Abstract
In recent years, microglial cells are becoming to stand in the limelight. They may play a major role not only under physiological condition but also under many pathophysiological conditions. Manipulating their function may lead to cure or attenuation of neurological diseases, traumatic injury, psychiatric disorders, or even sleepiness or aging. In this chapter, the properties of microglial cells including receptors for neurotransmitters and neurohormones/neuromodulators, transporters, and other signals which are related to therapeutic possibilities are summarized.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Microglia
- Inflammation
- Pain
- Neurodegenerative disorders
- Psychiatric disorders
- Microglia
- Neurotransmitter receptor
- Neuropeptide receptor
- Endocrine system
- Neurohormone
- Cytokine
- Chemokine
- Neuropathic pain
- Neurodegenerative diseases
- ALS
- Traumatic brain injury
- LPS
- Sleep loss
- Aging
- Obesity
- Psychiatric disorder
- Autism
13.1 Introduction
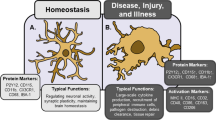
Microglial cells are immune cells in the central nervous system (CNS) . Their contributions towards physiological and pathophysiological processes have been well documented (Kettenmann et al. 2011; Eyo and Dailey 2013; Verkhratsky and Butt 2013) . In pathophysiological condition, microglial cells rapidly respond to any kind of damage signals accompanying poly-etiological insults to the CNS. Microglial responses in the CNS may occur as a result of trauma, ischemia, infection or neurodegenerative stimuli. Microglial cells have been implicated, to some extent, in the pathogenesis of all of the common neurodegenerative disorders involving protein aggregation such as Alzheimer’s disease (AD) , Parkinson’s disease (PD) and amyotrophic lateral sclerosis (ALS) . However, the precise role they play in the development of these pathologies remains unclear and it seems that they contribute to the pathological process in different ways in a disease-specific manner. A better understanding of their varied roles is essential if they are to be the target for novel therapeutic strategies (Gentleman 2013) .
Activated microglia proliferate rapidly, migrate to the site of injury or infection and elicit defense response by phagocytosis of cell debris, production of cytokines , chemokines and reactive oxygen species, and presentation of antigens to other immune cells. In addition, microglia participate in tissue repair by producing neurotrophic factors. However, chronically activated microglia become neurotoxic to the surrounding CNS parenchyma.
Chronic activation of microglia has been shown to augment neurodegeneration in PD, AD, brain injury and number of other CNS pathologies. Identification of factors that control microglial activation , therefore, has become the major focus of recent research (Rangarajan et al. 2013) . Microglial dysfunction may also contribute to genetic neurobehavioral disorders (Benarroch 2013) . At the sites of injury or any pathological area, classical “activated” microglia, with an amoeboid morphology, show different molecular and genetic characteristics compared to those in resting microglia. Recently, this subdivision into two main types (resting and activated microglia) is regarded as oversimplification and more sophisticated microglial activity states throughout an activation process were proposed (Hanisch and Kettenmann 2007; Eyo and Dailey 2013) .
Microglial activation is also regarded as a biomarker for traumatic brain injury (Hernandez-Ontiveros et al. 2013) . During a cascade of secondary cell death and multiple inflammatory responses accompanying traumatic brain injury, inflammatory cytokines and chemokines spreads to normal brain areas juxtaposed to the core impacted tissue. Among immune cells involved, microglia is a key player in propagating inflammation to tissues neighboring the core site of injury.
Multiple sclerosis (MS) exhibits many of the hallmarks of an inflammatory autoimmune disorder including breakdown of the blood-brain barrier (BBB) , the recruitment of lymphocytes, microglia, and macrophages to lesion sites. Inflammatory cascade is amplified by pro-inflammatory cytokines , compromising the BBB, recruiting immune cells from the periphery, and activating resident microglia (see review; Ortiz et al. 2013) . Genome-wide microarray analysis comparing micro-dissected active cortical MS lesions with those of tuberculous meningitis (inflammatory control) showed that more than 80% of the identified MS-specific genes were related to T cell-mediated inflammation, microglia activation, oxidative injury, DNA damage and repair, remyelination and regenerative processes (Fischer et al. 2013) .
Correlation between neurodegenerative diseases and oxidative stress is also suggested since mitochondrial dysfunction, i.e. cell energy impairment, apoptosis and overproduction of reactive oxygen species (ROS), is a final common pathogenic mechanism in aging and in neurodegenerative disease such as AD, PD and ALS (Emerit et al. 2004) .
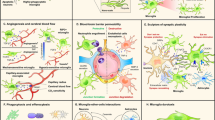
In this chapter, receptors, and other molecules, which are expressed or upregulated in activated microglia and possibly contributing to pathological conditions, are discussed as potential targets for medical intervention in microglia (Fig. 13.1).
13.2 Neurotransmitter Receptors
Microglia express various types of neurotransmitter receptors (Kettenmann et al. 2011; Verkhratsky and Butt 2013; Kettenmann and Ransom 2013; Lee 2013) , each of which can have a specific pathological role(s) which are discussed below .
13.2.1 Glutamate Receptors
13.2.1.1 AMPA Receptors
Microglia express functionally heterogeneous AMPA (α-amino-hydroxy-5-methyl-isoxazole-4-propionate)-type of receptors (Noda et al. 2000; Hagino et al. 2004) and contribute to glutamate (Glu)-mediated neuron-glia interaction (Bezzi et al. 2001) . Microglia express highly Ca2+ impermeable AMPA receptors due to the expression of GluA2 subunit . In addition, expression of plasmalemmal GluA2 subunits significantly increases upon activation of microglia, for example after treatment by lipopolysaccharide (LPS) (Beppu et al. 2013) . Since a decreased expression of GluA2 was reported in some of the neurodegenerative diseases such as AD and Creutzfeldt-Jakob disease (Ferrer and Puig 2003; Carter et al. 2004), functional change in GluA2-deficient microglia was investigated, showing higher Ca2+-permeability, consequently inducing significant increase in the release of pro-inflammatory cytokine, such as tumor necrosis factor-α (TNF-α). GluA2-deficient mouse brain also showed more neurotoxicity in response to kainic acid (Beppu et al. 2013) . Therefore, involvement of microglia in glutamatergic synaptic transmission may be also important to understand the mechanism of some neurodegeneration in which low GluA2 is suggested (Noda and Beppu 2013) .
In the hypothalamus and neurohypophysis of rats with streptozotocin (STZ)-induced diabetes, the expression of GluA2/3 was progressively down-regulated, being hardly detected at 4 months of STZ-diabetes, together with up-regulation of N-methyl D-aspartate receptor (NMDAR) subunit GluN1 and neuronal nitric oxide synthase (nNOS). In addition, both astrocytes and microglia appeared activated. Therefore, it was speculated that the glutamate receptors and NO are linked to overactivation of paraventricular and supraoptic nuclei leading ultimately to cell death (Luo et al. 2002) . The lack of GluA2/3 in microglia may also contribute to the cell death .
13.2.1.2 Kainate Receptors
Microglia express kainate (KA) receptor subunits (Yamada et al. 2006; Noda et al. 2000) and KA-induced current was affected by application of concanavalin A which is a positive modulator selective for KA-preferring receptors (subunits GluR5–GluR7 and KA-1 or KA-2) (Huettner 1990; Partin et al. 1993; Wong and Mayer 1993) , suggesting that only some of the KA-responsive cells express KA-preferring receptors and not only AMPA-preferring receptors (Noda et al. 2000). It was indicative of heterogeneous distribution of AMPA- and KA-preferring receptors among microglial cells; some cells express predominantly AMPA-preferring receptors, some cells express predominantly KA-preferring receptors, and some cells express both. The functional difference has not been identified yet .
13.2.1.3 NMDA Receptors
Although NMDA-induced membrane currents were not observed in primary cultured microglial cells (Noda et al. 2000), it was reported that cortical microglia express NMDAR subunits in vitro and in vivo and activation of the microglial NMDARs plays a pivotal role in neuronal cell death in the perinatal and adult brain (Kaindl et al. 2012) . Microglia have unique NMDAR subunit expression with a high level of the GluN2D subunit that is distinct from that found in neurons, indicating distinct NMDAR subunit assembly. The pathophysiologic relevance of microglial NMDARs is particularly high, as they confer sensitivity to excitotoxic cortical injury that is likely to be significant for a large variety of neurological diseases . For example, after stroke , glutamate released by damaged neurons in the infarct core can diffuse to nearby cells, and this can lead to an activation of microglia via their NMDARs and render them neurotoxic. Conversely, loss of function of the microglial NMDARs protects from gray matter damage .
