Abstract
Cardiovascular disease remains the leading cause of death in American adult men and women. In the United States, it is estimated that nearly one in every four deaths is attributable to heart disease, amounting to more than 600,000 people annually. Smoking cessation efforts, tight control of blood pressure, and widespread use of cholesterol-lowering medications are examples of commonly practiced approaches to lowering CAD risk of the asymptomatic population, based on the increasing understanding of the markers of disease and its pathophysiology. As more specific markers that correlate with cardiovascular and coronary artery disease (CAD) are identified, the predictive value is improved, the understanding of the disease is enhanced, and its relationship to other disease states—specifically obesity—is elucidated. Although long-term cardiac-specific survival data after intentional weight loss is lacking, there is strong data to support that weight loss in the overweight and obese population decreases nearly all identified risk factors for cardiovascular disease. To date, bariatric surgery has proven to be the only durable treatment method for significant weight loss in the obese population. Thus, weight loss surgery is a rational approach to the treatment of obesity in patients at risk for cardiovascular disease.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Cholesterol
- Obesity
- Overweight
- Toxicity
- Angiotensin
- Cardiomyopathy
- Bariatric
- Diabetes
- Survival
- Risk
- Gastrectomy
-
1.
Review the factors that contribute to cardiovascular disease, focusing on obesity and overweight.
-
2.
Discuss preoperative cardiac evaluation for prospective bariatric surgery patients.
-
3.
Discuss how weight loss and bariatric surgery improve cardiac function and decrease risk factors among associated comorbidities.
Introduction
Cardiovascular disease remains the leading cause of death in the United States. Nearly one in every four deaths is attributable to heart disease, amounting to more than 600,000 people annually [1], and more than 700,000 Americans have a heart attack every year due to coronary artery disease (CAD) or coronary heart disease (CHD) [2]. Not surprisingly, the economic health-care burden due to CAD in the United States is significant—the annual direct medical care expenditure exceeds $5 billion—and the 10-year expenditure of patients with CAD is approximately eight times that of patients without CAD [3]. As a result, there is an interest in identifying modifiable risk factors as rational points for early intervention.
The Framingham Heart Study, initiated in the mid-twentieth century, examined populations regularly for the development of cardiovascular disease and introduced the notion that lifestyle practices and environmental influences along with genetic traits influence overall cardiac health. From this, major risk factors for the development of CAD were identified, initially determined by following the asymptomatic population cohort for approximately 10 years. The factors that were determined to be most predictive included age, gender, tobacco use, high blood pressure or need for antihypertensive medication , total cholesterol , and high-density lipoprotein (HDL) concentrations [4]. Based on these data, tools for calculating estimated 10-year risk for myocardial infarction are available online (e.g., http://hp2010.nhibihin.net/atpiii/calculator.asp). Consequently, smoking cessation, tight control of blood pressure, and widespread use of cholesterol-lowering medications are examples of commonly practiced approaches to lowering CAD risk of the asymptomatic population. This is based on the increasing understanding of the markers of cardiovascular disease and its pathophysiology.
With the advancement of biomedical technology, there is a continued effort to refine and identify new physiologic, genetic, and image-derived biomarkers to more accurately identify the at-risk population. For biomarkers to be effective, they should be measured precisely and reproducibly and accurately reflect the time course of disease. In addition, the significance of various biomarkers may change in value at different times. Therefore, the calculated absolute risk of CAD is dependent on age-nonspecific risk scores. For example, a calculated 10-year risk of <10% is considered “low risk,” while a calculated risk of >20% is considered “high risk.”
Novel serum biomarkers have been isolated to enhance the sensitivity of traditional population-wide risk assessment tools and to potentially improve disease risk. Biomarkers for heart disease include C-reactive protein (CRP) , low-density lipoprotein (LDL) and very low-density lipoprotein (VLDL) , B-type natriuretic peptide (BNP) , apolipoprotein B (apoB), lipoprotein (a), homocysteine, and troponin I. These biomarkers may be used in detection and prevention of CAD but also in enhancing our understanding of pathophysiology [4,5,6]. Indeed, the association between dyslipidemia—elevated LDL, triglycerides, total cholesterol, and low HDL—and coronary artery disease is now well established [7]. Similarly, the efficacy of statin therapy in lowering CAD risk is also known [8]. Elevated low-density lipoprotein (LDL) cholesterol , low concentrations of high-density lipoprotein (HDL) cholesterol , and elevated triglyceride levels are now commonly used markers for an increased risk of CAD.
A reduction in LDL cholesterol with the use of statin medications (HMG-CoA reductase inhibitor) reduces mortality from CAD, likely by improving levels of cholesterol homeostasis and the dependent inflammatory state. Atherosclerosis, including coronary artery disease, has been recognized as an inflammatory disease [9, 10]. In fact, LDL, homocysteine, and high blood pressure are believed to directly or indirectly lead to arterial endothelial injury, subsequent local inflammation, smooth muscle activation, and the development of coronary atherosclerosis.
Thus, inflammatory markers have also become relevant as CAD biomarkers. C-reactive peptide (CRP) has emerged as a strong independent predictor of future myocardial infarction, stroke, and peripheral artery disease [5]. CRP, in association with LDL levels, was found to be a strong predictor of cardiovascular events, while the total cholesterol to HDL ratio as well as CRP was found to be predictive of future myocardial infarction [11]. Similarly, lipoprotein (a) and homocysteine levels reflect an increased inflammatory state and an increased risk of developing CAD in an asymptomatic population.
