Abstract
Losses caused by the coffee berry borer, Hypothenemus hampei (Ferrari) have been decreasing coffee production. This pest causes annual losses over U.S. $500 million. The chemical insecticides have been used to Coffee berry borer control. Due to their effects of pesticides on human and environmental health, some countries decided to ban the use of endosulfan. Another hand, biological insecticide based on entomopathogenic fungus Beauveria bassiana (Balsamo) Vuillemin became one of the key measures in coffee integrate pest management. Release by spray application requires high concentrations of B. bassiana, and this enhances the cost of CBB control. Thus, developing new environmentally friendly cultivation and cost-efficient methods to manage the coffee berry borer has become essential. In response to this, a novel method for the application of microbial control agents against agricultural pests using bee pollinators has been developed. In this chapter, we discuss the potential use of managed bees as vectors of B. bassiana to coffee berry borer control and challenges.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Coffee is already part of people’s routine life. Around 2.58 billion cups of coffee are daily consumed (Bacon 2005). Most of the coffee produced in the world comes from smallholding, and it is considered the main source of economic resources for many poor families that lives in rural communities (FAO 2015). Nearly five hundred million people are involved on coffee trade, right from the plantation of coffee to final consumption (DaMatta et al. 2007).
Despite high production and demand from consumers, coffee production around the world is strongly affected by disease and pest attacks. Actually, this is considered as one of the primary factors that lead to coffee yield reduction in the main coffee-producing countries (Oliveira et al. 2014). For instance in Brazil, the world’s largest producer of Arabica coffee, annual losses due to pests and diseases are around 0.4 million tons (Oliveira et al. 2014). To compensate losses and to raise agricultural production and productivity, many farmers increase the use of chemical inputs (Wilson and Tisdell 2001). However, this can result in direct and indirect economic losses related to obtaining and using pesticides which can harm the human health and natural enviroment.
The deleterious effects of pesticides on human and environmental health, including wild pollinators, have been discussed in the scientific literature (Fischer and Moriarty 2011; Janssen 2011), in relation to the development of resistance to major coffee diseases and pests like leaf rust (caused by Hemileia vastatrix Berk. and Br) (Silva et al. 2006) and the Coffee Berry Borer, Hypothenemus hampei (Ferrari) (Coleoptera: Curculionidae: Scolytinae) (Brun et al. 1989). Henry and Feola (2013), studying pesticide use among smallholder coffee producers in Jamaica, found that the majority of farmers suffer from at least one health symptom associated with pesticide handling, because safety practices were scarcely adopted. According to them, there was also the risk that other household members and the wider local community be exposed to pesticides. Despite that, the cost related to chemical control associated with this type of management makes clear the need of a new concept in agriculture involving a severe reduction in the use of chemical inputs (Nicolopoulou-Stamati et al. 2016), especially to control pests and diseases in coffee agrosystem. The implementation of environmentally friendly practices which are safer for the environment and human health and biodiversity, and capable of increasing crop yields quantity and quality is necessary to ensure long-term food security and profitability for coffee production.
In the early 1990s, a novel sustainable method for pest control using bees as a vector of microbial control agents against agricultural pests and diseases was proposed (Peng et al. 1992). The Bee vectoring Technology (BVT) used managed bees to deliver microbial control agents to plants against plant pathogens and insect pests of crops (Peng et al. 1992; Kevan et al. 2008). Bee vectoring technology has several advantages over spraying, it requires low amounts of inoculum, decrease the need of external inputs, it reduces the cost and labor-intensive and minimizes non-target organisms exposure (Kevan et al. 2003). This technology combines two complementary ecosystem services, pollination, and pest control, and it might increase the potential for ‘win–win’ scenarios contribute to increasing crop yields, and ensure environmental safety. The most studies about bee vectoring focus in pest and disease that affects the flowers and leaves, few studies reveal the potential of these approaches in pest and disease that affects the fruits directly. Therefore, based in some studies results and a couple of information we believe that BVT can be a technique that contributes to pollination and at the same time with pest control in coffee crops as demonstrated in other crops. In this chapter, we discuss the potential use of managed bees as vectors of microbial agents to coffee berry borer control and challenges.
2 Bee Vectoring Technology (BVT)
Bee vectoring is a technology that uses managed pollinating bees to disperse beneficial microbial agents to flowering plants for the control of insect pests and suppression of plant diseases (Peng et al. 1992; Kevan et al. 2008). This approach is possible due to the interaction between the following components: the crop, the pest (weed, disease, or herbivore), the pollinator (vector), the biocontrol agents, the powdery product, the dispensers, and the security for the environment and the human health (Kevan et al. 2008). The vector is the bee species that has a high rate of flower visitation and deposition capacity of the microbial control agent (MCA) on the target crop. The selection of MCA depends on target crop pest or disease, and it must be safe for bees and the environment. In general, the powdery MCA formulations of a commercial product is often used in BVT approach (Mommaerts and Smagghe 2011). The powdery MCA formulations are often mixed with a carrier or diluted to reduce concentration and maximize the contact with MCA and bee bodies (Kevan et al. 2008; Al-Mazra’awi et al. 2007). Designed dispensers fitted in front of the beehives make possible the contact between bees and MCA. Thus, when bees pass through the control agent provided in dispensers fitted in the beehive entrance, they pick up the inoculum of microbial agents control (fungi, bacteria, and viruses) on their bodies and hairs. Then, when bees visit flowers to collect nectar and pollen and during self-grooming on the leaves of plants, they deposit the inoculum powder on the flowers and leaves of the target crops (Kevan et al. 2008).
