Abstract
The management of patients receiving sedation anesthesia and analgesia for surgeries and procedures requires an understanding of the unique challenges involved. Absence of such a clear understanding could lead to harm or permanent injury to patients. Sedation anesthesia, is provided in many different locations outside of the operating room and hospital settings. Professional anesthesia providers are anesthesiologists and nurse anesthetists who remain the experts in the perioperative care and delivery of anesthesia and education. However, many non- anesthesia providers have been privileged to deliver sedation anesthesia. These individuals include but are not limited to nurses, dentists, and proceduralists. The spectrum of the continuum of depth of consciousness, the lack of or insufficient training, and inadequate preoperative patient selection and monitoring, are some of the factors that have resulted in adverse patient outcomes.
The goal of this chapter is to provide anesthesiologists and non-anesthesiologist providers with the principles of sedation anesthesia and analgesia and the American Society of Anesthesiology sedation guidelines and requirements. Sedation anesthesia, as with general anesthesia, requires the highest level of vigilance and patient care.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Sedation anesthesia
- Sedation guidelines
- Sedation monitoring
- ASA sedation guidelines
- Moderate sedation
- Depth of consciousness
- Oral sedation
- Sedation and patient selection
- Nitrous oxide
Introduction
Sedation for painful procedures predates general anesthesia. Before the discovery of nitrous oxide, surgeons used agents as varied as alcohol and opium to reduce the pain and anxiety related to injury and surgery. In modern terms, these patients were not anesthetized but were semiconscious or awake but somnolent. It was not until the mid-nineteenth century, when a dentist by the name of Horace Wells noted that nitrous oxide could have a profound effect on consciousness and perceived pain, that clinicians began to offer patients a level of sedation that approached general anesthesia [1]. With the discovery of local anesthetics and their widespread use as a means to relieve pain during surgical procedures, the use of nitrous oxide as a sedating agent became less popular. Subsequently, nitrous oxide was combined with compounds such as chloroform and halothane, producing an unconscious state well beyond sedation. Nitrous oxide, by reducing anxiety and providing analgesia, then became an adjuvant to pain control when used with more potent agents.
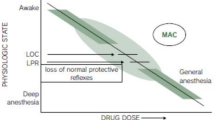
Today healthcare providers administer sedation to patients via a variety of routes. The management of patients receiving sedation for surgical and non-surgical procedures requires a deep understanding of the continuum of consciousness associated with the administration of sedating drugs. Consciousness can be lost during attempts to sedate patients because the activity of agents that depress the central nervous system is not always predictable. This continuum of sedation, inadequate monitoring, poor patient selection, and insufficient training can lead to adverse events when both anesthesia and non-anesthesia providers are involved. With the migration of inpatient surgeries to outpatient and office-based practices, the development of newer anesthetic drugs, the use of regional anesthesia , and new surgical techniques, sedation anesthesia and analgesia will continue to evolve, but will always require a complete understanding of applicable guidelines, regulations, and facility categories (Figure 14.1).
Shows the different levels of sedation and general anesthesia. The optimal level of sedation varies with each surgical procedure. Anesthesia providers must be capable of effectively performing a continuum of anesthesia planes, sometimes for the same case to achieve the ideal level of patient comfort and safety
The purpose of this chapter is to define levels of sedation, to identify the risks of sedation, to address the requirements for patient safety in all clinical circumstances, and to evaluate current sedation guidelines. The most recent updates on sedation and its applicability in the ambulatory setting will be discussed.
Definition of Sedation Levels
Consciousness reflects a biologic continuum. Sedation with agents that depress the CNS interrupts that continuum in ways that are not always predictable based on the agents themselves or the unique and highly individualized response in each subject. Though sedation is divided into three categories, light, moderate, or deep, in fact, this categorization does not reflect the reality of a lack of readily identifiable stop points in the movement of a patient from one level to the next. This lack of a dividing line is significant because the safety of the patient hangs in the balance. Deepening of sedation is associated with declining physiological self-protections, primarily within the airway. The point at which the circumstance is unsafe for the patient is variable. The conscious patient is capable of protecting the airway from foreign matter regurgitated from the esophagus and stomach. The sedated patient at some point in the continuum of consciousness cannot, and clinicians are rarely able to identify associated levels of risk despite experience in sedation management. The American Society of Anesthesiology (ASA) describes and defines three levels of sedation anesthesia [2]. Four parameters for each level of sedation, including general anesthesia, are used to define each level. It is paramount that the provider of sedation anesthesia completely understands these definitions for the following reasons; (1) each clinician and non-clinician that administers sedation anesthesia have different requirements, degrees, training, and limitations; (2) facility categories also differ in regulations at both the state, local and federal level as to what type of anesthesia can be provided to patients.