13.2.1.4 Metabotropic Glutamate Receptors
Microglial cells also bear metabotropic glutamate receptors (mGluRs) (Biber et al. 1999; Farso et al. 2009; Taylor et al. 2002) ; mGluR1,5 (Group I) is linked to intracellular Ca2+ signaling, while mGluR2,3 (Group II) (Taylor et al. 2002) and mGluR4,6,8 (Group III) (Taylor et al. 2003) are coupled to adenosine 3′:5′ cyclic monophosphate (cAMP) and involved in regulation of TNF-α release and microglial cytotoxicity (Taylor et al. 2005). Since mGluRs is proposed as targets for multipotential treatment of neurological disorders (Byrnes et al. 2009) , microglial mGluRs may be critical regulators in neuron to glia transmissions especially under pathological state .
13.2.2 Purinergic Receptors
The most widespread and possibly most functionally important receptors for microglia are purinoceptors (Verkhratsky et al. 2009b) .
13.2.2.1 Ionotropic Purinoceptors (P2X Receptors)
The main type of ionotropic purinoceptors expressed in mature microglial cells areP2X4 and P2X7 receptors. The P2X4 receptors are also constitutively expressed in microglia and contribute to microglial activation in particular in the context of neuropathic pain . Increased levels of P2X4 receptors was found in activated microglia (Tsuda et al. 2003) , whereas intrathecal injection of cultured microglia bearing P2X4 receptors induced allodynia in the absence of peripheral nerve damage (Inoue and Tsuda 2009) . Involvement of P2X4 in KA-induced status epilepticus was also reported (Ulmann et al. 2013) .
The P2X7 receptors are involved in various neuropathologies (Franke et al. 2012) and contribute to various aspects of microglial reactions. P2X7 receptors are abundantly present in immune cells and mediate many immune reactions, including the processing and the release of various cytokines . Microglial cells constitutively express P2X7 receptors and various brain lesions and neuropathologies (such as, for example, MS, ALS and AD) induce substantial up-regulation of P2X7 receptors expression (Sperlagh et al. 2006; Verkhratsky et al. 2009a) . Activation of P2X7 receptors regulates multiple microglial processes from microglial activation to apoptotic death. Stimulation of P2X7 receptors was reported to be necessary for microglial activation by amyloid-β (Aβ) protein (Sanz et al. 2009) , and P2X7 receptors control microglial secretion of pro-inflammatory factors (see (Kettenmann et al. 2011) and references therein). Incidentally, over-expression of P2X7 receptors in microglia triggers their activation in the in vitro system in complete absence of any other exogenous factors (Monif et al. 2009) . Recently P2X7 receptor was reported to regulate microglial survival in living brain tissues (Eyo et al. 2013) . Removal of extracellular Ca2+ or application of a potent P2X7 receptor antagonist, protected microglial cell death. These pharmacological results were complemented by studies in tissue slices from P2X7 receptor null mice, in which oxygen-glucose deprivation (OGD)-induced microglial cell death was reduced by nearly half. These results indicate that stroke-like conditions induce Ca2+-dependent microglial cell death that is mediated in part by P2X7 receptor. Increased neocortical expression of the P2X7 receptor after status epilepticus and anticonvulsant effect of P2X7 receptor antagonist were also reported (Jimenez-Pacheco et al. 2013) . From a therapeutic standpoint, these findings could help direct novel approaches to enhance microglial survival and function following stroke and other neuropathological conditions .
13.2.2.2 Metabotropic Purinoceptors (P2Y Receptors)
Microglia express several metabotropic purinoceptors with predominant appearance of P2Y2, P2Y6, P2Y12, and P2Y13 receptors. Stimulation of these receptors as a rule triggers Ca2+ signals that often involve endoplasmic reticulum Ca2+release and store-operated Ca2+ influx; overstimulation of P2Y pathways can produce a long-lasting activation of the latter that can contribute to various aspects of microglial activation (Toescu et al. 1998) . The P2Y6 receptors, distinctively sensitive to UDP, regulate microglial phagocytosis (Koizumi et al. 2007) , whereas ADP-preferring P2Y12 receptors are fundamental for acute microglial responses to pathological insults, for morphological activation, membrane ruffling and chemotaxis (Verkhratsky et al. 2009a) . In addition P2Y12 receptors are linked to integrin-β1 signaling, which regulates extension of microglial processes (Ohsawa et al. 2010; Eyo and Wu 2013) . In the spinal cord P2Y12 receptors are involved in the genesis of neuropathic pain (Inoue and Tsuda 2009) .
As mentioned above, neuron-to-microglia purinergic signaling regulates microglial extension and retraction (see review (Eyo and Wu 2013)). In the event of neuronal injury, neurons release purines including ATP which can be degraded by endogenous enzymes into ADP and adenosine. Released purines diffuse in the extracellular space and activate P1 adenosine (A3) and P2Y (P2Y12) receptors on microglia that act in concert.
Recently, it was shown that soluble Aβ peptide 1–42 induced self-uptake in microglia through pinocytosis, a process involving activation of P2Y4 receptors by autocrine ATP signaling. It demonstrates a previously unknown function of ATP as a “drink me” signal for microglia and P2Y4 receptors as a potential therapeutic target for the treatment of AD (Li et al. 2013) .
13.2.2.3 Adenosine (P1) Receptors
Adenosine receptors of P1 type are represented by classical seven-transmembrane spanning G protein-coupled receptors and are subclassified into adenosine A1, A2A, A2B, and A3 receptors with distinct pharmacological and functional properties. All four types of adenosine receptors were identified at the mRNA level in cultured rat microglia (Fiebich et al. 1996b) . As mentioned above, purinergic activation leads to extension of microglial processes towards the injury site. Following microglial activation , adenosine can also activate A2A receptors that mediate microglial branch retraction. First, a role for the adenosine A2A receptor in microglial cytoskeletal rearrangements was suggested (Orr et al. 2009) . Adenosine A2B receptors mediate an increase in interleukin (IL)-6 in human astroglioma cells (Fiebich et al. 1996a) . Recently, there was a report that microglia also express A1 receptor which is up-regulated upon ATP treatment. Moreover, selective stimulation of A1 receptors inhibits morphological activation of microglia, possibly by suppressing the Ca2+ influx induced by ATP. Microglial cells, pretreated with the A1 receptor agonist, exhibit lower capability to facilitate the nociceptive neurons, as compared with the cells treated with ATP alone (Luongo et al. 2014) .
13.2.3 GABA (γ-Aminobutyric Acid) Receptors
Functional GABAB receptors were identified in a subpopulation of microglial cells in culture (Kuhn et al. 2004) . The physiological role of microglial GABAB receptors are described in Kettenmann et al. (2011) , but their pathophysiological role needs to be identified.
13.2.4 Cholinergic Receptors
The expression of neuronal α7 nicotinic receptors (nACh) was initially found in cultured mouse microglia (De Simone et al. 2005; Shytle et al. 2004) . The activation of nACh receptors generally inhibits the immune response of microglial cells, thus representing endogenous “cholinergic anti-inflammatory pathway” (Shytle et al. 2004). On example is that nicotine was shown to inhibit P2X7-mediated radical oxygen species (ROS) production in Aβ peptide 1–42 stimulated cultured microglial cells (Moon et al. 2008) . Also, long-term (15 days) incubation of corticostriatal organotypic slices with nicotine reduced the thrombin-dependent activation of microglia (Ohnishi et al. 2009) . More recently, the α7 nACh receptor ligands reduced LPS-induced TNF-α release from microglia (Thomsen and Mikkelsen 2012) .
