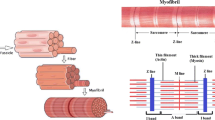
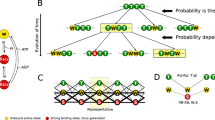
Abstract
Demembranated insect or rabbit striated muscle fibres at equilibrium (i.e. in the absence of ATP hydrolysis) were modified either by substituting ethylene glycol for water or by adding AMPPNP. The resultant states observed by electron microscopy and X-ray diffraction appeared to contain a mixture of at least two distinct types of crossbridge, which were not randomly mixed. The crossbridges held tension for a great deal longer than they remained attached to actin in solution. In the presence of AMPPNP the muscle fibres relaxed at a critical glycol concentration. These properties indicate that the crossbridges interacted with one another.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Arata, T. and Podolsky, R.J. (1982). Crossbridge flexibility derived from the influence of lattice spacing on mechanical properties of muscles fibres in rigor. Biophys. J. 37: 362a.
Barrington-Leigh, J., Goody, R.S., Holman, W., Holmes, K.C., Rosenbaum, G. and Tregear, R.T. (1977). In: Insect Flight Muscle ed. R.T. Tregear. Amsterdam: North-Holland pp 137–146.
Clarke, M.L., Marston, S.B. and Tregear, R.T. (1980). An attempt to assess the biochemically effective actin concentration in rabbit skeletal muscle. J. Muscle Res. Cell. Motil. 1: 447–448.
Clarke, M.L. (1982). The attachment of myosin heads to actin in the presence and absence of an unhydrolysable analogue of ATP. D. Phil. Thesis, Oxford.
Clarke, M.L. and Tregear, R.T. (1982). Tension maintenance and crossbridge detachment. FEES Letters (in press).
Ford, L.E., Huxley, A.F. and Simmons, R.M. (1981). The relation between stiffness and filament overlap in stimulated frog muscle fibres. J. Physiol. 311: 219–249.
Goody, R.S., Barrington-Leigh, J., Mannherz, H.G., Tregear, R.T. and Rosenbaum, G. (1976). X-ray titration of binding of β, γ, imido ATP to myosin in insect flight muscle. Nature, 262: 613–615.
Holmes, K.C., Tregear, R.T. and Barrington-Leigh, J. (1980). Interpretation of the low angle X-ray diffraction from insect flight muscle in rigor. Proc. Roy. Soc. B. 207: 13–33.
Kuhn, H.J. (1973). Transformation of chemical energy into mechanical energy by glycerol-extracted fibres of insect flight muscle in the absence of nucleosidetriphosphate hydrolysis. Experientia, 29: 1086–1088.
Marston, S.B. (1973). Kinetic studies of the contractile mechanism of muscle. D. Phil. Thesis; University of Oxford.
Marston, S.B. (1982). The rates of formation and dissociation of actin-myosin complexes. Biochem. J. 203: 453–480.
Marston, S.B. and Weber, A.M. (1975). The dissociation constant of the actin-heavy meromyosin subfragment-1 complex. Biochemistry 14: 3868–3873.
Marston, S.B., Rodger, C.D. and Tregear, R.T. (1976). Changes in muscle crossbridges when β, γ - imido-ATP binds to myosin. J. Mol. Biol. 104: 263–276.
Marston, S.B., Tregear, R.T., Rodger, C.D. and Clarke, M.L. (1979). Coupling between the enzymatic site of myosin and the mechanical output of muscle. J. Mol. Biol. 128: 111–126.
Meisner, D. and Beinbrech, G. (1979). Alterations of crossbridge angle induced by β, γ-imido-adenosine-triphosphate. Electron microscope and optical diffraction studies on myofibrillar fragments of abdominal muscles of the crayfish Orconectes limosus. Eur. J. Cell. Biol. 19: 189–195.
Miller A. and Tregear R.T. (1972). Structure of insect fibrillar flight muscle in the presence and absence of ATP. J. Mol. Biol. 70: 85–104.
Reedy, M.K. (1968). Ultrastructure of insect flight muscle. L screw sense and structural grouping in the rigor cross-bridge lattice. J. Mol. Biol. 31: 155–178.
Reedy, M.K., Leonard, K.R., Freeman, R and Arad, T. (1981). Thick myofilament mass determination by electron scattering measurements with the scanning transmission electron microscope. J. Musc. Res. Cell. Motil. 2: 45–64.
Reedy, M., Reedy, M.K. and Goody, R.S. (1981). Crossbridge structure in rigor and AMP.PNP states of insect flight muscle. Biophys. J. 33: 22a.
Tregear, R.T., Milch, J., Goody, R.S., Holmes, K.C., and Rodger, C.D. (1979). X-ray diffraction of insect flight muscle. In: Crossbridge Mechanism in Muscle Contraction, pp 407–423, ed. by H. Sugi and G. Pollack, University of Tokyo Press.
Tregear, R.T., Clarke, M.L., Marston, S.B., Rodger, C.D., Bordas, J., and Koch, M. (1982). A study of demembranated muscle fibres under equilibrium conditions. In: Basic Biology of Muscles: a Comparative Approach. Ed B. Twarog and R. Levine. Raven Press, NY..
Wells, J.A., Sheldon, M. and Yount, R.G. (1980). Magnesium nucleotide is stoicheometrically trapped at the active site of myosin and its active proteolytic fragments by thíol cross-linking reagents. J. Biol. Chem. 255: 1598–1602.
Williams, D.L. and Greene L.E. (1982). Comparison of the effect of tropomyosin and tropomin-tropomyosin on acto-51. Biophys. J. 37: 50a.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1984 Plenum Press, New York
About this chapter
Cite this chapter
Tregear, R.T., Clarke, M.L. (1984). On the Possibility of Interaction between Neighbouring Crossbridges. In: Pollack, G.H., Sugi, H. (eds) Contractile Mechanisms in Muscle. Advances in Experimental Medicine and Biology, vol 37. Springer, Boston, MA. https://doi.org/10.1007/978-1-4684-4703-3_15
Download citation
DOI: https://doi.org/10.1007/978-1-4684-4703-3_15
Publisher Name: Springer, Boston, MA
Print ISBN: 978-1-4684-4705-7
Online ISBN: 978-1-4684-4703-3
eBook Packages: Springer Book Archive