Minimal sedation (anxiolysis) is a drug-induced state defined as a patient that has a standard verbal or non-verbal response to vocal stimulation, and the cardiovascular function, spontaneous ventilation, and airway remain intact and unaffected. Moderate sedation (conscious sedation) is a drug-induced state defined as a patient who has a “purposeful” response to vocal stimulation alone or in addition to tactile stimulation. Vital signs, spontaneous ventilation, and airway systems may remain unaffected in moderate sedation. Deep sedation is a drug-induced state defined as a patient who responds purposefully only to repeated painful stimuli because normal vocal or tactile stimulation do not easily arouse them. These patients may require intervention for airway and ventilation management; however, cardiovascular parameters usually remain intact. Patients under general anesthesia are unarousable to painful stimuli and need cardiovascular, ventilatory, and airway support (Table 14.1).
As simple as these definitions may seem, providing sedation can be challenging and uncertain. Sedation anesthesia and analgesia often occur in remote locations such as offices, MRI suites, dental clinics, hospital bedsides, endoscopy suites, emergency rooms, in addition to operating rooms. This dilemma of variable locations and providers poses a real hazard to patient safety,
Patient Safety
Variability in levels of sedation and the perception that the actual procedures are short and inconsequential has led some to suggest that requirements for providing safe sedation can be curtailed without danger to the patient. These observations have often been exacerbated by the lack of complete knowledge of outcomes in patients sedated outside of medical centers. The lack of published reports of deaths and neurological injuries has been interpreted to mean that sedation without proper monitoring, training, and vigilance is safe. Recent closed claims analyses suggest otherwise.
Bhananker, Posner, and others reviewed all medical claims associated with Monitored Anesthesia Care (MAC) in the ASA Closed Claims Database. They found that over forty percent of claims related to sedation involved death and permanent neurological brain injury and was similar to the statistics for general anesthesia. They concluded that over-sedation, respiratory depression, and inadequate monitoring were the primary causes for mortality and morbidity during MAC cases [3]. Hug and others have suggested that patient injuries during MAC cases are due to the attitudes of anesthesia and surgical personnel. The notion that MAC is “safe” can lead to less vigilance and diligence on the part of the surgical and anesthesia team [4]. Hug proposed that MAC should stand for “Maximum Anesthesia Caution” [4].
The number of MAC sedation adverse events is unknown. Findings by Bhananker, Hug, and others were limited to the ASA Closed Claims Database. Sedation anesthesia was provided to patients in a vast number of locations and by a variety of non-anesthesia providers. The safety of these practices is currently not possible to estimate given that there are limited reported adverse events and clinical outcomes in the literature in these locations and by non-anesthesia providers. It is not until celebrities, such as Joan Rivers, suffered damage or death that the topics of patient safety, sedation guidelines, and provider credentials have come to national attention.
The Center for Medicare and Medicaid Services (CMS) issued a change in 2009 to its guidelines requiring that an anesthesiologist must oversee deep sedation anesthesia and analgesia [5]. Massachusetts General Hospital’s department of Anesthesiology reviewed a historical cohort of endoscopic procedures. They studied the incidence of adverse events comparing patients who received sedation by gastroenterologist-supervised RNs to those who received sedation by anesthesiologists or anesthesiologist-supervised CRNAs. They found a statistically significant reduction in reported sedation-related adverse events when anesthesia personnel were involved (.38% RN vs. 08%CRNA) [6].
With the increase in complex surgical procedures, increasing numbers of elderly and pediatric patients scheduled in non-OR locations for surgical and diagnostic procedures requiring sedation, the American Society of Anesthesiologists and other societies have developed clinical guidelines to ensure safe practices for administration of sedation.
The ASA guidelines clearly define which practitioners should be granted privileges for the administration of moderate sedation [7].
-
(i).
Anesthesia Professional: anesthesiologist, nurse anesthetist or anesthesiologist assistant
-
(ii).
Non-anesthesiologist Sedation Practitioner: Licensed physician, dentist, or podiatrist who has not completed postgraduate training in anesthesiology but trained to administer and supervise the administration of moderate sedation.