13.2.5 Adrenergic Receptors
Accumulation of intracellular cAMP following stimulation of β2 receptors provided the first evidence for the expression of adrenergic receptors in microglia (Prinz et al. 2001) . While β2 receptor stimulation suppressed LPS-induced release of IL-12p40 in cultured microglia (Prinz et al. 2001), stimulation of β1 receptors increased expression of IL-1β mRNA (Tanaka et al. 2002) . Increasing intracellular cAMP suppressed proliferation of microglia (Fujita et al. 1998) , inhibited ATP-induced release of TNF-α (Morioka et al. 2009) and modulate microglial migration and phagocytosis. Micoglial adrenergic receptors could be relevant in pathogenesis of AD as early degeneration of locus coeruleus in AD and depletion of adrenergic input to the brain affects the ability of microglia to provide for effective clearance of Aβ (Heneka et al. 2010) . Recently, it became obvious that norepinephrine modulates the motility of resting and activated microglia via different adrenergic receptors; β2 receptors mediate microglial process dynamics in resting cells whereas α2A receptors do in activated cells. In addition, the presence of cross-talk between adrenergic and purinergic signaling in microglia was suggested (Gyoneva and Traynelis 2013) . These data show that norepinephrine can modulate microglial motility, which could affect microglial functions in pathogenic situations of either elevated or reduced norepinephrine levels.
13.2.6 Dopamine Receptors
The D1,2,3,4 (but not D5) receptors were identified on translational level in microglia in culture and visualized by immunostaining in substantia nigra from Parkinsonian brains, suggesting that activation of microglia in PD triggers expression of dopamine receptors, which may explain an increase in microglia-related toxicity towards dopamine-producing neurons (Mastroeni et al. 2009) . In PD, microglial cells in substantia nigra are activated and concentrated around dystrophic dopaminergic neurons (Rogers et al. 2007) ; the role of dopamine receptors in this specific activation and migration of microglia needs to be clarified further. The inhibition of D4 receptors by specific agonist suppressed microglial activation in the spinal cord of superoxide dismutase 1 transgenic animal model of ALS; this reduced microglial activation coincided with slowing down the disease progression (Tanaka et al. 2008) .
13.2.7 Serotonin Receptor
Microglia express 5-HT2 serotonin receptors (Kettenmann et al. 2011; Kato et al. 2013b) . There is a possibility that modulating microglia serotonin receptors may be a key target in the treatment of various psychiatric disorders , which likely will be a hot topic in the near future.
13.3 Neuropeptide Receptors
In addition to neurotransmitter receptors , microglia has receptors for a wide variety of neuropeptides, such as endothelin, bradykinin, angiotensin II, somatostatin,neurokinin-1, vasoactive intestinal peptide (VIP), neuropeptide Y (see (Kettenmann et al. 2011) (Kettenmann and Ransom 2013) for details). Bradykinin receptors, for example, have distinct effects on microglia from those on neurons. Inducible B1 receptor stimulate microglial migration (Ifuku et al. 2007) and have rather neuroprotective effects against LPS-induced activation and inflammatory responses (Noda et al. 2006, 2007) . Pituitary adenylate cyclase activating polypeptide (PACAP) activates a quinine-sensitive K+ outward current and modulates activities in microglia (Ichinose et al. 1998) , VIP and PACAP inhibit the MEKK1/MEK4/JNK signaling pathway (Delgado 2002b) and CREB binding protein (CBP)-nuclear factor (NF)-κB interaction (Delgado 2002a) in endotoxin-activated microglia. Neuropeptide receptors in microglia could become a strong therapeutic target, especially for AD.
13.4 Hormones and Hormone Receptors
It was indicated that glial cells may play a significant role in the link between the endocrine and nervous systems (Garcia-Segura et al. 1996; Chowen et al. 1996) . Glial cells express nuclear receptors for both thyroid and steroid hormones and participate in the metabolism of these hormones, resulting in the production of neuroactive metabolites. Furthermore, glial cells synthesize endogenous neuroactive steroids, including pregnenolone and progesterone, from cholesterol. It is known that thyroid hormones, glucocorticoids, gonadal steroids, and neurosteroids affect glial functions. Under physiological conditions, hormonal effects on glia may have important consequences for neuronal development, metabolism , and activity and for the formation and plasticity of synaptic connections. In addition, glucocorticoids, gonadal steroids, and neurosteroids may affect regenerative processes in neurons by modulating glial responses after injury. These effects include the activation of microglia, which is regulated by glucocorticoids. Therefore, endocrine impairments affect microglial function as well as other cell types in the CNS.
13.4.1 Sex Hormones
Estrogen has been shown to be neuroprotective in stroke and other neural injury models. The presence of estrogen receptor-β (ER-β) in rat microglial cells was reported a decade ago (Mor et al. 1999) . Estrogen-mediated neuroprotection is critically dependent on insulin-like growth factor (IGF)-1 signaling. Microglia appears as the source of IGF-1 and the locus of estrogen-IGF-1 interactions in stroke neuroprotection (Sohrabji and Williams 2013) .
Microglial cell line BV-2, responded to hypoxia by enhanced pro-inflammatory cytokine secretion and reduced phagocytic activity, which is prevented by sex steroids resulting in a switch of microglial cells from a pro-inflammatory to a more anti-inflammatory phenotype. Anti-inflammatory effects of gonadal steroids might directly be mediated through hormone-microglia interactions in addition to known effects via astroglial regulation (Habib et al. 2013a, 2013b) . Estrogen is also neuroprotective in the MS model (Wisdom et al. 2013) and encephalomyelitis (Spence et al. 2013; Wu et al. 2013) . Also, testosterone, being metabolized to estradiol and dihydrotestosterone, decreases reactive astroglia and reactive microglia after brain injury in male rats: role of its metabolites, estradiol and dihydrotestosterone (Barreto et al. 2007) .
Neuronal plasticity is regulated by the ovarian steroids estradiol and progesterone in many normal brain functions , as well as in acute response to injury and chronic neurodegenerative disease. In a female rat model of axotomy, the estradiol-dependent compensatory neuronal sprouting is antagonized by progesterone (Bali et al. 2013) .
In developing rat brain, microglia are essential to masculinization of brain and behavior (Lenz et al. 2013) . Estradiol up-regulates the synthesis of prostaglandin E2 (PGE2), originally in neurons, and then activate microglia and stimulates their own release of PGE2. This feed-forward loop of PGE2 production is required for the masculinization of the preoptic area and therefore of sexual behavior(Welberg 2013) . Microglial dysfunction may be also involved in abnormalities of brain development and sexual behavior.
13.4.2 Cortisol Receptor
Glucocorticoid- and mineralocorticoid receptors in microglia were reported (Tanaka et al. 1997) . Corticosterone suppresses the proliferation of BV2 microglia cells via glucocorticoid, but not mineralocorticoid receptor (Nakatani et al. 2012) . Dexamethasone inhibits the Nox-dependent ROS production via suppression of MKP-1-dependent MAPK pathways in activated microglia, suggesting down-regulation of Nox-2 and overexpression of MKP-1 that regulate ROS and NO may form the potential therapeutic strategy for the treatment of neuroinflammation in neurodegenerative diseases (Huo et al. 2011) .
13.4.3 Oxytocin Receptors
Oxytocin mediates social neuroprotection after cerebral ischemia. There is evidence for a direct suppressive action of oxytocin on cultured microglia, which is a key instigator in the development of neuroinflammation after cerebral ischemia (Karelina et al. 2011) . Since oxytocin is important for social behavior (Eckstein and Hurlemann 2013) , possible involvement of microglia via oxytocin needs to be investigated.
13.5 Cytokine and Chemokine Receptors
Microglial cells are in possession of several types of cytokine and chemokines receptors which regulate multitude of immune responses (see (Kettenmann et al. 2011; Kettenmann and Ransom 2013) for details). Microglia express multiple interleukin receptors (IL-1RI, IL-1RII, IL-5R, IL-6R, IL-8R, IL-9R, IL-10R, IL-12R, IL-13R, and IL-15R) (Lee et al. 2002) and the activation or cultured mouse microglia with LPS induced expression of IL-2 receptors (Sawada et al. 1995) . Spinal cord microglia showed sensitivity to IL-6, connected with a signal transducer and activator of transcription (STAT) pathway, contributing to development of mechanical allodynia and neuropathic pain (Dominguez et al. 2008) .
