Abstract
With increasing evidence that senescent cells are detrimental towards a range of age-associated diseases and physiologic declines, there is rising urgency to develop interventions to suppress their adverse effects. Most senolytic approaches aim to eliminate senescent cells by rendering them vulnerable to apoptosis, while senostatic (senomorphic) approaches do not destroy the cell and instead suppress a specific senescent trait. In both senolysis and senostasis, the major goals include reducing the senescence-associated secretory phenotype (SASP) and to enhance the immunogenicity of the senescent compartment. These therapeutic aims are best elicited from the plasma membrane, although efforts to identify plasma membrane targets are only now beginning. We discuss several plasma membrane proteins expressed preferentially in senescent cells and their roles in neutralizing senescent cells by immune-mediated senolysis (as reported for DPP4, VIM, and NKFB2 ligands) and by suppressing the SASP (as reported for SCAMP4 and CD36). We identify the advantages and challenges of developing therapeutic approaches directed at the plasma membrane of senescent cells.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
1.1 Cellular Senescence in Physiology and Disease
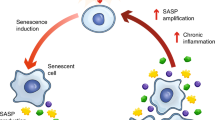
Cellular senescence is a specific response to cell damage that leads to long-term, generally irreversible growth arrest. Senescence can be triggered by stresses such as telomere attrition, DNA damage, oncogene activation, oxidative injury, and inflammation (von Zglinicki 2002; Guerra et al. 2003; Herbig et al. 2004; van Deursen 2014; Gorgoulis et al. 2019). In young persons, senescence is considered to be tumor-suppressive as it prevents the propagation of potentially transformed cells (Lowe et al. 2004; Collado and Serrano 2010; Muñoz-Espín and Serrano 2014; Chen et al. 2005; Kirkland and Tchkonia 2017). However, in older organisms, an excessive accumulation of senescent cells can become detrimental to tissues and organs, leading to organ dysfunction and promoting pathologies such as neurodegeneration, cardiovascular disease, diabetes, cancer, arthritis, liver disease, and sarcopenia (van Deursen 2014; Campisi 2005). Accordingly, in young persons, cellular senescence is recognized as a homeostatic response to sublethal damage, but in older persons it promotes aging-associated decline and disease (López-Otín et al. 2013). Additionally, senescence improves tissue repair by promoting wound healing (Demaria et al. 2014; Krizhanovsky et al. 2008).
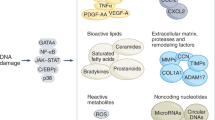
Senescent cells display distinct features. Compared with proliferating cells, senescent cells are larger, contain cytoplasmic vacuoles, exhibit increased senescence-associated β-galactosidase (SA-βGal) activity, and have higher levels of reactive oxygen species (ROS) (Gorgoulis et al. 2019; Ben-Porath and Weinberg 2004). In addition, senescent cells express different subsets of RNAs, including noncoding transcripts such as PURPL, and proteins such as cyclin-dependent kinase (CDK) inhibitors p21 (CDKN1A) and p16 (CDKN2A) (Alcorta et al. 1996; Casella et al. 2019; Wiley et al. 2017). They also exhibit a trait known as the senescence-associated secretory phenotype (SASP), whereby they display increased secretion of distinct subsets of cytokines, growth factors, and matrix remodeling proteins, including interleukin (IL)6 and IL8, vascular endothelial growth factor (VEGF), tumor necrosis factor (TNF), and matrix metalloproteinase 9 (MMP9) (Coppe et al. 2008; Parrinello et al. 2003; Dimri et al. 1995; Narita et al. 2003).
Among the differentially expressed proteins, the suppressor of growth and tumorigenesis p16 [an activator of the protein retinoblastoma (RB)], has been considered one of the most promising triggers and biomarkers of senescence in tissues and organs, as well as in cultured cells (Alcorta et al. 1996; Krishnamurthy et al. 2004; Benanti and Galloway 2004; Zindy et al. 1997). Supporting this notion, prolonged p16 expression promoted cellular senescence and decreased regenerative capacity in aging tissues, including the hematopoietic, pancreatic, and neural stem cell compartments (Kim and Sharpless 2006). Recent studies also showed that the numbers of p16-positive cells correlate with biological (but not chronological) aging in different tissues (Liu et al. 2009; Baker et al. 2011; Waaijer et al. 2012) and served as a surrogate marker to evaluate clinical outcomes following therapeutic interventions (Nelson et al. 2012). However, p16 appears not to be a universal biomarker of senescence (reviewed in Hernandez-Segura et al. 2018) and its low abundance and intracellular location pose further limitations on its usefulness as a marker. Thus, efforts have intensified in recent years to identify senescent markers that (1) improve the detection of senescent cells including live cells, (2) identify all senescent cells universally or subsets of senescent cells, and (3) provide a means to isolate senescent cells for elimination or for further analysis. Such markers are particularly valuable in therapeutic efforts aimed at protecting or destroying senescent cells.