Some studies report the success of bee vector technology (Carreck et al. 2007; Mommaerts et al. 2010). Hokkanen et al. (2015) conducted a study in five countries on the management of strawberry grey mold caused by B. cinerea with the biocontrol fungus, Gliocladium catenulatum vectored by honey bees or bumble bees targeting strawberry cultivation in open fields. By the results, under heavy disease pressure bee vectoring provided on average a 47% disease reduction, which was a similar result to multiple fungicide sprays. However, under light disease pressure, biocontrol decreased grey mold by an average of 66%, which was more efficient than fungicide sprays. Other studies found similar results, where the use of bees as vectors of MCA was effective against pest or disease in many crops (Kovach et al. 2000; Maccagnani et al. 2005; Shafir et al. 2006).
3 Coffee Market
Coffee is considered the second most important commodity in the world after oil (Daviron and Ponte 2005). Brazil is the most significant world producer and international trade of coffee, followed by Vietnam and Colombia (FAO 2015). According to the ICO 2016 report, the total consumption of all importing countries was estimated at 104.9 million bags (60 kilograms or 132.276 pounds of coffee). The world consumption in 2015 suggests a steady increase to 152.1 million bags (ICO 2016). The average annual growth rate remains at a healthy 2% over since 2014, highlighted by an increase in consumption in exporting countries. The world’s largest consumers are the European Union and the United States, both demanding around 42 and 24.4 million bags, respectively. The European Union shows an average consumption growth of 0.8% per year since 2012, but the USA continues to show an even more significant increase in coffee consumption by an estimated average rate of 3.2 % (ICO 2016).
4 Coffee Botany
All natural Coffea species are native to tropical and subtropical Africa. The genus Coffea is a member of the family Rubiaceae (Davis et al. 2006). Three species of Coffea are most commercially traded, Coffea arabica, Coffea canephora (commonly known as “robusta” coffee), Coffea liberica (liberica) and var. dewevrei (excelsa) (Davis et al. 2006, Ngo et al. 2011; FAO 2015). Coffea arabica is responsible for approximately 60% of the global coffee production, while the other 40% Coffea canephora (FAO 2015). Coffea liberica and other forms represent an irrelevant proportion of the entire global production (Donald 2004).
The C. arabica species is native to southwestern Ethiopia. Production is successful at elevation ranging of 900–1500 m (Davis et al. 2006). C. canephora originated in the lowlands of equatorial Africa where it grows naturally between (50–)250–1500 m (Davis et al. 2006; DaMatta et al. 2007).
Arabica coffee typically presents one main trunk, and Robusta coffee is typically multi-trunked (Vieira 2008). In both species, the trunks develop above the soil and the plant produces horizontal plagiotropic branches, on which blooming and production occur (Fig. 1) (Vieira 2008). The flowers are produced in inflorescences on the axes of plagiotropic branches (Vieira 2008). The flowers of both species are hermaphrodite, and they have five white petals, an elongated corolla tube (Klein et al. 2003b). There are five stamens, two-branched stigma, and an inferior ovary of two chambers and one ovule per chamber (Klein et al. 2003b). Coffea arabica is allotetraploid, self-fertile and this species does not need cross-pollination. On the other hand, Robusta coffee is diploid and self-sterile (requires cross-pollination). The flower opens in the morning and the stigma is already receptive when anthesis occurs (Free 1993; Klein et al. 2003b). After that, the pollen starts shedding (Ngo et al. 2011).
The flowering phenology and the number of plants blooming per year are influenced by precipitation and region’s latitude (Vieira 2008). The flowering period is stimulated by first rainfall events in the seasons followed by a dry period, and it may result in more than one bloom (Alvim 1985; Vieira 2008). In Brazil blooming occurs during the spring (e.g., from September to December in the main Chapada Diamantina coffee production areas) (Fig. 2).
The fruit of coffee is an ellipsoid drupe, their size vary with the cultivar or variety planted and cultivation conditions (Vieira 2008). In arabica coffee, ripe fruits are red or yellow (Fig. 3), in robusta plants, more hues occur (Vieira 2008). Robusta less susceptible to attacks by pests and disease, produces more berries, and the quality of the beverage is lower when compared to Arabica. (Willson 1999; DaMatta et al. 2007; Reiger 2006; Ngo et al. 2011).
5 Coffee Pollination
Robusta coffee is self-incompatible, and C. arabica is self-fertile and many studies have recorded that wild and managed bees play an important role in pollination of both species (Fig. 4) (Willmer and Stone 1989; Badilla and Ramírez 1991; Raw and Free 1977; Klein et al. 2003a, b; Ricketts 2004; Ngo et al. 2011; Saturni et al. 2016; Nunes 2017; Hipólito et al. 2018). Honey bees and stingless bees are the most abundant flower visitors during mass-flowering (Willmer and Stone 1989; Ngo et al. 2011). Krishnan et al. (2012) have conducted an experiment to compare the contribution of self, wind and insect pollination to fruit set in C. canephora. This author’s reported that the number of flowers that development in fruits was highest when hand cross-pollinated (44%), followed by open- (insect and wind combined; 33%) and wind- (22.1%) pollination treatments. The flowers from open-pollinated treatments received almost the double of pollen grains than wind-pollinated flowers. The pollination provided by bees increased fruit production by 50% in C. canephora. In India, Boreux and collaborators (2013) found that bees contributed significantly to coffee production by increasing the number of berries produced in C. canephora. However, this is related to the initial flower number. The visitation by bees can increase berry production by more than 25%. According to Classen et al. (2014), bees contribute significantly increased fruit weight of coffee by an average of 7.4% in C. arabica. Bagging experiments conducted by Nunes (2017) with C. arabica in Brazil show that the rates of pollen deposition on stigmas and growth of the pollen tube were higher when the flower was visited by Apis mellifera scutellata Lepeletier, 1836 than those by spontaneous self-pollination. Thus, a single visit from A. mellifera contributes to fruit development with weight, height, and width more regular. A recent study on coffee farms of C. arabica in Chapada Diamantina shows similar results, coffee flower visitors improved the yield on average 30% (Hipólito et al. 2018). All results reveal the importance of pollination services providing by management and wild bees to increase the yields.