Among chemokine receptors (CCR) which are expressed in the cells of the CNS, microglial cells in particular express CCR1, CCR2, CCR7 and CCR5 (rodents) and CXCR1, CXCR3 and CCR3 (humans) (Boddeke et al. 1999a, b; Flynn et al. 2003) . A major regulator of microglial activation and neurotoxicity in the CNS is the CX3CL1 (fractalkine, i.e. ligand)/CX3CR1 (fractalkine receptor) signaling axis (Zujovic et al. 2000; Cardona et al. 2006) . On the other hand, it was suggested that CCR2/CCR5 antagonist has the potential for broad clinical use in neuropathic pain treatment (Padi et al. 2012) and decreased pain responses of CCR5 knockout mice to chemical or inflammatory stimuli was reported (Lee et al. 2013) . In addition, another important chemokine, chemokine (C-C motif) ligand 1 (CCL-1), originally a well-characterized chemokine secreted by activated T cells, also plays an important role in neuropathic pain induced after partial sciatic nerve injury (Akimoto et al. 2013a) . CCL-1 plays an important role in neuropathic pain induced by nerve injury and is also produced in various cell types in the CNS, especially in dorsal root ganglia. The specific receptor for CCL-1, CCR8, was found mainly in neurons but also in astrocytes and microglia. CCL-1 has multiple effects on microglia, i.e. increasing chemotaxis, motility, proliferation and phagocytosis. CCL-1 also activated microglia morphologically, promoted mRNA levels for brain-derived neurotrophic factor (BDNF) and IL-6, and increased the release of nitrite from microglia (Akimoto et al. 2013b) . These indicate that CCL-1 may contribute to the development of neurological diseases, especially in neuropathic pain.
13.6 Other Receptors and Molecules
13.6.1 Toll Like-Receptors
Toll-like receptors (TLR1–9) are particularly diversified in microglia being involved in regulation of activation in response to multiple exogenous pathological factors. Activation of TLRs triggers several complex signaling cascades that often involve adaptor protein MyD88, transcription factors AP-1 and NF-κB (Hansson and Edfeldt 2005; Leulier and Lemaitre 2008) . Different TLRs detect different infectious agents. Stimulation of TLRs initiates activation of microglia, and the type or receptor involved may define specific activation programs. It was recently reported that microglia works as key mediators of post-traumatic brain edema and was suggested that high-mobility group box protein 1 (HMGB1)-TLR4 signaling promotes neurovascular dysfunction after traumatic brain injury (Laird et al. 2014) .
13.6.2 Sigma Receptor
Sigma-1 receptor (S1R), an endoplasmic reticulum-resident receptor with chaperone-like activity, has recently attracted great interest. S1R is involved in several processes leading to acute and chronic neurodegeneration, including ALS pathology. Pharmacological manipulation of S1R seems to be a promising strategy to cure ALS. It pointed to increased availability of growth factors and modulation of astrocytosis and of macrophage/microglia as part of the mechanisms (Peviani et al. 2013) .
13.6.3 Ion Transporters
Microglial plasmalemmal transporters such as Na+/Ca2+ exchanger (Ifuku et al. 2007) , Na+/HCO3 − co-transporter and/or Na+-dependent Cl−/HCO3 − exchanger (Schwab 2001) are also important in microglial migration.
13.6.4 Macrophage Colony Stimulating Factor
Increased levels of macrophage colony stimulating factor, increased microglia and microglial activation are found in many different CNS pathologies including tumors, neurodegenerative diseases and injury (reviewed in Charles et al. (2012); El Khoury and Luster (2008); Imai and Kohsaka (2002); Loane and Byrnes (2010)) .
13.7 Pathological Conditions Relating to Microglial Activation
13.7.1 Sleep Deprivation
Deficit of sleep triggers changes in inflammatory signaling pathways in the brain and periphery. The TLR4 receptors activate inflammatory signaling cascades in response to endogenous and pathogen-associated ligands known to be elevated in association with sleep loss . TLR4 is therefore a possible mediator of some of the inflammation-related effects of sleep loss (Wisor et al. 2011a) . In agreement to this observation, microglial changes in the cerebral response were observed after sleep loss, which was modulated by minocycline , a compound that attenuates microglial activation occurring in association with neuroinflammatory events (Wisor et al. 2011c) . Actually changes in the sleep/wake cycle that occur subsequent to administration of D-methamphetamine are modulated by cerebral microglia; the effects of D-methamphetamine on sleep/wake cycles are mediated in part by actions on microglia, including possibly nNOS activity and cytokine synthesis (Wisor et al. 2011b) .
13.7.2 Obesity
In addition to neurodegenerative disorders , obesity is also considered to be associated with a state of chronic neuroinflammation. High-fat-diet exposure induces significant microglial activation not only in the hypothalamus (Yi et al. 2012) but also in the cerebellar white matter (Tapia-Gonzalez et al. 2011) . Free fatty acid palmitate on its own induces activation of BV2 microglia cell line. Further, pre-exposure to palmitate changed the response of microglia to LPS, affecting the mRNA levels of the pro-inflammatory cytokines IL-1β and IL-6 (Tracy et al. 2013) .
13.7.3 Aging
With aging, microglia change their morphology and may display diminished capacity for normal functions related to migration , clearance, and the ability to shift from a pro-inflammatory to an anti-inflammatory state to regulate injury and repair. This shift in microglia potentially contributes to increased susceptibility and neurodegeneration as a function of age (Harry 2013) , though aging itself it not a disease. However, age-related macular degeneration, as well as neurodegenerative diseases such as AD, PD, share two characteristics in common: (1) a disease prevalence that increases markedly with advancing age, and (2) microglia-related neuroinflammation. These characteristics have led to the hypothesis that pathogenic mechanisms underlying age-related neurodegenerative disease involve aging changes in microglia (Wong 2013) .
As mentioned above, microglia have been implicated in disease progression for several age-related brain disorders (Lai et al. 2013) . Also, hypothalamic inflammation and control of systemic ageing was reported. The hypothalamus is important for the development of whole-body ageing in mice, and that the underlying basis involves hypothalamic immunity mediated by IκB kinase-β, NF-κB and related microglia–neuron immune crosstalk (Gabuzda and Yankner 2013; Zhang et al. 2013) .
Considering these evidence mentioned above, understanding and controlling microglial aging and age-associated microglial priming, being increased inflammatory status, may represent an opportunity for elucidating disease mechanisms and for formulating novel therapies (Wong 2013; Norden and Godbout 2013) .
13.7.4 Psychiatric Disorders Including Autism Spectrum Disorder
Microglial cells, are more and more considered to contribute to multiple neuropathologies, including neuropsychiatric and developmental disorders. Growing evidence indicates a role for deregulation or malfunction of glial cells and their neuroinflammatory response in the brains of autistic patients (Theoharides et al. 2013) .
Recently neurobiological basis of oligodendroglial abnormalities in schizophrenia and microglial activity in the context of the disease, in neonatal brain injury and in various experimental models of white matter damage were described (Chew et al. 2013) . Microglia, have recently been also suggested to play important roles in neuropsychiatric disorders (Kato et al. 2013a) and possibility that modulating microglia may be a key target in the treatment of various psychiatric disorders has been proposed (Kato et al. 2013b) . Future investigations to clarify the correlation between neuroendocrine factors and microglia may contribute to a novel understanding of the pathophysiology of neuropsychiatric disorders.
13.8 Conclusion
Microglial cells are fundamental elements of neuropathology. Microglial cells are constantly scanning the brain tissue for any signs of damage and when these latter occur microglial launch a defensive response generally known as microglial activation. Microglia express variety of receptors and respond to many signals. Identification of those signaling and manipulating microglial functions may lead to promising therapeutic strategy for neuropathological processes.