2 Senolytics
2.1 Strategies to Reduce the Burden of Senescent Cells During Age
‘Senolysis’, or the elimination of senescent cells, is a strategy that has been actively pursued in recent years. A key landmark in this effort was a report by Baker and colleagues of a genetic mouse model (INK-ATTAC) in which activation of caspase 8 in p16-expressing cells eliminated senescent cells and ameliorated age-related losses in strength, adiposity, and vision (Baker et al. 2011). Recapitulating this genetic approach and similar mouse models (Demaria et al. 2014), a handful of chemical compounds named ‘senolytics’ have been identified that selectively eliminate senescent cells (Hernandez-Segura et al. 2018). Typically, senolytic drugs potently block pathways that protect senescent cells, thereby enabling their destruction (Ovadya and Krizhanovsky 2018). For example, various studies suggested that antiapoptotic protein members of the BCL2 family are significantly increased during senescence; accordingly, inhibition of those proteins in senescent cells activates programmed cell death (Yosef et al. 2016). Following similar strategies, approaches to augment apoptotic signals have been effective in achieving senolysis. Inhibitors of PI3K (phosphotidyl inositol 3 kinase) such as quercetin, inhibitors of tyrosine kinases including EPHA2 (the ephryn receptor A2) such as dasatinib, inhibitors of glucose homeostasis, such as 2-deoxyglucose (2-DG), as well as the aforementioned BCL2 inhibitors (ABT263, ABT737) were found to remove senescent cells selectively from heterogeneous cell populations (Czabotar et al. 2014; Zhu et al. 2016; Chang et al. 2016; Tse et al. 2008).
Given the detrimental impact of senescent cells in many aspects of aging pathophysiology, as mentioned above, efforts to identify specific and effective senolytics have intensified in aging research (López-Otín et al. 2013; Childs et al. 2014; Newgard and Sharpless 2013). However, although senolytics show promise in cultured cells and in some animal models, their broad application in age-related pathologies faces important obstacles. Top among them is the fact that since most senolytic compounds are strong inducers of apoptosis, they are often toxic also for proliferating healthy cells and can delay wound healing (Demaria et al. 2014; Cang et al. 2015; Zhu et al. 2017). Thus, it is critical that advanced approaches be developed in which senescent cells are targeted more specifically. An earlier study suggested alternative drug therapies targeting multiple cellular pathways simultaneously (Zhu et al. 2015). Along these lines, combination therapies such as administration of dasatinib + quercetin (D + Q) have shown synergistic cytotoxicity on senescent cells in different age-related pathologic conditions (Farr et al. 2017; Lehmann et al. 2017; Roos et al. 2016; Ogrodnik et al. 2017). However, senolytic combinations do not appear sufficient to solve the problem of specificity, and the mechanisms of action of senolytics remain poorly understood at present. A number of alternative approaches are being explored to thwart the damage of senescent cells.
‘Senostasis’ (or ‘senomorphism’) refers to the suppression of a senescent trait to reduce the detrimental impact of senescent cells, as proposed in the case of lung senescence by Birch et al. (Birch et al. 2018). Initiatives to identify senostatic approaches focused on two properties of senescent cells have emerged in recent times. First, as mentioned above, senescent cells secrete high amounts of proinflammatory factors (the SASP, as mentioned above) and extracellular matrix proteases (Coppe et al. 2008; Kuilman and Peeper 2009). Given that SASP factors are capable of remodeling tissue and promoting a pro-inflammatory phenotype, they can exacerbate age-related pathology (Coppé et al. 2010). In a paracrine fashion, SASP factors enhance senescence in neighboring cells. Therefore, neutralizing SASP factors or preventing their expression has been proposed as a possible strategy to reduce the levels and deleterious actions of senescent cells in culture and in vivo (Moiseeva et al. 2013). In this regard, mTOR inhibition decreased the secretion of IL1-dependent cytokines, including IL6 (Laberge et al. 2015; Herranz et al. 2015; Noh et al. 2019), and the use of neutralizing antibodies against the IL1 receptor (IL1R) and soluble protein ILlA was sufficient to reduce the downstream activity of transcription factor NF-kB, thereby abolishing the transcription of SASP factors (Orjalo et al. 2009). Second, senescent cells are inherently immunogenic and are therefore targets of immunological surveillance (Sagiv and Krizhanovsky 2013; Sagiv et al. 2016; Biran et al. 2015), pointing to possible immune-associated interventions in senostatic therapy.