6 Coffee Pests
The natural characteristic perennial coffee plant (Coffea spp.) facilitates attacks by some insects and diseases (Barrera 2008). The coffee root, trunk, foliage, and berry are susceptible to attack both in plantation and post-harvesting. In most cases, the pests weaken the plant, reducing yield or affecting the quality of grains (Barrera 2008).
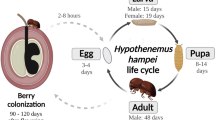
Among the pest that attacks coffee plants, the coffee berry borer (Hypothenemus hampei Ferrari (Coleoptera: Curculionidae: Scolytinae) is the worst pest threatening coffee crop throughout the worldwide (Barrera 2008; Vega et al. 2015). Hypothenemus hampei originating from Africa now is considered cosmopolitan (Barrera 2008). This beetle causes direct damage to the coffee because attacking berries in all development phases, especially those with more than 20% dry matter (Damon 2000). Fruits attacked show a little hole in its apical portion, located at the center or ring of the berry’s ostiole (Barrera 2008; Vega et al. 2015). Usually, injured fruits fall and rot prematurely. All these damages lead to a reduction in yield and affecting bean quality (Barrera 2008). In Brazil, this pest causes annual losses around at US$215–358 million (Oliveira et al. 2013; Infante et al. 2013; Vega et al. 2015).
For some years, the synthetic insecticide Endosulfan (C9H6Cl6O3S) was used in many countries against CBB. The application of the CBB population’s levels up to 80% (Aristizábal et al. 2016). Despite the ease of application in the field and insecticidal efficacy, the misuse of Endosulfan resulted in indirect economic losses, leading to social and environmental consequences (Lubick 2010; Infante 2018). Due to the effects of pesticides on human and environmental health, some countries have banned the use of endosulfan (Janssen 2011). Since Brazil forbade the use of Endosulfan, the infestation levels of CBB have reached alarming levels (Brazil 2015). Alternatively, another insecticides such as pirimiphos-methyl, fenitrothion, chlorpyrifos, and fenthion, have been used with success against the CBB (Bustillo-Pardey 2002). A variety of strategies have been proposed to reduce the infestation levels of CBB (Vega et al. 2015; Infante 2018). Many studies revealed the efficacy of adopting the Integrated Pest Management (IPM) methods to control CBB (Aristizábal 2005; Benavides et al. 2012). Infante (2018) summarized several additional methods.
Among the techniques used in IPM, the biological control with the entomopathogenic fungus Beauveria bassiana plays a major role in controlling CBB. This fungus is considered a natural controller of CBB because it is found infecting the H. hampei in all coffee plantations where CBB has arrived (Benavides et al. 2012). Beauveria bassiana is considered as an environmentally safe bioinsecticide, no deleterious effects on humans and the environment and has a low impact on non-target organisms including CBB natural enemies (Zimmermann 2007; Aristizábal et al. 2016). This fungus attack their host insects usually percutaneously (Zimmermann 2007).
The use of B. bassiana for CBB control is carried out through one or more flood applications of large numbers of aerial conidia in dry or liquid formulation (Mascarin and Jaronski 2016). The inundative application is performed using traditional spray methods and recently by autoinoculation traps (Mota et al. 2017). Despite that the autoinoculation trap provided high levels of H. hampei mortality in the field, the traps only attract a small amount portion of the insects in the field (Mota et al. 2017; Infante 2018). The efficacy of autoinoculation traps at long-term control of H. hampei and the cost-benefit of this strategy need to be investigated.
In inundative applications of B. bassiana by spray application, a high concentration of conidia ranging from 1× 1011 to 1 × 1012 conidia/ha in aqueous suspension has been used (Benavides et al. 2012; Mascarin and Jaronski 2016; Nakai and Lacey 2017). As summarized for Nakai and Lacey (2017) the mortalities rates of CBB by spray application of B. bassiana in fields trials ranged from 10% to 90%. A variety of factors influence the sucess of B. bassiana against CBB, such as the temperature, altitude, humidity, formulation, application equipment, strain, concentration, virulence, infestation level and location of CBB (inside or outside of fruit) (Mascarin and Jaronski 2016; Nakai and Lacey 2017).
According to Vega et al. (2015) most of the studies about mortality rates of CBB by spray application of B. bassiana in fields trials do not include the cost-benefit analysis. In the field, high concentrations of B. bassiana are spread by spray application, and this increases the cost of CBB control (Benavides et al. 2012). Besides that, spray applications cause negative impact on conidia viability of the microbial control agent (Nilsson and Gripwall 1999). This method requires ready access and much water throughout the plantation, labor work, and machinery (Vega et al. 2015). Thus, it is essential to develop cost-effective and low impact practices for B. bassiana field application on coffee. Below we discuss how the adoption of the Bee Vectoring Technology can improve the efficacy to delivery B. bassiana spores against CBB, and improve the initial, maturation and harvest fruit set.
