Abstract
Comorbidity presents significant problems to investigators engaged in the search for biomarkers of post-traumatic stress disorder (PTSD) because it means that multiple overlapping psychiatric phenotypes may be present in any PTSD sample. This chapter reviews research on an internalizing/externalizing model of post-traumatic psychopathology and discusses its relevance to the search for PTSD biomarkers. It focuses on two candidate biomarkers that have been studied extensively in relation to PTSD—basal cortisol level and startle reflex amplitude—but have yielded complicated and mixed results. Our review of the cortisol literature finds evidence for links between disorders of the internalizing spectrum and elevated levels of cortisol and links between externalizing psychopathology and low levels of cortisol. The review of the startle reflex literature reveals an association between exaggerated startle and fear-related disorders of the internalizing spectrum. These findings illustrate how individual differences in internalizing and externalizing comorbidity may moderate associations between these measures and PTSD diagnostic status. The chapter concludes with a discussion of the advantages of studying latent dimensions of comorbidity in future PTSD biomarker research.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
Introduction
Post-traumatic stress disorder (PTSD) is the product of an environmental pathogen (i.e., a traumatic stressor) operating on a variety of individual diatheses or predispositions. These diatheses span the spectrum of human variation in vulnerability to psychopathology and result in extensive heterogeneity in the phenotypic expression of the disorder. One manifestation of this is a diverse pattern of diagnostic comorbidity associated with PTSD. Brown et al. (1) assessed the co-occurrence of current and lifetime Diagnostic and Statistical Manual of Mental Disorders, fourth edition (DSM-IV) anxiety and unipolar mood disorders in 1,126 community outpatients and found that, of all the disorders assessed, PTSD showed the most prevalent and varied pattern of comorbidity. Of individuals with a current diagnosis of PTSD, 92% met criteria for another current Axis I disorder, with the most frequent being major depressive disorder (77%), generalized anxiety disorder (38%), and alcohol abuse/dependence (31%). Similarly, in studies of veterans receiving clinical services, 82% of those with a current diagnosis of PTSD met criteria for another Axis I disorder (2), and the National Vietnam Veterans Readjustment Study showed that 50% of veterans with PTSD had an additional Axis I diagnosis (3 ).
Comorbidity presents significant problems to investigators engaged in the search for biomarkers of PTSD. It undermines the assumption that PTSD is a discrete syndrome that is qualitatively and mechanistically distinct from other DSM-IV disorders. It also means that multiple overlapping psychiatric phenotypes may be present in any PTSD sample, which can obscure the search for markers that reliably distinguish cases from controls. The aims of this chapter are to review research on an internalizing/externalizing model of the phenotypic heterogeneity (i.e., clinical presentations) among individuals with PTSD and to discuss the relevance of this model for the search for PTSD biomarkers. We review the literature on two measures that have been the focus of extensive PTSD biomarker research—basal cortisol levels and startle reflex amplitude—and illustrate how individual differences in internalizing and externalizing comorbidity may moderate associations between these measures and PTSD diagnostic status.
The Internalizing and Externalizing Model of Psychiatric Comorbidity
The internalizing/externalizing model of psychiatric comorbidity proposes that patterns of behavioral disturbance and psychiatric symptoms among common mental disorders cohere along two underlying, or latent, dimensions. It is rooted in over 30 years of research in the area of childhood behavior disorders (cf.4,5) and has recently come to the fore in the adult psychopathology literature as the result of a series of influential factor-analytic studies of the structure of adult mental illness (6–12 ). Krueger, Kendler, Kessler, and others have shown that comorbid disorders tend to cohere along these dimensions, with the alcohol and substance-related disorders and antisocial personality disorder loading on the externalizing dimension and the unipolar mood and anxiety disorders falling on the internalizing dimension. In several studies, the latter has been subdivided into correlated factors termed anxious-misery (defined by major depression, dysthymia, generalized anxiety disorder) and fear (comprised of panic and phobic disorders; 8,12–14). This structure has demonstrated invariance across genders and multiple samples drawn at random from a larger sample (6,8) and has also been shown to account for patterns of diagnostic comorbidity in combat veterans (15 ). Twin and adoption studies have implicated genetic factors in the etiology of the externalizing and internalizing latent factors (16,17 ), and the magnitude of these genetic effects increases with the severity of the behavior problems on a given dimension (18 ).
The externalizing/internalizing model is consistent with other major models of comorbidity that posit that the co-occurrence of disorders among broad classes (e.g., the anxiety and unipolar mood disorders) is due largely to the fact that they emerge from a common diathesis (e.g., 19,20). This concept is supported by evidence suggesting that whereas much overlap exists in terms of the predisposing factors within a given spectrum of psychopathology, the manifestations of these diatheses differ considerably as a function of exposure to various environmental factors (e.g., trauma exposure, other life stressors, or developmental experiences). In other words, the different manifestations of these shared vulnerability dimensions are represented by the various DSM-IV diagnoses. This concept is in accord with a leading theoretical explanation for the high rate of co-occurrence of disorders within a spectrum of psychopathology (i.e., “Disorders A and B co-occur because they are both influenced by another underlying or causal factor C”; cf. 21), although further empirical work is needed to explore the influence of other possibilities (e.g., disorder A predisposes or causes disorder B; disorders A and B are associated because they share overlapping definitional criteria; disorders A and B should not be considered comorbid because they can be subsumed into a larger category that has been artificially split by the classification system).
Internalizing and Externalizing Ptsd Subtypes
Recent studies suggest that the internalizing/externalizing model may also be relevant to the understanding of patterns of comorbidity (i.e., phenotypic heterogeneity) among individuals with PTSD and their links to underlying biologic mechanisms and biomarkers (15,22–24 ). Specifically, through a series of cluster analytic studies of personality inventories completed by individuals with PTSD, Miller and colleagues found evidence of internalizing and externalizing subtypes of PTSD in both male and female samples totaling over 1,000 subjects. Summarizing across these three studies, Miller et al. found that one subtype, termed externalizing, was characterized by the tendency to express post-traumatic distress outwardly through antagonistic interactions with others and conflict with societal norms and values. Individuals in this subgroup had elevated rates of antisocial personality and substance-related disorders, endorsed high levels of anger and aggression, and produced personality inventory profiles defined by high disconstraint (i.e., impulsivity) coupled with high negative emotionality. They described themselves as easily upset, chronically stressed, and prone to act impulsively with little regard for the consequences of their actions. On measures of personality disorder features, they described themselves as tending toward exhibitionistic, manipulative, and unconventional behavior. In both studies of veterans in which data on premilitary characteristics were available, externalizers reported elevated rates of premilitary delinquency, suggesting that these characteristics may reflect the influence of externalizing personality traits that were present prior to the trauma.
