Abstract
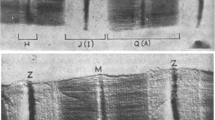
In situ, ultrastructural measurements of diameters of contractile filaments in skeletal and heart muscle differ considerably from those previously reported. Past measurements have been made in thin, transverse epoxy sections that were non-specifically stained with heavy metal salts to overcome background scattering of epoxy polymer. In our images from transverse de-embedded sections, the hexagonal lattice has some considerable differences from that seen in epoxy sections. Muscle samples from rat atrium and frog sartorius were fixed, dehydrated, embedded in polyethylene glycol, and sectioned. Sections were de-embedded in graded polyethylene glycol/ethanol, mounted on coated grids, critical point dried, and viewed in the electron microscope without staining. The backbone diameters of thick filaments were measured in the M band region and have an average value, after correction for shrinkage, of 25 nm. Thin filament diameters range from 6.5–9.5 nm. In regions of overlap of thin and thick filaments, the thick filament profiles varied from circular to asymmetric; diameters range up to 36 nm and yield eccentricity ratios varying from 1.5 to 1.0 (circular profiles). Portions of thick filaments touch or partially envelope neighboring thin filaments. The relative contributions of cytoskeletal components to these images of overlap regions remains to be determined, but the backbone diameters in glycerinated frog sartorius are not significantly different from control samples. The present results are consistent with those reported for rotary shadowed thick filaments; from recent experiments in muscles whose myofilament lattice is osmotically compressed; and with estimates of A band mass. This lattice geometry yields relatively low surface-to-surface distances between filaments. Steric considerations and their implications for cross bridge theory are discussed.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Brown, L.M. (1978). Calibration of a commercial electron microscope with a grating replica to an acuracy of better than 1%. J. Microsc. 113 (pt2): 149–160.
Eisenberg, B.R. and Mobley, B.A. (1975). Size changes in single muscle fibers during fixation and embedding. Tissue and Cell 7 (2): 383–387.
Elliot A. and Offer G. (1978). Shape and flexibility of the myosin molecule. J. Molec. Biol. 123: 505–519.
Franzini-Armstrong, C. (1973). The structure of a simple Z line. J. Cell Biol. 58: 630–642. Gerdes, A.M., Kriseman, J. and Bishop, S.P. (1982). Morphometric Study of Cardiac Muscle: The problem of tissue shrinkage. Lab. Invest. 48 (3): 271–274.
Heuser, J.E. and Salpeter, S.R. (1979). Organization of acetylcholine receptors in quick-frozen, deep-etched, and rotary-replicated Torpedo postsynaptic membrane. J. Cell Biol. 82: 150–173.
Huxley, H.E. (1972). Molecular basis of contraction in cross-striated muscles. In: The Structure and Function of Muscle, 2nd ed., vol. 1, pt. 1, 301–387, Bourne, G.H. (ed.) Academic Press. New York and London.
Magid, A., Ting-Beall, H.P., Carvell, M., Kontis, T. and Lucaveche, C. (1982). Connecting filaments, core filaments, and side struts: A proposal to add three new load-bearing structures to the sliding filament model. ( This volume).
Millman, B.M. and Nickel, B.G. (1980). Electrostatic forces in muscle and cylindrical gel systems. Biophys. J. 32: 49–63.
Millman, B.M., Racey, T.J. and Matsubara, I. (1981). Effects of hyperosmotic solutions on the filament lattice of intact frog skeletal muscle. Biophys. J. 33: 189–202.
Page, S.G. and Huxley, H.E. (1963). Filament lengths in striated muscle J. Cell Biol. 19: 369390.
Page, S.G. (1974). Measurements of structural parameters in cardiac muscle. In: The Physiological Basis of Starlings Law of the Heart. Ciba Foundation Symposium 24 (new series) 13–30 Elsevier Exerpta Medica, North Holland.
Reedy, M.D. and Lucaveche, C. (1982). A-Band mass exceeds mass of its filament components by 30–45%. (This volume).
Robinson, T.F. (1980). Lateral connections between heart muscle cells as revealed by conventional and high voltage transmission electron microscopy. Cell and Tiss. Res. 211 (3): 353–359.
Robinson, T.F., Hayward, B.S., Krueger, J.K., Sonnenblick, E.H., and Wittenberg, B.A. (1981). Isolated heart myocytes: ultrastructural case study technique. J. Microsc. 124 Pt. 2: 135–142.
Spurr, A.R. (1969). A low viscosity epoxy resin embedding medium for electron microscopy. J. Ultrastruc. Res. 26: 31–43.
Stempel, J.G. and Ward, R.T. (1984). An improved staining method for E.M. J. Cell Biol. 22: 697–701.
Trinick, J. and Elliott, A. (1979). Electron microscope studies of thick filaments from vertebrate skeletal muscle. J. Molec. Biol. 131: 133–136.
Venable, J.H. and Coggeshall, R. (1965). A simplified lead citrate stain for use in electron microscopy. J. Cell Biol. 25(2) Pt. 1: 407.
Wang, K. (1982). Are there new types of longitudinal filaments in the sarcomeres of striated muscles? (This volume).
Wolosewick, J.J. and Porter, K.R. (1979). Polyethylene glycol (PEG) and its application in electron microscopy. J. Cell Biol 83: 303a.
Wolosewiek, J.J. (1980). The application of polyethylene glycol (PEG) to electron microscopy. J. Cell Biol. 86: 675–681.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1984 Plenum Press, New York
About this chapter
Cite this chapter
Robinson, T.F., Cohen-Gould, L. (1984). Myofilament Diameters: An Ultrastructural Re-Evaluation. In: Pollack, G.H., Sugi, H. (eds) Contractile Mechanisms in Muscle. Advances in Experimental Medicine and Biology, vol 37. Springer, Boston, MA. https://doi.org/10.1007/978-1-4684-4703-3_5
Download citation
DOI: https://doi.org/10.1007/978-1-4684-4703-3_5
Publisher Name: Springer, Boston, MA
Print ISBN: 978-1-4684-4705-7
Online ISBN: 978-1-4684-4703-3
eBook Packages: Springer Book Archive