-
(iii).
Supervised Sedation Professional: a licensed registered nurse or advanced practice nurse or physician assistant who is trained to administer medications and monitors patients during moderate sedation under the direct supervision of an anesthesiologist or a non-anesthesiologist sedation practitioner
This statement applies to any facility type: dentistry offices, physician offices, hospitals, surgery centers, MRI suites, endoscopy suites. Therefore, all non-anesthesia professionsals that administer medications via any route across the continuum of sedation anesthesia and analgesia should have the training and skills to rescue and resuscitate any patients whose level of sedation reaches an unintended state. They should be able to perform a complete patient history and physical examination, pre-operative anesthesia evaluation, anesthetic assessment plan and acknowledge any risks factors that may pose a risk to the patient. Anesthesiologists remain the experts in the physiology, pharmacology and clinical care of patients that are administered sedation anesthesia, analgesia, regional anesthesia, and general anesthesia. A non-anesthesia professional should initiate a consultation with an anesthesiologist for questions and patient concerns, and a request for patient care when the anesthetic care is outside the capability of the professional’s skill set. The ASA clearly states that an anesthesia professional should be consulted to provide sedation anesthesia if a practitioner is not trained in the rescue of patients from general anesthesia, or for prolonged or therapeutic procedures requiring deep sedation, anticipated intolerance to standard sedatives or ASA class IV or V patients (Table 14.2).
Patient Selection and Pre-operative Evaluation
The first phase of sedation anesthesia and analgesia is patient selection and evaluation. A preoperative assessment is defined as the process of clinical evaluation that precedes the delivery of anesthesia for surgical and diagnostic procedures [8]. Anesthesia professionals are familiar with pre-operative anesthesia evaluation, however, this is not a usual practice or learned knowledge for non-anesthesia professionals. The preoperative anesthesia evaluation is an inherent part of the standard of care for anesthesiologists, regardless of the type of anesthetic delivered. The Joint Commission requires that all non-anesthesia professionals perform a pre-operative assessment when there is a potential for a compromised airway, respiratory depression, and cardiovascular dysfunction, as in the case of moderate or deep sedation [9]. The pre-operative anesthesia assessment should include a comprehensive review of the patient’s past and current medical records, diagnostic tests, laboratory results, previous surgeries and anesthetics, and any related complications and a complete physical examination.. A thorough and comprehensive review of systems for each patient should be performed to assess the patient’s ASA Classification and the imminent risk for anesthesia and surgical complications.
Preoperative evaluation deficiencies may lead to unanticipated adverse events, morbidity, and mortality. The Australian Incident Monitoring Study (AIMS) found that 11% of reported adverse anesthesia events in the Australian claim database were attributed to a pre-operative evaluation not being performed, and 23% of adverse events were because the anesthesiologist in the surgical case had not conducted a pre-operative anesthesia assessment (Table 14.3). Fifty-seven percent of these events were reported to be preventable with better preoperative anesthesia assessment and evaluation [10]. Metzner and colleagues looked at the ASA Closed Claims Database from 1990 and later to assess the patterns of injury and liability in office settings versus operating rooms (Table 14.4).
Nearly fifty percent of sedation anesthesia and MAC cases occur outside of the operating room. Metzner and others concluded that patients over the age of seventy (27%), patients with an ASA Classification of three and four (57%) and obese patients (56%) had complications related to sedation, anesthesia, and analgesia [11]. Adverse events, such as cardiopulmonary decompensation evident by desaturation, hypoxemia, hypercarbia, hypotension, and unintended moderate and deep sedation can occur in patients with increased co-morbidities. The ASA Task Force recommends that all major organ systems, previous anesthesia experience with sedation, general and regional anesthesia, allergies, medications, smoking history, and a focused physical and airway examination be performed and thoroughly reviewed. The current and updated ASA 2018 sedation guidelines for moderate sedation now recommend that the pre-operative anesthesia evaluation be done days to weeks before the day of surgery and again immediately before the procedure. This differs from the original ASA Task Force 2002 sedation guidelines that required that a pre-anesthetic evaluation be done immediately before surgery regardless of the level of anesthesia. The pre-anesthesia evaluation recommendations for deep sedation have not been published to date.