7 Can Pollinators Help to Control Diseases/Pests on Coffee?
The close relationship between coffee and bees has been described above. Among coffee flower visitors, the honeybee is the most frequently reported one in the literature as an important pollinator for C. arabica and C. canephora. Overall, the terms “increased production”, “the most dominant visitor”, “the most frequent flower visitor”, “the primary pollinators” and “important pollinator” are frequently cited in studies that investigated the role of honey bees to improve coffee yields in many sites around the world (Roubik 2002a, 2002b; Ricketts 2004; Veddeler et al. 2006; Bos et al. 2007; Vergara et al. 2008; Ngo et al. 2011). Raw and Free (1977) reported that coffee brushes caged with honeybees showed higher yields of berries in C. arabica. In C. canephora, Klein et al. (2003a) and Krishnan et al. 2012 concluded that Apis spp., not only A. mellifera, are the most common visitors to coffee flowers.
As mentioned above, honeybees visit the coffee crop efficiently. Their interactions with the coffee plant covers one crucial assumption for BVT success: the close vector-plant interactions. Moreover, honeybees have a large foraging range (up to 3 km in radius) facilitating the spreading of biocontrol agents in large areas (Mommaerts and Smagghe 2011; Abou-Shaara 2014). Thus, honeybees have the needed requirements to be employed as a vector for disseminating microbial agents control on coffee crops.
Honey bees have been used in many studies to investigate their ability to disseminate some microbial control agents in both greenhouse conditions and open field cultivation (Peng et al. 1992; Butt et al. 1998; Carreck et al. 2007; Johnson et al. 1993). In the study of Dedej et al. (2004) using honey bees as a vector of the bacterium Bacillus subtilis against mummy berry disease incidence in flower infection by Monilinia vaccinii-corymbosa, they found that bee-vectored agent Serenade reduced the incidence of mummy berry disease. Combining the results available in the literature on the success of honey bees for coffee pollination and vectoring of other crops we believe that the honey bee has a high potential as a vector of microbial agents control against the pest and disease in coffee.
BVT also requires that microbial control agents need to be safe to pollinator/vector. Regarding coffee pests, the entomopathogenic fungus Beauveria bassiana can be used for dissemination by honeybees for coffee berry borer control. Several studies reported the effects of B. bassiana on A. mellifera (Alves et al. 1996; Al-Mazra’awi et al. 2007; Meikle et al. 2008). These effects are conditioned to conidia concentration of B. bassiana, the strain and the types of exposition (Al-Mazra’awi et al. 2007; Potrich et al. 2018). It is necessary to quantify the effects of this fungus on the honeybees before the trial to better understand the optimal concentration of B. bassiana that poses the least for this vector and causes high mortality of CBB.
Some studies demonstrate the efficacy of honeybees to vectoring B. bassiana (at rates 1 ×109 conidia/g) against some pests. The potential of dissemination of B. bassiana by honeybees for control of Tarnished Plant Bug Lygus lineolaris (Al-Mazra’awi et al. 2007; Palisot de Beauvois) on canola was investigated by Al-Mazra’awi et al. (2006). The bees effectively vectored the inoculum from the hives to the crop and these results indicated that bees might provide a novel means for applying B. bassiana to manage L. lineolaris in canola. According to these authors, the benefits are better pollination, reduction in pest pressure of L. lineolaris, and reduced reliance on insecticides.
The results mentioned above show the capacity of honey bees to spread the fungus B. bassiana to many crops around the world against some pests. These results refer to pests and diseases that attack the flowers. It is surprising that one fundamental question remains unanswered: how bees can help control a pest that has a cryptic life? The process that leads to bee-vectoring B. bassiana to infecting the CBB remains unclear. Almost all life cycle of the H. hampei occurs inside of the coffee berry which difficult their control (Barrera 2008). In the field, post-harvest fallen berries in the ground are a source of new infestations because they are reservoirs for adult insects and larvae (Castaño et al. 2005; Benavides et al. 2012). Few months after plants are blooming, when conditions are appropriate occurs the massive adult emergence of the old coffee berries (Barrera 2008). Those adults mate with their siblings and fly, repeating the entire cycle (Benavides et al. 2012). According to Cure et al. (1998), control measures need to be carried out between the end of harvesting and the appearance of the first fruits of the early maturation of the crop. Generally, B. bassiana is applied when female H. hampei are just starting to penetrate the berries at the beginning of the year or in fallen berries on the ground (Damon 2000; Aristizábal et al. 2016). According to Alves (1998), insect vectors are essential to inoculation and infection of others insects that live in sheltered places as CBB, because the former insect is capable of dispersing the fungus across the farm.
Bees spread the inoculum on the flowers and leaves of the crop, maybe in the soil too. A coffee stand has one or more blooming periods, and sometimes the vectorization of the fungus by bees can happen more than once. These repeated applications might increase the natural population of B. bassiana in an agrosystem. The adults of CBB are infected by bee-vectored B. bassiana through: (1) fallen berries contaminated with bee-vectored biocontrol agent; (2) Other insects visiting the coffee plants and then disseminating B. bassiana between host insects; (3) Alternative hosts may be infected and produce spores that also infect the CBB; (4) by wind currents.
