Abstract
The efficacy of several nanoparticles [silica (Si), nanoclays, fly ash and iron oxide (IO)] in stabilizing CO2 foam is studied via flow experiments in a microfluidic device. The resulting foam is characterized using modified bulk foam tests. Size and uniformity of nanoparticle (NP) dispersions are quantified using dynamic light scattering. Results indicate that the size distribution and surface charge of the particles are influential parameters on the stability and formability of the foam, which in turn have a direct relationship with oil recovery performance. Si, nanoclays and fly ash NPs assisted by surfactant mixtures generate stable foams and result in high ultimate oil recoveries (over 90%). Even though IO-surfactant mixtures generate foams with relatively inferior stability characteristics and ultimate recovery, approximately three quarters of the IO NPs are recovered once exposed to a magnetic field. Unlike nanoclays and fly ash, the use of Si and IO NPs as foam stabilizers results in significant improvements in recovery at much smaller pore-volumes injected (~10 PVIs).
Access provided by Autonomous University of Puebla. Download conference paper PDF
Similar content being viewed by others
Keywords
1 Introduction
Carbon dioxide (CO2) Enhanced Oil Recovery (EOR) provides potential opportunities for CO2 utilization and long-term storage [1, 2]. The relative low density and viscosity of CO2 may contribute to unfavorable volumetric sweep efficiencies resulting from severe channeling and gravity override [3]. CO2 foam has been proposed as an alternative to mitigate these shortcomings [4] but has not been widely implemented in field applications due to concerns related to foam instability. NPs may serve as effective foam stabilizers with minimal losses in the subsurface due to their relatively high adsorption energy values under reservoir conditions [5, 6]. Foam stability and formability impact foam mobility and sweep efficiency in porous media and result in various oil recovery performances [7]. In this work, a microfluidic device featuring a porous medium, fabricated to represent a Berea sandstone [8], is used to visualize and characterize NP-stabilized CO2 foam flow behavior, using silica (Si), nanoclays, by-product fly ash, and recyclable iron-oxide (IO) NPs. Foam properties are investigated using modified bulk foam tests, and their efficacy as an injectant is studied using displacement experiments in the microfluidic device.
2 Experiments
Four different types of nanoparticles are investigated: amorphous fumed silica T30 (unmodified, 100% SiOH coverage), nanoclays, fly-ash (two stage wet-grinded), and IO NPs (synthesized by a co-precipitation method). NPs are dispersed in deionized (DI) water and mixed with lauramidopropyl betaine (LAPB) and alpha-olefin sulfonate (AOS) surfactant mixtures. The NP/surfactant solution, CO2 gas and a 30,000 ppm sodium chloride brine are used as injectants. A Gullfaks Blend crude oil with a viscosity of 45.9 cp serves as the resident fluid. A Brookhaven Zeta PALS instrument is used to characterize particle size distribution and zeta potential at 20 °C. The stability and formability of foams stabilized by various NPs are quantified via modified bulk foam tests, where foam generation as a function of time is measured using a transparent graduated cylinder [8, 9]. The microfluidic device features a pore network with an area of 1.6 in. × 1.4 in., and has a porosity of approximately 40%, and a permeability of approximately 0.35 Darcy. The relationship between oil recovery and foam stability is analyzed using high resolution images of the porous medium during experiments. The recovered IO NPs are quantified using a HACH DR/4000U spectrophotometer.
3 Results
Particle diameters and polydispersity indices are presented in Table 1 [8, 9]. All NP samples have net charges (zeta-potential) lower than −30 mV, which indicates a high stability [10]. Results from modified bulk foam experiments are presented in Table 2. The porous medium is initially saturated with brine, followed by the injection of 40 pore volumes (PVs) of the crude oil. Prior to foam flooding, 8 PVs of brine are injected at a rate of 4 µL/min, resulting in a recovery of approximately 40%.
4 Discussion
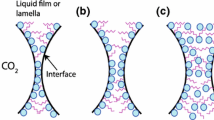
All four NPs improve foam stability compared to the use of surfactants alone. Differences in stability of the four NP-assisted foams may be explained by differences in partial particle flocculation, contact angle and the resulting rearrangements at the foam lamella [11]. The absorption of the surfactant mixtures on Si and fly ash NP surfaces may result in contact angles between CO2, aqueous phase and NP surfaces (60°–70°) that lead to an improved foam stability [12, 13]. In addition, arrangement of Si and fly ash NPs at the interfaces in the CO2-surfactant solution has a significant effect on maximum capillary pressure, which contributes to foam stability [14, 15]. In general, the improved stability of CO2 foams due to the use of NPs results in higher oil recoveries compared to foams assisted by surfactants only [16]. The addition of Si, fly ash and nanoclay NPs in the surfactant mixtures results in stable foams and relatively high ultimate recoveries of over 90% (Fig. 1). Even though the use of IO NPs in surfactant mixtures is not as effective in generating stable foams and recovering the resident fluid, approximately three quarters of the IO NPs are recovered at the outlet using a magnetic field. As seen in Fig. 1, the use of Si and IO NPs as foam stabilizers results in significant improvements in recovery after approximately ten pore-volumes injected (PVI), whereas significantly larger PVIs are needed to achieve similar recovery gains using fly ash and nanoclays.
