Abstract
During the twentieth century, the overall life expectancy of the human population is rapidly increasing worldwide. The global share of older people (aged 60 years or over) increased from 9.2% in 1990 to 11.7% in 2013, and it is predicted that by 2050, it will reach 21.1% of the world population, with 392 million persons aged 80 years or over, more than three times the present [1]. Aging is associated with a progressive loss of tissue and organ function over time [2]. With aging, there is an increased risk of unfavorable changes in body composition, including a decrease in muscle and an increase in fat mass [3].
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
During the twentieth century, the overall life expectancy of the human population is rapidly increasing worldwide. The global share of older people (aged 60 years or over) increased from 9.2% in 1990 to 11.7% in 2013, and it is predicted that by 2050, it will reach 21.1% of the world population, with 392 million persons aged 80 years or over, more than three times the present [1]. Aging is associated with a progressive loss of tissue and organ function over time [2]. With aging, there is an increased risk of unfavorable changes in body composition, including a decrease in muscle and an increase in fat mass [3].
27.1 Obesity
Overweight and obesity represent worldwide phenomena, which are associated with a risk to develop several chronic diseases such as type 2 diabetes and metabolic syndrome, cancer, rheumatoid arthritis and osteoarthritis, cognitive impairment and dementia, gallbladder disease, and those affecting the cardiovascular system [4, 5]. Besides, both obesity and aging impose various functional limitations on the human body, resulting in a severe burden on quality of life [6], and it is estimated that a 2.3% of global disability-adjusted life years are caused by overweight or obesity [7].
With increasing longevity, the proportion of postmenopausal women is also on the rise. Obesity and central adiposity are major health problems during the postmenopausal years. Estrogen deficiency linked to the menopausal transition has been shown to be associated with the increase in visceral fat mass and waist to hip ratio changing from a gynoid to an android body fat distribution pattern, where fat accumulates on the upper portion of the abdomen instead of the hips [8, 9]. However other authors have reported that chronological aging but not menopause was associated with the increase in weight and waist circumference during the fifth and sixth decades of life, while menopausal status was not associated with these changes [10]. As well as hormonal changes and aging, weight gain at midlife has been also attributed to the reduction in energy expenditure of women who have undergone menopause compared with premenopausal women, mainly as a result of the reduction in physical activity [11].
Body mass index (BMI) is the most commonly employed instrument to assess overweight and obesity in global population-based adult research and is assessed by dividing an individual’s weight (kg) by her height (m2). A BMI < 25 kg/m2 indicates normal weight, 25 ≤ BMI < 30 kg/m2 overweight, and BMI ≥ 30 kg/m2 obesity [12]. However, several issues regarding BMI as a tool for obesity assessment have been reported. Its validity to assess obesity status in older adults has been recently questioned [13, 14], especially in older women due to those menopause-related changes in body composition and height decrease associated with kyphosis, shortening of the spinal vertebrae, or thinning of weight-bearing cartilage [15], which may result in BMI overestimation, causing weight category misclassifications. Moreover, BMI has little correlation with adipose mass, which in the second half of life tends to become troncular. To this respect waist circumference has been used to assess abdominal obesity. Overweight status and obesity have been reliably linked to coronary artery disease, congestive heart failure, and gallbladder disease using waist circumference to determine overweight and obese status [16]. Waist circumference of 102 cm and over for men and of 88 cm and over for women has been described to assess abdominal obesity [17].
BMI also fails to differentiate between lean and fat tissue. Imaging techniques such as magnetic resonance imaging (MRI) or computed tomography (CT) are the gold standard for evaluating the distribution of body fat, but the high cost and low availability make it difficult to use in large population studies. In clinical practice, dual-energy X-ray absorptiometry (DEXA) and bioelectrical impedance analysis (BIA) are the most frequently used methods to assess body composition and calculate body fat percentage. When DEXA was employed for body composition assessment, a body fat percentage of more than 28% and 40% of body fat percent for elderly men and women, respectively, have been used to define obesity [18]. A cutoff point of 27% and 38% of body fat percentage for elderly men and women, respectively, has been described to determine obesity with BIA [19].
27.2 Sarcopenia
One of the dramatic changes associated with human aging is the progressive decline of skeletal muscle mass. As the human body ages, the skeletal muscle mass declines annually approximately by 0.1–0.5% starting from age 30, and this gradual decrease, which has been suggested to be approximately 6% per decade after midlife, is accompanied by a simultaneous reduction of strength [20, 21]. There are significant differences among individuals in peak muscle mass, the age at which muscle loss begins, and the amount of muscle that is lost over time [22]. In women there a sharp decline in muscle mass after menopause has been described [23].
The term “sarcopenia” was first proposed by Rosenberg in 1989 to describe the decline of muscle mass associated with aging [24]. Since then, sarcopenia has been defined as the loss of skeletal muscle mass and strength that occurs with advancing age [25].
In the last 20 years, several different definitions of sarcopenia have been proposed, but no consensus has been reached. For instance, Baumgartner et al. [26] used a measure of relative muscle mass, since absolute muscle mass is correlated strongly with height and thus calculated an index of relative skeletal muscle mass as appendicular skeletal muscle mass (kg)/height2 (m2). They defined sarcopenia as the index of relative skeletal muscle mass being less than two standard deviations below the mean of a young reference group. On the other hand, Janssen et al. [27] converted absolute skeletal muscle mass (kg) to percentage skeletal muscle mass (muscle mass/body mass × 100) and termed the skeletal muscle index. They defined class I and II sarcopenia when values were within 1–2 and <2 standard deviations of young adult values.
Since then, muscle strength and physical performance have incorporated to muscle mass in the criteria for sarcopenia diagnosis. In 2010, the European Working Group on Sarcopenia in Older People (EWGSOP) defined sarcopenia as “a syndrome characterized by progressive and generalized loss of skeletal muscle mass and strength with a risk of adverse outcomes such as physical disability, poor quality of life and death” [28]. Therefore, diagnostic criteria included low muscle mass (presarcopenia) together with low muscle strength or physical function (sarcopenia). The presence of the three diagnostic criteria refers to severe sarcopenia. Several groups such as the Foundation for the National Institutes of Health (FNIH) Sarcopenia Project [29]; International Working Group (IWG) [30]; Society of Sarcopenia, Cachexia, and Wasting Disorders (SCWD) [31]; and the European Society for Clinical Nutrition and Metabolism Special Interest Group on cachexia-anorexia in chronic wasting diseases (ESPEN) [32] have published operational criteria to define sarcopenia, using different assessment tools or cutoff points. For instance, while the EWGSOP employed appendicular skeletal muscle mass/height2 as skeletal muscle mass index, the FNIH Sarcopenia Project used appendicular lean mass divided by body size (ALMBMI). SCWD and ESPEN criteria are similar to those described by the EWGSOP, while IWG took a different cutoff point for gait speed when assessing physical performance.
As for muscle mass assessment, CT and MRI are also considered as the gold standard for estimating muscle mass given that they allow distinguishing fat from other soft tissues [33]. DEXA has been shown to be the preferred alternative method to CT and MRI for research and clinical use since it may discriminate between fat, bone mineral, and lean tissues and estimate appendicular skeletal muscle mass [28]. Bioelectrical impedance analysis estimates lean and fat body mass at the molecular level with good correlation with MRI [34] and DEXA [35]. Although positive correlations have been described between calf circumference and appendicular skeletal muscle mass and skeletal muscle index, and thus it could be used as a surrogate marker of muscle mass for diagnosing sarcopenia [36], anthropometric measurements such as this one or calf circumference are not recommended for diagnosing sarcopenia given that they are prone to error [28].