In contrast, the internalizing subtype was characterized by tendencies to direct post-traumatic distress inwardly through shame, self-defeating/deprecating and anxious processes, avoidance, depression, and withdrawal. Across these three studies, individuals in this subtype were characterized by high rates of comorbid major depression and panic disorder, schizoid and avoidant personality disorder features, and personality profiles defined by high negative emotionality combined with low positive emotionality. Individuals in this subtype further described themselves as unenthusiastic, uninspired, easily fatigued, and lacking interests, but like externalizers, prone to experiencing frequent and intense negative emotions. They reported having few friends, feeling aloof and distant from others, and preferring to spend time alone. In contrast with the externalizers, internalizers reported that they tended to be self-effacing and humble and did not feel particularly special, admirable, or talented. They endorsed a restricted range of emotions in interpersonal settings and feelings of social inhibition, inadequacy, and hypersensitivity to negative evaluation.
These findings suggest that the internalizing/externalizing model of psychopathology, originally developed to account for covariation among broad classes of mental disorders (9; c.f.11), is relevant to the understanding of the heterogeneity of PTSD comorbidity. Moreover, similar patterns that can now be interpreted as reflecting individual differences in internalizing and externalizing processes were described in earlier cluster analytic studies of U.S. and Australian veterans with PTSD (25–27 ). Taken together, these findings suggest that the internalizing/externalizing model may be a useful heuristic for studying the influence of comorbidity on the relationship between PTSD and candidate biomarkers.
Relationship Between Ptsd and Cortisol and the Moderating Influence of Internalizing and Externalizing Comorbidity
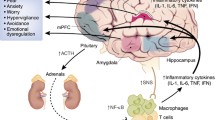
The hypothalamic-pituitary-adrenal (HPA) axis is a key neurobiological substrate of the stress response, and abnormalities in its functioning have long been implicated in the pathophysiology of PTSD (for reviews, see Refs. 28–30). Activity of this system is initiated by the release of corticotropin-releasing hormone (CRH) from the paraventricular nucleus of the hypothalamus. CRH acts as a neuropeptide in the production of adrenocorticotropic hormone (ACTH) in the pituitary and as a neurotransmitter in an elaborate network of interconnected neurons in the limbic system, brain stem, and cortex that are reactive to exogenous challenge and endogenous distress (for a review, see Ref. 31). Once CRH is released from the paraventricular nucleus into the hypophyseal portal blood of the median eminence, it is transported to the anterior lobe of the pituitary, where it stimulates the secretion of ACTH. ACTH in turn enters the general circulation and triggers the adrenal cortex to begin the synthesis of cortisol from low-density lipoprotein (LDL) cholesterol.
Cortisol has many peripheral effects on metabolic and immune processes. Centrally, it plays an important role in negative-feedback control of HPA axis activity by binding to glucocorticoid receptors of the hypothalamus and pituitary, which inhibits the release of CRH and ACTH, respectively (32,33 ). There is also evidence that adrenalectomy, which leads to a loss of negative feedback from peripheral cortisol, results in greatly increased numbers of CRH-containing storage vesicles in the central nucleus of the amygdala and bed nucleus of the stria terminalis, suggesting that cortisol also exerts inhibitory effects on CRH production in a variety of extrahypothalamic regions (34 ).
Cortisol has been the most commonly studied component of the HPA axis as a potential biomarker for PTSD because it is easily sampled, can be measured in urine, plasma, or saliva, and is a well-established marker of the body's response to stress. Unfortunately, 20 years of clinical studies that have examined the relationship between cortisol levels and PTSD have yielded complicated and mixed results. Many investigations have found lower levels of cortisol under baseline or nonstressful conditions using both urinary (35,36 ) and plasma samples (37–40 ) in individuals with PTSD compared to controls. Investigators have also reported evidence for a reduced rise in salivary cortisol on awakening in PTSD (41,42 ), and patients with PTSD have been found in many studies to respond to dexamethasone administration with enhanced cortisol suppression, suggesting a heightened sensitivity to cortisol in PTSD (38,41,43,44 ). These findings are not uniform, however. Other studies have found higher levels of cortisol in individuals with PTSD compared to controls in cerebral spinal fluid (CSF;45), urine (46–48 ), plasma (49 ), and saliva (50 ). Still others have shown no significant differences in salivary cortisol between groups differing in PTSD diagnostic status (51,52).
In an effort to integrate these discrepant findings, investigators have focused primarily on demographic differences between samples (e.g., age, sex, chronicity of PTSD, type of trauma) and variability in the methods used to assess cortisol, such as the time of day of sample collection, the source of the sample (e.g., plasma, urine, saliva, or CSF), or the conditions under which the sample was collected (baseline vs. pre- or post-psychological or physical stress). These factors account for a large proportion of variation in cortisol levels, and a recent meta-analysis of over 100 studies linking stress to HPA axis function showed that such factors contribute significantly to differential findings across studies (53 ). Results of that analysis also showed that, across studies, PTSD was associated with significantly lower daily levels of cortisol (Cohen's d = −0.34) and lower levels of cortisol post-dexamethasone administration (Cohen's d = −0.25) compared to control subjects exposed to the same stress.
One explanation for variation in study findings that has received little direct attention to date is the hypothesis that internalizing and externalizing comorbidity moderates the association between PTSD and cortisol levels. In many studies, the influence of comorbid disorders has not been well assessed or examined in the data analyses. When comorbidity has been factored into study designs or analyses, the focus has generally been on major depressive disorder because it is highly comorbid with PTSD and in non-PTSD samples generally associated with hyper- as opposed to hypo-cortisolism. In the section that follows, we review the literature on the differential relations between internalizing and externalizing psychopathology and cortisol levels to illustrate the moderating influences that these dimensions of comorbid psychopathology may have on the relationship between cortisol and PTSD.
Evidence for the Link Between Internalizing Disorders and Elevated Cortisol
Research on childhood behavior disorders has been the source of considerable evidence for a link between internalizing symptoms and elevated cortisol. Internalizing symptoms, such as social withdrawal and anxiety, have been associated with greater salivary cortisol levels under baseline conditions (54 ) and in response to a social stressor in children and adolescents (55,56 ). Behavioral inhibition, a trait related to shyness, withdrawal, and anxiety, evidences a similar association (57,58 ), and infants whose mothers have panic disorder have been found to have higher salivary cortisol levels compared to controls (59). Greater salivary cortisol reactivity in response to a social stressor has also been shown to prospectively predict the development of subsequent anxiety disorders (60 ).