Importantly, the above-mentioned features of senescent cells are centered on the plasma membrane. In the following section, we review several membrane proteins that were identified as distinguishing senescent from non-senescent cells and we address the advantages of alternative senolytic and senostatic strategies aiming at cell surface proteins. We discuss their impact as well as future challenges to transform proof-of-concept evidence into successful therapies.
3 Targeting the Membrane of Senescent Cells
3.1 Biomarkers on the Plasma Membrane of Senescent Cells
Efforts are increasing to find novel senescence biomarkers associated with the outside of the plasma membrane because intracellular (nuclear or cytoplasmic) markers are believed to be less useful candidates for therapeutic interventions, particularly those directed at live cells. Plasma membrane markers are especially desirable for strategies aimed at removing senescent cells through the immune system (Krizhanovsky et al. 2008; Xue et al. 2007).
A number of proteins were found to be elevated on the plasma membrane of senescent cells, including DCR2, ICAM1, and NOTCH3 (Collado et al. 2005; Gorgoulis et al. 2005; Cui et al. 2013) (Table 7.1). Althubiti and coworkers (Althubiti et al. 2014) subsequently identified a number of plasma membrane-associated proteins including DEP1 and B2MG as potential biomarkers of senescence in cell culture and tissues. In a recent screen for senescent biomarkers using mice immunized with senescent lung fibroblasts, Frescas et al. (2017) found antibodies that recognized senescence-associated cell surface antigens. Among them, IgM clone 9H4 specifically detected a posttranslationally modified form of the protein vimentin, present in high levels on the plasma membrane of senescent cells (Frescas et al. 2017). The fact that the modified vimentin was also detectable in the plasma of aged mice (SAMP8, senescence-accelerated mouse prone 8) compared to wild-type (C57BL6) suggested that this protein could both accumulate on the cell surface and be secreted into the extracellular space (Frescas et al. 2017).
3.2 Cell Surface Proteins Enhancing Immune Surveillance
Besides the studies mentioned above, there have been other recent attempts to identify and remove senescent cells by targeting cell surface proteins specifically expressed in senescent cells. Sagiv et al. (2016) discovered that the expression of two ligands of NKG2D, MICA and ULBP2, increased greatly in cells rendered senescent following DNA damage, expression of oncogenes, or replicative exhaustion. It was already known that the two NKG2D ligands were more highly expressed in response to cellular stresses (Spear et al. 2013), and their accumulation on the surface of senescent fibroblasts facilitated NK cell-mediated cytotoxicity through a mechanism previously described (Sagiv et al. 2016; Soriani et al. 2009; Iannello et al. 2013; Schmiedel and Mandelboim 2018). The introduction of antibodies directed at MICA or ULBP2 as well as the ablation of Nkg2d genes in mice suppressed the elimination of senescent cells by NK cells, demonstrating that cell surface antigens can drive the removal of senescent cells by the immune system (Sagiv et al. 2016). In a follow-up study, shedding of NKG2D ligands from the cell surface was found to enable severely damaged senescent cells to escape immunosurveillance (Muñoz et al. 2019).
Kim et al. (2017) reported evidence that senescent cells could be selectively eliminated by exploiting the presence of another plasma membrane protein, DPP4 (dipeptidyl peptidase 4, also known as CD26). A screen of cell surface markers uncovered > 100 proteins more highly expressed on the plasma membrane of senescent fibroblasts than proliferating fibroblasts; among them, DPP4 was most highly increased in several senescent cells and was studied as a potential biomarker (Kim et al. 2017). DPP4 had been studied extensively in the context of glucose metabolism and type 2 diabetes (T2D) (Mentlein and Dipeptidyl-peptidase 1999; Röhrborn et al. 2015), as DPP4 cleaves and thereby inactivates glucagon-like peptide-1 (GLP-1), a protein that stimulates insulin production and helps to remove intestinal glucose. Members of the gliptin family of DPP4 inhibitors (e.g., sitagliptin, saxagliptin, and vildagliptin) have been used in combination with insulin for T2D therapy (Gomez-Peralta et al. 2018). Although they are effective for T2D therapy and are generally safe (Scheen 2018), their impact on senescence has not been examined at present. However, as DPP4 was highly abundant on the plasma membrane of senescent cells but not proliferating cells, antibody-dependent cell-mediated cytotoxicity (ADCC) was used to eliminate senescent cells selectively. A humanized anti-DPP4 antibody was employed to tag senescent (DPP4-positive) fibroblasts, and these were subsequently eliminated by using NK cells, while proliferating cells (DPP4-negative) were preserved (Kim et al. 2017).