Ureña and Chuncho (2008) investigated the ability of honey bees to deliver B. bassiana to coffee crops targeting H. hampei. Their results show that honey bees vectoring of B. bassiana spores can provide a coffee berry protection against berry borer infestation in coffee fields. The inoculum of B. bassiana used in the experiment had a concentration at 6.5 × 1010 colony forming units (CFU) per gram. The average percentages of infested berries with B. bassiana infection in field trial increased after the fungal dispersion by bees, but the inoculum distribution was not homogeneous. Ureña and Chuncho (2008) also found that when bees vectored the fung, the average number of infested berries with B. bassiana infection was 43%, exceeding that provided by spray fungal suspension (23–30%). In some sample plots, when bees vectored the fung, the population of B. bassiana increased and reduced the population of coffee berry borer. Moreover, the bees vectored B. bassiana spores at a distance up to 200 m from the hives. According to Ureña and Chuncho (2008), an apiary of 4 bee hives can cover 12.5 ha of coffee homogeneously.
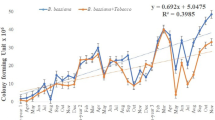
In Brazil (Macedo, personal communication, December 25, 2017), some experiments are developing (Fig. 4). Honey bee hives, A. mellifera scutellata, were used during the experiment. Dispensers similar to those used by Peng et al. (1992) were used in field trials (Fig. 5). Preliminary results show that honey bees can deliver B. bassiana spores to coffee at a distance up to 350 meters from hives (Fig. 6). The low percentage of fungal colony forming units (CFU) of B. bassiana were observed on the leaves and flowers sampled in a field trial. The low amount of conidia on coffee flowers and leaves (Fig. 7) might be attributed to rain during the field trial. Overall, the results show that that microbial biocontrol can be vectored at long distances by bees into coffee fields and the dissemination of B. basssiana spores by bees during blooming can contribute to the regulation of CBB populations. As a result, rise the CBB control, and increase the fruit set by pollination service, and protect the coffee berries during maturation/ripening yield.
The inoculum dispenser (see Peng et al. 1992). The dispenser is loaded with inoculum (Beauveria bassiana + Vectorite) and attached to the beehive in field trial in coffee crop (Chapada Diamantina-Brazil) (Photo: Juliana Macedo)
8 Challenges
Although the evidence compiled in this chapter points out that using BVT on coffee can be considered a suitable management method in pest control, there are many gaps to explore in this field of science in order to better understand the multitrophic relationships (between agent and vectors), and dynamics of this practice.
As mentioned above, the use of bees as a vector of biocontrol agents for crop protection is possible. The bees can disseminate the inoculum to flowers and leaves, but we need to assess the distribution, deposition of bee-vectored B. bassiana in coffee plants a long time. This helps to estimate the persistence and recovery of this fungus in the coffee plants and environment. The development of research is essential to study the efficacy of mixing strains of B. bassiana with other agents (e.g. Metarhizium anisopliae) for bee-vectoring and their effects on the health of bees. This can help improve CBB control.
Coffee plantations around the world are grown under a wide range of conditions (e.g., shade levels and sun light). These conditions can affect the viability of the control agents used (positively or negatively), as well as the foraging of bees, and this topic need further investigation. In unshaded coffee production, sunlight and warmth affect the post-application persistence of Beauveria bassiana. Spore shelf-life and longevity need to be improved to enhance their persistence in the field. The implementation of BVT in different spatial and temporal scales, as well as landscapes effects on the effectiveness of BVT use, and different management techniques, also need to be investigated. The evidence compiled in this chapter points out that BVT can be considered a suitable management method to coffee IPM.
9 Final Considerations
The impacts of intensive agriculture are clear. Thus, a new approach like BVT is necessary. BVT increases pest control efficiency and crop productivity. This approach should be employed as a part of the Integrated Pest Management (IPM) combined with other non-chemical pest control methods for coffee berry borer control. Some years ago, BVT could be considered only as a management perspective, but nowadays BVT is a reliable method for pest control in some crops (apple, strawberry, canola), and has a great potential for coffee plantations.