B-type natriuretic peptide (BNP) is released directly from the heart. Preferentially synthesized in ventricular myocardium, BNP responds to acute ventricular volume expansion and serves to counteract the effects of the renin-angiotensin-aldosterone system [12]. Not only a marker for ventricular dysfunction and heart failure, BNP, and its N-terminal propeptide , NTproBNP , has been shown to be strong predictive indicators of future cardiovascular events in patients with stable CAD.
An additional marker for cardiovascular disease that has gained special interest in the setting of obesity is leptin . Leptin is derived from adipocytes and is thought to be involved in weight regulation and insulin homeostasis. High leptin concentrations were found to be predictive of first acute myocardial infarction, along with higher body mass index (BMI) , plasma insulin, and diastolic blood pressure [13, 14]. As more specific markers that correlate with cardiovascular and coronary artery disease are identified, the predictive value is improved, the understanding of the disease is enhanced, and its relationship to other disease states—specifically obesity—is elucidated.
Obesity and Cardiovascular Disease
I have more flesh than another man; and therefore more frailty.
William Shakespeare (Henry IV Part I, Act III Scene III)
The link between obesity and physical frailty has long been recognized. Obesity is associated with multiple comorbid conditions and an increased mortality. The overall relative risk of mortality is approximately 2.00 for men and 1.65 for women with a BMI > 35 kg/m2, compared to the normal weight population [15]. In fact, trends suggest that obesity will overtake smoking as the primary preventable cause of death in the United States [16]. Thus, it is not surprising that obesity in an otherwise healthy population in the fifth decade of life is also associated with frailty decades later [17].
Obesity is associated with significant comorbid conditions. These maladies affect almost every system in the body including the musculoskeletal, pulmonary, endocrine, immune, and cardiovascular systems. The cardiovascular system is especially affected with multiple physiologic changes that often lead to adverse outcomes. Thus, concurrent with the dramatic increase in the prevalence of obesity in the United States and worldwide over the past several decades, reaching epidemic proportions [18, 19], the American Heart Association (AHA) issued a “call to action ” in response [20]. Since then, the AHA and American College of Cardiology recognized obesity as raising the risk of hypertension, stroke, and coronary artery disease, as well as being associated in and of itself with increased risk mortality from coronary vascular disease [21].
One of the early physiologic changes with obesity is an increase in total blood volume, with subsequent increase in ventricular filling pressures, cardiac output, and cardiac workload [22]. Preexisting or subsequent development of hypertension further encourages cardiac chamber dilation , left ventricular hypertrophy , and remodeling [23]. It is not surprising then that the obese population is at risk for ventricular dysfunction , although its impact on mortality is not clear. Indeed, obesity alone is considered an independent predictor of developing CAD and a higher risk of systolic and diastolic left ventricular dysfunction [24, 25].
However, the relationship between obesity and cardiovascular disease appears to be much more complex. In fact, obesity actually lowers systemic vascular resistance and thus may provide a protective component, leading to the “obesity paradox .” Multiple studies suggest that overweight and obese patients with known cardiovascular disease are predicted to have a better event-free survival compared to normal or underweight cohorts [25,26,27,28]. Thus, it seems that obesity increases the initial risk for cardiovascular disease, but it may confer some protective benefit from mortality in those already afflicted with heart failure or CAD. The mechanism for this observation is not clear, although there is some evidence to suggest that BMI is not the proper parameter to be measured. Rather, waist circumference (WC) and waist-hip ratio (WHR) , as proxies for central adiposity, may be more predictive of mortality in patients with CAD. In addition, it is uncertain whether these findings in obese individuals still hold true in the severely or morbidly obese. A large systematic review and collaborative analysis found that increasing central obesity was associated with higher mortality in patients with CAD and the use of WC in addition to WHR is a more effective indicator than either measurement alone [29].
Nonetheless, the PDAY (Pathobiological Determinants of Atherosclerosis in Youth ) study provided convincing data to suggest that obesity in adolescents and young adults (aged 15–34 years) accelerates the progression of atherosclerosis 10–20 years before any clinical symptoms [30]. From available evidence, it is clear that earlier onset of obesity, corresponding to a longer period of time spent with obesity, leads to a lifetime increased risk of cardiovascular disease and mortality. A younger obese population has a higher mortality than a BMI-matched older population [31, 32].
It has long been recognized that fat tissue is not a simple storage cell of fat but is metabolically highly active and is responsible for systemic secretion of molecules that impact cardiovascular homeostasis [33]. Of these factors, the concentrations of plasminogen activator inhibitor-1, angiotensin II, CRP, fibrinogen, and TNF-α (alpha) are directly affected by BMI. Similarly, the concentrations of IL-6 directly affect CRP levels [34].
Along with obesity, insulin resistance has been implicated in the development of CAD. The insulin resistance syndrome or metabolic syndrome concepts reflect the concurrent pathophysiology of obesity, insulin resistance, dyslipidemia, and cardiovascular disease. Obesity is a leading cause of insulin resistance, which implies a peripheral tissue resistance to the effects of circulating insulin, mainly glucose metabolism [35]. In the adipocytes, lipogenesis and lipid storage and metabolism are directly affected, resulting in an increased systemic load of free fatty acids. Interestingly, central adiposity has the greatest impact on insulin resistance and its effects. The hyperinsulinemic state is also implicated as a growth factor with direct deleterious cardiac effects. While the exact mechanism is not yet fully understood, it is clear that insulin resistance is part of a syndrome that significantly increases the risk of cardiovascular disease and whose hallmark is obesity [36].