Studies with adult participants have paralleled these findings. Specifically, adults with panic disorder have been found to exhibit higher levels of baseline plasma (61,62) and urinary and salivary ( 63) cortisol compared to controls. Numerous studies have linked basal hypercortisolism and dexamethasone nonsuppression to major depression (for a review, see Ref.64), and Miller, Chen, and Zhou's (53) meta-analysis estimated that the size of the depressed versus control group difference in postdexamethasone cortisol levels to be large (d = 1.13). Similar effects have been observed in individuals with generalized anxiety disorder (65,66). Taken together, these studies provide strong evidence for a link between disorders of the internalizing spectrum and elevated levels of cortisol.
Evidence for the Link Between Externalizing Disorders and Low Cortisol
A contrasting body of research suggests an association between disorders of the externalizing spectrum and abnormally low levels of cortisol. Theorists have long conjectured that the neural substrate for externalizing disorders involves diminished arousal (67–69), and since cortisol secretion is thought to partially reflect the arousal state of the organism, many investigators have focused on cortisol as a potential biomarker for this class of psychopathology. For example, Vanyukov et al., (70) found that young boys with antisocial fathers had lower levels of baseline salivary cortisol compared to controls, and their total number of conduct disorder symptoms was inversely correlated (r = −.20) with their cortisol levels. Similarly, investigators have reported associations between low basal cortisol concentrations and aggression toward peers (71), hostility to the teacher (72), and conduct disorder severity (70). Finally, salivary (73) and serum (74) cortisol has been shown to be inversely associated with psychopathic traits (i.e., callousness, lack of remorse) in older male adolescents/young adults.
Evidence for the Link Between Externalizing Disorders and Low Cortisol
A contrasting body of research suggests an association between disorders of the externalizing spectrum and abnormally low levels of cortisol. Theorists have long conjectured that the neural substrate for externalizing disorders involves diminished arousal (67–69), and since cortisol secretion is thought to partially reflect the arousal state of the organism, many investigators have focused on cortisol as a potential biomarker for this class of psychopathology. For example, Vanyukov et al., (70) found that young boys with antisocial fathers had lower levels of baseline salivary cortisol compared to controls, and their total number of conduct disorder symptoms was inversely correlated (r = −.20) with their cortisol levels. Similarly, investigators have reported associations between low basal cortisol concentrations and aggression toward peers (71), hostility to the teacher (72), and conduct disorder severity (70). Finally, salivary (73) and serum (74) cortisol has been shown to be inversely associated with psychopathic traits (i.e., callousness, lack of remorse) in older male adolescents/young adults.
Similar effects have also been observed in research with adults with externalizing disorders. For example, King, Jones, Scheuer, Curtis, and Zarcone (75) found that inpatient adults with substance use diagnoses exhibited lower baseline levels of plasma cortisol than controls. Interestingly, in that sample no participants met criteria for major depressive disorder, while in a second study of substance users with comorbid depression, elevated serum cortisol levels were found (76). There is also evidence that trait impulsivity, which is the personality dimension thought to underlie propensities toward externalizing disorders, may be inversely associated with cortisol levels. For example, Wang et al. (77) found that novelty seeking (a personality construct closely related to impulsivity) was inversely associated with urinary cortisol (r = −.43) in male Vietnam veterans with PTSD. King et al. (75) reported similar findings in a community sample control group. In sum, considerable evidence suggests that children and adults with problems in the externalizing domain exhibit low basal cortisol, and these findings align with theories that emphasize the role of hypo-arousal in the etiology of disorders in this spectrum (e.g.,69). On the other hand, high levels of basal cortisol have often been found in association with disorders of the anxious-misery spectrum (e.g., major depression and generalized anxiety disorder) and problems in the domain of excessive behavioral inhibition.
The Startle Response
Like basal cortisol, the startle response has been the focus of extensive research as a possible biomarker for PTSD. Exaggerated startle responding has been recognized as a core symptom of PTSD since the earliest descriptions of combat soldiers suffering adverse effects of exposure to the stress of combat. Many individuals with PTSD report extreme reactions to startling events in the environment. They may report flinching or recoiling in response to an unexpected touch or springing awake in response to an unexpected noise in the night and being unable to fall back to sleep. Combat veterans describe “hitting the dirt” or “ducking for cover” at the sound of a car backfiring. In each instance, the initial startle response may be followed by a cascade of anxious arousal symptoms, with recovery taking minutes to hours.
From a psychophysiological perspective, the startle response is a constellation of reflexive motor movements, phasic autonomic responses, and voluntary orienting responses that occur in response to any sudden, intense change in stimulus intensity. The reflexive component of the reaction begins with an eyeblink between 20 and 50 ms after the onset of a startle-eliciting stimulus (e.g., car backfiring) and spreads distally throughout the body. In humans, it is measured via electromyography (EMG) recordings of the contraction of the orbicularis oculi muscle, which closes the eyelid. Startle-eliciting stimuli are typically loud noises presented over headphones, and the magnitude of the muscle contraction is the primary measure of interest. Secondary, longer latency autonomic responses include heart rate acceleration and skin conductance increases. These begin within a second after the onset of a startling stimulus and typically peak several seconds later.
The startle response has attracted considerable attention as a potential PTSD biomarker. Exaggerated startle is unique among the PTSD symptoms in terms of the degree of correspondence between the clinical symptom that occurs in humans and the behavioral analogue that can studied in the clinical psychophysiology lab or using animal models. Perhaps because of this, more is known about the neurocircuitry and neuromodulators of startle than for any other symptom of the disorder. Two primary systems in the brain have been implicated as possible mechanisms for the symptom of exaggerated startle in PTSD: the locus coeruleus/norepinephrine system and the hypothalamic/CRH system. Evidence for these links includes studies showing that lesions of the locus coeruleus and drugs that inhibit its activity decrease startle reactivity, whereas drugs that increase locus coeruleus activity have the opposite effect (78). Likewise, CRH administration produces a pronounced, dose-dependent enhancement of startle that can be blocked by pretreatment with a CRH receptor antagonist (e.g.,79,80).