Regarding muscle strength, handgrip strength is commonly used in research and clinical practice [37], since it has been reported to be highly correlated with lower extremity muscle strength [38], which is important in gait, posture, and physical function. Knee flexion and extension techniques may be suitable for research studies, but their use in clinical practice is limited by equipment and training special requirements [28].
There are a number of methods to evaluate physical performance. The Short Physical Performance Battery test, which combines gait speed, chair-rise time, and balance assessment, is one of the most commonly used tools in the research of sarcopenia [39]. Among other tests, usual gait speed, which is a part of the Short Physical Performance Battery test, but can also be obtained from the timed up-and-go test, can be used as a single parameter to provide a predictive value for disability [40].
27.3 Sarcopenic Obesity
Aging-related decrease in muscle and fat mass increase may be masked by a stable body weight, resulting in a phenotype called sarcopenic obesity which is id defined as the coexistence of both sarcopenia and obesity [41]. Although it has gained significant attention from the scientific community in recent years, there is no universally accepted definition. Therefore, true prevalence estimations are unclear due to several factors such as the lack of consensus regarding diagnostic criteria or definitions (i.e., muscle mass alone or together with muscle strength and/or physical performance for sarcopenia) or differences associated with ethnicity in study populations [42]. For instance, Kemmler et al. [43] reported a SO prevalence that ranged from 4.1% (EWGSOP criteria + body fat >25%) to 2.1% (IWGS criteria + body fat >30%) in community-dwelling ≥ 70 years men. There is also divergence in terms of the chosen outcome to define sarcopenia (i.e., appendicular skeletal muscle mass, skeletal muscle mass, or fat-free mass) and obesity (i.e., body mass index, waist circumference, or body fat percentage). To this respect, fat accumulation and redistribution associated with muscle loss do not necessarily lead to BMI increase, and Han et al. [44] suggested that waist circumference gives a better indication of adiposity and sarcopenic obesity than BMI.
The prevalence of sarcopenic obesity also varies with regard to the cutoff points and methods used for its diagnosis (Table 27.1). For instance, with DEXA, Baumgartner et al. [41] reported that the prevalence of SO was 2% in 60–69 years old people from New Mexico and 10% for those ≥80 years. When using a Relative Skeletal Muscle Index, 8.9% of men and 7.1% of women (70–79 years old, USA) sarcopenic obese (appendicular lean mass/height2) [45], but prevalence was higher (60 years and older, USA) when using appendicular lean mass/BMI, ranging from 16 to 40% [46].
27.3.1 Etiopathogenesis
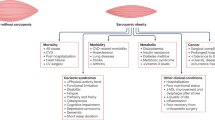
The etiopathogenesis of sarcopenic obesity is a complex process where several common pathophysiological mechanisms interplay (Fig. 27.1). These factors involve lifestyle factors such as diet, low physical activity and sedentary behavior, smoking, hormone changes (insulin, growth factors, vitamin D), oxidative stress, neuromuscular changes, or immunological (proinflammatory cytokines) factors [51].
Aging-related changes in body composition seem to be strongly interconnected. With age, and together with muscle mass decrease, there is an alteration in body fat distribution where visceral fat and waist circumference increase, whereas subcutaneous fat decreases. Besides, there is an important increase in muscle fat infiltration [52].
The aging process is characterized by a state of chronic inflammation, known as “inflammaging.” It is an important link among obesity, insulin resistance, aging, and age-associated diseases [4]. Inflammation leads to local and systemic increment of proinflammatory makers, such as tumor necrosis factor-α, interleukin-1β or 6, interferon-γ, inflammatory adipokines, chemokines, and free fatty acids [53].
Obesity and sarcopenia are interconnected. Abdominal fat deposit has been described to be more proinflammatory than general obesity. Increased intramuscular and intrahepatic fat contribute to insulin resistance [54] which may affect protein degradation, protein synthesis, and muscle growth through locally released adipokines and free fatty acids. Adipokines secreted from fat tissue could lead to muscle wasting and fatty infiltration [55] which may cause an inflammatory state within the muscle. With this proinflammatory environment, obese people preferentially mobilize muscle, not fat, leading to fat increase and muscle loss and thus sarcopenic obesity.
27.3.2 Consequences
Obesity-related consequences are widely studied and debated modern epidemics and are related to a substantial and rising percentage of healthcare costs [56]. Among others, obesity is associated with an increased risk of type 2 diabetes, hypertension, dyslipidemia, and cardiovascular disease [57]. Sarcopenia is also known to be linked with adverse glucose metabolism and metabolic syndrome, in middle-aged and older nonobese people [58].
Sarcopenia also represents a significant influence on healthcare charges, mainly attributed to its strong effect on disability, and the increased healthcare expenditures in disabled persons [59]. Compared with sarcopenia or obesity alone, individuals with sarcopenic obesity have been reported to have poorer physical function and 2.5 times higher risk of reporting disability regarding instrumental daily living activities [18]. Obesity has been also associated with disability, and BMI and waist circumference have been described as important predictors of the onset or worsening of mobility disability in the older adult [60].
In sarcopenic obese people, both sarcopenia and obesity might synergistically increase their health-related deleterious effects [42], with, among others, worse cardiovascular risk profiles [61]. However, there are conflicting results, and several studies have reported that, compared with sarcopenic obesity, obesity has more cardiovascular risk factors in older women [62]. Sarcopenic obesity also presents a negative effect on lower functional capacity, a higher risk of falls [35], and a loss of independence [63].
Regarding mortality rates, Janssen et al. [64] reported that higher BMI values may be related to a lower risk, but if obesity is combined with low muscle strength, the risk of mortality may overcome the protective effect. Elderly people with sarcopenia have decreased survival rates following acute illness and with a doubled risk of nosocomial infection [65]. As for sarcopenic obesity, it has been associated with significantly higher risk of all-cause mortality compared to nonsarcopenic, nonobese subjects [66]. Nevertheless, in the study published by Batsis et al. [19] with data extracted from NHANES III study, older women with sarcopenia had a higher risk of all-cause mortality, independent of obesity.
27.3.3 Management
Differences regarding sarcopenic obesity definition and diagnose lead to difficulties in comparing the effectiveness of the strategies that target this entity [67]. Taking into account the complexity of sarcopenic obesity etiopathogenesis, its management requires a multifactorial approach [68].
Obesity management has traditionally centered on decreasing weight rather than increasing muscle. It mainly aims to reduce intra-abdominal fat through caloric restriction. As mentioned above, obesity is associated with several chronic illnesses, and in this way, intentional weight loss in obese patients can have important clinical benefits or prevent many of the obesity-related risk factors for cardiovascular diseases as well as improvements in osteoarthritis or type 2 diabetes mellitus [69]. It has been also described that a moderate weight loss determines a significant improvement in insulin resistance, in fat distribution, and, more importantly, in muscle lipid infiltration [70]. Besides, excess caloric intake that results in obesity may lead to abnormal surges in serum-free fatty acids and glucose levels, which are linked high levels of oxidative stress, accelerating sarcopenia [71].