Obstructive sleep apnea (OSA) is directly associated with obesity and impacts cardiac function. Obesity, in and of itself, is a common cause of alveolar hypoventilation and is considered the most important modifiable risk factor for sleep-disordered breathing [36, 37]. Obesity and OSA so often coexist in the same population that it can be difficult to identify diagnoses that are correlated with one and not the other. Those patients with sleep apnea have an increased mortality as well as a risk of diurnal hypertension, nocturnal dysrhythmias, pulmonary hypertension, right and left ventricular failure, myocardial infarction, and stroke [38, 39].
Whether related to OSA or not, the risk of acute arrhythmia leading to sudden death is markedly increased in the obese population. In the Framingham study, obesity was found to be a major modifiable risk factor for sudden cardiac death in both men and women [40]. Prolongation of the QTc interval seen on ECG (electrocardiogram) seen in these subjects increases the risk of arrhythmia and cardiovascular mortality. In fact, both prolongation and shortening of the QT interval were found to be associated with all-cause mortality, cardiovascular disease and ventricular arrhythmia, and sudden death. For prolonged QT, the increased risk is seen for both males and females at an interval of 420–450 ms [41, 42].
Meanwhile, weight loss in the morbidly obese population, and specifically loss of central adiposity, is associated with a reduction in cardiovascular risk factors and overall mortality. Despite this, there is uncertainty whether intentional weight loss is beneficial to patients with known cardiovascular disease. This point of controversy is based on several studies that have shown increased risk of arrhythmia and negative outcomes in patients with aggressive dietary restrictions, meal replacements, and very-low-calorie diets or pharmacotherapy with cardiac toxicity [22]. This is typically manifested through the prolongation of the QTc interval often observed in these situations. Fatal or life-threatening arrhythmias have been observed after radical dieting or liquid protein meal replacements.
Nonetheless, it is clear that weight loss in the obese population improves obesity-related risk factors for cardiovascular disease. Even modest medical weight loss in overweight and obese patients directly correlates with a reduction of CRP, lipids, and glucose, with a lower total mortality as well [43]. In general, weight loss by any means leads to improvement of cardiovascular risk factors and is strongly recommended for the asymptomatic obese population [44]. Total blood volume, stroke volume, and systemic arterial pressure all decline with weight loss. Consequently, filling pressures decrease, left ventricular stroke work diminishes, and oxygen consumption decreases [45].
Finally, weight loss also benefits the symptomatic obese population already diagnosed with cardiovascular disease. A healthy eating protocol along with structured exercise training is paramount in this population. A study of 377 patients with cardiovascular disease who lost weight had a 38% relative risk reduction relative to patients who did not lose weight, in all-cause mortality and cardiovascular events—including myocardial infarction, stroke, and heart failure [45, 46]. Although long-term cardiac-specific survival data after intentional weight loss is lacking, there is strong data to support that weight loss in the overweight and obese population decreases nearly all identified risk factors for cardiovascular disease. Yet, there is some conflicting data to suggest that change in weight (positive or negative) may be associated with adverse outcomes in all-comers with coronary heart disease [47]. Nonetheless, it is widely reported that weight gain is closely associated with adverse cardiovascular risk and suggested that in the severely and morbidly obese, weight loss is of significant benefit.
To date, bariatric surgery has proven to be the only durable treatment method for significant weight loss in the obese population. Thus, weight loss surgery is a rational approach to the treatment of obesity in patients at risk for cardiovascular disease. Due to their higher risk for cardiac events, the preoperative evaluation is especially significant.
Preoperative Cardiac Evaluation
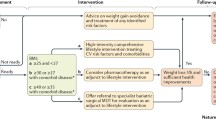
Obesity independently increases cardiovascular risk factors, and many obese patients may have underlying cardiovascular disease but are asymptomatic when they present for bariatric surgery evaluation. Thus, the cardiac evaluation of these patients is especially important. In fact, the National Institutes of Health Consensus Development Program Statement specifically advocates a multidisciplinary approach , suggesting a thorough medical evaluation as part of the preoperative work-up, and it is common practice to include an evaluation by a cardiologist to assess for cardiac risks [48].
There is no consensus approach to the preoperative work-up of the bariatric patient, but general guidelines are followed by most. A thorough history and physical examination are used to identify comorbid conditions with specific surgical implications and those that can affect cardiovascular risk, such as the presence of dyspnea on exertion, daytime sleepiness, and nighttime snoring; the presence of hypertension, diabetes, or insulin resistance; or the use of medications that may prolong the QT interval. The degree of central obesity should be assessed by physical examination, as it relates to cardiovascular risk more strongly than BMI.
A routine preoperative laboratory evaluation should also include the known biochemical markers previously discussed, including a full lipid profile, CRP, and fasting insulin levels. Other biochemical markers that may be informative preoperatively and followed in the postoperative period include lipoprotein (a) and homocysteine.
