Abstract
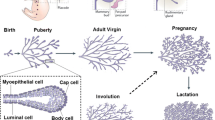
Development of the mammary gland in females is a dynamic, orchestrated process that occurs throughout postnatal development. Initiated during embryogenesis, epithelial cells advance into the underlying stromal matrix to form a primitive rudimentary structure. With the onset of puberty this anlage then responds to hormonal and local cues to rapidly establish a ductal network. Whereas in mice this network is relatively simple, in humans there is significantly more branching morphogenesis to develop terminal duct lobular unit structures. With the onset of pregnancy and associated changes in the hormonal and local environment, alveolar development progresses to establish a gland that is densely filled with alveolar structures by the end of pregnancy. Concomitantly, mammary epithelial cells within the gland begin to attain their unique ability to synthesize various milk constituents, such that by parturition, functional lactogenesis can be realized.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Anderson E, Clarke RB, Howell A. Estrogen responsiveness and control of normal human breast proliferation. J Mammary Gland Biol Neoplasia 1998;3:23–35.
Atwood CS, Hovey RC, Glover JP, Chepko G, Ginsburg E, Robison WG, Vonderhaar BK. Progesterone induces side-branching of the ductal epithelium in the mammary glands of peripubertal mice. J Endocrinol 2000;167:39–52.
Brisken C. Hormonal control of alveolar development and its implications for breast carcinogenesis. J Mammary Gland Biol Neoplasia 2002;7:39–48.
Couse JF, Korach KS. Estrogen receptor null mice: what have we learned and where will they lead us? Endocr Rev 1999;20:358–417.
Daniel CW, Silberstein G. Postnatal development of the rodent mammary gland. In: Neville MC, Daniel CW, editors. The Mammary Gland: Development, Regulation, and Function. New York: Plenum Press, 1987; pp 3–36.
Djonov V, Andres AC, Ziemiecki A. Vascular remodelling during the normal and malignant life cycle of the mammary gland. Microsc Res Tech 2001;52:182–189.
Flux DS. Growth of the mammary duct system in intact and ovariectomized mice of the chi strain. J Endocrinol 1954;11:223–237.
Grimm SL, Seagroves TN, Kabotyanski EB, Hovey RC, Vonderhaar BK, Lydon JP, Miyoshi K, Hennighausen L, Ormandy CJ, Lee AV, Stull MA, Wood TL, Rosen JM. Disruption of steroid and prolactin receptor patterning in the mammary gland correlates with a block in lobuloalveolar development. Mol Endocrinol 2002;16:2675–2691.
Hadsell DL. Genetic manipulation of mammary gland development and lacation. In: Pickering LK, Morrow AL, Ruiz-Palacios GM, Schanler RJ. Protecting Infants through Human Milk: Advancing the Scientific Evidence. Advances in Experimental Medicine and Biology Series. New York: Kluwer Academic/Plenum Publishers, 2004; this volume.
Haslam SZ, Osuch JR, Raafat AM, Hofseth LJ. Postmenopausal hormone replacement therapy: effects on normal mammary gland in humans and in a mouse postmenopausal model. J Mammary Gland Biol Neoplasia 2002;7:93–105.
Horseman ND, Zhao W, Montecino-Rodriquez E, Tanaka M, Nakashima K, Engle SJ, Smith F, Markoff E, Dorskind K. Defective mammopoiesis, but normal hematopoiesis, in mice with a targeted disruption of the prolactin gene. EMBO J 1997;16:6926–6935.
Hovey RC, McFadden TB, Akers RM. Regulation of mammary gland growth and morphogenesis by the mammary fat pad: a species comparison. J Mammary Gland Biol Neoplasia 1999;4:53–68.
Hovey RC, Goldhar AS, Baffi J, Vonderhaar BK. Transcriptional regulation of vascular endothelial growth factor expression in epithelial and stromal cells during mouse mammary gland development. Mol Endocrinol 2001a;15:819–831.
Hovey RC, Trott JF, Ginsburg E, Goldhar A, Sasaki MM, Fountain SJ, Sundararajan K, Vonderhaar BK. Transcriptional and spatiotemporal regulation of prolactin receptor mRNA and cooperativity with progesterone receptor function during ductal branch growth in the mammary gland. Dev Dyn 2001b;222:192–205.
Hovey RC, Trott JF, Vonderhaar BK. Establishing a framework for the functional mammary gland: from endocrinology to morphology. J Mammary Gland Biol Neoplasia 2002;7:17–38.
Hovey RC, Harris J, Hadsell DL, Lee AV, Ormandy CJ, Vonderhaar BK. Local insulin-like growth factor-II mediates prolactin-induced mammary gland development. Mol Endocrinol 2003;17:460–471.
Humphreys RC. Programmed cell death in the terminal end bud. J Mammary Gland Biol Neoplasia 1999;4:213–220.
Ichinose RR, Nandi S. Influence of hormones on lobulo-alveolar differentiation of mouse mammary glands in vitro. J Endocrinol 1966;35:331–340.