Therefore, a correct diet approach is of great importance in obesity management. A recent study analyzed several diet patterns in postmenopausal women and showed that with independence of age, menopausal age, total daily caloric intake and daily physical activity, and high consumption of unrefined cereals and legumes together with low intake of refined cereals were associated with lower BMI, waist circumference, and waist to height ratio. Nevertheless, greater consumption of red meat and potatoes, and low consumption of nuts, coffee, and tea, was associated with an increase in these three obesity parameters.
In postmenopausal women, physical exercise benefits in age-related diseases such as disability and falls, metabolic syndrome, cardiovascular diseases, or dementia, and cognitive function impairment has been widely described [72]. Nevertheless, the addition of exercise to energy restriction does not appear to have an additive effect on the amount of weight lost [42]. Moreover, weight loss induced by energy restriction alone may be costly in terms of losses in fat-free mass, and thus, muscle mass may decrease together with total body weight.
Several strategies have been described in the recent years regarding the treatment and management of sarcopenia, mainly aimed to improve muscle mass and function [73]. The most important approaches include nutritional and physical exercise interventions, either alone or combined, which have been shown as effective strategies not only for sarcopenia treatment but also to prevent the onset and the development of risk factors for sarcopenia, such as obesity, diabetes, chronic low-grade inflammatory state, cardiovascular accidents, and hormonal deficit. In a recent review, Sgro et al. [74] concluded that that even in the case of overt hormonal deficiency, replacement therapy may be recommended not only because of the direct effects on muscle mass and performance but also because a good hormone milieu represents a requisite for adaptive process of supercompensation to training-induced stimulus. At this respect, recent studies in rats have shown that hormone replacement therapy with growth and/or parathyroid hormone may be an effective strategy in sarcopenia prevention and treatment [75, 76].
27.3.3.1 Physical Exercise
The benefits of distinct forms of programmed physical exercise programs on muscle mass and function have been demonstrated in different age groups. For instance, Crane et al. [77] studied muscle strength in three different age groups (20–39, 40–64, and 64–86 years), concluding that muscle strength significantly decreased with age, and people who practiced long-term aerobic exercise had significantly higher muscle strength assessed by several methods such as grip strength, relative maximal isometric knee extension torque, and absolute and relative 1-RM knee extension. In addition, it has been shown that, in well-trained older adults, skeletal muscle structure analyzed by muscle biopsies is more similar to the active young men than to that of the age-matched sedentary men [78].
It has been widely described that prolonged resistance exercise, which has been shown to be effective and safe even in very old and frail subjects, leads to specific type II muscle fiber hypertrophy in healthy young and older men [79]. Aerobic exercise training has also important benefits, and it is more suitable to maintain and/or increase aerobic and cardiovascular fitness and conserves muscle mass by improving muscle blood flow and decreasing oxidative stress [80].
Resistance exercise is currently described as the most effective exercise strategy to improve muscle mass, strength, and function in older people, but most of these studies have been performed among older, nonsarcopenic obese adults [81, 82]. When analyzing the effects of programmed physical exercise on people with sarcopenic obesity, only a few randomized controlled trials have been published. At this respect, Gadelha et al. [83] found improvements in both fat-free mass and sarcopenic obesity index that considered appendicular FFM based on height and fat mass, after 24 weeks of progressive resistance training.
The benefits of both aerobic and resistance exercises combined in sarcopenic obesity people have been studied, and thus Park et al. [84] found improvements in waist circumference, physical performance, fat mass, and muscle strength in postmenopausal women aged 65 years and over. Chen et al. [85], in a study performed in men and women aged 65–75 years, found an increase in muscle mass and a decrease in total fat mass and visceral fat area after resistance and aerobic exercises alone or combined (concurrent exercise), compared with participants who did not train. Muscle strength performance and serum IGF-1 level were also superior in the trained groups, especially after resistance exercises. Nevertheless, despite these improvements in sarcopenic obesity parameters, a clear conclusion cannot be drawn due to the disparity of the results obtained and the heterogeneity in the assessment of the sarcopenic obesity-related outcomes and criteria [86].
Between the recent training modalities, whole-body electromyostimulation (WB-EMS) has been proven to be effective in increasing muscle thickness, muscle fiber cross-sectional area, and knee extension strength in healthy human skeletal muscles [87]. More specifically, WB-EMS has shown benefits in muscle strength in different target populations such as elite soccer players [88] or postmenopausal women [89]. In 2016, Kemmler et al. [90] reported that, in middle-aged men, WB-EMS effects on general strength and body composition are similar to those obtained by high-intensity resistance training exercise. Nevertheless, WB-EMS studies carried out in sarcopenic obese people usually employee a combination of this exercises and dietary supplementation and will be described below.
27.3.3.2 Nutrition
Nutrition is a key factor in the development of both sarcopenia and obesity, although while sarcopenia is associated with an inadequate nutritional intake, obesity may be a result of an excess in the energy intake consumption (together with low energy expenditure) [91]. Consequently, nutritional strategies focused on sarcopenic obese people should target an optimal nutrient intake to increase skeletal muscle mass or prevent muscle mass loss, as well as to decrease excess fat mass.
Hypocaloric diets are very effective for losing weight in obese older adults. However, this strategy is highly undesirable in sarcopenic obese people, since it is estimated that about 25% of this weight loss is skeletal muscle mass, and can also have harmful effects for the micronutrient status and bone mineral density. Thus, in sarcopenic obesity, a weight loss diet should always focus on the preservation of muscle mass and could be combined with a high-protein diet and/or micronutrient supplementation [67].
Dietary amino acids have been demonstrated to have a positive regulatory effect on the muscle protein synthesis [92]. Protein intake may also be a significant factor for sarcopenia prevention and management. In older adults (>65 years), a dietary protein intake of 1.0–1.2 g/kg is recommended to maintain and regain the muscle mass and function in the long term, while it should be higher (1.2–1.5 g/kg) in individuals that suffer from chronic diseases [93]. This can reduce the risk of chronic diseases and improve outcomes [94]. Nevertheless it has been reported that an excessive intake of protein could be very harmful for the 65 and younger population [95].
The type of protein and the amino acid composition are also relevant for muscle mass preservation or gain during weight loss. Whey protein, the soluble protein fractions extracted from diary milk [96], can stimulate whole-body and muscle protein synthesis [97] and could represent an effective countermeasure to prevent muscle atrophy associated with physical inactivity and muscle unloading during aging [42]. Nevertheless, it has been reported that whey protein ingestion has a greater anabolic effect in the elderly than the essential amino acids that it contains [98]. An intake of leucine (2.0–2.5 g/day), mainly derived from animal sources, has been reported to improve the postprandial muscle protein synthesis in elderly men [99]. Beta-hydroxy-beta-methylbutyrate, a metabolite of leucine, can improve this muscle loss, but to date only a small number of studies have shown increases in lean (muscle) mass and some muscle function and physical performance parameters in older people with or without resistance exercise and in muscle mass preservation during bed rest [100]. Other essential amino acids such as l-arginine [101] or l-cysteine [102] have been shown to have beneficial effects regarding insulin resistance in type 2 diabetic patients.
Protein source (plant or animal) and intake timing are also of importance. Overall, animal-derived dietary protein seems most effective in eliciting muscle protein synthesis [103]. Regarding the timing of protein intake, a more evenly distribution of dietary protein intake (every 3–4 h), instead of protein consumption during the three main meals, has been associated with higher muscle strength, physical performance, and skeletal muscle mass in older adults [104, 105].