The preoperative fasting rules should also apply to sedation anesthesia and analgesia as recommended by the ASA. Individual hospitals, diagnostic centers, office suites and non-operating room departments may have their own fasting guidelines. It is essential to become familiar with and review these guidelines to be in compliance with hospital policy and to be sure that these guidelines comply with the ASA guidelines. Currently, the literature is inconclusive as to the safety and efficacy of preoperative fasting. Some studies have concluded that longer fasting times are less efficacious and do not improve patient safety [12]. Green, Mason, and Krauss conducted the first systematic review of published pulmonary aspiration incidence during sedation anesthesia and analgesia in patients of all ages. Nearly 3 percent of the identified records described one or more occurrences of pulmonary aspiration during sedation anesthesia and analgesia. The gastrointestinal endoscopy suite was found to have a significantly higher incidence of aspiration morbidity and mortality [13]. Observational studies have indicated the nil per os (NPO) status and the prevalence of aspiration during sedation is relatively infrequent, and even rare [13,14,15]. Despite the ongoing debate to rethink the current NPO guidelines, the ASA Task Force recommends the following guidelines to be used in sedation, general and regional anesthesia, and analgesia (Table 14.5).
Sedation and Monitoring
Serious adverse events have been reported related to monitored anesthesia care. Cardiovascular decompensation, respiratory depression, and arrest, permanent neurological brain injury and death are associated with surgical and diagnostic procedures during and after sedation in both adults and children [3]. The severity of sedation anesthesia adverse events is comparable to those of general anesthesia (Figure 14.2). Nearly half of these MAC-associated adverse events were deemed preventable with better monitoring.
The severity of injury in monitored anesthesia care (MAC), general, and regional anesthesia claims. The proportion of claims for death (14%) and permanent brain damage (7%) was reduced in regional anesthesia compared with MAC (33% death and 8% brain damage). In contrast, the severity of the injury was similar between MAC claims and those associated with general anesthesia (27% death and 10% brain damage). ∗ P < 0.025 MAC versus regional. (Figure from Bhananker et al. [16]. (Used with permission of Wolters Kluwer Health, Inc.))
The monitoring of patients undergoing surgical and diagnostic procedures with sedation requires the same level of vigilance for measuring and recording physiologic parameters as with patients administered general anesthesia. Patient monitoring described by the ASA Task Force consists of five measured parameters; (1) Level of consciousness (2) Oxygenation and ventilation (3) Hemodynamic monitoring (4) Recording of monitored parameters (5) Availability or presence of an individual for patient monitoring.
Although there is insufficient evidence to support that qualitative monitoring of a patient’s level of consciousness improves patient safety or decreases adverse events, the ASA Task Force recommends a five-minute interval monitoring of a patient’s response to verbal commands and tactile stimulation. Some studies have shown no conclusive evidence to support the use of depth of sedation monitoring devices such as the processed electroencephalogram (EEG) monitor, bispectral index (BIS) monitor, or the Anesthesia Responsiveness Monitor. A systematic review and meta-analysis (total of 16 clinical trials; 2138 participants) of the depth of monitoring devices used in sedation and analgesia, found no substantial evidence to support improved patient outcomes and decreased adverse events in patients monitored with processed EEG compared to those who were not. However, the total dose of Propofol used was significantly lower in the group of patients monitored with EEG compared to those who were not [16]. Other studies have also found no correlations with BIS monitoring and the level of sedation.
Oxygenation monitoring via pulse oximetry has been a staple for patient care for all levels of sedation and general anesthesia practices.
Capnography , the measurement of the concentration or the partial pressure of carbon dioxide via infrared spectroscopy, has proved beneficial in improving patient safety during sedation.
The respiratory function may become impaired and inadequate, during moderate and deep sedation secondary to opioids and benzodiazepines.. The marked decrease of the genioglossus nerve activity demonstrated by electromyography during the transition from consciousness to deeper levels of sedation may lead to airway obstruction, and aspiration secondary to pharyngeal dysfunction [17, 18]. Depression of the central nervous system leading to hypoventilation theoretically precedes hypoxemia. It’s hypothesized that the early detection of respiratory compromise may lead to earlier clinical interventions. Although the review of literature regarding the safety and effectiveness of capnography during sedation is scarce, current research demonstrates that the addition of capnography does reduce the incidence of hypoxia-related events [19,20,21]. Deitch showed that by using capnography during procedural sedation, inadequate ventilation was identified before the onset of hypoxemia by an average time of sixty seconds [19]. Another essential benefit of capnography, which is the graphical waveform measurement of the respiratory ventilation cycle, is that it can provide clinical diagnostic information to the clinician, which is otherwise unavailable with only a numerical ETCO2 value by capnometry. The height, width, and shape of the capnography waveform verify the quality of the patient’s ventilatory status. Understanding the four phases of the CO2 waveform and associated disease patterns such as hypopneic hypoventilation, bradypneic-hypoventilation, central apnea, obstructive apnea, and bronchospasm, the sedation provider can more readily treat the underlying cause. Use of capnography for continual monitoring of ventilatory function to supplement standard monitoring by observation and pulse oximetry is one of the new recommendations in the recently updated the guidelines on sedation and analgesia by the American Society of Anesthesiologists (ASA) Task Force by in March 2018 [2].