References
Abou-Shaara HF (2014) The foraging behaviour of honey bees, Apis mellifera: a review. Vet Med 59:1–10
Al-Mazra’awi MS (2007) Impact of the entomopathogenic fungus Beauveria bassiana on the honey bee, Apis mellifera (Hymenoptera: Apidae). World J Agric Sci 3(1):7–11
Al-Mazra’awi MS, Shipp JL, Broadbent AB, Kevan PG (2006) Dissemination of Beauveria bassiana by honey bees (Hymenoptera: Apidae) for control of tarnished plant bug (Hemiptera: Miridae) on canola. Environ Entomol 35(6):1569–1577
Al-Mazra’awi MS, Kevan PG, Shipp L (2007) Development of Beauveria bassiana dry formulation for vectoring by honey bees Apis mellifera (Hymenoptera: Apidae) to the flowers of crops for pest control. Biocontrol Sci Tech 17(7):733–741
Alves SB (1998) Fungos entomopatogênicos. In: Alves SB (ed) Controle microbiano de insetos. FEALQ. Piracicaba, Brazil, pp 289–381
Alves SB, Marchini LC, Pereira RM, Baumgratz LL (1996) Effects of some insect pathogens on the Africanized honey bee, Apis mellifera L. (Hym., Apidae). J Appl Entomol 120:559–564
Alvim PT (1985) Coffee. In: Halevy AH (ed) Handbook of flowering, vol 2. CRC Press, Boca Raton, pp 308–316
Aristizábal LF (2005) Investigación participativa en el manejo integrado de la broca del café. In: Memorias XXXII congreso sociedad colombiana de entomología. Socolen, Ibagué, pp 27–29
Aristizábal LF, Bustillo AE, Arthurs SP (2016) Integrated pest management of coffee berry borer: strategies from Latin America that could be useful for coffee farmers in Hawaii. Insects 7:E6
Bacon C (2005) Confronting the coffee crisis: can fair trade, organic and specialty coffees reduce small-scale farmer vulnerability in northern Nicaragua? World Dev 33(3):497–511. https://doi.org/10.1016/j.worlddev.2004.10.002
Badilla F, Ramírez BW (1991) Polinización de café por Apis mellifera L. y otros insectos en Costa Rica. Turrialba 41(3):285–288
Barrera JF (2008) Coffee pests and their management. In: Encyclopedia of entomology. Springer Netherlands, Dordrecht, pp 961–998
Benavides P, Góngora C, Bustillo A (2012) IPM program to control coffee berry borer Hypothenemus hampei, with emphasis on highly pathogenic mixed strains of Beauveria bassiana, to overcome insecticide resistance in Colombia. In: Perveen F (ed) Insecticides – advances in integrated pest management. InTech, Rijeka, pp 511–540. https://doi.org/10.5772/28740
Boreux V, Kushalappa CG, Vaast P, Ghazoul J (2013) Interactive effects among ecosystem services and management practices on crop production: pollination in coffee agroforestry systems. Proc Natl Acad Sci 110(21):8387–8392
Bos MM, Veddeler D, Bogdanski AK, Klein AM, Tscharntke T, Steffan-Dewenter I, Tylianakis JM (2007) Caveats to quantifying ecosystem services: fruit abortion blurs benefits from crop pollination. Ecol Appl 17(6):1841–1849
Brazil (2015) Ministério da Agricultura, Pecuária e Abastecimento. Portarias no 11 e 12 de 23 de janeiro de 2015. Diário Oficial da República Federativa do Brasil, Brasília, 26 Jan. 2015. Seção 1. http://pesquisa.in.gov.br/imprensa/servlet/INPDFViewer?jornal=1&pagina=2&data=26/01/2015&captchafield=firistAccess. Accessed 17 Jan 2018
Brun LO, Marcillaud C, Gaudichon V, Suckling DM (1989) Endosulfan resistance in Hypothenemus hampei (Coleoptera: Scolytidae) in New Caledonia. J Econ Entomol 82(5):1312–1316
Bustillo-Pardey AE (2002) El manejo de cafetales y su relacion con el control de la broca del café en Colombia. Boletin Técnico Cenicafé 24:40
Butt TM, Carreck NL, Ibrahim L, Williams IH (1998) Honey bee-mediated infection of pollen beetle (Meligethes aeneus Fab.) by the insect-pathogenic fungus, Metarhizium anisopliae. Biocontrol Sci Tech 8:533–538
Carreck NL, Butt TM, Clark SJ, Ibrahim L, Isger EA, Pell JK, Williams IH (2007) Honey bees can disseminate a microbial control agent to more than one inflorescence pest of oilseed rape. Biocontrol Sci Tech 17(2):179–191
Castaño A, Benavides P, Baker PS (2005) Dispersión de Hypothenemus hampei en cafetales zoqueados. Revista Cenicafe (Colombia) 56(2):142–150
Classen A, Peters MK, Ferger SW, Helbig-Bonitz M, Schmack JM, Maassen G, Steffan-Dewenter I (2014) Complementary ecosystem services provided by pest predators and pollinators increase quantity and quality of coffee yields. Proc R Soc Lond B Biol Sci 281(1779):20133148
Cure JR, Santos RHS, Moraes JC, Vilela EF, Gutierrez AP (1998) Phenology and population dynamics of the coffee berry borer Hypothenemus hampei (Ferr.) in relation to the phenological stages of the berry. Anais da Sociedade Entomológica do Brasil 27:325–335
DaMatta FM, Ronch CP, Maestri M, Barros RS (2007) Ecophysiology of coffee growth and production. Braz J Plant Physiol 19(4):485–510
Damon A (2000) A review of the biology and control of the coffee berry borer, Hypothenemus hampei (Coleoptera: Scolytidae). Bull Entomol Res 90:453–465
Daviron B, Ponte S (2005) The coffee paradox: global markets, commodity trade and the elusive promise of development. Zed Books, London
Davis AP, Govaerts R, Bridson DM, Stoffelen P (2006) Annotated taxonomic conspectus of the genus Coffea (Rubiaceae). Bot J Linn Soc 152(4):465–512. https://doi.org/10.1111/j.1095-8339.2006.00584.x