It is common for the obese patient to undergo at least one adjunct study as part of the preoperative work-up. Although the utility of routine chest X-rays is debated, it may be useful to obtain in the bariatric population, which has a high prevalence of asymptomatic cardiopulmonary disease. Findings of cardiomegaly or cephalization of pulmonary vessels may suggest cardiovascular disease and prompt further work-up.
The electrocardiogram (ECG) , however, should be considered a mandatory part of the preoperative evaluation of the bariatric patient. Structural cardiac changes that are induced by obesity may influence the appearance of the ECG. These include cardiac displacement, ventricular hypertrophy , a distant heart due to chest wall adiposity, and elevated pulmonary pressures [33]. As a result, multiple findings are expected in the obese patient who has cardiac morphological changes. Of patients preparing for bariatric surgery, 62% were found to have specific ECG abnormalities [49]. Included were LVH, RVH, short PR, bundle branch block, ST-T wave abnormalities, AV block, tachycardia, ectopy, and prolonged QT. Therefore, the finding of an abnormal echocardiogram may predict a more complicated postoperative course and need for intensive care utilization [50].
Those patients with a significant cardiac history, cardiac symptoms, an especially low functional capacity, or abnormalities uncovered in the initial evaluation should undergo a noninvasive functional cardiac evaluation or cardiac stress testing. While myocardial nuclear perfusion imaging may provide valuable information, table weight and torso diameter limitations may make it impractical in some of these patients [51]. Stress echocardiography is widely used to evaluate known or suspected coronary artery disease. However, the image quality in the obese population may be significantly compromised, decreasing the efficacy of the test. Subepicardial fatty tissue, for example, may commonly result false positive for a pericardial effusion or conversely underestimate the amount of pericardial fluid [52]. On the other hand, many have advocated for the use of dobutamine stress echocardiography. It can provide the practicality of an echocardiogram, with enhanced image quality. Echocardiograph contrast agents improve the fidelity of the test and may be used in stress and nonstress testing. In a study of 590 morbidly obese patients, the transthoracic dobutamine stress echocardiograph was inconclusive in only 6.4% of patients and was found to be safe and effective [51, 53].
Other noninvasive cardiac imaging studies include the single-photon emission computed tomography (SPECT) , positron emission tomography (PET) , and coronary computed tomographic angiography (CTA) . The utility and cost-effectiveness of these studies are unclear, and it is not currently recommended for routine risk stratification in the asymptomatic patient [54].
Bariatric Surgery and Cardiac Risk Factors
Obesity and tobacco smoking remain the most impactful, modifiable risk factors for cardiovascular disease. With the prevalence of morbid obesity increasing to epidemic proportions, it is an increasingly important target for preventive treatment.
Conventional weight loss approaches include dietary modifications, behavioral and exercise therapy, and pharmacotherapy. Although the greatest weight loss effect is seen when these modalities are used in combination, surgery remains the only treatment modality that provides significant and durable weight loss, especially in the population with BMI >35 kg/m2. For example, in a large meta-analysis of patients undergoing gastric banding, gastric bypass, gastroplasty, biliopancreatic diversion, or duodenal switch, Buchwald et al. demonstrated a total percent excess weight loss of >60% [55]. In addition, long-term studies demonstrate the durability of such weight loss results after bariatric surgery [56, 57]. There is no combination of medical treatment to produce similarly robust results.
Hypertension or the use of antihypertensive medication, in the obese population, is significantly improved following weight loss surgery [55, 58, 59]. A meta-analysis of more than 16,000 bariatric patients demonstrated a resolution or reduction of hypertension in 68% after bariatric surgery [60].
Similarly, insulin resistance and diabetes are markedly improved, and often enter remission, after surgery. The positive effect of bariatric surgery on type 2 diabetes is now well established [61,62,63,64], and a decrease in insulin resistance through to occur early after surgery [65]. The benefits to patients with diabetes are sustained in long-term studies of 5 and 10 years [66, 67]. The early decrease in insulin resistance and improvement in diabetes seen after bariatric surgery cannot be explained by calorie restriction alone or by the amount of weight loss [68]. The incretin effect, changes in bile acid metabolism, and changes in the gut microbiome have all been implicated; and it is likely that multiple gastrointestinal regulatory mechanisms are responsible for this change in physiology [69,70,71].
Dyslipidemia is present in 40–50% of preoperative bariatric patients and is also a major modifiable risk factor for CAD [72]. Elevated total cholesterol, LDL, triglycerides, and low HDL levels are all markers for coronary artery risk. Bariatric surgery results in an improvement in each of these parameters. One of the early studies to demonstrate an improvement in dyslipidemia after a weight loss procedure was based on POSCH (Program on the Surgical Control of the Hyperlipidemias), a prospective randomized trial [73]. A partial ileal bypass significantly reduced total and LDL cholesterol and reduced mortality from CAD after a 5-year follow-up.
Since then, multiple studies have shown other bariatric operations are also effective at treating dyslipidemia and improving total cholesterol, LDL, triglyceride, and HDL levels. Often lipid levels return to normal after surgery, and in parallel, medication usage for treating hyperlipidemia decreases [74,75,76,77]. While the SOS failed to show a decrease in total cholesterol in all patients, they also consistently demonstrated a decrease in triglycerides and an increase in HDL cholesterol [57]. These results have persisted in long-term studies as well [67]. Overall, weight loss surgery leads to an improvement in all measures of dyslipidemia in multiple different bariatric operations.