Summary of the Updated Guidelines
The updated guidelines specifically address moderate sedation/analgesia, and separate guidelines for deep sedation are underway. The guidelines were developed by a multidisciplinary task force of physicians including dental surgeons with the intent of addressing moderate procedural sedation provided by any medical specialty in any location. These guidelines do not specify certification requirements for providers.
The guidelines suggest that ASA Standards, Guidelines, and Policies should be adhered to except when not applicable to outpatient care. The facility should have a medical director or governing body that establishes policy and is responsible for the activities of the facility and its staff. The facility must comply with the local, state and federal laws and regulations with specific reference to the ASA “Statement on Nonoperating Room Anesthetizing Locations.”
Preoperative Care and Patient Evaluation
Patient and procedure selection should be such that duration and complexity of the procedure will permit the procedure to be completed under sedation, will not require general anesthesia and the patient be recovered and discharged from the facility. Preprocedure patient evaluation should preferably be done well ahead in advance. Before the procedure, patients or legal guardians should be informed of the benefits, risks, and limitations of sedation/analgesia and possible alternatives. Providers of sedation/analgesia must elicit patient preferences and verify that patients are adequately fasted as per the facility’s guidelines. There must be appropriate education, training and licensure, by the facility when non-anesthesiologist physicians are administering or supervising the administration of sedation/analgesia.
Personnel
When administering moderate sedation, it is likely that the level of intended sedation may be exceeded. Therefore, a designated individual, not from the team performing the procedure, should be present to monitor the patient throughout the procedure. There must be adequate training for the responsible individual in recognition and treatment of apnea and airway obstruction and this individual must be authorized to seek additional help.
Monitoring and Equipment
At a minimum, all facilities should have a reliable source of oxygen, suction, resuscitation equipment and emergency drugs. When providing deep sedation, an ECG monitor and a defibrillator should be readily available. Patients should be monitored for level of consciousness assessed by the response of patients; adequacy of ventilation assessed by clinical signs, capnography and pulse oximetry and hemodynamic stability assessed by measuring blood pressure, heart rate and electrocardiography. There should be a recording of monitored parameters and availability/presence of an individual responsible for patient monitoring. The frequency of monitoring depends on the depth of sedation, the type and amount of medications administered, the length of the procedure and the general condition of the patient.
During the Procedure
Studies show that the use of supplemental oxygen during the procedure reduces the frequency of hypoxemia. Therefore, supplemental oxygen should be administered unless specifically contraindicated for a particular patient or procedure. The name, route, site, time of administration and dosage of all drugs administered should be documented.
Recovery
Decreased stimulation after the procedure, delayed drug absorption after nonintravenous sedation and slow drug elimination may contribute to residual sedation during the recovery period. The facility must have appropriate staff and an equipped area to monitor the patients at regular intervals until (e.g., every 5–15 min) they are at the baseline level of consciousness and are not at increased risk of cardiorespiratory depression. Providers must design discharge criteria to minimize the risk of central nervous system or cardiorespiratory depression after discharge from observation by trained personnel. In the event of an emergency or need for extended care, written protocols should be in place to safely transfer patients to a prespecified facility in a timely manner.
Creation and Implementation of Patient Safety Processes
Facilities should have quality improvement processes and emergency preparedness plans to ensure patient safety. Quality improvement processes should be based on national, regional or institutional reporting protocols and should be periodically updated to keep up with the advances in sedation practices and technology. Team training, simulation drills and regular education of personnel should be utilized to prepare for rare events. An emergency response plan (e.g., activating the “code blue” team or activating the emergency medical response system: 911 or equivalent) should be created.