Although these findings point to the promise of the startle response as a possible biomarker of PTSD, clinical laboratory studies have yielded mixed findings regarding the validity of exaggerated startle as a symptom of PTSD. Pole (81) recently conducted a meta-analysis of 20 studies that compared samples of individuals with and without PTSD on measures of startle responding and found that the effect size for the eyeblink reflex, weighted to control for sample size differences, was significant but modest (r =.13). Of those 20 studies, approximately half showed significant positive group differences in eyeblink startle amplitude, with some showing quite large effects (i.e., r >.50; 82,83). These findings suggest that there may be one or more important moderating variables that have not been consistently addressed by procedures used in past startle studies.
One variable that has received considerable research attention in studies of startle in PTSD is contextual anxiety, with the idea being that exaggerated startle in PTSD is a context- or state-dependent phenomenon related to anxiety processes (84,85). This hypothesis follows from the seminal research of Michael Davis and colleagues (e.g., 86,87) on the neurobiology of fear, anxiety, and startle, which showed that the amplitude of the startle response is potentiated by both exposure to contextual threat (i.e., anxiety; as in returning to the location of previous aversive conditioning) and explicit threat (i.e., fear; as in exposure to a conditioned stimulus signaling imminent shock). In line with this, a growing body of clinical research with PTSD patients suggests that differences between PTSD and non-PTSD groups are most reliably observed under test conditions involving the distal anticipation of an aversive stimulus and are not observed under conditions involving proximal threat (for a review, see Ref. 88). For example, Grillon et al. (85) examined startle responses in veterans with and without PTSD during an initial laboratory session that involved no aversive manipulation followed several days later by startle testing during an aversive conditioning procedure that involved anticipation of a mild shock. Significant group differences in baseline startle amplitude were observed only during session 2, suggesting that group effects were linked to the anxiogenic context in which the shock conditioning took place. Consistent with the possibility that exaggerated startle in PTSD is linked exclusively to the neurobiological system underlying contextual anxiety and not to the system underlying fear, no group differences in the fear response to presentation of conditioned threat cues (i.e., a CS+) were found.
Effects of Comorbidity on Baseline and Overall Startle Amplitude
Comorbidity may also serve a moderating role in the relationship between PTSD and amplitude of the startle response. Several studies have found exaggerated startle responding in samples of individuals with other anxiety disorders, which raises questions about the specificity of the measure as a possible biomarker of PTSD. For example, exaggerated baseline startle has been observed in several studies of patients with panic disorder (89–93). Grillion and colleagues (94) examined startle as a possible vulnerability marker among offspring of parents with anxiety disorders or alcoholism and found that the magnitude of baseline startle was greater in children with a parental history of anxiety than in children without a parental history of anxiety. Similar findings were observed in a sample of individuals at risk for depression by virtue of their parents' depression diagnoses (95). In addition, Cuthbert et al. (96) reported that baseline startle responses were significantly larger among anxiety disorder patients with depression than for anxiety patients without depression, suggesting an additive effect of internalizing comorbidity on startle amplitude.
Other studies suggested that startle amplitude may tap individual differences in level of trait fearfulness. For example, rats that differ in fearfulness can be distinguished on the basis of their behavioral reactions to startling stimuli, with fearful rats exhibiting more extreme responses (97). In humans, individuals who score high on Cloninger's measure of harm avoidance (i.e., “a heritable tendency to respond intensely to aversive stimuli and to learn to avoid punishment, novelty and non-reward passively”; 98) exhibit larger overall startle response amplitudes compared to their counterparts who score low on the measure (99). Together, these findings suggest a link between exaggerated startle and psychopathology of the internalizing spectrum broadly and fear-related disorders more specifically.
Conclusions and Directions for Future Research
Comorbidity of PTSD presents problems on multiple levels for clinicians and researchers alike. In the clinical arena, individuals with comorbid Axis I or II disorders have been shown to have more severe PTSD symptoms (e.g., 100–102) and poorer responses to treatment (e.g.,103,104). For researchers, PTSD comorbidity often represents a nuisance variable addressed through post hoc statistical controls or challenging (and arguably ill-advised) efforts to select “pure” PTSD cases from highly comorbid samples. The heterogeneity within PTSD also means that there is potential for multiple overlapping phenotypes to occur within any sample of individuals with the disorder. As the foregoing literature review suggests, the diagnostic composition of any sample and the proportion of cases with predominantly internalizing versus externalizing comorbidity can have a substantial influence on associations between candidate biomarkers and measures of PTSD.
PTSD is not alone among psychiatric disorders in this regard. Psychiatric epidemiology studies suggest that similar heterogeneity exists in some, but not all, mental disorders with the determining factor being the breadth of comorbidity associated with the index disorder. PTSD shows a particularly severe and diverse pattern of diagnostic comorbidity with frequently co-occurring conditions ranging from those of the internalizing spectrum to the externalizing spectrum. Certain other disorders, such as alcohol abuse/dependence, show similar patterns of comorbidity. Cloninger (98) emphasized this when he advanced a two-class typology of alcohol dependence. In his model, type I alcoholics (i.e., an internalizing type) were defined as individuals who use alcohol in association with heightened negative affectivity to avoid, dampen, or escape those states. In type II alcoholism (i.e., an externalizing type), alcohol problems were thought to stem from propensities toward sensation seeking and risky or uninhibited behavior. On the other hand, other psychiatric disorders with a narrower range of comorbidity—and more distinct and circumscribed phenotypes—rarely co-occur with each other. For example, Kessler and colleagues (7) reported that the internalizing spectrum disorders obsessive-compulsive disorder and separation anxiety disorder were negatively correlated with the externalizing syndromes drug dependence and conduct disorder. Generalizing from this, one would not expect to find large numbers of externalizers among samples composed of individuals with protoypic internalizing diagnoses, especially those related to pathological fear or vice versa (e.g., antisocial personality disorder is not a problem commonly found among individuals with simple phobia).
The foregoing literature review suggests that exaggerated startle, low basal cortisol, and perhaps any candidate biomarkers for PTSD are pleiotropic, meaning that the same marker may relate to the occurrence of several different diagnostic phenotypes. We know that psychiatric disorders do not co-occur by chance but rather cohere along latent dimensions of psychopathology. By extension, we should expect most psychiatric biomarkers to correlate with disorders within a spectrum of psychopathology rather than with individual diagnoses, as basal cortisol and startle appear to. In future studies, it may be advantageous to model associations between biomarkers and phenotypes using analytic approaches that better map onto the complex nature of these associations. Latent variable methods would be well suited for the study of pleiotropic biomarkers because they can be used to model the common factor underlying a broad class of disorders as well as the unique factors that predict individual disorders. This permits the investigator to examine separately (1) the strength of associations between the candidate biomarker and the common factor underlying a set of diagnostic indicators and (2) the strength of the association between the biomarker and individual diagnostic indicators. Furthermore, the latent factors underlying co-occurring diagnoses can be conceptualized as endophenotypic traits, which represent purer, error-free measures of the dimension underlying the diagnoses of interest. As a result, one can expect latent dimensions of psychopathology to map more directly and completely onto their biologic substrate and yield substantially increased predictive power for biomarker association analyses compared to analyses that focus on identifying biomarkers of individual disorders.