Other biochemical risk factors also improve after bariatric surgery. One of the most predictive cardiac risk factor, high-sensitivity CRP (hsCRP), as well as homocysteine levels, decreases significantly after weight loss surgery [60, 74]. These results have been shown to persist for at least 1 year after surgery and were observed in different weight loss operations as well [78]. Of the other emerging cardiac risk factors, homocysteine and lipoprotein (a) levels also significantly improve and trend toward normal. Similarly, B-type natriuretic peptide, a marker for current altered cardiac dynamics, changes after bariatric surgery in a manner directly correlated with weight loss [79].
Taken together, bariatric surgery was shown to decrease the Framingham risk score for at least 7 years after surgery [80], absolute risk of a cardiovascular event, and mortality from all causes in general and from cardiovascular causes specifically. Weight loss surgery resulted in a sustained reduction of 10-year cardiovascular risk, with the impact greatest in those patients with preexisting risk factors [81].
It is not surprising, therefore, that in addition to effective weight loss, bariatric surgery results in a decrease in all-cause mortality [82, 83], as well as mortality from CAD. In fact, Adams et al. demonstrated a 56% reduction in mortality from coronary artery disease after bariatric surgery [84]. With long-term follow-up extending beyond a decade, bariatric surgery in general was associated with reduced number of fatal cardiovascular deaths and lower incidence of first-time myocardial infarction or stroke [85]. Finally, obese patients with already known clinically significant cardiomyopathy seemed to benefit from weight loss surgery [86].
Conclusion
Cardiovascular disease is a leading cause of death in American adults. Obesity has emerged as a major modifiable risk factor for future fatal and nonfatal cardiovascular events. Other than obesity, multiple factors have been identified as predictors of cardiovascular risk. Bariatric surgery is the most effective treatment of obesity and results in improvement in all known modifiable risk factors for cardiovascular disease.
Question Section
-
1.
Does gastric bypass surgery increase HDL?
-
A.
True
-
B.
False
-
A.
-
2.
Which procedure improves cardiac risk factors?
-
A.
Gastric band
-
B.
Sleeve gastrectomy
-
C.
Gastric bypass
-
D.
All of the above
-
A.
-
3.
Which cardiac risk factors are improved following bariatric surgery?
-
A.
Insulin resistance
-
B.
Dyslipidemia
-
C.
CRP
-
D.
Hypertension
-
E.
All of the above
-
A.
-
4.
Long-term weight loss is similar between patients who undergo weight loss surgery and patients treated non-operatively (diet modification and exercise programs).
-
A.
True
-
B.
False
-
A.
References
Miniño AM, Murphy SL, Xu J, Kochanek KD. Deaths: final data for 2008, national vital statistics reports, 59 10. Hyattsville: National Center for Health Statistics; 2011.
https://www.cdc.gov/heartdisease/facts.htm. Last cited July 2018.
Russell MW, Huse DM, Drowns S, Hamel EC, Hartz SC. Direct medical costs of coronary artery disease in the United States. Am J Cardiol. 1998;81:1110–5.
Wilkins JT, Lloyd-Jones DM. Are novel serum biomarkers informative? Med Clin North Am. 2012;96:1–11.
Ridker PM, Rifai N, Rose L, Buring JE, Cook NR. Comparison of C-reactive protein and low-density lipoprotein cholesterol levels in the prediction of first cardiovascular event. N Engl J Med. 2002;347:1557–65.
Field PA, Vasan RS. Clin Pharmacol Ther 2018 [Epub ahead of print].
Prospective Studies Collaboration, Lewington S, Whitlock G, Clarke R, Sherliker P, Emberson J, Halsey J, et al. Blood cholesterol and vascular mortality by age, sex, and blood pressure: a meta-analysis of individual data from 61 prospective studies with 55,000 vascular deaths. Lancet. 2007;370:1829–39.
Colhoun HM, Betteridge DJ, Durrington PN, Hitman GA, Neil HA, Livingstone SJ, CARDS investigators, et al. Primary prevention of cardiovascular disease with atorvastatin in type 2 diabetes in the Collaborative Atorvastatin Diabetes Study (CARDS): multicentre randomized placebo-controlled trial. Lancet. 2004;364:685–96.
Ross R. Atherosclerosis-an inflammatory disease. N Engl J Med. 1999;340:115–26.
Mullenix PS, Andersen CA, Starnes BW. Atherosclerosis as inflammation. Ann Vasc Surg. 2005;19:130–8.
Ridker PM. Evaluating novel cardiovascular risk factors: can we better predict heart attacks? Ann Intern Med. 1999;130:933–7.
Daniels LB, Maisel AS. Natriuretic peptides. J Am Coll Cardiol. 2007;50:2357–68.
Soderberg S, Ahren B, Jansson JH, Johnson O, Hallmans G, Asplund K, Olsson T. Leptin is associated with increased risk of myocardial infarction. J Intern Med. 1999;246:409–18.
Wallerstedt S, Eriksson AL, Niklason A, Ohlsson C, Hedner T. Serum leptin and myocardial infarction in hypertension. Blood Press. 2004;13:243–6.
Pischon T, Boeing H, Hoffmann K, Bergmann M, Schulze MB, Overvad K, et al. General and abdominal adiposity and risk of death in Europe. N Engl J Med. 2008;359:2105–20.