Sedation by Oral Route
With the intent of providing cost-effective medical care, there has been a tremendous increase in office-based medical and surgical procedures [22, 23]. This has changed the administration of anesthesia with increased use of sedation techniques and local anesthesia. Dental surgeons, faciomaxillary, and cosmetic plastic surgeons in office-based settings frequently administer oral sedation for office-based procedures [24, 25]. More recently, ophthalmologists have performed cataract surgery using oral sedation in combination with topical anesthesia [26]. However, the literature on the practice of oral sedation is very sparse.
Oral sedation offers the advantages of better patient compliance, efficiency, decreased cost for patients and affords more control. Oral sedation is easy to administer, convenient and painless. There is minimal downtime and recovery is quick. Unlike intravenous sedation, an effective level of sedation is not guaranteed, nor can a deeper level of sedation be prevented. Therefore, patient safety must be the prime consideration [25].
Though the level of sedation is independent of the route of administration, the goal of oral sedation is usually to provide minimal sedation along with reduced pain , anxiety and reduced patient-recall of the procedure [27]. According to the American Dental Association (ADA), administration of a drug by enteral route usually provides minimal sedation as long as the maximum FDA-recommended dose is not exceeded. When the maximum recommended dose is exceeded or more than one enteral drug is administered to achieve the desired sedation effect, guidelines for moderate sedation must apply [28].
Dental surgeons frequently perform oral sedation in their office. The ADA has guidelines for minimal sedation stating that patients considered for minimal sedation must be suitably evaluated preoperatively by a thorough history, focused clinical examination, and appropriate consultation with their primary care physician or medical specialist [28]. Informed consent for the proposed sedation must be obtained from the patient, parent or legal guardian. They must also be advised regarding the procedure associated with the delivery of any sedative agents. As it is likely that depth of sedation may increase more than the intended level, an adequate oxygen supply and equipment necessary to deliver oxygen under positive pressure must be available. Preoperative dietary restrictions must be considered, based on the sedative techniques. In addition to the physician/dentist performing the procedure, at least one additional person trained in Basic Life Support for health care providers must be present. The patient must be monitored by either the physician/dentist or an appropriately trained individual under the direction of the physician/dentist during the procedure. Monitoring should include the level of consciousness, pulse oximetry, respiratory rate, chest excursions, blood pressure, and heart rate. There should be full documentation of drugs used, dosage, vital signs monitored, adverse drug reactions, and if applicable emergency measures were employed during the procedure and in the recovery period.
A qualified physician must determine and document satisfactory recovery of the patient prior to discharge from the facility. The patient, parent, legal guardian, or caregiver must receive postoperative instructions.
Safety of Oral Sedation
As the volume of office-based medical and surgical procedures continues to rise, so does the use of oral sedation for these procedures [24, 25]. It is important to ensure that patients receive a level of anesthetic care comparable to that provided in a hospital. There is a lack of studies to show how this affects patient morbidity and mortality [26].
Butz et al. studied the safety of oral sedation in facial aesthetic surgery in an office-based facility from 2008 to 2014. This retrospective analysis studied 199 patients undergoing 283 surgical procedures using oral sedation and local anesthesia. All patients received a combination of diazepam, diphenhydramine and hydrocodone/acetaminophen or acetaminophen alone. Tramadol was substituted as an analgesic in patients with hydrocodone allergy. The investigators reported no major complications related to oral sedation and attributed their success to careful patient selection and appropriate medication and dosage selection. They state that patients with ASA physical status III or greater are not ideal candidates for office-based procedures. Patients with poorly controlled hypertension should not be offered this option. Also, patients with obstructive sleep apnea, history of a difficult airway and prior anesthesia complications should be cleared medically prior to considering the sedation protocol. They also stress the importance of titrating medications to presenting vital signs, avoiding large doses of opioids, liberal use of local anesthetics and that the surgeon must discuss the procedure with the patient at appropriate times and reassure the patient throughout the procedure [25].
Routinely, cataract surgeries are performed as outpatient procedures under topical anesthesia. Successful outcomes require not only precision with surgical techniques but also patient cooperation at appropriate times. Intravenous sedation is commonly used during the procedure. Chen et al., in a prospective randomized control trial compared oral diazepam with intravenous midazolam for conscious sedation during cataract surgery under topical anesthesia. Outcomes studied were undesired movement, pain, anxiety, and the inability of the patient to cooperate with surgeon instructions. There were no significant intraoperative complications in any case. Undesired movement was statistically significant in the IV midazolam group. There was no statistically significant difference in any other primary outcomes between the two groups. Based on this and the relative cost difference, the authors’ institution has changed to oral diazepam as the first-line agent for sedation during cataract surgeries. They do reiterate that careful patient selection is crucial for successful outcomes [27]. Improvements in care can be made with national standardization of care, safety checklists, and development and adherence to professional practice guidelines.