References
Brown, T. A. , Campbell, L. A. , Lehman, C. L. , Grisham, J. R. , and Mancill, R. B. (2001) Current and lifetime comorbidity of the DSM-IV anxiety and mood disorders in a large clinical sample. J Abnorm Psychol 110, 585–99.
Orsillo, S. M. , Weathers, F. W. , Litz, B. T. , Steinberg, H. R. , Huska, J. A. , and Keane, T. M. (1996) Current and lifetime psychiatric disorders among veterans with war zone-related posttraumatic stress disorder. J Nerv Ment Dis 184, 307–13.
Kulka, R. A. , Schlenger, W. E. , Fairbank, J. A. , Hough, R. L. , Jordan, B. K. , Marmar, C. R. , and Weiss, D. S. (1990). Trauma and the Vietnam War Generation: Report on the Findings from the National Vietnam Veterans Readjustment Study. New York: Brunner/Mazel.
Achenbach, T. M. , and Edelbrock, C. S. (1978) The classification of child psychopathology: a review and analysis of empirical efforts. Psychol Bull 85, 1275–1301.
Achenbach, T. M. , and Edelbrock, C. S. (1984) Psychopathology of childhood. Annu Rev Psychol 35, 227–56.
Kendler, K. S. , Prescott, C. A. , Myers, J. , and Neale, M. C. (2003) The structure of genetic and environmental risk factors for common psychiatric and substance use disorders in men and women. Arch Gen Psychiatry 60, 929–37.
Kessler, R. C. , Chiu, W. T. , Demler, O. , Merikangas, K. R. , and Walters, E. E. (2005) Prevalence, severity, and comorbidity of 12-month DSM-IV disorders in the National Comorbidity Survey Replication. Arch Gen Psychiatry 62, 617–27.
Krueger, R. F. (1999) The structure of common mental disorders. Arch Gen Psychiatry 56, 921–26.
Krueger, R. F. , Caspi, A. , Moffitt, T. E. , and Silva, P. A. (1998) The structure and stability of common mental disorders (DSM-III-R): a longitudinal-epidemiological study. J Abnorm Psychol 107, 216–27.
Krueger, R. F. , Chentsova-Dutton, Y. E. , Markon, K. E. , Goldberg, D. , and Ormel, J. (2003) A cross-cultural study of the structure of comorbidity among common psychopathological syndromes in the general health care setting. J Abnorm Psychol 112, 437–47.
Krueger, R. F. , McGue, M. , and Iacono, W. G. (2001) The higher-order structure of common DSM mental disorders: internalization, externalization, and their connections to personality. Pers Indiv Differ 30, 1245–59.
Vollebergh, W. A. M. , Idema, J. , Bijl, R. V. , de Graaf, R. , Smit, F. , and Ormel, J. (2001) The structure and stability of common mental disorders: the NEMESIS Study. Arch Gen Psychiatry 58, 597–603.
Cox, B. J. , Clara, I. P. , and Enns, M. W. (2002) Posttraumatic stress disorder and the structure of common mental disorders. Depress Anxiety 15, 168–71.
Slade, T. , and Watson, D. (2006) The structure of common DSM-IV and ICD-10 mental disorders in the Australian general population. Psychol Med 36, 1593–1600.
Miller, M. W. , Fogler, J. M. , Wolf, E. J. , Kaloupek, D. G. , and Keane, T. M. , (2008) The internalizing and externalizing structure of psychiatric comorbidity in combat veterans. J Trauma Stress 21, 58–65.
Krueger, R. F. , Hicks, B. M. , Patrick, C. J. , Carlson, S. R. , Iacono, W. G. , and McGue, M. (2002) Etiologic connections among substance dependence, antisocial behavior and personality: modeling the externalizing spectrum. J Abnorm Psychol 111, 411–24.
Deater-Deckard, K. , and Plomin, R. (1999) An adoption study of the etiology of teacher and parent reports of externalizing behavior problems in middle childhood. Child Dev 70, 144–54.
Gjone, H. , Stevenson, J. , and Sundet, J. M. (1996) Genetic influence on parent-reported attention-related problems in a Norwegian general population twin sample. J Am Acad Child Adolesc Psychiatry 35, 588–96.
Barlow, D. H. , Chorpita, B. F. , and Turovsky, J. (1996) Fear, panic, and disorders of emotion. In D. A. Hope, Current Theory and Research in Motivation. Nebraska Symposium on Motivation 43. Lincoln: University of Nebraska Press.
Clark, D. A. , Steer, R. A. , and Beck, A. T. (1994) Common and specific dimensions of self-reported anxiety and depression: implications for the cognitive and tripartite models. J Abnorm Psychol 103, 645–54.
Frances, A. J. , Widiger, T. A. , and Fyer, M. R. (1990) The influence of classification methods on comorbidity. In J. D. Maser and C. R. Cloninger, Comorbidity of Mood and Anxiety Disorders (pp. 41–59). Washington, DC: American Psychiatric Association.
Miller, M. W. , Greif, J. L. , and Smith, A. A. (2003) Multidimensional Personality Questionnaire profiles of veterans with traumatic combat exposure: internalizing and externalizing subtypes. Psychol Assessment 15, 205–15.
Miller, M. W. , Kaloupek, D. G. , Dillon, A. L. , and Keane, T. M. (2004) Externalizing and internalizing subtypes of combat-related PTSD: a replication and extension using the PSY-5 Scales. J Abnorm Psychol 113, 636–45.
Miller, M. W. , and Resick, P. A. (2007) Internalizing and externalizing subtypes of female sexual assault survivors: implications for the understanding of complex PTSD. Behav Ther 38, 58–71.
Forbes, D. , Creamer, M. , Allen, N. , Elliott, P. , McHugh, T. , Debenham, P. , and Hopwood, M. (2003) MMPI-2 based subgroups of veterans with combat-related PTSD. J Nerv Ment Dis 191, 531–537.