References
Akimoto N, Honda K, Uta D, Beppu K, Ushijima Y, Matsuzaki Y, Nakashima S, Kido MA, Imoto K, Takano Y, Noda M (2013a) CCL-1 in the spinal cord contributes to neuropathic pain induced by nerve injury. Cell Death Dis 4:e679. doi:10.1038/cddis.2013.198
Akimoto N, Ifuku M, Mori Y, Noda M (2013b) Effects of chemokine (C-C motif) ligand 1 on microglial function. Biochem Biophys Res Commun 436(3):455–461. doi:10.1016/j.bbrc.2013.05.126
Bali N, Arimoto JM, Morgan TE, Finch CE (2013) Progesterone antagonism of neurite outgrowth depends on microglial activation via Pgrmc1/S2R. Endocrinology 154(7):2468–2480. doi:10.1210/en.2012–2109
Barreto G, Veiga S, Azcoitia I, Garcia-Segura LM, Garcia-Ovejero D (2007) Testosterone decreases reactive astroglia and reactive microglia after brain injury in male rats: role of its metabolites, oestradiol and dihydrotestosterone. Eur J Neurosci 25(10):3039–3046. doi:10.1111/j.1460–9568.2007.05563.x
Benarroch EE (2013) Microglia: multiple roles in surveillance, circuit shaping, and response to injury. Neurology 81(12):1079–1088. doi:10.1212/WNL.0b013e3182a4a577
Beppu K, Kosai Y, Kido MA, Akimoto N, Mori Y, Kojima Y, Fujita K, Okuno Y, Yamakawa Y, Ifuku M, Shinagawa R, Nabekura J, Sprengel R, Noda M (2013) Expression, subunit composition, and function of AMPA-type glutamate receptors are changed in activated microglia; possible contribution of GluA2 (GluR-B)-deficiency under pathological conditions. Glia 61(6):881–891. doi:10.1002/glia.22481
Bezzi P, Domercq M, Brambilla L, Galli R, Schols D, De Clercq E, Vescovi A, Bagetta G, Kollias G, Meldolesi J, Volterra A (2001) CXCR4-activated astrocyte glutamate release via TNFalpha: amplification by microglia triggers neurotoxicity. Nat Neurosci 4(7):702–710. doi:10.1038/8949089490 [pii]
Biber K, Laurie DJ, Berthele A, Sommer B, Tolle TR, Gebicke-Harter PJ, van Calker D, Boddeke HW (1999) Expression and signaling of group I metabotropic glutamate receptors in astrocytes and microglia. J Neurochem 72(4):1671–1680
Boddeke EW, Meigel I, Frentzel S, Biber K, Renn LQ, Gebicke-Harter P (1999a) Functional expression of the fractalkine (CX3C) receptor and its regulation by lipopolysaccharide in rat microglia. Eur J Pharmacol 374(2):309–313
Boddeke EW, Meigel I, Frentzel S, Gourmala NG, Harrison JK, Buttini M, Spleiss O, Gebicke-Harter P (1999b) Cultured rat microglia express functional beta-chemokine receptors. J Neuroimmunol 98(2):176–184
Byrnes KR, Loane DJ, Faden AI (2009) Metabotropic glutamate receptors as targets for multipotential treatment of neurological disorders. Neurotherapeutics 6(1):94–107. doi:10.1016/j.nurt.2008.10.038
Cardona AE, Pioro EP, Sasse ME, Kostenko V, Cardona SM, Dijkstra IM, Huang D, Kidd G, Dombrowski S, Dutta R, Lee JC, Cook DN, Jung S, Lira SA, Littman DR, Ransohoff RM (2006) Control of microglial neurotoxicity by the fractalkine receptor. Nat Neurosci 9(7):917–924. doi:10.1038/nn1715
Charles NA, Holland EC, Gilbertson R, Glass R, Kettenmann H (2012) The brain tumor microenvironment. Glia 60(3):502–514
Chew LJ, Fusar-Poli P, Schmitz T (2013) Oligodendroglial alterations and the role of microglia in white matter injury: relevance to schizophrenia. Dev Neurosci 35(2–3):102–129. doi:10.1159/000346157
Chowen JA, Argente J, Busiguina S, Garcia-Segura LM (1996) The role of glia in the neuroendocrine hypothalamus: possible implications in hormone secretion. Horm Res 45(Suppl 1):15–18
De Simone R, Ajmone-Cat MA, Carnevale D, Minghetti L (2005) Activation of alpha7 nicotinic acetylcholine receptor by nicotine selectively up-regulates cyclooxygenase-2 and prostaglandin E2 in rat microglial cultures. J Neuroinflam 2(1):4. doi:10.1186/1742-2094-2-4
Delgado M (2002a) Vasoactive intestinal peptide and pituitary adenylate cyclase-activating polypeptide inhibit CBP-NF-kappaB interaction in activated microglia. Biochem Biophys Res Commun 297(5):1181–1185
Delgado M (2002b) Vasoactive intestinal peptide and pituitary adenylate cyclase-activating polypeptide inhibit the MEKK1/MEK4/JNK signaling pathway in endotoxin-activated microglia. Biochem Biophys Res Commun 293(2):771–776. doi:10.1016/S0006-291x(02)00283-8
Dominguez E, Rivat C, Pommier B, Mauborgne A, Pohl M (2008) JAK/STAT3 pathway is activated in spinal cord microglia after peripheral nerve injury and contributes to neuropathic pain development in rat. J Neurochem 107(1):50–60. doi:10.1111/j.1471-4159.2008.05566.x
Eckstein M, Hurlemann R (2013) Oxytocin: evidence for a therapeutic potential of the social neuromodulator. Nervenarzt 84(11):1321–1328. doi:10.1007/s00115-013-3832-6
El Khoury J, Luster AD (2008) Mechanisms of microglia accumulation in Alzheimer’s disease: therapeutic implications. Trends Pharmacol Sci 29(12):626–632. doi:10.1016/j.tips.2008.08.004
Emerit J, Edeas M, Bricaire F (2004) Neurodegenerative diseases and oxidative stress. Biomed Pharmacother Biomed Pharmacother 58(1):39–46
Eyo UB, Dailey ME (2013) Microglia: key elements in neural development, plasticity, and pathology. J Neuroimmune Pharmacol 8(3):494–509. doi:10.1007/s11481-013-9434-z
Eyo UB, Wu LJ (2013) Bidirectional microglia-neuron communication in the healthy brain. Neural Plast 2013:456857. doi:10.1155/2013/456857
Eyo UB, Miner SA, Ahlers KE, Wu LJ, Dailey ME (2013) P2 × 7 receptor activation regulates microglial cell death during oxygen-glucose deprivation. Neuropharmacology 73:311–319. doi:10.1016/j.neuropharm.2013.05.032
Farso MC, O’Shea RD, Beart PM (2009) Evidence group I mGluR drugs modulate the activation profile of lipopolysaccharide-exposed microglia in culture. Neurochem Res 34(10):1721–1728. doi:10.1007/s11064-009-9999-3
Fiebich BL, Biber K, Gyufko K, Berger M, Bauer J, van Calker D (1996a) Adenosine A2b receptors mediate an increase in interleukin (IL)-6 mRNA and IL-6 protein synthesis in human astroglioma cells. J Neurochem 66(4):1426–1431
Fiebich BL, Biber K, Lieb K, van Calker D, Berger M, Bauer J, Gebicke-Haerter PJ (1996b) Cyclooxygenase-2 expression in rat microglia is induced by adenosine A2a-receptors. Glia 18(2):152–160.doi:10.1002/(SICI)1098-1136(199610)18:2<152::AID-GLIA7>3.0.CO;2-2
Fischer MT, Wimmer I, Hoftberger R, Gerlach S, Haider L, Zrzavy T, Hametner S, Mahad D, Binder CJ, Krumbholz M, Bauer J, Bradl M, Lassmann H (2013) Disease-specific molecular events in cortical multiple sclerosis lesions. Brain 136(Pt 6):1799–1815. doi:10.1093/brain/awt110
Flynn G, Maru S, Loughlin J, Romero IA, Male D (2003) Regulation of chemokine receptor expression in human microglia and astrocytes. J Neuroimmunol 136(1–2):84–93
Franke H, Verkhratsky A, Burnstock G, Illes P (2012) Pathophysiology of astroglial purinergic signalling. Purinerg Signal 8(3):629–657. doi:10.1007/s11302-012-9300-0