As for older people with sarcopenic obesity, hypocaloric diets should be supplied with adequate protein, which may help to prevent muscle loss [106] and improve adherence to low energy intake [107]. Ensuring adequate dietary protein intake using high-quality proteins should be of importance in adults with sarcopenic obesity, with the quality of the protein being more important than the quantity [42]. It has been described that whey protein and essential amino acid meal replacement during weight loss induced by caloric restriction diet promote a reduction of adipose tissue and a modest loss of lean tissue in the elderly population people [108]. However, although the combination of a hypocaloric high-protein diet seems to be effective in the prevention of sarcopenic obesity, this strategy does not seem to be effective for its treatment [67].
Another strategy is to combine different anabolic nutrients. It has been recently reported that the combination of vitamin D with whey protein, which has been enriched with leucine, could increase protein synthesis and finally promote muscle mass gain in older adults [109]. The risk of developing micronutrient deficiencies such as 25-hydroxyvitamin D; vitamin B6, C, and E; selenium; magnesium; or zinc is relatively high in obese adults, who, in addition, are especially at risk for micronutrient deficiencies when following a weight loss diet [110]. On the other hand, some of these minerals as well as low 25-hydroxy-vitamin D status are associated with the development of sarcopenia [111, 112], and vitamin D supplementation has been demonstrated to improve several sarcopenic parameters in older adults [113].
27.3.3.3 Exercise + Nutrition
As mentioned above, a combination of different approaches seems to be more appropriate than one single strategy, whether it is nutrition or exercise, in sarcopenic obesity management.
The addition of exercise to a hypocaloric diet in obese older adults attenuates the loss of skeletal muscle mass [114], and it also improves muscle strength and performance [67]. In frail obese adults, a hypocaloric diet together with the combination of aerobic and resistance exercise is more effective for improving the functional status than the combination of low energy diet with either aerobic or resistance exercises alone [115].
As for the combination of exercise and nutrition, Liao et al. [116] reported that, in overweight and obese elderly people, higher increases in lean mass, muscular volume, and leg strength are observed after resistance exercise combined with protein supplementation, in comparison with resistance exercise alone. However, they found that obese participants did not show greater change in muscle volume and grip strength compared with nonobese. Diet supplementation (particularly high protein intake or protein supplementation) combined with exercise is the most common strategy to counteract sarcopenic obesity. A recently published meta-analysis [117] focused on sarcopenic obese people has concluded that exercise alone and together with dietary supplementation improve muscle-related outcomes and reduce fat-related outcomes. However, the authors state that there is a need for better-designed randomized controlled trials with systematic evaluation of the different types of exercise and dietary supplements. More specifically, Kim et al. [118] analyzed the effects of progressive sequence of resistance and aerobic training and amino acid supplementation with tea catechin alone and combined. They observed that, although exercise and nutrition have beneficial effects on individual variables of body composition and physical function, improvements in muscle mass and variable combinations such as skeletal muscle mass index and fat percentage or physical performance and fat percentage were not found. When analyzing randomized controlled trials with combined nutritional supplements and exercise on sarcopenic obese population, WB-EMS is the most commonly used form of exercise. In a project performed in sarcopenic obesity community-dwelling women aged over 70 years, WB-EMS increased muscle mass and functional capacity, but the effect on body fat was minor, and the addition of protein-enriched supplements did not increase the effects of WB-EMS alone [43]. In the same project, when studying the effects on the metabolic syndrome [119], isolated WB-EMS did not induce significant improvements, but combined with low-dose protein supplementation, more favorable results were observed. In a similar project carried out in men (>70 years), improvements in fat and muscle mass, muscle strength, and gait velocity were observed in both WB-EMS alone and combined with protein supplements [120, 121].
27.4 Conclusions
Aging-related decrease in muscle and fat mass increase may be masked by a stable body weight, called sarcopenic obesity. Estimation of the prevalence is difficult due to the lack of consensus regarding sarcopenia definition and the heterogeneity in the assessment of the sarcopenic obesity-related outcomes. Different common pathophysiological mechanisms, such as lifestyle, immunological, endocrine, or vascular factors, are involved in its pathogenesis. A proinflammatory environment seems to play an important role in the association between sarcopenia and obesity. Postmenopause is associated with an increase in visceral adipose tissue, an important source of inflammatory markers. Sarcopenic obesity management requires a multifactorial approach, and several strategies, such as physical exercise or diet, either alone or combined have been studied in literature, but the limited number of studies focused on sarcopenic obesity people and the variability regarding the outcome measures and study designs preclude firm conclusions. An adequate combination of hypocaloric diet focused on losing weight in obese older adults and an appropriate protein intake have been described to be important factors for sarcopenic obesity prevention and treatment. Among the different forms of exercise, resistance and aerobic exercise programs, as well as more recent modalities such as whole-body electromyostimulation, seem to be effective in sarcopenic obesity management.
References
United Nations. World population ageing 2013 [Internet]. New York, USA: Department of Economic and Social Affairs, Population Division; 2013. Report No.: ST/ESA/SER.A/348. http://www.un.org/en/development/desa/population/publications/pdf/ageing/WorldPopulationAgeing2013.pdf.
Flatt T. A new definition of aging? Front Genet. 2012;3:148.
Ding J, Kritchevsky SB, Newman AB, Taaffe DR, Nicklas BJ, Visser M, Lee JS, Nevitt M, Tylavsky FA, Rubin SM, Pahor M, Harris TB. Health ABC study, effects of birth cohort and age on body composition in a sample of community-based elderly. Am J Clin Nutr. 2007;85(2):405–10.
Frasca D, Blomberg BB, Paganelli R. Aging, obesity, and inflammatory age-related diseases. Front Immunol. 2017;8:1745.
Guh DP, Zhang W, Bansback N, Amarsi Z, Birmingham CL, Anis AH. The incidence of co-morbidities related to obesity and overweight: a systematic review and meta-analysis. BMC Public Health. 2009;9:88.
Zambon M, Mazzali G, Zoico E, Harris TB, Meigs JB, Di Francesco V, Fantin F, Bissoli L, Bosello O. Health consequences of obesity in the elderly: a review of four unresolved questions. Int J Obes. 2016;29:1011–29.
Global Health Observatory (GHO) Obesity. Situation and trends. Geneva: World Health Organization; 2013.
Berdah J. Staying fit after fifty. Gynecol Obstet Fertil. 2006;34:920–6.
Davis SR, Lambrinoudaki I, Lumsden M, Mishra GD, Pal L, Rees M, Santoro N, Simoncini T. Menopause. Nat Rev Dis Primers. 2015;1:15004.
Sternfeld B, Wang H, Quesenberry CP Jr, Abrams B, Everson-Rose SA, Greendale GA, Matthews KA, Torrens JI, Sowers M. Physical activity and changes in weight and waist circumference in midlife women: findings from the Study of Women’s Health Across the Nation. Am J Epidemiol. 2004;160:912e22.
Lovejoy JC, Champagne CM, de Jonge L, Xie H, Smith SR. Increased visceral fat and decreased energy expenditure during the menopausal transition. Int J Obes. 2008;32:949–58.
World Health Organization. Obesity: preventing and management of the global epidemic. Report of the WHO consultation. Technical report series. No. 894. Geneva: World Health Organization; 2000.
Rothman KJ. BMI-related errors in the measurement of obesity. Int J Obes. 2008;3(s3):s56–9.
Hita-Contreras F. Traditional body mass index cut-offs in older people: time for a rethink with altered fat distribution, sarcopenia and shrinking height. Maturitas. 2018;113:A1–2.