Conclusions
Sedation and analgesia can be provided for ambulatory procedures without exposing the patient to the risks of general anesthesia. However, the incidence of adverse events including death and permanent brain damage in patients receiving MAC for predominantly elective surgeries mostly in outpatient settings were similar to that undergoing general anesthesia. Respiratory depression from oversedation was stated as an important mechanism of injuries [3]. There is always a possibility of unintended escalation of depth of sedation. Different scientific societies have offered guidelines with respect to appropriate patient selection, skilled personnel administering the sedation, appropriate drug selection, careful monitoring of the patient during and immediately after the procedure and regular evaluation of practices with the aim of providing sedation while maintaining patient safety. Implementation of these guidelines into regular practice has the potential to reduce adverse events and improve outcomes in providing sedation and analgesia.
References
Haridas RP. Horace wells’ demonstration of nitrous oxide in Boston. Anesthesiology. 2013;119(5):1014–22.
Apfelbaum JL, Gross JB, Connis RT, Agarkar M, Arnold DE, Coté CJ, Tung A. Practice guidelines for moderate procedural sedation and analgesia 2018: a report by the American Society of Anesthesiologists Task Force on Moderate Procedural Sedation and Analgesia, the American Association of Oral and Maxillofacial Surgeons, American College of Radiology, American Dental Association, American Society of Dentist Anesthesiologists, and Society of Interventional Radiology. Anesthesiology. 2018;128(3):437–79.
Bhananker SM, Posner K, Cheney FW, Caplan RA, Lee LA, Domino KB. Injury and liability associated with monitored anesthesia care: a closed claims analysis. Anesthesiology. 2006;104(2):228–34.
Hug CC Jr. MAC should stand for Maximum Anesthesia Caution, not Minimal Anesthesiology Care. Anesthesiology. 2006;104(2):221–3.
Centers for Medicare & Medicaid Services (CMS). Clarifications of the interpretive guidelines for the anesthesia services condition of participation, and revised hospital anesthesia services interpretive guidelines–State Operations Manual (SOM) Appendix A section 482.52. CMS publication no. 100–07, transmittal 59, effective date May 21, 2010.
Guimaraes ES, Campbell EJ, Richter JM. The safety of nurse-administered procedural sedation compared to anesthesia care in a historical cohort of advanced endoscopy patients. Anesth Analg. 2014;119(2):349–56.
American Society of Anesthesiologists. Statement on granting privileges for administration of moderate sedation to practitioners who are not anesthesia professionals [Internet]2005 Oct 25 [updated 2016 Oct 26; cited 2018 Aug 8]. Available from http://www.asahq.org/quality-and-practice-management/standards-guidelines-and-related-resources/statement-of-granting-privileges-for-administration-of-moderate-sedation-to-practitioners
Apfelbaum JL, Connis RT, Nickinovich DG, Pasternak LR, Arens JF, Caplan RA, Fleisher LA, Flowerdew R, Gold BS, Mayhew JF, Rice LJ. Practice advisory for preanesthesia evaluation: an updated report by the American Society of Anesthesiologists Task Force on Preanesthesia Evaluation. Practice parameter | March 2012. Anesthesiology. 2012;116(3):522–38. https://doi.org/10.1097/ALN.0b013e31823c1067.
Urman RD, Kaye AD, editors. Moderate and deep sedation in clinical practice. 1st ed. Cambridge/New York: Cambridge University Press; 2012.
Kluger MT, Tham EJ, Coleman NA, Runciman WB, Bullock MF. Inadequate pre-operative evaluation and preparation: a review of 197 reports from the Australian incident monitoring study. Anaesthesia. 2000;55(12):1173–8.
Metzner J, Posner KL, Domino KB. The risk and safety of anesthesia at remote locations: the US closed claims analysis. Curr Opin Anaesthesiol. 2009;22(4):502–8.
Ghaffar S, Haverland C, Ramaciotti C, Scott WA, Lemler MS. Sedation for pediatric echocardiography: evaluation of preprocedure fasting guidelines. J Am Soc Echocardiogr. 2002;15(9):980–3.
