Abstract
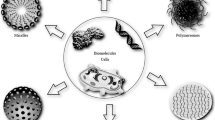
Unlike the other three major methods of enzyme immobilization, which emphasize the “microenvironment,” encapsulation of enzymes emphasizes the “intracellular environment” of enzymes and proteins (Figure 1). Here, the enzyme solution or suspension is encapsulated or enveloped within a membrane system in such a way that the membrane creates an intracellular environment for the enzymes, preventing them from leaking out or coming into direct contact with the external environment. Large molecules such as proteins and cells cannot cross the membrane to interact with the enclosed enzymes. Substrates that are permeable can equilibrate rapidly across the membrane to be acted on by the enzymes inside, and the product can diffuse out. Unlike the case of gel entrapment, which involves the entrapping of individual molecules of enzymes in polymer lattices, in encapsulation, any concentration, any volume, and any amount of enzymes can be enclosed within membrane envelopes of different configurations. This principle of allowing any type or concentration of enzymes, cells, or cell extracts to be encapsulated within membrane envelopes allows for an extremely large variation in the membrane composition, configuration, and content. This chapter is a brief review of the general principles and methods of preparation for encapsulation of enzymes and proteins, with emphasis on artificial cells. Only a brief review is made of other forms of encapsulation since they will be dealt with in detail in later chapters.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Apple, M., 1971, Hemodialysis against enzymes as a method of “gene replacement” in cases of inherited metabolic diseases, Proc. West. Pharmarol. Soc. 14: 125.
Boguslaski, R. C., and Janik, A. M., 1971, A kinetic study of microencapsulated bovine carbonic anhydrase, Biochim. Biophys. Acta 250: 266.
Broun, G., Thomas, D., Gellf, G., Domurado, D., Berjonnearu, A. M., and Gillon, T., 1973, New method for binding emzyme molecules into a water insoluble matrix—properties after insolubilization, Biotechnol. Bioeng. 15: 359.
Campbell, J., and Chang, T. M. S., 1975, Enzymatic recycling of coenzymes by a multi-enzyme system immobilized within semipermeable collodion microcapsules, Biochim. Biophys. Acta 397: 101.
Campbell, J., and Chang, T. M. S., 1976, The recycling of NAD+ (free and immobilized) within semipermeable aqueous microcapsules containing a multi-enzyme system, Biochem. Biophys. Res. Commun. 69: 562.
Chambers, R. P., Ford, J. R., Allender, J. H., Baricos, W. H., and Cohen, W., 1974, Continuous processing with cofactors requiring enzymes, coenzyme retention, and regeneration in: Enzyme Engineering (E. K. Pye and L. B. Wingard, Jr., eds.), p. 195, Plenum Press, New York.
Chang. T. M. S., 1957, Hemoglogin corpuscles, report of research project for B. Sc. Honours, McGill University.
Chang, T. M. S., 1964, Semipermeable microcapsules, Science 146: 524.
Chang, T. M. S., 1965, Semipermeable aqueous microcapsules, Ph.D. thesis, McGill University.
Chang, T. M. S., 1966, Semipermeable aqueous microcapsules (“artificial cells”): with emphasis on experiments in an extracorporeal shunt system, Trans. Amer. Soc. ArtifecialInternal Organs 12: 13.
Chang, T. M. S., 1969a, Clinical potential of enzyme technology, Science Tools 16: 33.
Chang, T. M. S., 1969b, Lipid-coated spherical ultrathin membranes of polymer or cross-linked protein as possible cell membrane models, Federation Proc. 28: 461.
Chang, T. M. S., 1971, Stabilization of enzymes by microencapsulation with a concentrated protein solution or by microencapsulation followed by cross-linking with glutaraldehyde, Biochem. Biophys. Res. Commun. 44: 1531.
Chang, T. M. S., 1972a, Artificial Cells, Charles C. Thomas, Publisher, Springfield, Ill.
Chang, T. M. S., 1972b, A new approach to separation using semipermeable microcapsules (artificial cells): combined dialysis, catalysis, and absorption, in: “Recent Development in Separation Science,” Vol. I ( N. N. Li, ed.), p. 203, CRC Press, Cleveland, Ohio.
Chang, T. M. S., 1973, L-asparaginase immobilized within semipermeable microcapsules: in-vitro and in-vivo stability, Enzyme 14: 95.
Chang, T. M. S., 1974, A comparison of semipermeable microcapsules and standard dialysers for use in separation, Separ. Purif Methods 3: 245.
Chang, T. M. S., 1976, Biodegradable semipermeable microcapsules containing enzymes, hormones, vaccines and biologicals, J. Bioengineering,(in press).
Chang, T. M. S., and Poznansky, M. J., 1968, Semipermeable aqueous microcapsules (artificial cells): V. Permeability characteristics, J. Biomed. Mater. Res. 2: 187.
Chang, T. M. S., MacIntosh, F. C., and Mason, S. G., 1963, Semipermeable aqueous microcapsules, Proc. Can. Federation Biol. Soc. 6: 16.