3.3 Membrane-Associated Proteins Modulating the SASP
Among the unique features of senescence, the rise in SASP is particularly detrimental, as the ensuing proinflammatory phenotype and debilitated extracellular matrix compromise tissue and organ function and exacerbate age-related pathology (Hernandez-Segura et al. 2018; Campisi 2011). Thus, the SASP trait itself represents an attractive therapeutic target, as suppressing SASP can ameliorate the harmful impact of senescent cells. Along these lines, compounds such as rapamycin, metformin, or inhibitors of NFKB1 (NF-κB) or p38 have been developed to suppress SASP in senescent cells (Xie et al. 2016; Nacarelli and Sell 2017). However, as mentioned earlier, the long-term use of chemical compounds has inherent side effects.
Studies highlighting the role of plasma membrane proteins in promoting the SASP are beginning to emerge. The levels of the plasma membrane protein CD36 were found to increase strongly in response to different senescent stimuli in a range of cell types (Chong et al. 2018). The interaction of several ligands, including Aβ42 and oxidized low-density lipoprotein (oxLDL), with CD36 triggered signaling through SRC and p38 that culminated with the transcription of NF-κB-driven SASP factors. In this manner, CD36 was found to promote the SASP trait and to foment senescence (Chong et al. 2018).
An earlier report showed that IL1A (IL-1α) accumulated on the senescent cell surface and that the levels of major proinflammatory factors IL6 and IL8 decreased when IL1A was blocked by the IL1 receptor antagonist (Orjalo et al. 2009). This study suggested that IL1 was a major upstream regulator of the SASP program, and further indicated that regulation was elicited from the cell membrane (or the extracellular region). In agreement with this form of regulation, a surfaceome analysis of proteins on the plasma membrane by Kim et al. (2018) led the authors to propose a novel function for the membrane-associated protein SCAMP4 (secreted carrier membrane protein 4) in the SASP. As reported earlier for the related member SCAMP5, which promotes the calcium-regulated exocytosis of signal peptide-containing cytokines such as CCL5 in carcinoma cells and monocytes (Han et al. 2009), SCAMP4 was also found to be capable of enhancing the secretion of SASP factors in senescent cells (Kim et al. 2018), although the specific mechanism was not elucidated. At the molecular level, SCAMP4 protein accumulated on the surface of senescent fibroblasts due to the greater stability of SCAMP4 in senescent cells, opening a window for possible therapeutic actions. The authors concluded that SCAMP4 promotes the secretion of SASP factors including IL6, IL8, CCL2, CXCL1 and MIF, with a possible autocrine influence on spreading and further strengthening the senescent phenotype (Kim et al. 2018).
4 Future Directions
4.1 Future Directions in Senolysis and Senostasis Focused on the Plasma Membrane
The initial examples presented here provide proof of concept that plasma membrane proteins are suitable targets of senolytic interventions, aimed at killing senescent cells (as with DPP4, modified VIM, and NKG2D ligands) as well as senostatic interventions, aimed at preventing a detrimental senescent phenotype (as with SCAMP4 and CD36) (Fig. 7.1).
Proteins present on the surface of senescent cells present opportunities for the elimination of senescent cells (senolysis) or the inhibition of senescent cell function (senostasis). Proof-of-principle examples include several plasma membrane proteins recently shown to be drastically elevated in senescent cells. DPP4, NFKB2 ligands, and modified VIM were found to serve as antigens that triggered strong immunosurveillance, while suppression of SCAMP4 and CD36 potently reduced the senescence-associated secretory trait
Universal markers of senescent on the plasma membrane appear particularly attractive for both senolytic and senostatic interventions. Many of the membrane markers listed in Table 7.1 were found elevated primarily in fibroblasts and endothelial cells that were rendered senescent via different mechanisms. However, other senescent cells residing in liver, muscle, pancreas, lung, epithelial tissues, immune system, etc., should also be investigated in order to identify truly ubiquitous plasma membrane targets. At the same time, plasma membrane markers of senescence that are specific to given cell types or to specific senescence programs may also be desirable for interventions aimed at select senescent cell populations. Whether universal or specific, more comprehensive knowledge of the plasma membrane proteome in a wide range of senescent cell types and senescence inducers is needed. Moreover, in principle other plasma membrane-associated molecules (e.g., lipids, carbohydrates, and nucleic acids) may also be suitable targets of universal or specific senescence-modulatory interventions.