5 Conclusions
This study focuses on the efficacy of four types of NPs in stabilizing CO2 foams and on the resulting improvements in oil recovery due to foam injection in a microfluidic device. Fly ash NPs, a by-product from coal fired power plants, are a promising stabilizer of CO2 foams. Their use in EOR processes may serve as a venue for their underground disposal and may improve the economics of CO2 utilization and storage by replacing more expensive materials. IO NPs do not appear to be as effective as fly ash in stabilizing CO2 foams; nevertheless, a significant portion of IO NPs may be recovered by subjecting the outflow stream to a magnetic field. The optimal choice of NPs as a foam stabilizer may depend not only on the stability of the resulting foam, but also on the PVI needed to recover the resident fluid and the associated material cost and operational expenditure.
References
Kalyanaraman, N., Arnold, C., Gupta, A., Tsau, J.S., Ghahfarokhi, R.B.: Stability improvement of CO2 foam for enhanced oil-recovery applications using polyelectrolytes and polyelectrolyte complex nanoparticles. J. Appl. Polym. Sci. 134(6) (2017)
Worthen, A.J., Bagaria, H.G., Chen, Y., Bryant, S.L., Huh, C., Johnston, K.P.: Nanoparticle-stabilized carbon dioxide-in-water foams with fine texture. J. Colloid Interface Sci. 391, 142–151 (2013)
Kim, I., Worthen, A.J., Johnston, K.P., DiCarlo, D.A., Huh, C.: Size-dependent properties of silica nanoparticles for Pickering stabilization of emulsions and foams. J. Nanopart. Res. 18(4) (2016)
Guo, H., Zitha, P.L.J., Faber, R., Buijse, M.: A novel alkaline/surfactant/foam enhanced oil recovery process. SPE J. 17(04), 1186–1195 (2012)
Yekeen, N., Manan, M.A., Idris, A.K., Padmanabhan, E., Junin, R., Samin, A.M.: A comprehensive review of experimental studies of nanoparticles-stabilized foam for enhanced oil recovery. J. Petrol. Sci. Eng. 164, 43–74 (2018)
Hua, X., Michael, A., Bevan, A.M., Frechette, J.: Competitive adsorption between nanoparticles and surface active ions for the oil-water interface. Langmuir 34(16), 4830–4842 (2018)
Yousef, Z.A., Almobarky, M.A., Schechter, D.S.: The effect of nanoparticle aggregation on surfactant foam stability. J. Colloid Interface Sci. 511, 365–373 (2018)
Guo, F., He, J., Johnson, P.A., Aryana, S.A.: Stabilization of CO2 foam using by-product fly ash and recyclable iron oxide nanoparticles to improve carbon utilization in EOR processes. Sustain. Energy Fuels 1, 814–822 (2017)
Guo, F., Aryana, S.A.: An experimental investigation of nanoparticle-stabilized CO2 foam used in enhanced oil recovery. Fuel 186, 430–442 (2016)
Hirasaki, G.J., Lawson, J.B.: Mechanisms of foam flow in porous media: apparent viscosity in smooth capillaries. Soc. Petrol. Eng. J. 25(02), 176–190 (1985)
Hunter, T.N., Pugh, R.J., Franks, G.V., Jameson, G.J.: The role of particles in stabilizing foams and emulsions. Adv. Coll. Interface. Sci. 137, 57–81 (2008)
Shaw, D.: Introduction to Colloid and Surface Chemistry, 4th edn. Butterworth-Heinemann (1992)
Denkov, D.A., Lvanov, B.I., Kralchevsky, A., Wasan, T.D.: A possible mechanism of stabilization of emulsions by solid particles. J. Colloid Interface Sci. 150, 589–593 (1992)
Kaptay, G.: Interfacial criteria for stabilization of liquid foams by solid particles. Colloids Surf. A Physicochem. Eng. Asp. 230(1–3), 67–80 (2004)
Singh, R., Mohanty, K.K.: Synergy between nanoparticles and surfactants in stabilizing foams for oil recovery. Energy Fuels 29(2), 467–479 (2015)
Worthen, A.J., Foster, L.M., Dong, J., Bollinger, J.A., Peterman, A.H., Pastora, L.E.: Synergistic formation and stabilization of oil-in-water emulsions by a weakly interacting mixture of zwitterionic surfactant and silica nanoparticles. Langmuir 30(4), 984–994 (2014)
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Nature Switzerland AG
About this paper
Cite this paper
Guo, F., Aryana, S.A. (2019). Nanoparticle-Stabilized CO2 Foam Flooding. In: Banerjee, S., Barati, R., Patil, S. (eds) Advances in Petroleum Engineering and Petroleum Geochemistry. CAJG 2018. Advances in Science, Technology & Innovation. Springer, Cham. https://doi.org/10.1007/978-3-030-01578-7_15
Download citation
DOI: https://doi.org/10.1007/978-3-030-01578-7_15
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-01577-0
Online ISBN: 978-3-030-01578-7
eBook Packages: Earth and Environmental ScienceEarth and Environmental Science (R0)