Sorkin JD, Muller DC, Andres R. Longitudinal change in the heights of men and women: consequential effects on body mass index. Epidemiol Rev. 1999;21(2):247–60.
Alberti KG, Eckel RH, Grundy SM, Zimmet PZ, Cleeman JI, Donato KA, Fruchart JC, James WP, Loria CM, Smith SC Jr, International Diabetes Federation Task Force on Epidemiology and Prevention, National Heart, Lung, and Blood Institute; American Heart Association; World Heart Federation, International Atherosclerosis Society; International Association for the Study of Obesity. Harmonizing the metabolic syndrome: a joint interim statement of the International Diabetes Federation Task Force on Epidemiology and Prevention; National Heart, Lung, and Blood Institute; American Heart Association; World Heart Federation; International Atherosclerosis Society; and International Association for the Study of Obesity. Circulation. 2009;120:1640–5.
Rollins KE, Javanmard-Emamghissi H, Awwad A, Macdonald IA, Fearon KCH, Lobo DN. Body composition measurement using computed tomography: does the phase of the scan matter? Nutrition. 2017;41:37–44.
Baumgartner RN, Wayne SJ, Waters DL, Janssen I, Gallagher D, Morley JE. Sarcopenic obesity predicts instrumental activities of daily living disability in the elderly. Obes Res. 2004;12:1995–2004.
Batsis JA, Mackenzie TA, Barre LK, Lopez-Jimenez F, Bartels SJ. Sarcopenia, sarcopenic obesity and mortality in older adults: results from the National Health and Nutrition Examination Survey III. Eur J Clin Nutr. 2014;68:1001–7.
Janssen I. Evolution of sarcopenia research. Appl Physiol Nutr Metab. 2010;35:707–12.
Curcio F, Ferro G, Basile C, Liguori I, Parrella P, Pirozzi F, Della-Morte D, Gargiulo G, Testa G, Tocchetti CG, Bonaduce D, Abete P. Biomarkers in sarcopenia: a multifactorial approach. Exp Gerontol. 2016;85:1–8.
Janssen I. The epidemiology of sarcopenia. Clin Geriatr Med. 2011;27:355–63.
Montero-Fernández N, Serra-Rexach JA. Role of exercise on sarcopenia in the elderly. Eur J Phys Rehabil Med. 2013;49:131.
Rosenberg I. Summary comments: epidemiological and methodological problems in determining nutritional status of older persons. Am J Clin Nutr. 1989;50:1231–3.
Morley JE, Baumgartner RN, Roubenoff R, Mayer J, Nair KS. Sarcopenia. J Lab Clin Med. 2001;137:231–43.
Baumgartner RN, Koehler KM, Gallagher D, Romero L, Heymsfield SB, Ross RR, Garry PJ, Lindeman RD. Epidemiology of sarcopenia among the elderly in New Mexico. Am J Epidemiol. 1998;147:755–63.
Janssen I, Heymsfield SB, Ross R. Low relative skeletal muscle mass (sarcopenia) in older persons is associated with functional impairment and physical disability. J Am Geriatr Soc. 2002;50:889–96.
Cruz-Jentoft AJ, Baeyens JP, Bauer JM, Boirie Y, Cederholm T, Landi F, Martin FC, Michel JP, Rolland Y, Schneider SM, Topinková E, Vandewoude M, Zamboni M. European working group on sarcopenia in older people, sarcopenia: European consensus on definition and diagnosis: report of the European Working group on sarcopenia in older people. Age Ageing. 2010;39(4):412–23.
Dam TT, Peters KW, Fragala M, Cawthon PM, Harris TB, McLean R, Shardell M, Alley DE, Kenny A, Ferrucci L, Guralnik J, Kiel DP, Kritchevsky S, Vassileva MT, Studenski S. An evidence-based comparison of operational criteria for the presence of sarcopenia. J Gerontol A Biol Sci Med Sci. 2014;69:584–90.
Fielding RA, Bruno V, Evans WJ, Bhasin S, Morley JE, Newman AB, Abellan van Kan G, Andrieu S, Bauer J, Breuille D, Cederholm T, Chandler J, De Meynard C, Donini L, Harris T, Kannt A, Keime Guibert F, Onder G, Papanicolaou D, Rolland Y, Rooks D, Sieber C, Souhami E, Verlaan S, Zamboni M. Sarcopenia: an undiagnosed condition in older adults. Current consensus definition: prevalence, etiology, and consequences. International working group on sarcopenia. J Am Med Dir Assoc. 2011;12:1–9.
Morley JE, Abbatecola AM, Argiles JM, Baracos V, Bauer J, Bhasin S, Cederholm T, Coats AJ, Cummings SR, Evans WJ, Fearon K, Ferrucci L, Fielding RA, Guralnik JM, Harris TB, Inui A, Kalantar-Zadeh K, Kirwan BA, Mantovani G, Muscaritoli M, AB Newman F, Rosano GMR-F, Roubenoff R, Schambelan M, Sokol GH, Storer TW, Vellas B, von Haehling S, Yeh SS, Anker SD, Society on Sarcopenia, Cachexia and Wasting Disorders Trialist Workshop, Society on Sarcopenia, Cachexia and Wasting Disorders Trialist Workshop. Sarcopenia with limited mobility: an international consensus. J Am Med Dir Assoc. 2012;12:403–9.
Muscaritoli M, Anker SD, Argiles J, Aversa Z, Bauer JM, Biolo G, Boirie Y, Bosaeus I, Cederholm T, Costelli P, Fearon KC, Laviano A, Maggio M, Fanelli FR, Schneider SM, Schols A, Sieber CC. Consensus definition of sarcopenia, cachexia and pre-cachexia: joint document elaborated by special interest groups (SIG) “cachexia-anorexia in chronic wasting diseases” and “nutrition in geriatrics”. Clin Nutr. 2010;29:154–9.
Cesari M, Fielding RA, Pahor M, Goodpaster B, Hellerstein M, van Kan GA, Anker SD, Rutkove S, Vrijbloed JW, Isaac M, Rolland Y, M’rini C, Aubertin-Leheudre M, Cedarbaum JM, Zamboni M, Sieber CC, Laurent D, Evans WJ, Roubenoff R, Morley JE, Vellas B, International Working Group on Sarcopenia, International Working Group on Sarcopenia. Biomarkers of sarcopenia in clinical trials-recommendations from the International Working Group on Sarcopenia. J Cachexia Sarcopenia Muscle. 2012;3:181–90.
Chien MY, Huang TY, Wu YT. Prevalence of sarcopenia estimated using a bioelectrical impedance analysis prediction equation in community-dwelling elderly people in Taiwan. J Am Geriatr Soc. 2008;56:1710–5.
Malafarina V, Uriz-Otano F, Iniesta R, Gil-Guerrero L. Sarcopenia in the elderly: diagnosis, physiopathology and treatment. Maturitas. 2012;71:109–14.
Kawakami R, Murakami H, Sanada K, Tanaka N, Sawada SS, Tabata I, Higuchi M, Miyachi M. Calf circumference as a surrogate marker of muscle mass for diagnosing sarcopenia in Japanese men and women. Geriatr Gerontol Int. 2015;15:969–76.
Laurentani F, Russo C, Bandinelli S, Bartali B, Cavazzini C, Di Iorio A, Corsi AM, Rantanen T, Guralnik JM, Ferrucci L. Age-associated changes in skeletal muscles and their effect on mobility: an operational diagnosis of sarcopenia. J Appl Physiol. 2003;95:1851–60.