Additional attention should be paid to develop improved approaches (molecular, biochemical, pharmacological, etc.) for senolysis and senostasis. An immediate advance over ADCC is the technology known as antibody-drug conjugate (ADC), through which specific cell surface markers can be identified by an antibody linked to a highly cytotoxic drug (Sievers and Senter 2013). Developed for cancer therapy, ADC antibodies have been shown capability to deliver drugs that cause cell death, generally by eliciting DNA damage, but could be repurposed to deliver senolytic or senostatic agents.
In specific cases in which the plasma membrane markers of senescence are enzymes, select drugs capable of modulating their function could be exploited. The peptidase DPP4 is a putative example of this approach, as numerous safe and effective inhibitors of DPP4 are already in the market for T2D and warrant direct attention. With increased knowledge of senescence-associated plasma membrane markers, other proteins may also emerge that can be modulated pharmacologically.
In addition, superior models in which to test the therapeutic benefit of senostatic and senolytic interventions are needed. Mouse models have provided helpful information thus far, but improved human models, e.g., organoids and tissue grafts, will help to establish the experimental specifics of efficacy of such approaches.
Finally, methods to deliver therapeutic agents directed at factors on the plasma membrane of senescent cells, whether broad-spectrum of selective, represent an ongoing challenge. Senescent cells directly accessible via the circulatory system (immune cells, endothelial cells, etc.) may be readily targeted. However, strategies to reach senescent cells located deeper in organs are necessary in order to gain full benefit from interventions to modulate the senescent cell compartment and achieve favorable outcomes.
References
Alcorta DA, Xiong Y, Phelps D, Hannon G, Beach D, Barrett JC (1996) Involvement of the cyclin-dependent kinase inhibitor p16 (INK4a) in replicative senescence of normal human fibroblasts. Proc Natl Acad Sci USA 93(24):13742–13747
Althubiti M, Lezina L, Carrera S, Jukes-Jones R, Giblett SM, Antonov A et al (2014) Characterization of novel markers of senescence and their prognostic potential in cancer. Cell Death Dis 5(11):e1528
Baker DJ, Wijshake T, Tchkonia T, LeBrasseur NK, Childs BG, van de Sluis B et al (2011) Clearance of p16Ink4a-positive senescent cells delays ageing-associated disorders. Nature 479:232–236
Benanti JA, Galloway DA (2004) Normal human fibroblasts are resistant to RAS-induced senescence. Mol Cell Biol 24:2842–2852
Ben-Porath I, Weinberg RA (2004) When cells get stressed: an integrative view of cellular senescence. J Clin Invest 113(1):8–13
Biran A, Perelmutter M, Gal H, Burton DG, Ovadya Y, Vadai E et al (2015) Senescent cells communicate via intercellular protein transfer. Genes Dev 29(8):791–802
Birch J, Barnes PJ, Passos JF (2018) Mitochondria, telomeres and cell senescence: implications for lung ageing and disease. Pharmacol Ther 183:34–49
Campisi J (2005) Senescent cells, tumor suppression, and organismal aging: good citizens, bad neighbors. Cell 120(4):513–522
Campisi J (2011) Cellular senescence: putting the paradoxes in perspective. Curr Opin Genet Dev 21(1):107–112
Cang S, Iragavarapu C, Savooji J, Song Y, Liu D (2015) ABT-199 (venetoclax) and BCL-2 inhibitors in clinical development. J Hematol Oncol 8:129
Casella G, Munk R, Kim KM, Piao Y, De S, Abdelmohsen K et al (2019) Transcriptome signature of cellular senescence. Nucl Acids Res 47(14):7294–7305
Chang J, Wang Y, Shao L, Laberge RM, Demaria M, Campisi J et al (2016) Clearance of senescent cells by ABT263 rejuvenates aged hematopoietic stem cells in mice. Nat Med 22(1):78–83
Chen Z, Trotman LC, Shaffer D, Lin HK, Dotan ZA, Niki M et al (2005) Crucial role of p53-dependent cellular senescence in suppression of Pten-deficient tumorigenesis. Nature 436(7051):725–730