Green SM, Mason KP, Krauss BS. Pulmonary aspiration during procedural sedation: a comprehensive systematic review. Br J Anaesth. 2017;118(3):344–54.
Beach ML, Cohen DM, Gallagher SM, Cravero JP. Major adverse events and relationship to nil per Os status in pediatric sedation/anesthesia outside the operating room: a report of the Pediatric Sedation Research Consortium. Anesthesiology. 2016;124(1):80–8.
Besi E, Besi C, Lees R, Morrison A, O’Connor N. Comparison of fasting and non-fasting patients receiving intravenous (IV) sedation. Oral Surg. 2018;11(2):98–104.
Conway A, Sutherland J. Depth of anaesthesia monitoring during procedural sedation and analgesia: a systematic review and meta-analysis. Int J Nurs Stud. 2016;63:201–12.
Sheahan CG, Mathews DM. Monitoring and delivery of sedation. Br J Anaesth. 2014;113(Suppl 2):ii37–47.
Hillman DR, Walsh JH, Maddison KJ, Platt PR, Kirkness JP, Noffsinger WJ, Eastwood PR. Evolution of changes in upper airway collapsibility during slow induction of anesthesia with propofol. Anesthesiology. 2009;111(1):63–71.
Deitch K, Miner J. Does end-tidal CO2 monitoring during emergency department procedural sedation and analgesia with propofol decrease the incidence of hypoxic events? A randomized, controlled trial. Ann Emerg Med. 2010;55(3):258–64.
Waugh JB, Epps CA, Khodneva YA. Capnography enhances surveillance of respiratory events during procedural sedation: a meta-analysis. J Clin Anesth. 2011;23(3):189–96.
Rose Bovino L, Brainard C, Beaumier K, Concetti V, Lefurge N, Mittelstadt E, Wilson T, Langhan ML. Use of capnography to optimize procedural sedation in the Emergency Department Pediatric Population. J Emerg Nurs. 2018;44(2):110–6.
Koenig L, Doherty J, Dreyfus J, Xanthopoulos J, Coalition AS. An analysis of recent growth of ambulatory surgery centers. KNG Health Consulting: Rockville; 2009.
Shapiro FE, Punwani N, Rosenberg NM, Valedon A, Twersky R, Urman RD. Office-based anesthesia: safety and outcomes. Anesth Analg. 2014;119:276–85.
Corcuera-Flores JR, Silvestre-Rangil J, Cutando-Soriano A, López-Jiménez J. Current methods of sedation in dental patients – a systematic review of the literature. Med Oral Patol Oral Cir Bucal. 2016;21:e579–86.
Butz DR, Gill KK, Randle J, Kampf N, Few JW. Facial aesthetic surgery: the safe use of oral sedation in an office-based facility. Aesthet Surg J. 2016;36:127–31.
Chen M, Hill GM, Patrianakos TD, Ku ES, Chen ML. Oral diazepam versus intravenous midazolam for conscious sedation during cataract surgery performed using topical anesthesia. J Cataract Refract Surg. 2015;41:415–21.
Baker TJ, Gordon HL. Midazolam (Versed) in ambulatory surgery. Plast Reconstr Surg. 1988;82:244–6.
ADA guidelines for the use of sedation and general anesthesia by dentists. October 2016.
American Society of Anesthesiologists Task Force on Sedation and Analgesia by Non-Anesthesiologists. Practice guidelines for sedation and analgesia by non-anesthesiologists. Anesthesiology. 2002;96:1004–17.
Metzner J, et al. The risk and safety of anesthesia at remote locations: the US closed claims analysis. Curr Opin Anaesthesiol. 2009;22:502–8.
Practice guidelines for preoperative fasting and the use of pharmacologic agents to reduce the risk of pulmonary aspiration: application to healthy patients undergoing elective procedures: an updated report by the American Society of Anesthesiologists Task Force on Preoperative Fasting and the Use of Pharmacologic Agents to Reduce the Risk of Pulmonary Aspiration. Anesthesiology. 2017;126:376–93.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Ritchie-Dabney, R., Parekh, U.R. (2020). Sedation Guidelines. In: Rajan, N. (eds) Manual of Practice Management for Ambulatory Surgery Centers. Springer, Cham. https://doi.org/10.1007/978-3-030-19171-9_14
Download citation
DOI: https://doi.org/10.1007/978-3-030-19171-9_14
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-19170-2
Online ISBN: 978-3-030-19171-9
eBook Packages: MedicineMedicine (R0)