Allison DB, Fontaine KR, Manson JE, Stevens J, Van Itallie TB. Annual deaths attributable to obesity in the United States. JAMA. 1999;282:1530–8.
Strandberg TE, Sirola J, Pitkala KH, Tilvis RS, Standberg AY, Stenholm S. Association of midlife obesity and cardiovascular risk with old age frailty: a 26-year follow-up of initially healthy men. Int J Obes. 2012;36:1153–7.
Mokdad AH, Serdula MK, Dietz WH, Bowman BA, Marks JS, Koplan JP. The spread of the obesity epidemic in the United States 1991–1998. JAMA. 1999;282:1519–22.
Kuczmarski RJ, Flegal KM, Campbell SM, Johnson CL. Increasing prevalence of overweight among US adults: the National Health and Nutrition Examination Surveys, 1960 to 1991. JAMA. 1994;272:205–11.
Eckel RH, Krauss RM. American Heart Association call to action: obesity as a major risk factor for coronary heart disease: AHA Nutrition Committee. Circulation. 1998;97:2099–100.
Jensen MD, Ryan DH, Apovian CM, et al. 2013 AHA/ACC/TOS guideline for the management of overweight and obesity in adults: a report of the American College of Cardiology/American Heart Association task force on practice guidelines and The Obesity Society. J Am Coll Card. 2014;63:2985–3023.
Lavie CJ, Milani RV, Ventura HO. Obesity and cardiovascular disease. J Am Coll Cardiol. 2009;53:1925–32.
Zarich SW, Kowalchuk GJ, McGuire MP, Benotti PN, Mascioli EA, Nesto RW. Left ventricular filling abnormalities in asymptomatic morbid obesity. Am J Cardiol. 1991;68:377–81.
Garrison RJ, Castelli WP. Weight and thirty-year mortality of men in the Framingham study. Ann Intern Med. 1985;103:1006–9.
Lavie CJ, Alpert MA, Arena R, Mehra MR, Milani RV, Ventura HO. Impact of obesity and the obesity paradox on prevalence and prognosis in heart failure. JACC Heart Fail. 2013;1:93–102.
Horwich TB, Fonarow GC, Hamilton MA, MacLellan WR, Woo MA, Tillisch JH. The relationship between obesity and mortality in patients with heart failure. J Am Coll Cardiol. 2001;38:789–95.
Lavie CJ, Osman AF, Milani RV, Mehra MR. Body composition and prognosis in chronic systolic heart failure: the obesity paradox. Am J Cardiol. 2003;91:891–4.
Oreopoulos A, Padwal R, Kalantar-Zadeh K, Fonarow GC, Norris CM, McAlister FA. Body mass index and mortality in heart failure: a meta-analysis. Am Heart J. 2008;156:13–22.
Coutinho T, Goel K, CorrêadeSá D, Kragelund C, Kanaya AM, Zeller M, et al. Central obesity and survival in subjects with coronary artery disease: a systematic review of the literature and collaborative analysis with individual subject data. J Am Coll Cardiol. 2011;57(19):1877–86.
McGill HC Jr, McMahan CA, Herderick EE, Zieske AW, Malcom GT, Tracy RE, Pathobiological Determinants of Atherosclerosis in Youth (PDAY) Research Group, et al. Obesity accelerates the progression of coronary atherosclerosis in young men. Circulation. 2002;105:2712–8.
Fontaine KR, Redden DT, Wang C, Westfall AO, Allison DB. Years of life lost due to obesity. JAMA. 2003;289:187–93.
Drenick EJ, Bale GS, Seltzer F, Johnson DG. Excessive mortality and causes of death in morbidly obese men. JAMA. 1980;243:443–5.
Poirier P, Giles TD, Bray GA, Hong Y, Stern JS, Pi-Sunyer FX, American Heart Association; Obesity Committee of the Council on Nutrition, Physical Activity, and Metabolism, et al. An update of the 1997 American Heart Association scientific statement on obesity and heart disease from the obesity committee of the council on nutrition, physical activity, and metabolism. Circulation. 2006;113(6):898–918.
Cigolini M, Targher G, Bergamo AI, Tonoli M, Agostino G, De Sandre G. Visceral fat accumulation and its relation to plasma hemostatic factors in healthy men. Arterioscler Thromb Vasc Biol. 1996;16:368–74.
Kahn BB, Flier JS. Obesity and insulin resistance. J Clin Invest. 2000;106:473–81.
Ginsberg HN. Insulin and cardiovascular disease. J Clin Invest. 2000;106:453–8.
Young T, Palta M, Dempsey J, Skatrud J, Weber S, Badr S. The occurrence of sleep-disordered breathing among middle-aged adults. N Engl J Med. 1993;328:1230–5.
Partinen M, Jamieson A, Guilleminault C. Long-term outcome for obstructive sleep apnea syndrome patients. Mortal Chest. 1988;94:1200–4.
Strollo PJ Jr, Rogers RM. Obstructive sleep apnea. N Engl J Med. 1996;334:99–104.
Kannel WB, Plehn JF, Cupples LA. Cardiac failure and sudden death in the Framingham study. Am Heart J. 1988;115:869–75.
Robbins J, Nelson JC, Rautaharju PM, Gottdiener JS. The association between the length of the QT interval and mortality in the Cardiovascular Health Study. Am J Med. 2003;115:689–94.