Childs BG, Baker DJ, Kirkland JL, Campisi J, van Deursen JM (2014) Senescence and apoptosis: dueling or complementary cell fates? EMBO Rep 15(11):1139–1153
Chong M, Yin T, Chen R, Xiang H, Yuan L, Ding Y et al (2018) CD36 initiates the secretory phenotype during the establishment of cellular senescence. EMBO Rep 19(6):pii: e45274
Collado M, Serrano M (2010) Senescence in tumours: evidence from mice and humans. Nat Rev Cancer 10(1):51–57
Collado M, Gil J, Efeyan A, Guerra C, Schuhmacher AJ, Barradas M et al (2005) Tumour biology: senescence in premalignant tumours. Nature 436:642
Coppe JP, Patil CK, Rodier F, Sun Y, Munoz DP, Goldstein J et al (2008) Senescence-associated secretory phenotypes reveal cell-nonautonomous functions of oncogenic RAS and the p53 tumor suppressor. PLoS Biol 6:2853–2868
Coppé JP, Desprez PY, Krtolica A, Campisi J (2010) The senescence-associated secretory phenotype: the dark side of tumor suppression. Annu Rev Pathol 5:99–118
Cui H, Kong Y, Xu M, Zhang H (2013) Notch3 functions as a tumor suppressor by controlling cellular senescence. Cancer Res 73:3451–3459
Czabotar PE, Lessene G, Strasser A, Adams JM (2014) Control of apoptosis by the BCL-2 protein family: implications for physiology and therapy. Nat Rev Mol Cell Biol 15(1):49–63
Demaria M, Ohtani N, Youssef SA, Rodier F, Toussaint W, Mitchell JR et al (2014) An essential role for senescent cells in optimal wound healing through secretion of PDGF-AA. Dev Cell 31(6):722–733
Dimri GP, Lee X, Basile G, Acosta M, Scott G, Roskelley C et al (1995) A biomarker that identifies senescent human cells in culture and in aging skin in vivo. Proc Natl Acad Sci USA 92:9363–9367
Farr JN, Xu M, Weivoda MM, Monroe DG, Fraser DG, Onken JL et al (2017) Targeting cellular senescence prevents age-related bone loss in mice. Nat Med 23(9):1072–1079
Frescas D, Roux CM, Aygun-Sunar S, Gleiberman AS, Krasnov P, Kurnasov OV et al (2017) Senescent cells expose and secrete an oxidized form of membrane-bound vimentin as revealed by a natural polyreactive antibody. Proc Natl Acad Sci USA 114(9):E1668–E1677
Gomez-Peralta F, Abreu C, Gomez-Rodriguez S, Barranco RJ, Umpierrez GE (2018) Safety and efficacy of DPP4 inhibitor and basal insulin in type 2 diabetes: an updated review and challenging clinical scenarios. Diabetes Ther 9(5):1775–1789
Gorgoulis VG, Pratsinis H, Zacharatos P, Demoliou C, Sigala F, Asimacopoulos PJ et al (2005) p53-dependent ICAM-1 overexpression in senescent human cells identified in atherosclerotic lesions. Lab Invest 85:502–511
Gorgoulis V, Adams PD, Alimonti A, Bennett DC, Bischof O, Bishop C et al (2019) Cellular senescence: defining a path forward. Cell 179(4):813–827
Guerra C, Mijimolle N, Dhawahir A, Dubus P, Barradas M, Serrano M et al (2003) Tumor induction by an endogenous K-ras oncogene is highly dependent on cellular context. Cancer Cell 4:111–120
Han C, Chen T, Yang M, Li N, Liu H, Cao X (2009) Human SCAMP5, a novel secretory carrier membrane protein, facilitates calcium-triggered cytokine secretion by interaction with SNARE machinery. J Immunol 182(5):2986–2996
Herbig U, Jobling WA, Chen BP, Chen DJ, Sedivy JM (2004) Telomere shortening triggers senescence of human cells through a pathway involving ATM, p53, and p21(CIP1), but not p16(INK4a). Mol Cell 14(4):501–513
Hernandez-Segura A, Nehme J, Demaria M (2018) Hallmarks of Cellular Senescence. Trends Cell Biol 28(6):436–453
Herranz N, Gallage S, Mellone M, Wuestefeld T, Klotz S, Hanley CJ et al (2015) mTOR regulates MAPKAPK2 translation to control the senescence-associated secretory phenotype. Nat Cell Biol 17(9):1205–1217
Iannello A, Thompson TW, Ardolino M, Lowe SW, Raulet DH (2013) p53-dependent chemokine production by senescent tumor cells supports NKG2D-dependent tumor elimination by natural killer cells. J Exp Med 210(10):2057–2069
Kim WY, Sharpless NE (2006) The regulation of INK4/ARF in cancer and aging. Cell 127(2):265–275
