Abstract
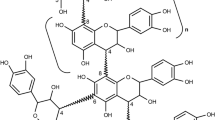
The chemistry of condensed tannins at alkaline pH is regulated by the formation of A- and/or B-ring quinone-methides as highly reactive intermediates. The former group of electrophilic species is of special significance in analogues with 5,7-dihydroxy A-ring functionality and may be successfully trapped by external nucleophiles such as phenolic nuclei and thiols. B-ring quinone-methides may similarly be trapped but are preferentially susceptible to intramolecular trapping to give analogues with rearranged pyran heterocycles. A mechanistic rationale to explain the base-catalyzed rearrangement reactions and the oxidative conversion of B- to A-type proanthocyanidins is proposed. These reactions are of relevance to the industrial application of condensed tannins.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Freudenberg, K.; Bohme, O.; Purrmann, L. Raumisomere catechine, II. Chem. Btr. 55:1734 (1922).
Freudenberg, K.; Purrmann, L. Raumisomere catechine, III. Chem. Ber. 56:1185 (1923); Raumisomere catechine, IV. Licbigs Ann. Chem. 437:274 (1924).
Whalley, W.B.; Mehta, P.P. The stereochemistry of some catechin derivatives. J. Chem. Soc.:5327 (1963).
Sears, K.D.; Casebier, R.L.; Hergert, H.L.; Stout, G.H.; McCandlish, L.E. The structure of catechinic acid. A base rearrangement product of catechin. J. Org. Chem. 39:3244(1974).
Courbat, P.; Weith, A.; Albert, A.; Pelter, A. Contribution to the study of the behavior of catechin in alkaline medium. Helv. Chem. Acta. 60:1665 (1977).
Kennedy, J.A.; Munro, M.H.G.; Powell, H.K.J.; Porter, L.J.; Foo, L.Y. The protonation reactions of catechin, epicatechin and related compounds. Aust. J. Chem. 37:885(1984).
Kuhnle, J.A.; Windle, J. J.; Waiss, A.C. Electron paramagnetic resonance spectra of flavonoid anion-radicals. J. Chem. Soc (B):613 (1969).
Jensen, O.H.; Pedersen, J.A. The oxidative transformations of (+)-catechin and (-)-epicatechin as studied by ESR. Tetrahedron 39:1609 (1983).
Foo, L.Y.; Porter, L.J. Synthesis and conformation of procyanidin diastereoisomers. J. Chem. Soc., Perkin Trans. 1:1535 (1983).
Kiatgrajai, P.; Wellons, J.D.; Gollob, L.; White, J.D. Kinetics of epimerization of (+)-catechin and its rearrangement to catechinic acid. J. Org. Chem. 47:2910 (1982).
Hemingway, R.W.; Foo, L.Y. Condensed tannins: quinone-methide intermediates in procyanidin synthesis. J. Chem. Soc., Chem. Commun.:1035 (1983).
Foo, L.Y.; Hemingway, R.W. Condensed tannins: synthesis of the first ‘branched ‘procyanidin trimer. J. Chem. Soc, Chem Commun.:85 (1984).
Laks, P.E.; Hemingway, R.W. Condensed Tannins: base-catalysed reactions of polymeric procyanidins with toluene-α-thiol. Lability of the interflavanoid bond and pyran ring. J. Chem. Soc, Perkin Trans. 1:465 (1987).
Laks, P.E.; Hemingway, R.W.; Conner, A.H. Condensed tannins. Base-catalyzed reactions of polymeric procyanidins with phloroglucinol: intramolecular rearrangements. j. Chem. Soc., Perkin Trans. 1:1875 (1987).
Attwood, M.R.; Brown, B.R.; Lisseter, S.G.; Torrero, C.L.; Weaver, P.M. Spectral evidence for the formation of quinone-methide intermediates from 5-and 7-hydroxyflavonoids. J. Chem. Soc., Chem. Commun.:177 (1984).
Hemingway, R.W.; Laks, P.E. Condensed tannins: a proposed route to 2R,3R-(2,3-cis)-proanthocyanidins. J. Chem. Soc., Chem. Commun.:746 (1985).
Jacques, D.; Opie, C.T.; Porter, L.J.; Haslam, E. Plant proanthocyanidins. Part 4. Biosynthesis of procyanidins and observations on the metabolism of cyanidin in plants. J. Chem. Soc., Perkin Trans. 1:1637 (1977).