Zhang Y, Post WS, Dalal D, Blasco-Colmenares E, Tomaselli GF, Guallar E. QT-interval duration and mortality rate. Arch Intern Med. 2011;171:1727–33.
Lavie CJ, Thomas RJ, Squires RW, Allison TG, Milani RV. Exercise training and cardiac rehabilitation in primary and secondary coronary prevention. Mayo Clin Proc. 2009;84:373–83.
Klein S, Burke LE, Bray GA, Blair S, Allison DB, Pi-Sunyer X, American Heart Association Council on Nutrition, Physical Activity, and Metabolism, et al. Clinical implications of obesity with specific focus on cardiovascular disease: a statement for professionals from the American Heart Association Council on Nutrition, Physical Activity, and Metabolism: endorsed by the American College of Cardiology Foundation. Circulation. 2004;110(18):2952–67.
Sierra-Johnson J, Romero-Corral A, Somers VK, Lopez-Jimenez F, Thomas RJ, Squires RW, et al. Prognostic importance of weight loss in patients with coronary heart disease regardless of initial body mass index. Eur J Cardiovasc Prev Rehabil. 2008;15:336–40.
Artham SM, Lavie CJ, Milani RV, Ventura HO. Value of weight reduction in patients with cardiovascular disease. Curr Treat Options Cardiovasc Med. 2010;12:21–35.
Dong SY, Yan ST, Wang ML, Li ZB, Fang LQ, Zeng Q. Associations of body weight and weight change with cardiovascular events and mortality in patients with coronary heart disease. Atherosclerosis. 2018;274:104–11.
Gastrointestinal surgery for severe obesity. http://consensus.nih.gov/1991/1991gisurgeryobesity084html.htm. Last accessed 12 Aug 2013.
Catheline JM, Bihan H, Le Quang T, Sadoun D, Charniot JC, Onnen I, et al. Preoperative cardiac and pulmonary assessment in bariatric surgery. Obes Surg. 2008;18:271–7.
Gonzalez R, Bowers SP, Venkatesh KR, Lin E, Smith CD. Preoperative factors predictive of complicated postoperative management after roux-en-Y gastric bypass for morbid obesity. Surg Endosc. 2003;17:1900–4.
Gugliotti D, Grant P, Jaber W, Aboussouan L, Bae C, Sessler D, et al. Challenges in cardiac risk assessment in bariatric surgery patients. Obes Surg. 2008;18:129–33.
Savage DD, Garrison RJ, Brand F, Anderson SJ, Castelli WP, Kannel WB, et al. Prevalence and correlates of posterior extra echocardiographic spaces in a free-living population based sample (the Framingham study). Am J Cardiol. 1983;51:1207–12.
Lerakis S, Kalogeropoulos AP, El-Chami MF, Georgiopoulou VV, Abraham A, Lynch SA, et al. Transthoracic dobutamine stress echocardiography in patients undergoing bariatric surgery. Obes Surg. 2007;17:1475–81.
Budoff MJ, Raggi P, Beller GA, Berman DS, Druz RS, Malik S, Rigolin VH, Wigold WG, Soman P. Noninvasive cardiovascular risk assessment of the asymptomatic diabetic patient. JACC Cardiovasc Imaging. 2016;9:176–92.
Buchwald H, Avidor Y, Braunwald E, Jensen MD, Pories W, Fahrbach K, et al. Bariatric surgery; a systematic review and meta-analysis. JAMA. 2004;292:1724–37.
Sjöström L, Lindroos AK, Peltonen M, Torgerson J, Bouchard C, Carlsson B, Swedish Obese Subjects Study Scientific Group, et al. Lifestyle, diabetes, and cardiovascular risk factors 10 years after bariatric surgery. N Engl J Med. 2004;351:2683–93.
Maciejewski M, Arterburn DE, Scoyoc LV, Smith VA, Yancy WS, Weidenbacher HJ, et al. Bariatric surgery and long-term durability of weight loss. JAMA. 2016;151(11):1046–55.
Hinojosa MW, Varela JE, Smith BR, Che F, Nguyen NT. Resolution of systemic hypertension after laparoscopic gastric bypass. J Gastrointest Surg. 2009;13:793–7.
Ghiassi S, Morton J, Bellatorre N, Eisenberg D. Short-term medication cost savings for treating hypertension and diabetes after gastric bypass. Surg Obes Relat Dis. 2012;8:269–74.
Heneghan HM, Meron-Eldar S, Brethauer SA, Schauer PR, Young JB. Effect of bariatric surgery on cardiovascular risk profile. Am J Cardiol. 2011;108:1499–507.
Pories WJ, Swanson MS, MacDonald KG, Long SB, Morris PG, Brown BM, et al. Who would have thought it? An operation proves to be the most effective therapy for adult-onset diabetes mellitus. Ann Surg. 1995;222:339–50.
Schauer PR, Burguera B, Ikramuddin S, Cottam D, Gourash W, Hamad G, et al. Effect of laparoscopic Roux-en-Y gastric bypass on type 2 diabetes mellitus. Ann Surg. 2003;238:467–84.
Mingrone G, Panunzi S, De Gaetano A. Bariatric surgery versus conventional medical therapy for type 2 diabetes. N Engl J Med. 2012;366:1577–85.