Dedej S, Delaplane KS, Scherm H (2004) Effectiveness of honey bees in delivering the biocontrol agent Bacillus subtilis to blueberry flowers to suppress mummy berry disease. Biol Control 31:422–427
Donald PF (2004) Biodiversity impacts of some agricultural commodity production systems. Conserv Biol 18(1):17–38. https://doi.org/10.1111/j.1523-1739.2004.01803.x
FAO (2015) Statistical pocketbook coffee. http://www.fao.org/3/a-i4985e.pdf
Fischer D, Moriarty T (2011) Pesticide risk assessment for pollinators: summary of a SETAC Pellston Workshop. Society of Environmental Toxicology and Chemistry (SETAC), Pensacola
Free JB (1993) Insect pollination of crops, 2nd edn. Academic, London
Henry D, Feola G (2013) Pesticide-handling practices of smallholder coffee farmers in eastern Jamaica. J Agric Rural Dev Trop Subtrop 114(1):59–67
Hipólito J, Boscolo D, Viana BF (2018) Landscape and crop management strategies to conserve pollination services and increase yields in tropical coffee farms. Agric Ecosyst Environ 256:218–225
Hokkanen HM, Menzler-Hokkanen I, Lahdenpera ML (2015) Managing bees for delivering biological control agents and improved pollination in berry and fruit cultivation. Sustain Agric Res 4(3):89
Infante F (2018) Pest management strategies against the coffee berry borer (Coleoptera: Curculionidae: Scolytinae). J Agric Food Chem 66(21):5275–5280
Infante F, Castillo A, Pérez J, Vega FE (2013) Field-cage evaluation of the parasitoid Phymastichus coffea as a natural enemy of the coffee berry borer, Hypothenemus hampei. Biol Control 67(3):446–450
ICO (International Coffee Organization) (2016) Coffee Market Report – February 2016. Available from: http://www.ico.org/documents/cy2015-16/cmr-0216-e.pdf
Janssen MPM (2011) Endosulfan. A closer look at the arguments against a worldwide phase out. RIVM letter report 601356002/2011, National Institute for Public Health and the Environment. Ministry of Health, Welfare and Sport: Bilthoven, The Netherlands
Johnson KB, Stockwell VO, Mclaughlin RJ (1993) Effect of antagonistic bacteria on the establishment of honey bee-dispersed Erwinia amylovora in pear blossoms and on fire blight control. Phytopathology 83:995–1002
Kevan PG, Sutton JC, Tam L, Boland G, Broadbent B, Thomson SV, Brewer GJ (2003, January) Using pollinators to deliver biological control agents against crop pests. In: Pesticide formulations and delivery systems: meeting the challenges of the current crop protection industry. ASTM International, West Conshohocken
Kevan PG, Kapongo J-P, Al-mazra’awi M, Shipp L (2008) Honey bees, bumble bees, and biocontrol: new alliances between old friends. In: James RR, PittsSinger TL (eds) Bee pollination in agricultural ecosystems. Oxford University Press, New York, pp 65–79
Klein AM, Steffan-Dewenter I, Tscharntke T (2003a) Pollination of Coffea canephora in relation to local and regional agroforestry management. J Appl Ecol 40(5):837–845. https://doi.org/10.1046/j.1365-2664.2003.00847.x
Klein AM, Steffan-Dewenter I, Tscharntke T (2003b) Bee pollination and fruit set of Coffea arabica and C. canephora (Rubiaceae). Am J Bot 90(1):153–157. https://doi.org/10.3732/ajb.90.1.153
Kovach J, Petzoldt R, Harman GE (2000) Use of honeybees and bumble bees to disseminate Trichoderma harzianum 1295–22 to strawberries for Botrytis control. Biol Control 18:235–242
Krishnan S, Kushalappa CG, Shaanker RU, Ghazoul J (2012) Status dos polinizadores e sua eficiência no café em um mosaico de paisagem fragmentada no sul da Índia. Ecologia Básica e Aplicada 13(3):277–285
Lubick N (2010) Endosulfan’s exit: US EPA pesticide review leads to a ban
Maccagnani B, Mocioni M, Ladurner E, Gullino ML, Maini S (2005) Investigation of hive mounted devices for the dissemination of microbiological preparations by Bombus terrestris. Bull Insectol 58:3–8
Mascarin GM, Jaronski ST (2016) The production and uses of Beauveria bassiana as a microbial insecticide. World J Microbiol Biotechnol 32(11):177
Meikle WG, Mercadier G, Holst N, Girod V (2008) Impact of two treatments of a formulation of Beauveria bassiana (Deuteromycota: Hyphomycetes) conidia on Varroa mites (Acari: Varroidae) and on honeybee (Hymenoptera: Apidae) colony health. Exp Appl Acarol 46:105–117
Mommaerts V, Smagghe G (2011) Entomovectoring in plant protection. Arthropod-Plant Interact 5:81–95
Mommaerts V, Put K, Vandeven J, Jans K, Sterk G, Hoffmann L, Smagghe G (2010) Development of a new dispenser for bumblebees and evaluation to disseminate microbiological control agents in strawberry in the greenhouse. Pest Manag Sci 66:1199–1207
Mota LHC, Silva WD, Sermarini RA, Demétrio CGB, Bento JMS, Delalibera Jr I (2017) Autoinoculation trap for management of Hypothenemus hampei (Ferrari) with Beauveria bassiana (Bals.) in coffee crops. Biol Control 111:32–39
Nakai M, Lacey L (2017) Microbial control of insect pests of tea and coffee. In: Burges HD, Hussey NW (eds) Microbial control of insect and mite pests from theory to practices. Academic, New York, p 275