Fujita H, Tanaka J, Maeda N, Sakanaka M (1998) Adrenergic agonists suppress the proliferation of microglia through beta 2-adrenergic receptor. Neurosci Lett 242(1):37–40
Gabuzda D, Yankner BA (2013) Physiology: inflammation links ageing to the brain. Nature 497(7448):197–198. doi:10.1038/nature12100
Garcia-Segura LM, Chowen JA, Naftolin F (1996) Endocrine glia: roles of glial cells in the brain actions of steroid and thyroid hormones and in the regulation of hormone secretion. Front Neuroendocrinol 17(2):180–211. doi:10.1006/frne.1996.0005
Gentleman SM (2013) Review: microglia in protein aggregation disorders: friend or foe? Neuropathol Appl Neurobiol 39(1):45–50. doi:10.1111/nan.12017
Gyoneva S, Traynelis SF (2013) Norepinephrine modulates the motility of resting and activated microglia via different adrenergic receptors. J Biol Chem 288(21):15291–15302. doi:10.1074/jbc.M113.458901
Habib P, Dreymueller D, Ludwig A, Beyer C, Dang J (2013a) Sex steroid hormone-mediated functional regulation of microglia-like BV-2 cells during hypoxia. J Steroid Biochem Mol Biology 138:195–205. doi:10.1016/j.jsbmb.2013.06.003
Habib P, Slowik A, Zendedel A, Johann S, Dang J, Beyer C (2013b) Regulation of hypoxia-induced inflammatory responses and M1-M2 phenotype switch of primary rat microglia by sex steroids. J Mol Neurosci. doi:10.1007/s12031-013-0137-y
Hagino Y, Kariura Y, Manago Y, Amano T, Wang B, Sekiguchi M, Nishikawa K, Aoki S, Wada K, Noda M (2004) Heterogeneity and potentiation of AMPA type of glutamate receptors in rat cultured microglia. Glia 47(1):68–77. doi:10.1002/glia.20034
Hanisch UK, Kettenmann H (2007) Microglia: active sensor and versatile effector cells in the normal and pathologic brain. Nat Neurosci 10(11):1387–1394. doi:10.1038/nn1997
Hansson GK, Edfeldt K (2005) Toll to be paid at the gateway to the vessel wall. Arterioscler Thromb Vasc Biol 25(6):1085–1087. doi:10.1161/01.ATV.0000168894.43759.47
Harry GJ (2013) Microglia during development and aging. Pharmacol Ther 139(3):313–326. doi:10.1016/j.pharmthera.2013.04.013
Heneka MT, Nadrigny F, Regen T, Martinez-Hernandez A, Dumitrescu-Ozimek L, Terwel D, Jardanhazi-Kurutz D, Walter J, Kirchhoff F, Hanisch UK, Kummer MP (2010) Locus ceruleus controls Alzheimer’s disease pathology by modulating microglial functions through norepinephrine. Proc Natl Acad Sci U S A 107(13):6058–6063. doi:10.1073/pnas.0909586107
Hernandez-Ontiveros DG, Tajiri N, Acosta S, Giunta B, Tan J, Borlongan CV (2013) Microglia activation as a biomarker for traumatic brain injury. Front Neurol 4:30. doi:10.3389/fneur.2013.00030
Huettner JE (1990) Glutamate receptor channels in rat DRG neurons: activation by kainate and quisqualate and blockade of desensitization by Con A. Neuron 5(3):255–266
Huo Y, Rangarajan P, Ling EA, Dheen ST (2011) Dexamethasone inhibits the Nox-dependent ROS production via suppression of MKP-1-dependent MAPK pathways in activated microglia. BMC Neurosci 12:49. doi:10.1186/1471–2202-12–49
Ichinose M, Asai M, Sawada M (1998) Activation of outward current by pituitary adenylate cyclase activating polypeptide in mouse microglial cells. J Neurosci Res 51(3):382–390
Ifuku M, Farber K, Okuno Y, Yamakawa Y, Miyamoto T, Nolte C, Merrino VF, Kita S, Iwamoto T, Komuro I, Wang B, Cheung G, Ishikawa E, Ooboshi H, Bader M, Wada K, Kettenmann H, Noda M (2007) Bradykinin-induced microglial migration mediated by B1-bradykinin receptors depends on Ca2+ influx via reverse-mode activity of the Na+/Ca2+ exchanger. J Neurosci 27(48):13065–13073. doi:10.1523/JNEUROSCI.3467-07.2007
Imai Y, Kohsaka S (2002) Intracellular signaling in M-CSF-induced microglia activation: role of Iba1. Glia 40(2):164–174. doi:10.1002/glia.10149
Inoue K, Tsuda M (2009) Microglia and neuropathic pain. Glia 57:1469–1479
Jimenez-Pacheco A, Mesuret G, Sanz-Rodriguez A, Tanaka K, Mooney C, Conroy R, Miras-Portugal MT, Diaz-Hernandez M, Henshall DC, Engel T (2013) Increased neocortical expression of the P2 × 7 receptor after status epilepticus and anticonvulsant effect of P2 × 7 receptor antagonist A-438079. Epilepsia 54(9):1551–1561. doi:10.1111/epi.12257
Kaindl AM, Degos V, Peineau S, Gouadon E, Chhor V, Loron G, Le Charpentier T, Josserand J, Ali C, Vivien D, Collingridge GL, Lombet A, Issa L, Rene F, Loeffler JP, Kavelaars A, Verney C, Mantz J, Gressens P (2012) Activation of microglial N-methyl-D-aspartate receptors triggers inflammation and neuronal cell death in the developing and mature brain. Ann Neurol 72(4):536–549. doi:10.1002/ana.23626
Karelina K, Stuller KA, Jarrett B, Zhang N, Wells J, Norman GJ, DeVries AC (2011) Oxytocin mediates social neuroprotection after cerebral ischemia. Stroke 42(12):3606–3611. doi:10.1161/STROKEAHA.111.628008
Kato TA, Hayakawa K, Monji A, Kanba S (2013a) Missing and possible link between neuroendocrine factors, neuropsychiatric disorders, and microglia. Front Integr Neurosci 7:53. doi:10.3389/fnint.2013.00053
Kato TA, Yamauchi Y, Horikawa H, Monji A, Mizoguchi Y, Seki Y, Hayakawa K, Utsumi H, Kanba S (2013b) Neurotransmitters, psychotropic drugs and microglia: clinical implications for psychiatry. Curr Med Chem 20(3):331–344
Kettenmann K, Ransom BR (2013) Neuroglia, 3rd edn. Oxford University Press, Oxford
Kettenmann H, Hanisch UK, Noda M, Verkhratsky A (2011) Physiology of microglia. Physiol Rev 91(2):461–553. doi:10.1152/physrev.00011.2010
Koizumi S, Shigemoto-Mogami Y, Nasu-Tada K, Shinozaki Y, Ohsawa K, Tsuda M, Joshi BV, Jacobson KA, Kohsaka S, Inoue K (2007) UDP acting at P2Y6 receptors is a mediator of microglial phagocytosis. Nature 446(7139):1091–1095
Kuhn SA, van Landeghem FK, Zacharias R, Farber K, Rappert A, Pavlovic S, Hoffmann A, Nolte C, Kettenmann H (2004) Microglia express GABA(B) receptors to modulate interleukin release. Mol Cell Neurosci 25(2):312–322. doi:10.1016/j.mcn.2003.10.023
Lai AY, Dibal CD, Armitage GA, Winship IR, Todd KG (2013) Distinct activation profiles in microglia of different ages: a systematic study in isolated embryonic to aged microglial cultures. Neuroscience 254:185–195. doi:10.1016/j.neuroscience.2013.09.010
Laird MD, Shields JS, Sukumari-Ramesh S, Kimbler DE, Fessler RD, Shakir B, Youssef P, Yanasak N, Vender JR, Dhandapani KM (2014) High mobility group box protein-1 promotes cerebral edema after traumatic brain injury via activation of toll-like receptor 4. Glia 62(1):26–38. doi:10.1002/glia.22581
Lee M (2013) Neurotransmitters and microglial-mediated neuroinflammation. Curr Protein Pept Sci 14(1):21–32
Lee YB, Nagai A, Kim SU (2002) Cytokines, chemokines, and cytokine receptors in human microglia. J Neurosci Res 69(1):94–103. doi:10.1002/jnr.10253