Hyer, L. , Davis, H. , Albrecht, W. , Boudewyns, P. , and Woods, G. (1994) Cluster analysis of MCMI and MCMI-II on chronic PTSD victims. J Clin Psychol 50, 502–15.
Piekarski, A. M. , Sherwood, R. , and Funari, D. J. (1993) Personality subgroups in an inpatient Vietnam veteran treatment program. Psychol Rep 72, 667–74.
Yehuda, R. (2001) Biology of posttraumatic stress disorder. J Clin Psychiat 62, 41–46.
Yehuda, R. (1997) Sensitization of the hypothalamic-pituitary-adrenal axis in posttraumatic stress disorder. Ann NY Acad Sci 821, 57–75.
Kaskow, J. W. , Baker, D. , and Geracioti, T. D. (2001) Corticotropin-releasing hormone in depression and post-traumatic stress disorder. Peptides 22, 845–51.
Lovallo, W. R. , and Thomas, T. L. (2000) Stress hormones in psychophysiological research. In J. T. Cacioppo, L. G. Tassinary, and G. G. Bernston, Handbook of Psychophysiology. 2nd ed. New York: Cambridge University Press.
McCann, S. M. (1988) The anterior pituitary and hypothalamus. In J. E. Griffin and S. R. Ojeda, eds. Textbook of Endocrine Physiology. New York: Oxford University Press.
Orth, D. N. , Kovacs, W. J. , and DeBold, C. R. (1992) The adrenal cortex. In J. D. and Wilson D. W. Foster, Textbook of Endocrinology. Philadelphia: Saunders.
Merchenthaler, I. (1984) Corticotropin-releasing factor (CRF)-like immunoreactivity in the rat central nervous system: extrahypothalamic distribution. Peptides 5, 53–69.
Yehuda, R. , Soutwick, S. M. , Nussbaum, G. , Wahby, V. , Giller, E. L. , and Mason, J. W. (1990) Low urinary cortisol excretion in patient with posttraumatic stress disorder. J Nerv Ment Dis 178, 366–69.
Yehuda, R. , Kahana, B. , Binder-Brynes, K. , Southwick, S. M. , Mason, J. W. , and Giller, E. L. (1995) Low urinary cortisol excretion in Holocaust survivors with posttraumatic stress disorder. Am J Psychiatry 152, 982–86.
Boscarino, J. A. (1996) Posttraumatic stress disorder, exposure to combat, and lower plasma cortisol among Vietnam veterans: findings and clinical impressions. J Consult Clin Psychol 64, 191–201.
Griffin, M. G. , Resick, P. A. , and Yehuda, R. (2005) Enhanced cortisol suppression following dexamethasone administration in domestic violence survivors. Am J Psychiatry 162, 1192–99.
Olff, M. , Güzelcan, Y. , de Vries, G. -J. , Assies, J. , and Gersons, B. P. R. (2006) HPA- and HPT-axis alterations in chronic posttraumatic stress disorder. Psychoneuroendocrinology 31, 1220–30.
Yehuda, R. , Teicher, M. H. , Trestman, R. L. , Levengood, R. A. , and Siever, L. J. (1996) Cortisol regulation in posttraumatic stress disorder and major depression: a chronobiological analysis. Biol Psychiatry 40, 79–88.
de Kloet, C. S. , Vermetten, E. , Heijnen, C. J. , Geuze, E. , Lentjes, E. G. W. M. , and Westenberg, H. G. M. (2007) Enhanced cortisol suppression in response to dexamethasone administration in traumatized veterans with and without posttraumatic stress disorder. Psychoneuroendocrinlogy 32, 215–26.
Wessa, M. , Rohleder, N. , Kirschbam, C. , and Flor, H. (2006) Altered cortisol awakening response in posttraumatic stress disorder. Psychoneuroendocrinology 31, 209–15.
Stein, M. B. , Yehuda, R. , Koverola, C. , and Hanna, C. (1997) Enhanced dexamethasone suppression of plasma cortisol in adult women traumatized by childhood sexual abuse. Biol Psychiatry 42, 680–86.
Yehuda, R. , Southwick, S. M. , Krystal, J. M. , Charney, D. S. , and Mason, J. W. (1993) Enhanced suppression of cortisol following dexamethasone administration in combat veterans with posttraumatic stress disorder and major depressive disorder. Am J Psychiatry 150, 83–86.
Baker, D. G. , Ekhator, N. N. , Kasckow, J. W. , Dashevsky, B. , Horn, P. S. , Bednarik, L. , and Geracioti, T. D. (2005) Higher levels of basal serial CSF cortisol in combat veterans with posttraumatic stress disorder. Am J Psychiatry 162, 992–94.
Lemieux, A. M. , and Coe, C. L. (1995) Abuse-related posttraumatic stress disorder: evidence for chronic neuroendocrine activation in women. Psychosom Med 57, 105–15.
Maes, M. , Lin, A. , Bonaccorso, S. , van Hunsel, F. , Van Gastel, A. , Delmeire, L. , Biondi, M. , Bosmans, E. , Kenis, G. , and Scharpe, S. (1998) Increased 24-hour urinary cortisol excretion in patients with post-traumatic stress disorder and patients with major depression, but not in patients with fibromyalgia. Acta Psychiatr Scand 98, 328–35.
Pitman, R. K. , and Orr, S. P. (1990) Twenty-four hour urinary cortisol and catecholamine excretion in combat-related posttraumatic stress disorder. Biol Psychiatry 27, 245–47.
Liberzon, I. , Abelson, J. L. , Flagel, S. B. , Raz, J. , and Young, E. A. (1999) Neuroendocrine and psychophysiologic responses in PTSD: a symptom provocation study. Neuropsychopharmacology 21, 40–50.
Lindley, S. E. , Carlson, E. B. , and Benoit, M. (2004) Basal and dexamethasone suppressed salivary cortisol concentrations in a community sample of patients with posttraumatic stress disorder. Biol Psychiatry 55, 940–45.
Young, E. A. , and Breslau, N. (2004) Cortisol and catecholamines in posttraumatic stress disorder: an epidemiologic community study. Arch Gen Psychiatry 61, 394–401.
Young, E. A. , and Breslau, N. (2004) Saliva cortisol in posttraumatic stress disorder: a community epidemiologic study. Biol Psychiatry 56, 205–9.