Haslam, E. Symmetry and promiscuity in procyanidin biochemistry. Phytochemistry 16:1625 (1977).
Platt, R.V.; Opie, CT.; Haslam, E. Biosynthesis of fiavan-3-ols and other secondary plant products from (2S)-phenylalanine. Phytochemistry 23:2211 (1984).
Czochanska, Z.; Foo, L.Y.; Newman, R.H.; Porter, L.J. Polymeric proanthocyanidins. Stereochemistry, structural units, and molecular weight. J. Chem. Soc., Perkin Trans. 1:2278 (1980).
Hemingway, R.W.; Foo. L.Y.; Porter, L.J. Linkage isomerism in trimeric and polymeric 2,3-cis procyanidins. J. Chem. Soc., Perkin Trans. 1:1209 (1982).
Laks, P.E.; Hemingway, R.W. Condensed tannins: structure of the ‘phenolic acids’. Holzforschnng 41:287 (1987).
Pizzi, A. Wood adhesives: chemistry and technology. Marcel Dekker, New York (1983).
Kreibich, R.E.; Hemingway, R.W. The use of tannins in structural laminating adhesives. In: Proceedings of IUFRO-NTRI Symposium on Wood Adhesives, CSIR, Pretoria, South Africa. 17-5:1 (1985).
Steenkamp, J.A.; Steynberg. J.P.; Brandt, E.V.; Ferreira, D.; Roux, D.G. Phlobatannins, a novel class of ring-isomerized condensed tannins. J. Chem. Soc., Chem. Commun.:1678 (1985).
Steynberg, J.P.; Burger, J.F.W.; Young, D.A.; Brandt, E.V.; Steenkamp, J.A.; Ferreira, D. Novel base-catalyzed rearrangements of (-)-fisetinidol-(+)-catechin profisetinidins with 2,3-trans-3,4-cis-flavan-3-ol constituent units. J. Chem. Soc., Chem. Commun.:1055 (1988).
Steynberg, J.P.; Burger, J.F.W.; Young, D.A.; Brandt, E.V.; Steenkamp, J.A.; Ferreira, D. Oligomeric flavanoids. Part 3. Structure and synthesis of phlobatannins related to (-)-fisetinidol-(4α,6)-and (4α,8)-(+)-catechin profisetinidins. J. Chem. Soc., Perkin Trans. 1:3323 (1988).
Steynberg, J.P.; Burger, J.F.W.; Young, D.A.; Brandt, E.V.; Steenkamp, J.A.; Ferreira, D. Oligomeric flavanoids. Part 4. Base-catalyzed conversions of (-)-fisetinidol-(+)-catechin profisetinidins with 2,3-trans-3,4-cis-flavan-3-ol constituent units. J. Chtm. Soc., Ptrkin Trans. 1:3331 (1988).
Malan, J.C.S.; Steenkamp, J.A.; Steynberg, J.P.; Young, D.A.; Brandt, E.V.; Ferreira, D. Oligomeric flavanoids. Part 8. The first profisetinidins and proguibourtinidins based on C-8 substituted (-)-fisetinidol units and related C-ring isomerized analogues. J. Chem. Soc., Ptrkin Trans. 1:209 (1990).
Malan, J.C.S.; Young, D.A.; Steynberg, J.P.; Ferreira, D. Oligomeric flavanoids. Part 10. Structure and synthesis of the first tetrahydropyrano[3,2-g]chromenes related to (4,6)-bis-(-)-fisetinidol profisetinidins. J. Chem. Soc., Ptrkin Trans. 1:227 (1990).
Steynberg, J.P.; Steenkamp, J.A.; Burger, J.F.W.; Young, D.A.; Ferreira, D. Oligomeric flavanoids. Part 11. Structure and synthesis of the first phlobatannins related to (4α,6:4α,8)-bis-(-)-fisetinidol-(-(-)-catechin profisetinidin triflavanoids. J. Chem. Soc., Perkin Trans. 1:235 (1990).
Steynberg, J.P.; Burger, J.F.W., Cronjé, A.; Bonnet, S.L.; Malan, J.C.S.; Young, D.A.; Ferreira, D. Structure and synthesis of phlobatannins related to (-)-fisetinidol-(-)-epicatechin profisetinidins. Phytochemistry 29:2979 (1990).