Chang, T. M. S., MacIntosh, F. C., and Mason, S. G., 1966, Semipermeable aqueous microcapsules: I. Preparation and properties, Can. J. Physiol. Pharmacol. 44: 115.
Chang, T. M. S., Johnson, L. J., and Ransome, O., 1967, Semipermeable aqueous microcapsules: IV. Nonthrombogenic microcapsules with heparin-complexed membranes, Can. J. Physiol. Pharmacol. 45: 705.
Choi, P. S. K., and Fan, L. T., 1973, Transient behaviour of encapsulated enzyme reactor systems, J. Apps. Chem. Biotechnol. 23: 531.
Dinelli, D., 1972, Fibre-entrapped enzymes, Process Biochem. 7: 9.
Gardner, D. L., Falb, R. D., Kim, B. C., and Emmerling, D. C., 1971, Possible uremic detoxification via oral-ingested microcapsules, Trans. Amer. Soc. Artificial Internal Organs 17: 239.
Gregoriadis, G., Leathwood, P. D., and Ryman, B. E., 1971, Enzyme-entrapment in liposomes, FEBS Letters 14: 95.
Ihler, G. M., Glew, R. H., and Schnure, F. W., 1973, Enzyme loading of erythrocytes, Proc. Natl. Acad. Sci. U.S. 70: 2663.
Kitajima, M., and Kondo, A., 1971, Fermentation without multiplication of cells using microcapsules that contain zymase complex and muscle enzyme extract, Bull. Chem. Soc. Japan 44: 3201.
Kitajima, M., Miyano, S., and Kondo, A., 1969, Studies on enzyme-containing microcapsules, J. Chem. Soc. Japan 72: 493.
Marconi, W., Guilinelli, S., and Morisi, F., 1974, Fiber-entrapped enzymes, in: Insolubilized Enzymes ( M. Salmona, C. Saronio, and S. Garattini, eds.), p. 51, Raven Press, New York.
May, S. W., and Landgraff, L. M., 1975, Cofactor recycling in liquid-membrane-enzyme systems, Biochem. Biophys. Res. Commun. 68: 786.
May, S. W., and Li, N. N., 1972, The immobilization of urease using liquid—surfactant membranes, Biochem. Biophys. Res. Commun. 47: 1179.
Mogensen, A. O., and Vieth, W. R., 1973, Mass transfer and biochemical reaction with semipermeable microcapsules, Biotechnol. Bioeng. 15: 467.
Mori, T., Tosa, T., and Chibata, I., 1973, Enzymatic properties of microcapsules containing asparagi-nase, Biochim. Biophys. Acta 321: 653.
Mosbach, K., and Mosbach, R., 1966, Entrapment of enzymes and micro-organisms in synthetic crosslinked polymers and their applications in volumn techniques, Acta Chem. Scan. 20: 2807.
Mueller, P., and Rudin, D. O., 1968, Resting and action potentials in experimental lipid membranes, J. Theoret. Biol. 18: 222.
Ostergaard, J. C. W., and Martiny, S. C., 1973, Immobilization of /3-galactosidase through encapsulation in water insoluble microcapsules, Biotchnol. Bioeng. 15: 561.
Rogers, S., 1968, Dialysis against enzymes, Nature 220: 1321.
Rony, P. K., 1971, Multiphase catalysts: II. Hollow fibre catalysts, Biotechnol. Bioeng. 13: 431.
Rosenthal, A. M., and Chang, T. M. S., 1971, The effect of ialinomycin on the movement of rubidium across lipid coated semipermeable microcapsules, Proc. Can. Federation Biol. Soc. 14: 44.
Sessa, G., and Weissman, G., 1970, Incorporation of lysozyme into liposomes, J. Biol. Chem. 245: 3295.
Shiba, M., Tomioka, S., Koishi, M., and Kondo, T., 1970, Studies on microcapsules: V. Preparation of polyamide microcapsules containing aqueous protein solution, Chem. Pharm. Bull. (Tokyo) 18: 803.
Sparks, R. E., Salemme, R. M., Meier, P. M., Litt, M. H., and Lindan, O., 1969, Removal of waste metabolites in uremia by microencapsulated reactants, Trans. Amer. Soc. Artificial Internal Organs 15: 353.
Sundaram, P. V., 1973, The kinetic properties of microencapsulated urease, Biochim. Biophys. Acta 321: 319.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1977 Plenum Press, New York
About this chapter
Cite this chapter
Chang, T.M.S. (1977). Encapsulation of Enzymes, Cell Contents, Cells, Vaccines, Antigens, Antiserum, Cofactors, Hormones, and Proteins. In: Chang, T.M.S. (eds) Biomedical Applications of Immobilized Enzymes and Proteins. Springer, Boston, MA. https://doi.org/10.1007/978-1-4684-2610-6_7
Download citation
DOI: https://doi.org/10.1007/978-1-4684-2610-6_7
Publisher Name: Springer, Boston, MA
Print ISBN: 978-1-4684-2612-0
Online ISBN: 978-1-4684-2610-6
eBook Packages: Springer Book Archive