Peolsson A, Hedlund R, Oberg B. Intra- and inter-tester reliability and reference values for hand strength. J Rehabil Med. 2001;33:36–41.
Guralnik JM, Simonsick EM, Ferrucci L, Glynn RJ, Berkman LF, Blazer DG, Scherr PA, Wallace RB. A short physical performance battery assessing lower extremity function: association with self-reported dis-ability and prediction of mortality and nursing home admission. J Gerontol. 1994;49:M85–94.
Mathias S, Nayak US, Isaacs B. Balance in elderly patients: the get-up and go test. Arch Phys Med Rehabil. 1986;67:387–9.
Baumgartner RN. Body composition in healthy aging. Ann N Y Acad Sci. 2000;904:437–48.
Molino S, Dossena M, Buonocore D, Verri M. Sarcopenic obesity: an appraisal of the current status of knowledge and management in elderly people. J Nutr Health Aging. 2016;20(7):780–8.
Kemmler W, Teschler M, Weißenfels A, Sieber C, Freiberger E, von Stengel S. Prevalence of sarcopenia and sarcopenic obesity in older German men using recognized definitions: high accordance but low overlap! Osteoporos Int. 2017;28:1881–91.
Han TS, Tajar A, Lean MEJ. Obesity and weight management in the elderly. Br Med Bull. 2011;97:169–96.
Newman AB, Kupelian V, Visser M, Simonsick E, Goodpaster B, Nevitt M, Kritchevsky SB, Tylavsky FA, Rubin SM, Harris TB, Health ABC Study Investigators. Sarcopenia: alternative definitions and associations with lower extremity function. J Am Geriatr Soc. 2003;51:1602–9.
Batsis JA, Mackenzie TA, Lopez-Jimenez F, Bartels SJ. Sarcopenia, sarcopenic obesity, and functional impairments in older adults: National Health and Nutrition Examination Surveys 1999–2004. Nutr Res. 2015;35:1031–9.
Bouchard DR, Dionne IJ, Brochu M. Sarcopenic/obesity and physical capacity in older men and women: data from the nutrition as a determinant of successful aging (NuAge)-the Quebec longitudinal study. Obesity (Silver Spring). 2009;17:2082–8.
Oh C, Jho S, No J-K, Kim H-S. Body composition changes were related to nutrient intakes in elderly men but elderly women had a higher prevalence of sarcopenic obesity in a population of Korean adults. Nutr Res. 2015;35:1–6.
Öztürk ZA, Türkbeyler İH, Abiyev A, Kul S, Edizer B, Yakaryılmaz FD, Soylu G. Health related quality of life and fall risk associated with age related body composition changes; sarcopenia, obesity and sarcopenic obesity. Intern Med J. 2018;48(8):973–81.
Moreira MA, Zunzunegui MV, Vafaei A, da Câmara SM, Oliveira TS, Maciel ÁC. Sarcopenic obesity and physical performance in middle aged women: a cross-sectional study in Northeast Brazil. BMC Public Health. 2016;16:43.
Prado CMM, Wells JCK, Smith SR, Stephan BCM, Siervo M. Sarcopenic obesity: a critical appraisal of the current evidence. Clin Nutr. 2012;31:583–601.
Delmonico MJ, Harris TB, Visser M, Park SW, Conroy MB, Velasquez-Mieyer P, Boudreau R, Manini TM, Nevitt M, Newman AB, Goodpaster BH, Health, Aging, and Body. Longitudinal study of muscle strength, quality, and adipose tissue infiltration. Am J Clin Nutr. 2009;90:1579–85.
Grant RW, Dixit VD. Adipose tissue as an immunological organ. Obesity (Silver Spring). 2015;23:512–8.
Guillet C, Boirie Y. Insulin resistance: a contributing factor to age-related muscle mass loss? Diabetes Metab. 2005;2:5S20–6.
Woodrow G. Body composition analysis techniques in the aged adult: indications and limitations. Curr Opin Clin Nutr Metab Care. 2009;12:8–14.
Biener A, Cawley J, Meyerhoefer C. The impact of obesity on medical care costs and labor market outcomes in the US. Clin Chem. 2018;64(1):108–17.
Kim J, Cho JJ, Park YS. Relationship between sarcopenic obesity and cardiovascular disease risk as estimated by the Framingham risk score. J Korean Med Sci. 2015;30:264–71.
Zhang H, Lin S, Gao T, Zhong F, Cai J, Sun Y, Ma A. Association between sarcopenia and metabolic syndrome in middle-aged and older non-obese adults: a systematic review and meta-analysis. Nutrients. 2018;10:E364.
Janssen I, Shepard DS, Katzmarzyk PT, Roubenoff R. The healthcare costs of sarcopenia in the United States. J Am Geriatr Soc. 2004;52:80–5.
Vincent HK, Vincent KR, Lamb KM. Obesity and mobility disability in the older adult. Obes Rev. 2010;11:568–79.
Lim S, Kim JH, Yoon JW, Kang SM, Choi SH, Park YJ, Kim KW, Lim JY, Park KS, Jang HC. Sarcopenic obesity: prevalence and association with metabolic syndrome in the Korean longitudinal study on health and aging (KLo-SHA). Diabetes Care. 2010;33:1652–4.
dos Santos EP, Gadelha AB, Safons MP, Nobrega OT, Oliveira RJ, Lima RM. Sarcopenia and sarcopenic obesity classifications and cardiometabolic risks in older women. Arch Gerontol Geriatr. 2014;59:56–61.
Aibar-Almazán A, Martínez-Amat A, Cruz-Díaz D, Jiménez-García JD, Achalandabaso A, Sánchez-Montesinos I, de la Torre-Cruz MM, Hita-Contreras F. Sarcopenia and sarcopenic obesity in Spanish community-dwelling middle-aged and older women: association with balance confidence, fear of falling and fall risk. Maturitas. 2018;107:26–32.
Janssen I, Katzmarzyk PT, Ross R. Body mass index is inversely related to mortality in older people after adjustment for waist circumference. J Am Geriatr Soc. 2005;53:2112–8.
Lang T, Streeper T, Cawthon P, Baldwin K, Taaffe DR, Harris TB. Sarcopenia: etiology, clinical consequences, intervention, and assessment. Osteoporos Int. 2010;21:543–59.
Tian S, Xu Y. Association of sarcopenic obesity with the risk of all-cause mortality: a meta-analysis of prospective cohort studies. Geriatr Gerontol Int. 2016;16:155–66.
Trouwborst I, Verreijen A, Memelink R, Massanet P, Boirie Y, Weijs P, Tieland M. Exercise and nutrition strategies to counteract sarcopenic obesity. Nutrients. 2018;10:E605.
Hita-Contreras F, Martínez-Amat A, Cruz-Díaz D, Pérez-López FR. Osteosarcopenic obesity and fall prevention strategies. Maturitas. 2015;80(2):126–32.
Poirier P, Giles TD, Bray GA, Hong Y, Stern JS, Pi-Sunyer FX, Eckel RH. Obesity and cardiovascular disease: pathophysiology, evaluation, and effect of weight loss: an update of the 1997 American Heart Association Scientific Statement on Obesity and Heart Disease from the Obesity Committee of the Council on Nutrition, Physical Activity and Metabolism. Circulation. 2006;113:898–918.
