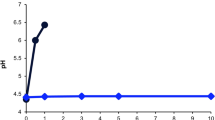
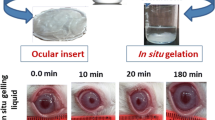
Abstract
Ophthalmic drugs are traditionally administered as solutions (collyria) or as anhydrous (petrolatum) ointments. However, it is well known that solutions do not result in administration of a precise and consistent dosage, since the induced lacrimation and reflex blinking that follow instillation quickly remove the drug from the precorneal area. Furthermore, eyedrop instillation typically results in a pulse entry mechanism, with undesirable transient peaks of drug concentration in the aqueous humour, and periods of nonmedication in the time intervals between pulses. Ointments, on the other hand, are somewhat better retained, but induce discomfort and blurred vision. Moreover, on account of their greasy nature they do not release effectively all types of drugs, and show poor patient compliance. These factors have stimulated the search for more sophisticated ophthalmic dosage forms, capable of delivering a precise drug dosage, and of maintaining an optimal concentration in the precorneal area for an extended period of time, thus rendering unnecessary a frequent administration of eyedrops.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
J. W. Shell and R. W. Baker, Diffusional Systems for Controlled Drug Release of Drugs to the Eye, Ann. Ophthalmol. 6:1037 (1974).
S. Podos, B. Becker, C. Asseff and J. Harstein, Pilocarpine Therapy with Soft Contact Lenses, Amer. J. Ophthalmol. 73:336 (1972).
A. Zaffaroni, Selective Administration of Drug with Ocular Therapeutic System, U.S. Pat. 4,186,184 (Jan 29, 1980) to Alza Corp.; through Chem. Abstr. 92:185937b (1980).
Y. F. Maichuk, Ophthalmic Drug Inserts, Invest. Ophthalmol. 14:87 (1975).
L. D. Dunn, B. S. Scott and E. D. Dorsey, Analysis of Pilocarpine and Isopilocarpine in Ophthalmic Solutions by Normal- Phase High-Performance Liquid Chromatography, J. Pharm. Sci. 70:446 (1981).
I. S. Gibbs and M. M. Tuckerman, Optimal Ferric Hydroxamate Method for Determination of Intact Pilocarpine, J. Pharm. Sci. 59:395 (1970).
G. L. Flynn, S. H. Yalkowsky and T. J. Roseman, Mass Transport Phenomena and Models, Theoretical Concepts, J. Pharm. Sci. 63: 479 (1974).
R. K. Hill and S. Barcza, Stereochemistry of the Jaborandi Alkaloids, Tetrahedron 22:2889 (1966).
S. P. Loucas and H. M. Haddad, Solid-State Ophthalmic Dosage Systems in Effecting Prolonged Release of Pilocarpine in the Cul-De-Sac, J. Pharm. Sci. 61:985 (1972).
S. P. Loucas and H. M. Haddad, Solid State Ophthalmic Dosage Systems II. Use of Polyuronic Acid in Effecting Prolonged Delivery of Pilocarpine in the Eye, Metabol. Ophthalmol. 1:27 (1976).
R. D. Schoenwald and R. E. Roehrs, Sustained Release Ophthalmic Drug Dosage, U.S. Pat. 4, 271,143 (May 9, 1979) to Alcon Laboratories, Inc.
M. F. Saettone, B. Giannaccini, A. Teneggi, P. Savigni and N. Tellini, Vehicle Effects on Ophthalmic Bioavailability: the Influence of Different Polymers on the Activity of Pilocarpine in Rabbit and Man, J. Pharm. Pharmacol. 34:464 (1982).
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1983 Plenum Press, New York
About this chapter
Cite this chapter
Saettone, M.F., Giannaccini, B., Chetoni, P., Galli, G., Chiellini, E. (1983). Polymeric Ophthalmic Drug Delivery Systems: Preparation and Evaluation of Pilocarpine-Containing Inserts. In: Chiellini, E., Giusti, P. (eds) Polymers in Medicine. Polymer Science and Technology. Springer, Boston, MA. https://doi.org/10.1007/978-1-4615-7643-3_13
Download citation
DOI: https://doi.org/10.1007/978-1-4615-7643-3_13
Publisher Name: Springer, Boston, MA
Print ISBN: 978-1-4615-7645-7
Online ISBN: 978-1-4615-7643-3
eBook Packages: Springer Book Archive