Miller, G. E. , Chen, E. , and Zhou, E. S. (2007) If it goes up, must it come down? Chronic stress and the hypothalamic-pituitary-adrenocortical axis in humans. Psychol Bull 133, 24–45.
Greaves-Lord, K. , Ferdinand, R. F. , Oldehinkel, A. J. , Sondeijker, F. E. P. L. , Ormel, J. , and Verhulst, F. C. (2007) Higher cortisol awakening response in young adolescents with persistent anxiety problems. Acta Psychiatr Scand 116, 137–44.
Klimes-Dougan, K. , Hastings, P. D. , Granger, D. A. , Usher, B. A. , and Zahn-Waxler, C. (2001) Adrenocortical activity in at-risk and normally developing adolescents: individual differences in salivary cortisol basal levels, diurnal variation, and responses to social challenges. Dev Psychopathol 13, 695–719.
Granger, D. A. , Weisz, J. R. , and Kauneckis, D. (1994) Neuroendocrine reactivity, internalizing behavior problems, and control-related cognitions in clinic-referred children and adolescents. J Abnorm Psychol 103, 267–76.
Kagan, J. , Reznick, J. S. , and Snidman, N. (1988) Biological bases of childhood shyness. Science 240, 167–71.
Schmidt, L. A. , Fox, N. A. , Rubin, K. H. , Sternberg, E. M. , Gold, P. W. , Smith, C. C. , and Schulkin, J. (1997) Behavioral and neuroendocrine responses in shy children. Dev Psychobiol 30, 127–140.
Warren, S. L. , Gunnar, M. R. , Kagan, J. , Anders, T. F. , Simmens, S. J. , Rones, M. , Wease, S. , Aron, E. , Dahl, R. , and Sroufe, A. (2003) Maternal panic disorder: infant temperament, neurophysiology, and parenting behaviors. J Am Acad Child Adolesc Psychiatry 42, 814–25.
Granger, D. A. , Weisz, J. R. , McCracken, J. T. , Ikeda, S. G. , and Douglas, P. (1996) Reciprocal influences among adrenocortical activation, psychosocial processes, and the behavioral adjustment of clinic-referred children. Child Dev 67, 3250–62.
Abelson, J. L. , Khan, S. , Liberzon, I. , and Young, E. A. (2007) HPA axis activity in patients with panic disorder: review and synthesis of four studies. Depress Anxiety 24, 66–76.
Marshall, R. D. , Blanco, C. , Printz, D. , Liebowitz, M. R. , Klein, D. F. , and Coplan, J. (2002) A pilot study of noradrenergic and HPA axis functioning in PTSD vs. panic disorder. Psychiatry Res 110, 219–30.
Bandelow, B. , Wedekind, D. , Sandvoss, V. , Broocks, A. , Hajak, G. , Pauls, J. , Peter, H. , and Rüther, E. (2000) Diurnal variation of cortisol in panic disorder. Psychiatry Res 95, 245–50.
Holsboer, F. (2000) The corticosteroid receptor hypothesis of depression. Neuropsychopharmacology 23, 477–501.
Schweizer, E. E. , Swenson, C. M. , Winokur, A. , Rickels, K. , and Maislin, G. (1986). The dexamethasone suppression test in generalised anxiety disorder. Br J Psychiatry 149, 320– 22.
Tiller, J. W. , Biddle, N. , Maguire, K. P. , and Davies, B. M. (1988) The dexamethasone suppression rest and plasma dexamethasone in generalized anxiety disorder. Biol Psychiatry 23, 261–70.
Quay, H. C. (1965) Psychopathic personality as pathological stimulation-seeking. Am J Psychiatry 122, 180–83.
Hare, R. D. (1970) Psychopathy: Theory and Research. New York: Wiley.
Zuckerman, M. (1999) Vulnerability to Psychopathology: A Biosocial Model. Washington, DC: American Psychiatric Press.
Vanyukov, M. M. , Moss, H. B. , Plail, J. A. , Blackson, T. , Mezzich, A. C. , and Tarter, R. E. (1993) Antisocial symptoms in preadolescent boys and their parents: associations with cortisol. Psychiatry Res 46, 9–17.
Tennes, K. , Kreye, M. , Avitable, N. , and Wells, R. (1986) Behavioral correlates of excreted catecholamines and cortisol in second-grade children. J Am Acad Child Adolesc Psychiatry 25, 764–70.
Tennes, K. , and Kreye, M. (1985) Children's adrenocortical responses to classroom activities and tests in elementary school. Psychosom Med 47, 451–60.
Burke, J. D. , Loeber, R. , and Lahey, B. B. (2007) Adolescent conduct disorder and interpersonal callousness as predictors of psychopathy in young adults. J Clin Child Adolesc Psychol 36, 334–46.
Holi, M. , Auvinen-Lintunen, L. , Lindberg, N. , Tani, P. , and Virkkunen, M. (2006) Inverse correlation between severity of psychopathic traits and serum cortisol levels in young adult violent male offenders. Psychopathology 39, 102–4.
King, R. J. , Jones, J. , Scheuer, J. W. , Curtis, D. , and Zarcone, V. P. (1990) Plasma cortisol correlates of impulsivity and substance abuse. Pers Indiv Differ 11, 287–91.
Wisniewski, A. B. , Brown, T. T. , John, M. , Cofranceso, J. , Golub, E. T. , Ricketts, E. P. , Wand, G. , and Dobs, A. S. (2006) Cortisol levels and depression in men and women using heroin and cocaine. Psychoneuroendocrinology 31, 250–55.
Wang, S. , Mason, J. , Charney, D. , Yehuda, R. , Riney, S. , and Southwick, S. (1997) Relationships between hormonal profile and novelty seeking in combat-related posttraumatic stress disorder. Biol Psychiatry 41, 145–51.
Davis, M. , Redmond, D. E. , Jr, and Baraban, J. M. (1979) Noradrenergic agonists and antagonists: effects on conditioned fear as measured by the potentiated startle paradigm. Psychopharmacology 65, 111–18.
Swerdlow, N. R. , Britton, K. T. , and Koob, G. F. (1989) Potentiation of acoustic startle by corticotropin-releasing factor (CRH) and by fear are both reversed by a-helical CRH(9–41). Neuropsychopharmacology 2, 285–92.
Risbrough V. B. , Hauger, R. L. , Roberts, A. J. , Vale, W. W. , and Geyer, M. A. (2004) Corticotropin-releasing factor receptors CRF1 and CRF2 exert both additive and opposing influences on defensive startle behavior. J Neurosci 24, 6545–52.
