, Faria CM, Almond EJ, Huggins TJ, Jordan WC, Bourke AF (2012) Reproductive conflict in bumblebees and the evolution of worker policing. Evolution 66:3765–3777
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Oi, C.A., Wenseleers, T., Oliveira, R.C. (2021). Causes and Consequences of Reproductive Conflicts in Wasp Societies. In: Prezoto, F., Nascimento, F.S., Barbosa, B.C., Somavilla, A. (eds) Neotropical Social Wasps. Springer, Cham. https://doi.org/10.1007/978-3-030-53510-0_8
Download citation
DOI: https://doi.org/10.1007/978-3-030-53510-0_8
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-53509-4
Online ISBN: 978-3-030-53510-0
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)