Steynberg, P.J.; Burger, J.F.W.; Bezuidenhoudt, B.C.B.; Steynberg, J.P.; Van Dyk, M.S.; Ferreira, D. The first natural condensed tannins with (-)-catechin ‘terminal’ units. Tetrahedron Lett. 31:2059 (1990).
Cronjé, A.; Young, D.A.; Steynberg. J.P.; Ferreira, D. [unpublished results]
Hundt, H.K.L.; Roux, D.G. Condensed tannins: determination of the point of linkage in ‘terminal’ (+)-catechin units and degradative bromination of 4-flavanylflavan-3,4-diols. J. Chem. Soc., Chem Commun.:696 (1978). Synthesis of condensed tannins. Part 3. Chemical shifts for determining the 6-and 8-bonding positions of ‘terminal’ (+)-catechin units. J. Chem. Soc., Perkin Trans. 1:1277 (1981).
Young, E.; Brandt, E.V.; Young, D.A.; Ferreira, D.; Roux, D.G. Synthesis of condensed tannins. Part 17. Oligomeric (2R,3S)-3,3′,4′,7,8-pentahydroxyflavans: atropisomerism and conformation of biphenyl and m-terphenyl analogues from Prosopis glandulosa (‘Mesquite’). J. Chem. Soc., Perkin Trans. 1:1737 (1986).
Young, D.A.; Cronjé, A.; Botes, A.L.; Ferreira, D.; Roux, D.G. Synthesis of condensed tannins. Part 14. Biflavanoid profisetinidins as synthons. The acid induced ‘phlobaphene’ reaction. J. Chem. Soc., Perkin Trans. 1:2521 (1985).
Steynberg, J.P.; Young, D.A.; Burger, J.F.W.; Ferreira, D.; Roux, D.G. Phlobatannins via facile ring isomerizations of profisetinidin and prorobinetinidin condensed tannin units. J. Chem. Soc., Chem. Commun.:1013 (1986).
Sweeny, G.J.; Iacobucci, G.A. Regiospecificity of (+)-catechin methylation. J. Org. Chem. 44:2298 (1979).
Ungaro, R.; Pochini, A.; Andreetti, G.D.; Domiano, P. Molecular inclusion in functionalized macrocycles. Part 9. The crystal and molecular structure of P-t-butylcalix[4]-anisole (2:1) complex: a new type of cage inclusion compound. J. Chem. Soc., Perkin Trans. 2:197 (1985).
Steynberg, J.P.; Brandt, E.V.; Ferreira, D. [unpublished results].
Bergman, W.R.; Viswanadhan, V.N.; Mattice, W.L. Conformations of polymeric proantho-cyanidins composed of (+)-catechin or (-)-epicatechin joined by 4-6 interflavan bonds. J. Chem. Soc., Perkin Trans. 2:45 (1988).
Botha, J.J.; Ferreira, D.; Roux, D.G. Condensed tannins. Circular dichroism method of assessing the absolute configuration at C-4 of 4-arylflavan-3-ols, and stereochemistry of their formation from flavan-3,4-diols. J. Chem. Soc., Chem. Commun.:699 (1979): Synthesis of condensed tannins. Part 1. Stereoselective and stereospecific syntheses of optically pure 4-arylflavan-3-ols, and assessment of their absolute stereochemistry at C-4 by means of circular dichroism. J. Chem. Soc., Perkin Trans. 1:1213 (1981).
Barrett, M.W.; Klyne, W.; Scopes, P.M.; Fletcher, A.C.; Porter, L.J.; Haslam, E. Plant proanthocyanidins. Part 6. Chiroptical studies. Part 95. Circular dichroism of procyani-dins. J. Chem. Soc., Perkin Trans. 1:2375 (1979).
Van der Westhuizen, J.H.; Ferreira, D.; Roux, D.G. Synthesis of condensed tannins. Part 2. Synthesis by photolytic rearrangement, stereochemistry, and circular dichroism of the first 2,3-cis-3,4-cis-4-arylflavan-3-ols. J. Chem. Soc., Perkin Trans. 1:122O (1981).
Porter, L.J.; Wong, R.Y.; Benson, M.; Chan, B.G.; Vishwanadhan, V.N.; Gandour, R.D.; Mattice, W.L. Conformational analysis of flavans: 1H NMR and molecular mechanical (MM2) studies of the benzopyran ring of 3′,4′,5,7-tetrahydroxyflavan-3-ols: the crystal and molecular structure of the procyanidin: (2R,3S,4R)-3′,4′,5,7-tetramethoxy-4(2,4,6-trimethoxyphenyl)-flavan-3-ol. J. Chem. Res. M:830 (1986); S:86 (1986).