Pole, N. (2007). The psychophysiology of posttraumatic stress disorder: a meta-analysis. Psychol Bull 137, 725–46.
Morgan, C. A. , Grillon, C. , Southwick, S. M. , Davis, M. , and Charney, D. S. (1995) Fear-potentiated startle in posttraumatic stress disorder. Biol Psychiatry 38, 378–85.
Morgan, A. , Grillon, C. A. , Southwick, S. M. , Davis, M. , and Charney, D. S. (1996) Exaggerated acoustic startle reflex in Gulf War veterans with posttraumatic stress disorder. Am J Psychiatry 153, 64–68.
Grillon, C. , and Morgan, C. A. (1999) Fear-potentiated startle conditioning to explicit and contextual cues in Gulf War veterans with posttraumatic stress disorder. J Abnorm Psychol 108, 134–42.
Grillon, C. , Morgan, C. A. , Davis, M. , and Southwick, S. M. (1998) Effect of darkness on acoustic startle in Vietnam veterans with PTSD. Am J Psychiatry 155, 812–17.
Davis, M. , Walker, D. L. , and Lee, Y. (1997) Role of the amygdala and bed nucleus of the stria terminalis in fear and anxiety measured with the acoustic startle reflex. Ann N Y Academy of Sci 821, 305–33.
Davis, M. Walker, D. L. , and Lee, Y. (1999) Neurophysiology and neuropharmacology of startle and its affective modification. In M. Dawson, A. Schell, and M. Bohmelt, eds. , Startle Modification: Implications for Neuroscience, Cognitive Science, and Clinical Science (pp. 95–113). Cambridge: Cambridge University Press.
Orr, S. P. , Metzger, L. J. , Miller, M. W. , and Kaloupek, D. G. (2004) Psychophysiological assessment of posttraumatic stress disorder. In J. P. Wilson and T. M. Keane, eds. , Assessing Psychological Trauma and PTSD. 2nd ed. , pp. 289–343. New York: Guilford.
Buhlmann, U. , Wilhelm, S. , Deckersbach, T. , Rauch, S. L. , Pitman, R. K. , and Orr, S. P. (2007) Physiologic responses to loud tones in individuals with obsessive-compulsive disorder. Psychosom Med 69, 166–72.
Kumai, V. , Kaviani, H. , Raven, P. W. , Gray, J. A. , and Checkley, S. A. (2001) Enhanced startle reactions to acoustic stimuli in patients with obsessive-compulsive disorder. Am J Psychiatry 158, 134–36.
Grillon, C. , Ameli, R. , Goddard, A. , Woods, S. W. , and Davis, M. (1994) Baseline and fear-potentiated startle in panic disorder patients. Biol Psychiatry 35, 431–39.
Ludewig, S. , Geyer, M. A. , Ramseier, M. , Vollenweider, F. X. , Rechsteiner, E. , and Cattapan-Ludewig, K. (2005) Information-processing deficits and cognitive dysfunction in panic disorder. J Psychiatry Neurosci 30, 37–43.
Melzig, C. A. , Weike, A. I. , Zimmerman, J. , and Hamm, A. O. (2007) Startle reflex modulation and autonomic responding during anxious apprehension in panic disorder patients. Psychophysiology 44, 846–54.
Grillon, C. , Dierker, L. , and Merikangas, K. R. (1997) Startle modulation in children at risk for anxiety disorders and/or alcoholism. J Am Acad Child Adolesc Psychiatry 36, 925–32.
Grillon, C. , Warner, V. , Hille, J. , Merikangas, K. R. , Bruder, G. E. , Tenke, C. E. , Nomura, Y. , Leite, P. , and Weissman, M. M. (2005) Families at high and low risk for depression: a three-generation startle study. Biol Psychiatry 57, 953–60.
Cuthbert, B. N. , Lang, P. J. , Strauss, C. , Drobes, D. , Patrick, C. J. , Bradley, M. M. , (2003) The psychophysiology of anxiety disorder: fear memory imagery. Psychophysiology 40, 407–22.
Plappert, C. F. , Pilz, P. K. D. , and Schnitzler, H. , (1993) Acoustic startle response and habituation in freezing and nonfreezing rats. Behav Neurosci 107, 981–87.
Cloninger, C. R. (1987) Neurogenetic adaptive mechanisms in alcoholism. Science 236, 410–16.
Corr, P. J. , Kumari, V. , Wilson, G. D. , Checkley, S. , and Gray, J. A. (1997) Harm avoidance and affective modulation of the startle reflex: a replication. Pers Indiv Differ 22, 591–93.
Back, S. E. , Sonne, S. C. , and Killeen, T. (2003) Comparative profiles of women with PTSD and comorbid cocaine or alcohol dependence. Am J Drug Alcohol Abuse 29, 169–89.
Brady, K. T. , and Clary, C. M. (2003) Affective and anxiety comorbidity in post-traumatic stress disorder treatment trials of sertraline. Compr Psychiatry 44, 360–69.
Zayfert, C. , Becker, C. B. , Unger, D. L. , and Shearer, D. K. (2002) Comorbid anxiety disorders in civilians seeking treatment for posttraumatic stress disorder. J Trauma Stress 15, 31–38.
Zlotnick, C. , Warshaw, M. , Shea, M. T. , Allsworth, J. Pearlstein, T. , and Keller, M. B. (1999) Chronicity in posttraumatic stress disorder (PTSD) and predictors of course of comorbid PTSD in patients with anxiety disorders. J Trauma Stress 12, 89–100.
Cloitre, M. , and Koenen, K. C. (2001) The impact of borderline personality disorder on the effectiveness of group treatment for women with PTSD related to childhood sexual abuse. Int J Group Psychother 53, 379–98.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2009 Humana Press, a part of Springer Science+Business Media, LLC
About this chapter
Cite this chapter
Miller, M., Wolf, E., Fabricant, L., Stein, N. (2009). Low Basal Cortisol and Startle Responding as Possible Biomarkers of PTSD: The Influence of Internalizing and Externalizing Comorbidity. In: LeDoux, J., Keane, T., Shiromani, P. (eds) Post-Traumatic Stress Disorder. Humana Press. https://doi.org/10.1007/978-1-60327-329-9_13
Download citation
DOI: https://doi.org/10.1007/978-1-60327-329-9_13
Published:
Publisher Name: Humana Press
Print ISBN: 978-1-60327-328-2
Online ISBN: 978-1-60327-329-9
eBook Packages: MedicineMedicine (R0)