Lee YK, Choi DY, Jung YY, Yun YW, Lee BJ, Han SB, Hong JT (2013) Decreased pain responses of C-C chemokine receptor 5 knockout mice to chemical or inflammatory stimuli. Neuropharmacology 67:57–65. doi:10.1016/j.neuropharm.2012.10.030
Lenz KM, Nugent BM, Haliyur R, McCarthy MM (2013) Microglia are essential to masculinization of brain and behavior. J Neurosci 33(7):2761–2772. doi:10.1523/JNEUROSCI.1268-12.2013
Leulier F, Lemaitre B (2008) Toll-like receptors–taking an evolutionary approach. Nat Rev Genet 9(3):165–178
Li HQ, Chen C, Dou Y, Wu HJ, Liu YJ, Lou HF, Zhang JM, Li XM, Wang H, Duan S (2013) P2Y4 receptor-mediated pinocytosis contributes to amyloid beta-induced self-uptake by microglia. Mol Cell Biol 33(21):4282–4293. doi:10.1128/MCB.00544-13
Loane DJ, Byrnes KR (2010) Role of microglia in neurotrauma. Neurotherapy 7(4):366–377. doi:10.1016/j.nurt.2010.07.002
Luo Y, Kaur C, Ling EA (2002) Neuronal and glial response in the rat hypothalamus-neurohypophysis complex with streptozotocin-induced diabetes. Brain Res 925(1):42–54
Luongo L, Guida F, Imperatore R, Napolitano F, Gatta L, Cristino L, Giordano C, Siniscalco D, Di Marzo V, Bellini G, Petrelli R, Cappellacci L, Usiello A, de Novellis V, Rossi F, Maione S (2014) The A1 adenosine receptor as a new player in microglia physiology. Glia 62(1):122–132. doi:10.1002/glia.22592
Mastroeni D, Grover A, Leonard B, Joyce JN, Coleman PD, Kozik B, Bellinger DL, Rogers J (2009) Microglial responses to dopamine in a cell culture model of Parkinson’s disease. Neurobiol Aging 30(11):1805–1817. doi:10.1016/j.neurobiolaging.2008.01.001
Monif M, Reid CA, Powell KL, Smart ML, Williams DA (2009) The P2X7 receptor drives microglial activation and proliferation: a trophic role for P2X7R pore. J Neurosci 29(12):3781–3791
Moon JH, Kim SY, Lee HG, Kim SU, Lee YB (2008) Activation of nicotinic acetylcholine receptor prevents the production of reactive oxygen species in fibrillar beta amyloid peptide (1–42)-stimulated microglia. Exp Mol Med 40(1):11–18. doi:10.3858/emm.2008.40.1.11
Mor G, Nilsen J, Horvath T, Bechmann I, Brown S, Garcia-Segura LM, Naftolin F (1999) Estrogen and microglia: a regulatory system that affects the brain. J Neurobiol 40(4):484–496
Morioka N, Tanabe H, Inoue A, Dohi T, Nakata Y (2009) Noradrenaline reduces the ATP-stimulated phosphorylation of p38 MAP kinase via beta-adrenergic receptors-cAMP-protein kinase A-dependent mechanism in cultured rat spinal microglia. Neurochem Int 55(4):226–234. doi:10.1016/j.neuint.2009.03.004
Nakatani Y, Amano T, Tsuji M, Takeda H (2012) Corticosterone suppresses the proliferation of BV2 microglia cells via glucocorticoid, but not mineralocorticoid receptor. Life Sci 91(15–16):761–770. doi:10.1016/j.lfs.2012.08.019
Noda M, Beppu K (2013) Possible contribution of microglial glutamate receptors to inflammatory response upon neurodegenerative diseases. J Neurol Disord 1. doi:10.4172/2329–6895.1000131
Noda M, Nakanishi H, Nabekura J, Akaike N (2000) AMPA-kainate subtypes of glutamate receptor in rat cerebral microglia. J Neurosci 20(1):251–258
Noda M, Kettenmann H, Wada K (2006) Anti-inflammatory effects of kinins via microglia in the central nervous system. Biol Chem 387(2):167–171. doi:10.1515/BC.2006.022
Noda M, Kariura Y, Pannasch U, Nishikawa K, Wang L, Seike T, Ifuku M, Kosai Y, Wang B, Nolte C, Aoki S, Kettenmann H, Wada K (2007) Neuroprotective role of bradykinin because of the attenuation of pro-inflammatory cytokine release from activated microglia. J Neurochem 101(2):397–410. doi:10.1111/j.1471-4159.2006.04339.x
Norden DM, Godbout JP (2013) Review: microglia of the aged brain: primed to be activated and resistant to regulation. Neuropathol Appl Neurobiol 39(1):19–34. doi:10.1111/j.1365-2990.2012.01306.x
Ohnishi M, Katsuki H, Takagi M, Kume T, Akaike A (2009) Long-term treatment with nicotine suppresses neurotoxicity of, and microglial activation by, thrombin in cortico-striatal slice cultures. Eur J Pharmacol 602(2–3):288–293. doi:10.1016/j.ejphar.2008.11.041
Ohsawa K, Irino Y, Sanagi T, Nakamura Y, Suzuki E, Inoue K, Kohsaka S (2010) P2Y12 receptor-mediated integrin-beta1 activation regulates microglial process extension induced by ATP. Glia 58:790–801
Orr AG, Orr AL, Li XJ, Gross RE, Traynelis SF (2009) Adenosine A(2A) receptor mediates microglial process retraction. Nat Neurosci 12(7):872–878. doi:10.1038/nn.2341
Ortiz GG, Pacheco-Moises FP, Bitzer-Quintero OK, Ramirez-Anguiano AC, Flores-Alvarado LJ, Ramirez-Ramirez V, Macias-Islas MA, Torres-Sanchez ED (2013) Immunology and oxidative stress in multiple sclerosis: clinical and basic approach. Clin Dev Immunol 2013:708659. doi:10.1155/2013/708659
Padi SS, Shi XQ, Zhao YQ, Ruff MR, Baichoo N, Pert CB, Zhang J (2012) Attenuation of rodent neuropathic pain by an orally active peptide, RAP-103, which potently blocks CCR2- and CCR5-mediated monocyte chemotaxis and inflammation. Pain 153(1):95–106. doi:10.1016/j.pain.2011.09.022
Partin KM, Patneau DK, Winters CA, Mayer ML, Buonanno A (1993) Selective modulation of desensitization at AMPA versus kainate receptors by cyclothiazide and concanavalin A. Neuron 11(6):1069–1082
Peviani M, Salvaneschi E, Bontempi L, Petese A, Manzo A, Rossi D, Salmona M, Collina S, Bigini P, Curti D (2013) Neuroprotective effects of the Sigma-1 receptor (S1R) agonist PRE-084, in a mouse model of motor neuron disease not linked to SOD1 mutation. Neurobiol Dis 62C:218–232. doi:10.1016/j.nbd.2013.10.010
Prinz M, Hausler KG, Kettenmann H, Hanisch U (2001) Beta-adrenergic receptor stimulation selectively inhibits IL-12p40 release in microglia. Brain Res 899(1–2):264–270
Rangarajan P, Eng-Ang L, Dheen ST (2013) Potential drugs targeting microglia: current knowledge and future prospects. CNS Neurol Disord Drug Targets 12(6):799–806
Rogers J, Mastroeni D, Leonard B, Joyce J, Grover A (2007) Neuroinflammation in Alzheimer’s disease and Parkinson’s disease: are microglia pathogenic in either disorder? Int Rev Neurobiol 82:235–246. doi:10.1016/S0074-7742(07)82012 - 5
Sanz JM, Chiozzi P, Ferrari D, Colaianna M, Idzko M, Falzoni S, Fellin R, Trabace L, Di Virgilio F (2009) Activation of microglia by amyloid b requires P2X7 receptor expression. J Immunol 182(7):4378–4385
Sawada M, Suzumura A, Marunouchi T (1995) Induction of functional interleukin-2 receptor in mouse microglia. J Neurochem 64(5):1973–1979
Schwab A (2001) Ion channels and transporters on the move. News Physiol Sci 16:29–33
Shytle RD, Mori T, Townsend K, Vendrame M, Sun N, Zeng J, Ehrhart J, Silver AA, Sanberg PR, Tan J (2004) Cholinergic modulation of microglial activation by alpha 7 nicotinic receptors. J Neurochem 89(2):337–343. doi:10.1046/j.1471–4159.2004.02347.x