Kim KM, Noh JH, Bodogai M, Martindale JL, Yang X, Indig FE et al (2017) Identification of senescent cell surface targetable protein DPP4. Genes Dev 31(15):1529–1534
Kim KM, Noh JH, Bodogai M, Martindale JL, Pandey PR, Yang X et al (2018) SCAMP4 enhances the senescent cell secretome. Genes Dev Jul 32(13–14):909–914
Kirkland JL, Tchkonia T (2017) Cellular senescence: a translational perspective. EBioMedicine 21:21–28
Krishnamurthy J, Torrice C, Ramsey MR, Kovalev GI, Al-Regaiey K, Su L et al (2004) Ink4a/Arf expression is a biomarker of aging. J Clin Invest 114:1299–1307
Krizhanovsky V, Yon M, Dickins RA, Hearn S, Simon J, Miething C et al (2008) Senescence of activated stellate cells limits liver fibrosis. Cell 134(4):657–667
Kuilman T, Peeper DS (2009) Senescence-messaging secretome: SMS-ing cellular stress. Nat Rev Cancer 9(2):81–94
Laberge RM, Sun Y, Orjalo AV, Patil CK, Freund A, Zhou L et al (2015) MTOR regulates the pro-tumorigenic senescence-associated secretory phenotype by promoting IL1A translation. Nat Cell Biol 17(8):1049–1061
Lehmann M, Korfei M, Mutze K, Klee S, Skronska-Wasek W et al (2017) Senolytic drugs target alveolar epithelial cell function and attenuate experimental lung fibrosis ex vivo. Eur Respir J 50(2):pii: 1602367
Liu Y, Sanoff HK, Cho H, Burd CE, Torrice C, Ibrahim JG et al (2009) Expression of p16(INK4a) in peripheral blood T-cells is a biomarker of human aging. Aging Cell 8:439–448
López-Otín C, Blasco MA, Partridge L, Serrano M, Kroemer G (2013) The hallmarks of aging. Cell 153(6):1194–1217
Lowe SW, Cepero E, Evan G (2004) Intrinsic tumour suppression. Nature 432(7015):307–315
Mentlein R, Dipeptidyl-peptidase IV (1999) (CD26)–role in the inactivation of regulatory peptides. Regul Pept 85(1):9–24
Moiseeva O, Deschênes-Simard X, St-Germain E, Igelmann S, Huot G, Cadar AE et al (2013) Metformin inhibits the senescence-associated secretory phenotype by interfering with IKK/NF-κB activation. Aging Cell 12(3):489–498
Muñoz DP, Yannone SM, Daemen A, Sun Y, Vakar-Lopez F, Kawahara M et al (2019) Targetable mechanisms driving immunoevasion of persistent senescent cells link chemotherapy-resistant cancer to aging. JCI Insight, pii: 124716
Muñoz-Espín D, Serrano M (2014) Cellular senescence: from physiology to pathology. Nat Rev Mol Cell Biol 15:482–496
Nacarelli T, Sell C (2017) Targeting metabolism in cellular senescence, a role for intervention. Mol Cell Endocrinol 455:83–92
Narita M, Nunez S, Heard E, Narita M, Lin AW, Hearn SA et al (2003) Rb-mediated heterochromatin formation and silencing of E2F target genes during cellular senescence. Cell Jun 113:703–716
Nelson JA, Krishnamurthy J, Menezes P, Liu Y, Hudgens MG, Sharpless NE et al (2012) Expression of p16(INK4a) as a biomarker of T-cell aging in HIV-infected patients prior to and during antiretroviral therapy. Aging Cell 11:916–918
Newgard CB, Sharpless NE (2013) Coming of age: molecular drivers of aging and therapeutic opportunities. J Clin Invest 123(3):946–950
Noh JH, Kim KM, Pandey PR, Noren Hooten N, Munk R, Kundu G et al (2019) Loss of RNA-binding protein GRSF1 activates mTOR to elicit a proinflammatory transcriptional program. Nucl Acids Res 47(5):2472–2486
Ogrodnik M, Miwa S, Tchkonia T, Tiniakos D, Wilson CL, Lahat A, Jurk D et al (2017) Cellular senescence drives age-dependent hepatic steatosis. Nat Commun 8:15691
Orjalo AV, Bhaumik D, Gengler BK, Scott GK, Campisi J (2009) Cell surface-bound IL-1alpha is an upstream regulator of the senescence-associated IL-6/IL-8 cytokine network. Proc Natl Acad Sci USA 106(40):17031–17036
Ovadya Y, Krizhanovsky V (2018) Strategies targeting cellular senescence. J Clin Invest 128(4):1247–1254
Parrinello S, Samper E, Krtolica A, Goldstein J, Melov S, Campisi J (2003) Oxygen sensitivity severely limits the replicative lifespan of murine fibroblasts. Nat Cell Biol 5:741–747
