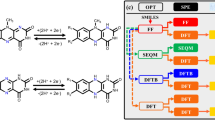
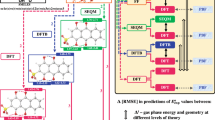
Abstract
Traditionally, chemists and material scientists rely on the slow and serendipitous trial-and-error process for discovering and developing new chemicals or materials. However, the conventional one-at-a-time, or one-by-one, methods are not capable of matching the pace of present-day material development. For example, in the area of drug discovery, it has been estimated theoretically that the number of possible drugs with molecular structures and weights attractive for pharmaceutical activity screening is 1018. This number is ~103 times larger than the number of chemical substances available from commercial sources or in-house collections. This number is also ~5×1010 times larger than that listed in the Chemical Abstracts Service (CAS) database. It is impossible to screen the pharmaceutical activities of such a large number of potential drugs by using traditional one-by-one methods. For organic substances, if each substance contains more than 30 atoms of H, C, O, N, and S elements, the number of potential stable molecular structures is expected to be ~1063. For inorganic substances, possibilities include ~3×103 binaries, ~7×104 ternaries, ~1×106 quaternaries, and ~6×1012 decanaries, which can be made from 75 useful and stable elements in the periodic table. These numbers exclude those with stoichiometric and structural diversity and different orders. Traditional one-by-one approaches will never be able to deal with the screening and optimization of these substances.
Access provided by Autonomous University of Puebla. Download to read the full chapter text
Chapter PDF
Similar content being viewed by others
Keywords
- Fuel Cell
- Oxygen Reduction Reaction
- Combinatorial Method
- Proton Exchange Membrane Fuel Cell
- Direct Methanol Fuel Cell
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
References
Potyrailo RA. High throughput screening of materials. Technical Report 2001; GE Research & Development Centre 2001CRD137.
Xiang XD, Sun X, Briceno G, Lou Y, Wang KA, Chang H et al. A combinatorial approach to materials discovery. Science 1995;268:1738–40.
Koinuma H, Takeuchi I. Combinatorial solid-state chemistry of inorganic materials. Nature 2004;3:429–38.
Maier WF, Stowe K, Sieg S. Combinatorial and high-throughput materials science. Angew Chem Int Ed 2007;46:6016–67.
Hogarth MP, Ralph TR. Catalysis for low temperature fuel cells, Part I. The cathode challenges. Platinum Metals Review 2002;46:3–14.
Hogarth MP, Ralph TR. Catalysis for low temperature fuel cells, Part II. The anode challenges. Platinum Metals Review 2002;46:117–35.
Ralph TR, Hogarth MP. Catalysis for low temperature fuel cells, Part III. The challenges of DMFCs. Platinum Metals Review 2002;46:146–64.
Liu HS, Song CJ, Zhang L, Zhang JJ, Wang HJ, Wilkinson DP. A review of anode catalysis in the direct methanol fuel cell. J Power Sources 2006;155:95–110.
Reddington E, Sapienza A, Gurau B, Viswanathan R, Sarangapani S, Smotkin ES, et al. Combinatorial electrochemistry: a highly parallel optical screening method for discovery of better electrocatalysts. Science 1998;280:1735–7.
Gurau B, Viswanathan R, Liu R, Lafrenz TJ, Ley KL, Smotkin ES. Structural and Electrochemcial characterization of binary, ternary, and quaternary platinum alloy catalysts for methanol electro-oxidation. J Phys Chem B 1998;102:9997–10003.
Jayaraman S, Hillier AC. Construction and reactivity screening of a surface composition gradient for combinatorial discovery of electro-oxidation catalysts. J Comb Chem 2004;6:27–31.
Jayaraman S, Hillier AC. Electrochemical synthesis and reactivity screening of a ternary composition gradient for combinatorial discovery of fuel cell catalysts. Meas Sci Technol 2005;16:5–13.
Strasser P, Fan Q, Devenney M, Weinberg WH. High throughput experimental and theoretical predictive screening of materials – a comparative study of search strategies for new fuel cell anode catalysts. J Phys Chem B 2003;107:11013–21.
Guerin S, Hayden BE, Lee CE, Mormiche C, Owen JR, Russell AE, et al. Combinatorial electrochemical screening of fuel cell electrocatalysts. J Comb Chem 2004;6:149–58.
Smotkin ES, Diaz-Morales RR. New electrocatalysts by combinatorial methods. Annu Rev Mater Res 2003;33:557–79.
Shah BC, Hillier AC. Imaging the reactivity of electro-oxidation catalysts with the scanning electrochemical microscope. J Electrochem Soc 2000;147:3043–8.
Fernandez JL, Bard AJ. Scanning electrochemical microscopy 47: Imaging electrocatalytic activity for oxygen reduction in an acidic medium by the tip generation-substrate collection mode. Anal Chem 2003;75:2967–74.
Jambunathan K, Hillier AC. Measuring electrocatalytic activity on a local scale with scanning differential electrochemical mass spectrometry. J Electrochem Soc 2003;150 (6):E312–20.
Jambunathan K, Jayaraman S, Hillier AC. A multielectrode electrochemical and scanning differential electrochemical mass spectrometry study of methanol oxidation on electrodeposited PtxRuy. Langmuir 2004;20:1856–63.
Shioyama H, Yamada Y, Ueda A, Kobayashi T. Screening of carbon supports for DMFC electrode catalysts by infrared thermography. Carbon 2003;41:579 –625.
Yamada Y, Ueda A, Shioyama H, Kobayashi T. High-throughput screening of PEMFC anode catalysts by IR thermography. Appl Surf Sci 2004;223:220–3.
Cooper JS, McGinn PJ. Combinatorial screening of thin film electrocatalysts for a direct methanol fuel cell anode. J Power Sources 2006;163:330–8.
Liu R, Smotkin ES. Array membrane electrode assemblies for high throughput screening of direct methanol fuel cell anode catalysts. J ElectroanalChem 2002;535:49–55.
He T, Kreidler E, Xiong L, Ding E. Combinatorial screening and nano-synthesis of platinum binary alloys for oxygen electroreduction. J Power Sources 2007;165:87–91.
Yang R, Bonakdarpour A, Bradley Eaton E, Stoffyn-Egli P, Dahn JR. Characterization and PEMFC testing of Pt1 xMx (M=Ru,Mo,Co,Ta,Au,Sn) anode electrocatalyst composition spreads. J Electrochem Soc 2007;154 (4):A275–82.
Liu JH, Jeon MK, Woo SI. High-throughput screening of binary catalysts for oxygen electroreduction. Appl Surf Sci 2006;252:2580–7.
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2008 Springer London
About this chapter
Cite this chapter
Liu, H., Zhang, J. (2008). Combinatorial Methods for PEM Fuel Cell Electrocatalysts. In: Zhang, J. (eds) PEM Fuel Cell Electrocatalysts and Catalyst Layers. Springer, London. https://doi.org/10.1007/978-1-84800-936-3_12
Download citation
DOI: https://doi.org/10.1007/978-1-84800-936-3_12
Publisher Name: Springer, London
Print ISBN: 978-1-84800-935-6
Online ISBN: 978-1-84800-936-3
eBook Packages: EngineeringEngineering (R0)