Sohrabji F, Williams M (2013) Stroke neuroprotection: estrogen and IGF-1 interactions and the role of microglia. J Neuroendocrinol. doi:10.1111/jne.12059
Spence RD, Wisdom AJ, Cao Y, Hill HM, Mongerson CR, Stapornkul B, Itoh N, Sofroniew MV, Voskuhl RR (2013) Estrogen mediates neuroprotection and anti-inflammatory effects during EAE through ERalpha signaling on astrocytes but not through ERbeta signaling on astrocytes or neurons. J Neurosci 33(26):10924–10933. doi:10.1523/JNEUROSCI.0886-13.2013
Sperlagh B, Vizi ES, Wirkner K, Illes P (2006) P2X7 receptors in the nervous system. Prog Neurobiol 78(6):327–346
Tanaka J, Fujita H, Matsuda S, Toku K, Sakanaka M, Maeda N (1997) Glucocorticoid- and mineralocorticoid receptors in microglial cells: the two receptors mediate differential effects of corticosteroids. Glia 20(1):23–37
Tanaka KF, Kashima H, Suzuki H, Ono K, Sawada M (2002) Existence of functional beta1- and beta2-adrenergic receptors on microglia. J Neurosci Res 70(2):232–237. doi:10.1002/jnr.10399
Tanaka K, Okada Y, Kanno T, Otomo A, Yanagisawa Y, Shouguchi-Miyata J, Suga E, Kohiki E, Onoe K, Osuga H, Aoki M, Hadano S, Itoyama Y, Ikeda JE (2008) A dopamine receptor antagonist L-745,870 suppresses microglia activation in spinal cord and mitigates the progression in ALS model mice. Exp Neurol 211(2):378–386. doi:10.1016/j.expneurol.2008.02.004
Tapia-Gonzalez S, Garcia-Segura LM, Tena-Sempere M, Frago LM, Castellano JM, Fuente-Martin E, Garcia-Caceres C, Argente J, Chowen JA (2011) Activation of microglia in specific hypothalamic nuclei and the cerebellum of adult rats exposed to neonatal overnutrition. J Neuroendocrinol 23(4):365–370. doi:10.1111/j.1365-2826.2011.02113.x
Taylor DL, Diemel LT, Cuzner ML, Pocock JM (2002) Activation of group II metabotropic glutamate receptors underlies microglial reactivity and neurotoxicity following stimulation with chromogranin A, a peptide up-regulated in Alzheimer’s disease. J Neurochem 82(5):1179–1191
Taylor DL, Diemel LT, Pocock JM (2003) Activation of microglial group III metabotropic glutamate receptors protects neurons against microglial neurotoxicity. J Neurosci 23(6):2150–2160
Taylor DL, Jones F, Kubota ES, Pocock JM (2005) Stimulation of microglial metabotropic glutamate receptor mGlu2 triggers tumor necrosis factor alpha-induced neurotoxicity in concert with microglial-derived Fas ligand. J Neurosci 25(11):2952–2964. doi:10.1523/JNEUROSCI.4456-04.2005
Theoharides TC, Asadi S, Patel AB (2013) Focal brain inflammation and autism. J Neuroinflam 10:46. doi:10.1186/1742–2094-10-46
Thomsen MS, Mikkelsen JD (2012) The alpha7 nicotinic acetylcholine receptor ligands methyllycaconitine, NS6740 and GTS-21 reduce lipopolysaccharide-induced TNF-alpha release from microglia. J Neuroimmunol 251(1–2):65–72. doi:10.1016/j.jneuroim.2012.07.006
Toescu EC, Moller T, Kettenmann H, Verkhratsky A (1998) Long-term activation of capacitative Ca2+ entry in mouse microglial cells. Neuroscience 86(3):925–935
Tracy LM, Bergqvist F, Ivanova EV, Jacobsen KT, Iverfeldt K (2013) Exposure to the saturated free fatty acid palmitate alters BV-2 microglia inflammatory response. J Mol Neurosci 51(3):805–812. doi:10.1007/s12031-013-0068-7
Tsuda M, Shigemoto-Mogami Y, Koizumi S, Mizokoshi A, Kohsaka S, Salter MW, Inoue K (2003) P2X4 receptors induced in spinal microglia gate tactile allodynia after nerve injury. Nature 424(6950):778–783
Ulmann L, Levavasseur F, Avignone E, Peyroutou R, Hirbec H, Audinat E, Rassendren F (2013) Involvement of P2 × 4 receptors in hippocampal microglial activation after status epilepticus. Glia 61(8):1306–1319. doi:10.1002/glia.22516
Verkhratsky A, Butt AM (2013) Glial physiology and pathophysiology, 1st edn. Wiley, New York
Verkhratsky A, Krishtal OA, Burnstock G (2009a) Purinoceptors on neuroglia. Mol Neurobiol 39(3):190–208. doi:10.1007/s12035-009-8063-2
Verkhratsky A, Krishtal OA, Burnstock G (2009b) Purinoceptors on neuroglia. Mol Neurobiol 39(3):190–208
Welberg L (2013) Microglia maketh the male. Nat Rev Neurosci 14(4):226. doi:10.1038/nrn3473
Wisdom AJ, Cao Y, Itoh N, Spence RD, Voskuhl RR (2013) Estrogen receptor-beta ligand treatment after disease onset is neuroprotective in the multiple sclerosis model. J Neurosci Res 91(7):901–908. doi:10.1002/jnr.23219
Wisor JP, Clegern WC, Schmidt MA (2011a) Toll-like receptor 4 is a regulator of monocyte and electroencephalographic responses to sleep loss. Sleep 34(10):1335–1345. doi:10.5665/SLEEP.1274
Wisor JP, Schmidt MA, Clegern WC (2011b) Cerebral microglia mediate sleep/wake and neuroinflammatory effects of methamphetamine. Brain Behav Immun 25(4):767–776. doi:10.1016/j.bbi.2011.02.002
Wisor JP, Schmidt MA, Clegern WC (2011c) Evidence for neuroinflammatory and microglial changes in the cerebral response to sleep loss. Sleep 34(3):261–272
Wong WT (2013) Microglial aging in the healthy CNS: phenotypes, drivers, and rejuvenation. Front Cell Neurosci 7:22. doi:10.3389/fncel.2013.00022
Wong LA, Mayer ML (1993) Differential modulation by cyclothiazide and concanavalin A of desensitization at native alpha-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid- and kainate-preferring glutamate receptors. Mol Pharmacol 44(3):504–510
Wu WF, Tan XJ, Dai YB, Krishnan V, Warner M, Gustafsson JA (2013) Targeting estrogen receptor beta in microglia and T cells to treat experimental autoimmune encephalomyelitis. Proc Natl Acad Sci U S A 110(9):3543–3548. doi:10.1073/pnas.1300313110
Yamada J, Sawada M, Nakanishi H (2006) Cell cycle-dependent regulation of kainate-induced inward currents in microglia. Biochem Biophys Res Commun 349(3):913–919. doi:10.1016/j.bbrc.2006.08.126
Yi CX, Tschop MH, Woods SC, Hofmann SM (2012) High-fat-diet exposure induces IgG accumulation in hypothalamic microglia. Dis Models Mech 5(5):686–690. doi:10.1242/dmm.009464
Zhang G, Li J, Purkayastha S, Tang Y, Zhang H, Yin Y, Li B, Liu G, Cai D (2013) Hypothalamic programming of systemic ageing involving IKK-beta, NF-kappaB and GnRH. Nature 497(7448):211–216. doi:10.1038/nature12143
Zujovic V, Benavides J, Vige X, Carter C, Taupin V (2000) Fractalkine modulates TNF-alpha secretion and neurotoxicity induced by microglial activation. Glia 29(4):305–315
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer Science+Business Media New York
About this chapter
Cite this chapter
Noda, M. (2014). Possible Therapeutic Targets in Microglia. In: Parpura, V., Verkhratsky, A. (eds) Pathological Potential of Neuroglia. Springer, New York, NY. https://doi.org/10.1007/978-1-4939-0974-2_13
Download citation
DOI: https://doi.org/10.1007/978-1-4939-0974-2_13
Published:
Publisher Name: Springer, New York, NY
Print ISBN: 978-1-4939-0973-5
Online ISBN: 978-1-4939-0974-2
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)