Schauer PR, Kashyap SR, Wolski K, Brethauer SA, Kirwan JP, Pothier CE, et al. Bariatric surgery versus intensive medical therapy in obese patients with diabetes. N Engl J Med. 2012;366:1567–76.
Rao RS, Yanagisawa R, Kini S. Insulin resistance and bariatric surgery. Obes Rev. 2012;13:316–28.
Thereaux J, Lesuffleur T, Czernichow S, Basdevant A, Msika S, Nocca D, et al. Association between bariatric surgery and rates of continuation, discontinuation, or initiation of antidiabetes treatment 6 years later. JAMA Surg. 2018;153(6):526–33.
Sjöström L. Bariatric surgery and reduction in morbidity and mortality: experiences from the SOS study. Int J Obes. 2008;32:S93–7.
Pournaras DJ, Osborne A, Hawkins SC, et al. Remission of type 2 diabetes after gastric bypass and banding: mechanism and 2 year outcomes. Ann Surg. 2010;252:966–71.
Mohammad S, Ramos LS, Buck J, Levin LR, Rubino F, McGraw TE. J Biol Chem. 2011;286:43062–70.
Park CW, Torquati A. Physiology of weight loss surgery. Surg Clin North Am. 2011;91:1149–61.
Batterham RL, Cummings DE. Mechanisms of diabetes improvement following bariatric/metabolic surgery. Diabetes Care. 2016;39:893–901.
Kaul A, Sharma J. Impact of bariatric surgery on co-morbidities. Surg Clin North Am. 2011;91:1295–312.
Buchwald H, Varco RL, Matts JP, et al. Effect of partial ileal bypass surgery on mortality and morbidity from coronary heart disease in patients with hypercholesterolemia: report of the Program on the Surgical Control of the Hyperlipidemias (POSCH). N Engl J Med. 1990;323:946–55.
Williams DB, Hagedorn JC, Lawson EH, et al. Gastric bypass reduces biochemical cardiac risk factors. Surg Obes Relat Dis. 2007;3:8–13.
Nguyen NT, Varela E, Sabio A, Tran CL, Stamos M, Wilson SE. Resolution of hyperlipidemia after laparoscopic Roux-en-Y gastric bypass. J Am Coll Surg. 2006;203:24–9.
Dixon JB, O’Brien PE. Lipid profile in the severely obese: changes with weight loss after lap-band surgery. Obes Res. 2002;10:903–10.
Segal JB, Clark JM, Shore AD. Prompt reduction in use of medications for comorbid conditions after bariatric surgery. Obes Surg. 2009;19:1646–56.
Woodard GA, Peraza J, Bravo S, Toplosky L, Hernandez-Boussard T, Morton JM. One year improvement in cardiovascular risk factors: a comparative trial of laparoscopic Roux-en-Y gastric bypass vs. adjustable gastric banding. Obes Surg. 2010;20:578–82.
Changchien EM, Ahmed S, Betti F, Higa J, Kiely K, Hernandez-Boussard T, et al. B-type natriuretic peptide increases after gastric bypass surgery and correlates with weight. Surg Endosc. 2011;25:2338–43.
Morton JM. ASMBS annual meeting 2012. http://asmbs.org/2012/06/new-stanford-university-study-shows-risk-factors-for-heart-attack-remain-low-seven-years-after-gastric-bypass/. Last accessed 15 Aug 2013.
Donadelli SP, Salgado W, Marchini JS, Schmidt A, Amato CA, Ceneviva R, et al. Change in predicted 10-year cardiovascular risk following Roux-en-Y gastric bypass surgery: who benefits? Obes Surg. 2011;21:569–73.
Christou NV, Sampalis JS, Liberman M, Look D, Auger S, McLean AP, et al. Surgery decreases long-term mortality, morbidity, and health care use in morbidly obese patients. Ann Surg. 2004;240:416–23.
Sjöström L, Narbro K, Sjöström CD, Karason K, Larsson B, Wedel H, Swedish Obese Subjects Study, et al. Effects of bariatric surgery on mortality in Swedish obese subjects. N Engl J Med. 2007;357:741–52.
Adams TD, Gress RE, Smith SC, Halverson RC, Simper SC, Rosamond WD, et al. Long-term mortality after gastric bypass surgery. N Engl J Med. 2007;357:753–61.
Sjöström L, Peltonen M, Jacobson P, Sjöström CD, Karason K, Wedel H, et al. Bariatric surgery and long-term cardiovascular events. JAMA. 2012;307:56–65.
McCloskey CA, Ramani GV, Mathier MA, Schauer PR, Eid GM, Mattar SG, et al. Bariatric surgery improves cardiac function in morbidly obese patients with severe cardiomyopathy. Surg Obes Relat Dis. 2007;3:503–7.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Kitamura, R.K., Morton, J.M., Eisenberg, D. (2020). Cardiac Risk Factor Improvement Following Bariatric Surgery. In: Nguyen, N., Brethauer, S., Morton, J., Ponce, J., Rosenthal, R. (eds) The ASMBS Textbook of Bariatric Surgery. Springer, Cham. https://doi.org/10.1007/978-3-030-27021-6_46
Download citation
DOI: https://doi.org/10.1007/978-3-030-27021-6_46
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-27020-9
Online ISBN: 978-3-030-27021-6
eBook Packages: MedicineMedicine (R0)