Röhrborn D, Wronkowitz N, Eckel J (2015) DPP4 in Diabetes. Front Immunol 6:386
Roos CM, Zhang B, Palmer AK, Ogrodnik MB, Pirtskhalava T, Thalji NM et al (2016) Chronic senolytic treatment alleviates established vasomotor dysfunction in aged or atherosclerotic mice. Aging Cell 15(5):973–977
Sagiv A, Krizhanovsky V (2013) Immunosurveillance of senescent cells: the bright side of the senescence program. Biogerontology 14(6):617–628
Sagiv A, Burton DG, Moshayev Z, Vadai E, Wensveen F, Ben-Dor S et al (2016) NKG2D ligands mediate immunosurveillance of senescent cells. Aging 8(2):328–344
Scheen AJ (2018) The safety of gliptins: updated data in 2018. Expert Opin Drug Saf 17(4):387–405
Schmiedel D, Mandelboim O (2018) NKG2D ligands-critical targets for cancer immune escape and therapy. Front Immunol 9:2040
Sievers EL, Senter PD (2013) Antibody-drug conjugates in cancer therapy. Annu Rev Med 64:15–29
Soriani A, Zingoni A, Cerboni C, Iannitto ML, Ricciardi MR, Di Gialleonardo V et al (2009) ATM-ATR-dependent up-regulation of DNAM-1 and NKG2D ligands on multiple myeloma cells by therapeutic agents results in enhanced NK-cell susceptibility and is associated with a senescent phenotype. Blood 113(15):3503–3511
Spear P, Wu MR, Sentman ML, Sentman CL (2013) NKG2D ligands as therapeutic targets. Cancer Immun 13:8
Tse C, Shoemaker AR, Adickes J, Anderson MG, Chen J, Jin S et al (2008) ABT-263: a potent and orally bioavailable Bcl-2 family inhibitor. Cancer Res 68(9):3421–3428
van Deursen JM (2014) The role of senescent cells in ageing. Nature 509(7501):439–446
von Zglinicki T (2002) Oxidative stress shortens telomeres. Trends Biochem Sci 27(7):339–344
Waaijer ME, Parish WE, Strongitharm BH, van Heemst D, Slagboom PE, de Craen AJ et al (2012) The number of p16INK4a positive cells in human skin reflects biological age. Aging Cell 11(4):722–725
Wiley CD, Flynn JM, Morrissey C, Lebofsky R, Shuga J, Dong X et al (2017) Analysis of individual cells identifies cell-to-cell variability following induction of cellular senescence. Aging Cell 16(5):1043–1050
Xie J, Wang X, Proud CG (2016) mTOR inhibitors in cancer therapy, F1000Res
Xue W, Zender L, Miething C, Dickins RA, Hernando E, Krizhanovsky V et al (2007) Senescence and tumour clearance is triggered by p53 restoration in murine liver carcinomas. Nature 445(7128):656–660
Yosef R, Pilpel N, Tokarsky-Amiel R, Biran A, Ovadya Y, Cohen S et al (2016) Directed elimination of senescent cells by inhibition of BCL-W and BCL-XL. Nat Commun 7:11190
Zhu Y, Tchkonia T, Pirtskhalava T, Gower AC, Ding H, Giorgadze N et al (2015) The Achilles’ heel of senescent cells: from transcriptome to senolytic drugs. Aging Cell 14(4):644–658
Zhu Y, Tchkonia T, Fuhrmann-Stroissnigg H, Dai HM, Ling YY, Stout MB et al (2016) Identification of a novel senolytic agent, navitoclax, targeting the Bcl-2 family of anti-apoptotic factors. Aging Cell 15(3):428–435
Zhu Y, Doornebal EJ, Pirtskhalava T, Giorgadze N, Wentworth M, Fuhrmann-Stroissnigg H et al (2017) New agents that target senescent cells: the flavone, fisetin, and the BCL-XL inhibitors, A1331852 and A1155463. Aging 9(3):955–963
Zindy F, Quelle DE, Roussel MF, Sherr CJ (1997) Expression of the p16INK4a tumor suppressor versus other INK4 family members during mouse development and aging. Oncogene 15:203–211
Acknowledgements
This work was supported by the NIA IRP, NIH, and by the Chungnam National University research fund.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Kim, K.M., Noh, J.H., Gorospe, M. (2020). Senolysis and Senostasis Through the Plasma Membrane. In: Muñoz-Espin, D., Demaria, M. (eds) Senolytics in Disease, Ageing and Longevity. Healthy Ageing and Longevity, vol 11. Springer, Cham. https://doi.org/10.1007/978-3-030-44903-2_7
Download citation
DOI: https://doi.org/10.1007/978-3-030-44903-2_7
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-44902-5
Online ISBN: 978-3-030-44903-2
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)