Kimata K, Sakakura T, Inaguma Y, Kato M, Nishizuka Y. Participation of two different mesenchymes in the developing mouse mammary gland: synthesis of basement membrane components by fat pad precursor cells. J Embryol Exp Morphol 1985;89:243–257.
Kleinberg DL, Feldman M, Ruan W. IGF-I: an essential factor in terminal end bud formation and ductal morphogenesis. J Mammary Gland Biol Neoplasia 2000;5:7–17.
Laidlaw IJ, Clarke RB, Howell A, Owen WMC, Pötten CS, Anderson E. Proliferation of normal human breast tissue implanted in athymic nude mice is stimulated by estrogen and not progesterone. Endocrinology 1995;136:164–171.
Lydon JP, DeMayo FJ, Funk CR, Mani SK, Hughes AR, Montgomery CA Jr, Shyamala G, Conneely OM, O’Malley BW. Mice lacking progesterone receptor exhibit pleiotropic reproductive abnormalities. Genes Dev 1995;9:2266–2278.
Lydon JP, Ge G, Kittrell FS, Medina O, O’Malley BW. Murine mammary gland carcinogenesis is critically dependent on progesterone receptor function. Cancer Res 1999;59:4276–4284.
Lyons WR, Li CH, Johnson RE. The hormonal control of mammary growth and lactation. Recent Prog Horm Res 1958;14:219–254.
Mueller SO, Clark JA, Myers PH, Korach KS. Mammary gland development in adult mice requires epithelial and stromal estrogen receptor alpha. Endocrinology 2002;143:2357–2365.
Neville MC, McFadden TB, Forsyth I. Hormonal regulation of mammary differentiation and milk secretion. J Mammary Gland Biol Neoplasia 2002;7;49–66.
Ormandy CJ, Camus A, Barra J, Damotte D, Lucas B, Buteau H, Edery M, Brousse N, Babinet C, Binait N, Kelly PA. Null mutation of the prolactin receptor gene produces multiple reproductive defects in the mouse. Genes Dev 1997;11:167–178.
Parmar H, Young P, Emerman JT, Neve RM, Dairkee S, Cunha GR. A novel method for growing human breast epithelium in vivo using mouse and human mammary fibroblasts. Endocrinology 2002; 143:4886–4888.
Robinson GW, McKnight RA, Smith GH, Hennighausen L. Mammary epithelial cells undergo secretory differentiation in cycling virgins but require pregnancy for the establishment of terminal differentiation. Development 1995;121:2079–2090.
Rose SR, Municchi G, Barnes KM, Kamp GA, Uriarte MM, Ross JL, Cassorla F, Cutler GB Jr. Spontaneous growth hormone secretion increases during puberty in normal girls and boys. J Clin Endocrinol Metab 1991;73:428–435.
Russo J, Russo IH. Development of the human mammary gland. In: Neville MC, Daniel CW, editors. The Mammary Gland: Development, Regulation, and Function. New York: Plenum Press, 1987; pp 67–93.
Russo J, Hu YF, Silva IDCG, Russo IH. Cancer risk related to mammary gland structure and development. Microsc Res Tech 2001;52:204–223.
Sakakura T. New aspects of stroma-parenchyma relations in mammary gland differentiation. Int Rev Cytol 1991;125:165–202.
Tobon H, Salazar H. Ultrastructure of the human mammary gland. I. Development of the fetal gland throughout gestation. J Clin Endocrinol Metab 1974;39:443–456.
Trott JF, Hovey RC, Koduri S, Vonderhaar BK. Alternative splicing to exon 11 of human prolactin receptor gene results in multiple isoforms including a secreted prolactin binding protein. J Mol Endocrinol 2003;30:31–47.
Vonderhaar BK. Hormones and growth factors in mammary gland development. In: Veneziale CM, editor. Control of Cell Growth and Proliferation. New York: Van Noostrand, Reinhold, 1984; pp 11–33.
Woodward TL, Xie JW, Haslam SZ. The role of mammary stroma in modulating the proliferative response to ovarian hormones in the normal mammary gland. J Mammary Gland Biol Neoplasia 1998;3:117–131.
Zhang HZ, Bennett JM, Smith KT, Sunil N, Haslam SZ. Estrogen mediates mammary epithelial cell proliferation in serum-free culture indirectly via mammary stroma-derived hepatocyte growth factor. Endocrinology 2002;143:3427–3434.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2004 Springer Science+Business Media New York
About this paper
Cite this paper
Hovey, R.C., Trott, J.F. (2004). Morphogenesis of Mammary Gland Development. In: Pickering, L.K., Morrow, A.L., Ruiz-Palacios, G.M., Schanler, R.J. (eds) Protecting Infants through Human Milk. Advances in Experimental Medicine and Biology, vol 554. Springer, Boston, MA. https://doi.org/10.1007/978-1-4757-4242-8_19
Download citation
DOI: https://doi.org/10.1007/978-1-4757-4242-8_19
Publisher Name: Springer, Boston, MA
Print ISBN: 978-1-4419-3461-1
Online ISBN: 978-1-4757-4242-8
eBook Packages: Springer Book Archive