Li Z, Heber D. Sarcopenic obesity in the elderly and strategies for weight management. Nutr Rev. 2012;70:57–64.
Maltais ML, Perreault K, Courchesne-Loyer A, Lagacé JC, Barsalani R, Dionne IJ. Effect of resistance training and various sources of protein supplementation on body fat mass and metabolic profile in Sarcopenic overweight older adult men: a pilot study. Int J Sport Nutr Exerc Metab. 2016;26:71–7.
Mendoza N, De Teresa C, Cano A, Godoy D, Hita-Contreras F, Lapotka M, Llaneza P, Manonelles P, Martínez-Amat A, Ocón O, Rodríguez-Alcalá L, Vélez M, Sánchez-Borrego R. Benefits of physical exercise in postmenopausal women. Maturitas. 2016;93:83–8.
Naseeb MA, Volpe SL. Protein and exercise in the prevention of sarcopenia and aging. Nutr Res. 2017;40:1–20.
Sgrò P, Sansone M, Sansone A, Sabatini S, Borrione P, Romanelli F, Di Luigi L. Physical exercise, nutrition and hormones: three pillars to fight sarcopenia. Aging Male. 2019;22(2):75–88.
Brioche T, Kireev RA, Cuesta S, Gratas-Delamarche A, Tresguerres JA, Gomez-Cabrera MC, Viña J. Growth hormone replacement therapy prevents sarcopenia by a dual mechanism: improvement of protein balance and of antioxidant defences. J Gerontol A Biol Sci Med Sci. 2014;69:1186–98.
Brent MB, Brüel A, Thomsen JS. PTH (1-34) and growth hormone in prevention of disuse osteopenia and sarcopenia in rats. Bone. 2018;110:244–53.
Crane JD, Macneil LG, Tarnopolsky MA. Long-term aerobic exercise is associated with greater muscle strength throughout the life span. J Gerontol A Biol Sci Med Sci. 2013;68:631–8.
Zampieri S, Pietrangelo L, Loefler S, Fruhmann H, Vogelauer M, Burggraf S, Pond A, Grim-Stieger M, Cvecka J, Sedliak M, Tirpáková V, Mayr W, Sarabon N, Rossini K, Barberi L, De Rossi M, Romanello V, Boncompagni S, Musarò A, Sandri M, Protasi F, Carraro U, Kern H. Lifelong physical exercise delays age associated skeletal muscle decline. J Gerontol A Biol Sci Med Sci. 2015;70:163–73.
Nilwik R, Snijders T, Leenders M, Groen BB, van Kranenburg J, Verdijk LB, van Loon LJ. The decline in skeletal muscle mass with aging is mainly attributed to a reduction in type II muscle fiber size. Exp Gerontol. 2013;48:492–8.
Donges CE, Duffield R, Drinkwater EJ. Effects of resistance or aerobic exercise training on interleukin-6, C-reactive protein, and body composition. Med Sci Sports Exerc. 2010;42:304–13.
Peterson MD, Sen A, Gordon PM. Influence of resistance exercise on lean body mass in aging adults: a meta-analysis. Med Sci Sports Exerc. 2011;43(2):249–58.
Liu CJ, Chang WP, Araujo de Carvalho I, Savage KEL, Radford LW, Amuthavalli Thiyagarajan J. Effects of physical exercise in older adults with reduced physical capacity: meta-analysis of resistance exercise and multimodal exercise. Int J Rehabil Res. 2017;40:303–14.
Gadelha AB, Paiva FM, Gauche R, De Oliveira RJ, Lima RM. Effects of resistance training on sarcopenic obesity index in older women: a randomized controlled trial. Arch Gerontol Geriatr. 2016;65:168–73.
Park J, Kwon Y, Park H. Effects of 24-week aerobic and resistance training on carotid artery intima-media thickness and flow velocity in elderly women with sarcopenic obesity. J Atheroscler Thromb. 2017;24(11):1117–24.
Chen HT, Chung YC, Chen YJ, Ho SY, Wu HJ. Effects of different types of exercise on body composition, muscle strength, and IGF-1 in the elderly with sarcopenic obesity. J Am Geriatr Soc. 2017;65(4):827–32.
Martínez-Amat A, Aibar-Almazán A, Fábrega-Cuadros R, Cruz-Díaz D, Jiménez-García JD, Pérez-López FR, Achalandabaso A, Barranco-Zafra R, Hita-Contreras F. Exercise alone or combined with dietary supplements for sarcopenic obesity in community-dwelling older people: a systematic review of randomized controlled trials. Maturitas. 2018;110:92–103.
Natsume T, Ozaki H, Kakigi R, Kobayashi H, Naito H. Effects of training intensity in electromyostimulation on human skeletal muscle. Eur J Appl Physiol. 2018;118:1339–47.
Filipovic A, Grau M, Kleinöder H, Zimmer P, Hollmann W, Bloch W. Effects of a whole-body electrostimulation program on strength, sprinting, jumping, and kicking capacity in elite soccer players. J Sports Sci Med. 2016;15:639–48.
Kemmler W, Schliffka R, Mayhew JL, von Stengel S. Effects of whole-body electromyostimulation on resting metabolic rate, body composition, and maximum strength in postmenopausal women: the training and electrostimulation trial. J Strength Cond Res. 2010;4:1880–7.
Kemmler W, Teschler M, Weissenfels A, Bebenek M, Fröhlich M, Kohl M, von Stengel S. Effects of whole-body electromyostimulation versus high intensity resistance exercise on body composition and strength: a randomized controlled study. Evid Based Complement Alternat Med. 2016;2016:9236809.
Stenholm S, Harris TB, Rantanen T, Visser M, Kritchevsky SB, Ferrucci L. Sarcopenic obesity: definition, cause and consequences. Curr Opin Clin Nutr Metab Care. 2008;11:693–700.
Malafarina V, Uriz-Otano F, Iniesta R, Gil-Guerrero L. Effectiveness of nutritional supplementation on muscle mass in treatment of sarcopenia in old age: a systematic review. J Am Med Dir Assoc. 2013;14:10–7.
Bauer J, Biolo G, Cederholm T, Cesari M, Cruz-Jentoft AJ, Morley JE, Phillips S, Sieber C, Stehle P, Teta D, Visvanathan R, Volpi E, Boirie Y. Evidence-based recommendations for optimal dietary protein intake in older people: a position paper from the PROT-AGE study group. J Am Med Dir Assoc. 2013;14:542–59.
Wolfe RR, Miller SL, Miller KB. Optimal protein intake in the elderly. Clin Nutr. 2008;27:675–84.
Levine ME, Suarez JA, Brandhorst S, Balasubramanian P, Cheng CW, Madia F, Fontana L, Mirisola MG, Guevara-Aguirre J, Wan J, Wan J, Passarino G, Kennedy BK, Wei M, Cohen P, Crimmins EM, Longo VD. Low protein intake is associated with a major reduction in IGF-1, cancer, and overall mortality in the 65 and younger but not older population. Cell Metab. 2014;19:407–17.
Hayes A, Cribb PJ. Effect of whey protein isolate on strength, body composition and muscle hypertrophy during resistance training. Curr Opin Clin Nutr Metab Care. 2008;11:40–4.
Antonione R, Caliandro E, Zorat F, Guarnieri G, Heer M, Biolo G. Whey protein ingestion enhances postprandial anabolism during short-term bed rest in young men. J Nutr. 2008;138:2212–6.