Ngo HT, Mojica AC, Packer L (2011) Coffee plant–pollinator interactions: a review. Can J Zool 89(8):647–660
Nicolopoulou-Stamati P, Maipas S, Kotampasi C, Stamatis P, Hens L (2016) Chemical pesticides and human health: the urgent need for a new concept in agriculture. Front Public Health 4:148. https://doi.org/10.3389/fpubh.2016.00148
Nilsson U, Gripwall E (1999) Influence of application technique on the viability of the biological control agents Verticillium lecanii and Stenernema feltiae. Crop Prot 18:53–59
Nunes DO (2017) Polinização de Coffea arabica: testando a efetividade e eficiência de Apis mellifera. Trabalho de Conclusão de curso- Instituto de Biologia, Universidade Federal da Bahia
Oliveira CM, Auad AM, Mendes SM, Frizzas MR (2013) Economic impact of exotic insect pests in Brazilian agriculture. J Appl Entomol 137:1–15
Oliveira CM, Auad AM, Mendes SM, Frizzas MR (2014) Crop losses and the economic impact of insect pests on Brazilian agriculture. Crop Prot 56:50–54
Peng G, Sutton JC, Kevan PG (1992) Effectiveness of honeybees for applying the biocontrol agent Gliocladium rosea to strawberry flowers to suppress Botrytis cinerea. Can J Plant Pathol 14:117–129
Potrich M, da Silva RT, Maia FM, Lozano ER, Rossi RM, Colombo FC et al (2018) Effect of entomopathogens on Africanized Apis mellifera L.(Hymenoptera: Apidae). Rev Bras Entomol 62(1):23–28
Raw A, Free JB (1977) The pollination of coffee (Coffea arabica) by honeybees. Trop Agric 54:365–370
Reiger M (2006) Introduction to fruit crops. Hawthorn Press Inc, New York
Ricketts TH (2004) Tropical forest fragments enhance pollinator activity in nearby coffee crops. Conserv Biol 18(5):1262–1271. https://doi.org/10.1111/j.1523-1739.2004.00227.x
Roubik DW (2002a) African honeybees augment neotropical coffee yield. In: Kevan PG, Imperatriz-Fonseca VL (eds) Pollinating bees: the conservation link between agriculture and nature. Ministry of Environment, Brasilia, pp 255–266
Roubik DW (2002b) The value of bees to the coffee harvest. Nature (London), 417(6890): 708. https://doi.org/10.1038/417708a. PMID: 12066176
Saturni FT, Jaffé R, Metzger JP (2016) Landscape structure influences bee community and coffee pollination at different spatial scales. Agric Ecosyst Environ 235:1–12. https://doi.org/10.1016/j.agee.2016.10.008
Shafir S, Dag A, Bilu A, Abu-Toamy M, Elad Y (2006) Honeybee dispersal of the biocontrol agent Trichoderma harzianum T39: effectiveness in suppressing Botrytis cinerea on strawberry under field conditions. Eur J Plant Pathol 116:119–128
Silva MDC, Varzea V, Guerra-Guimaraes L, Azinheira HG, Fernandez D, Petitot AS et al (2006) Coffee resistance to the main diseases: leaf rust and coffee berry disease. Braz J Plant Physiol 18(1):119–147
Ureña J, Chuncho CGM (2008) Utilización de abejas para la dispersión de Beauveria bassiana en el control biológico de la broca del café. Naturaleza y Desarrollo Agrario. Revista Científica del Área Agropecuaria y de Recursos Naturales Renovables, Universidad Nacional de Loja, Loja, Ecuador 1(1):30–37
Veddeler D, Klein AM, Tscharntke T (2006) Contrasting responses of bee communities to coffee flowering at different spatial scales. Oikos 112(3):594–601. https://doi.org/10.1111/j.0030-1299.2006.14111.x
Vega FE, Infante F, Johnson AJ (2015) The genus Hypothenemus, with emphasis on H. hampei, the coffee berry borer. In: Vega FE, Hoffstetter RW (eds) Bark beetles: biology and ecology of native and invasive species. Academic, San Diego, pp 427–494
Vergara C, Contreras J, Ferrari R, Paredes J (2008) Polinización entomófila. In: Manson RH, Hernandez-Ortiz V, Gallina S, Mehltreter K (eds) Agroecosistemas cafetaleros de Veracruz: biodiversidad, manejo y conservación. Instituto de Ecología A.C. (INECOL) e Instituto Nacional de Ecología, México
Vieira HD (2008) Chapter 1. Coffee: the plant and its cultivation. In: Souza RM (ed) Plant–parasitic nematodes of coffee. Springer Science, Dordrecht, pp 3–18
Willmer PG, Stone GN (1989) Incidence of entomophilous pollination of lowland coffee (Coffea canephora): the role of leaf cutter bees in Papua New Guinea. Entomol Exp Appl 50:113–124
Willson KC (1999) Coffee, cocoa and tea. CABI Publishing, Cambridge
Wilson C, Tisdell C (2001) Why farmers continue to use pesticides despite environmental, health and sustainability costs. Ecol Econ 39:449–462
Zimmermann G (2007) Review on safety of the entomopathogenic fungi Beauveria bassiana and Beauveria brongniartii. Biocontrol Sci Tech 17(6):553–596
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Macedo, J., Viana, B., Freitas, B., Medeiros, A., Kevan, P.G., Vergara, C.H. (2020). The Potential of Bee Vectoring on Coffee in Brazil. In: Smagghe, G., Boecking, O., Maccagnani, B., Mänd, M., Kevan, P. (eds) Entomovectoring for Precision Biocontrol and Enhanced Pollination of Crops. Springer, Cham. https://doi.org/10.1007/978-3-030-18917-4_10
Download citation
DOI: https://doi.org/10.1007/978-3-030-18917-4_10
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-18916-7
Online ISBN: 978-3-030-18917-4
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)