Katsanos CS, Chinkes DL, Paddon-Jones D, Zhang X, Aarsland A, Wolfe RR. Whey protein ingestion in elderly results in greater muscle protein accrual than ingestion of its constituent essential amino acid content. Nutr Res. 2008;28:651–8.
Wall BT, Hamer HM, de Lange A, Kiskini A, Groen BB, Senden JM, Gijsen AP, Verdijk, van Loon LJ. Leucine co-ingestion improves post-prandial muscle protein accretion in elderly men. Clin Nutr (Edinburgh, Scotland). 2013;32:412–9.
J. Cruz-Jentoft A. Beta-hydroxy-beta-methyl butyrate (HMB): from experimental data to clinical evidence in sarcopenia. Curr Protein Pept Sci. 2018;19:668–72.
Lucotti P, Setola E, Monti LD, Galluccio E, Costa S, Sandoli EP, Fermo I, Rabaiotti G, Gatti R, Piatti P. Beneficial effects of a long-term oral L-arginine treatment added to a hypocaloric diet and exercise training program in obese, insulin-resistant type 2 diabetic patients. Am J Physiol Endocrinol Metab. 2006;291:906–12.
Jain SK, Micinski D, Huning L, Kahlon G, Bass PF, Levine SN. Vitamin D and L-cysteine levels correlate positively with GSH and negatively with insulin resistance levels in the blood of type 2 diabetic patients. Eur J Clin Nutr. 2014;68:1148–53.
Van Vliet S, Burd NA, van Loon LJ. The skeletal muscle anabolic response to plant- versus animal-based protein consumption. J Nutr. 2015;145:1981–91.
Cardon-Thomas DK, Riviere T, Tieges Z, Greig CA. Dietary protein in older adults: adequate daily intake but potential for improved distribution. Nutrients. 2017;9:E184.
Farsijani S, Morais JA, Payette H, Gaudreau P, Shatenstein B, Gray-Donald K, Chevalier S. Relation between mealtime distribution of protein intake and lean mass loss in free-living older adults of the NuAge study. Am J Clin Nutr. 2016;104:694–703.
Farnsworth E, Luscombe ND, Noakes M, Wittert G, Argyiou E, Clifton PM. Effect of a high-protein, energy-restricted diet on body composition, glycemic control, and lipid concentrations in overweight and obese hyperinsulinemic men and women. Am J Clin Nutr. 2003;78:31–9.
Weigle DS, Breen PA, Matthys CC, Callahan HS, Meeuws KE, Burden VR, Purnell JQ. A high-protein diet induces sustained reductions in appetite, ad libitum caloric intake, and body weight despite compensatory changes in diurnal plasma leptin and ghrelin concentrations. Am J Clin Nutr. 2005;82:41–8.
Coker RH, Miller S, Schutzler S, Deutz N, Wolfe RR. Whey protein and essential amino acids promote the reduction of adipose tissue and increased muscle protein synthesis during caloric restriction-induced weight loss in elderly, obese individuals. Nutr J. 2012;11:105.
Chanet A, Verlaan S, Salles J, Giraudet C, Patrac V, Pidou V, Pouyet C, Hafnaoui N, Blot A, Cano N, Farigon N, Bongers A, Jourdan M, Luiking Y, Walrand S, Boirie Y. Supplementing breakfast with a vitamin D and leucine-enriched whey protein medical nutrition drink enhances postprandial muscle protein synthesis and muscle mass in healthy older men. J Nutr. 2017;147:2262–71.
Damms-Machado A, Weser G, Bischoff SC. Micronutrient deficiency in obese subjects undergoing low calorie diet. Nutr J. 2012;11:34.
Van Dronkelaar C, van Velzen A, Abdelrazek M, van der Steen A, Weijs PJM, Tieland M. Minerals and sarcopenia; the role of calcium, iron, magnesium, phosphorus, potassium, selenium, sodium, and zinc on muscle mass, muscle strength, and physical performance in older adults: a systematic review. J Am Med Dir Assoc. 2017;19:6–11.e3.
Scott D, Blizzard L, Fell J, Ding C, Winzenberg T, Jones GA. Prospective study of the associations between 25-hydroxy-vitamin D, sarcopenia progression and physical activity in older adults. Clin Endocrinol (Oxf). 2010;73:581–7.
Muir SW, Montero-Odasso M. Effect of vitamin D supplementation on muscle strength, gait and balance in older adults: a systematic review and meta-analysis. J Am Geriatr Soc. 2011;59:2291–300.
Weinheimer EM, Sands LP, Campbell WW. A systematic review of the separate and combined effects of energy restriction and exercise on fat-free mass in middle-aged and older adults: implications for sarcopenic obesity. Nutr Rev. 2010;68:375–88.
Villareal D, Aguirre L, Gurney B, Waters D, Colombo E, Armamento-Villareal R, Qualls C. Aerobic or resistance exercise, or both, in dieting obese older adults. N Engl J Med. 2017;376:1943–55.
Liao CD, Tsauo JY, Wu YT, Cheng CP, Chen HC, Huang YC, Chen HC, Liou TH. Effects of protein supplementation combined with resistance exercise on body composition and physical function in older adults: a systematic review and meta-analysis. Am J Clin Nutr. 2017;106:1078–91.
Hita-Contreras F, Bueno-Notivol J, Martínez-Amat A, Cruz-Díaz D, Hernandez AV, Pérez-López FR. Effect of exercise alone or combined with dietary supplements on anthropometric and physical function measures in community-dwelling elderly people with sarcopenic obesity: a meta-analysis of randomized controlled trials. Maturitas. 2018;116:24–35.
Kim H, Kim M, Kojima N, Fujino K, Hosoi E, Kobayashi H, Somekawa S, Niki Y, Yamashiro Y, Yoshida H. Exercise and nutritional supplementation on community dwelling elderly japanese women with sarcopenic obesity: a randomized controlled trial. J Am Med Dir Assoc. 2016;17(11):1011–9.
Wittmann K, Sieber C, von Stengel S, Kohl M, Freiberger E, Jakob F, Lell M, Engelke K, Kemmler W. Impact of whole body electromyostimulation on cardiometabolic risk factors in older women with sarcopenic obesity: the randomized controlled FORMOsA-sarcopenic obesity study. Clin Interv Aging. 2016;11:1697–706.
Kemmler, Weissenfels A, Teschler M, Willert S, Bebenek M, Shojaa M, Kohl M, Freiberger E, Sieber C, Von Stengel S. Whole-body electromyostimulation and protein supplementation favorably affect sarcopenic obesity in community dwelling older men at risk: the randomized controlled FranSO study. Clin Interv Aging. 2017;12:1503–13.
Kemmler W, Grimm A, Bebenek M, Kohl M, von Stengel S. Effects of combined whole-body electromyostimulation and protein supplementation on local and overall muscle/fat distribution in older men with sarcopenic obesity: the randomized controlled franconia sarcopenic obesity (FranSO) study. Calcif Tissue Int. 2018;103(3):266–77.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Hita-Contreras, F. (2019). Female Sarcopenic Obesity. In: Pérez-López, F. (eds) Postmenopausal Diseases and Disorders. Springer, Cham. https://doi.org/10.1007/978-3-030-13936-0_27
Download citation
DOI: https://doi.org/10.1007/978-3-030-13936-0_27
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-13935-3
Online ISBN: 978-3-030-13936-0
eBook Packages: MedicineMedicine (R0)