Steynberg, J.P.; Burger, J.F.W.; Young, D.A.; Brandt, E.V.; Ferreira, D. Oligomeric fla-vanoids. Part 6. Evidence supporting the inversion of absolute configuration at 3-C associated with base catalyzed A-/B-ring interchange of precursors having 2,3-trans-3,4-cis-flavan-3-ol constituent units. Heterocycles 28:923 (1989).
De Angelis, G.G.; Wildman, W.C. Circular dichroism studies-I. A quadrant rule for the optically active aromatic chromophore in rigid polycyclic systems. Tetrahedron 25:5099 (1969).
Burger, J.F.W.; Steynberg, J.P.; Young, D.A.; Brandt, E.V.; Ferreira, D. Oligomeric flavanoids. Part 5. Base-catalyzed C-ring isomerization of (+)-fisetinidol-(+)-catechinprofise-tinidins. J. Chem. Soc., Perkin Trans. 1:671 (1989).
Steynberg, J.P.; Burger, J.F.W.; Malan, J.C.S.; Cronjé, A.; Young, D.A.; Ferreira, D. Natural (-)-fisetinidol-(4,8)-(-)-epicatechin profisetinidins. Phytochemistry 29:275 (1990).
Slabbert, N.P. Ionisation of some flavanols and dihydrofiavanols. Tetrahedron 33:821 (1977).
Steenkamp, J.A.; Malan, J.C.S.; Roux, D.G.; Ferreira, D. Oligomeric flavanoids. Part 1. Novel dimeric profisetinidins from Colophospermum mopane. J. Chem. Soc., Perkin Trans. 1:1325 (1988).
Botha, J.J.; Ferreira, D.; Roux, D.G. Synthesis of condensed tannins. Part 4. A direct biomimetic approach to [4,6]-and [4,8]-biflavanoids. J. Chem. Soc., Perkin Trans. 1:1235 (1981).
Pelter, A.; Amenechi, P.I.; Warren, R.; Harper, S.H. The structure of two proanthocyanidins from Julbernardia globiflora. J. Chem. Soc. (C):2572 (1969).
Delle Monache, F.; Ferrari, F.; Poce-Tucci, A.; Marini-Bettolo, G.B. Catechins with (+)-epi-configuration in nature. Phytochemistry 11:2333 (1972).
Ellis, C.J.; Foo, L.Y.; Porter, L.J. Enantiomerism: a characteristic of the proanthocyanidin chemistry of the monocotyledonae, Phytochemistry 22:483 (1983).
Kashiwada, Y.; Iizuka, A.; Yoshioka, K.; Chen, R.; Nonaka, G.; Nishioka. I. Tannins and related compounds. XXCIII. Occurrence of enantiomeric proanthocyanidins in the Leguminosae plants, Cassia bistula L. and C. javanica L. Chem. Pharm. Bull. 38:888 (1990).
Dale, J.A.; Mosher, H.S. Nuclear magnetic resonance enantiomer reagents. Configurational correlations via nuclear magnetic resonance chemical shifts of diastereomeric mandelate, O-methylinandelate, and α-methoxy-α-trifluoromethylphenylacetate (MTPA) esters. J. Am. Chem. Soc. 95:512 (1973).
Sullivan, G.R.; Dale, J.A.; Mosher, H.S. Correlation of configuration and 19F chemical shifts of α-methoxy-α-trifluoromethylphenylacetate derivatives. J. Org. Chem. 38:2143 (1973).
Hundt, A.F.; Burger, J.F.W.; Steynberg, J.P.; Steenkamp, J.A.; Ferreira, D. Absolute configurations of flavan-3-ols and 4-arylflavan-3-ols via a modified Mosher’s method. Tetrahedron Lett. 31:5073 (1990).
Burger, J.F.W.; Kolodziej, H.; Hemingway, R.W.; Steynberg, J.P.; Young, D.A.; Ferreira, D. Oligomeric flavanoids. Part 15. Base-catalyzed pyran rearrangements of procyanidin B-2, and evidence for the oxidative transformation of B-to A-type procyanidins. Tetrahedron 46:5733 (1990).
Steynberg, J.P.; Bezuidenhoudt, B.C.B.; Burger, J.F.W.; Young, D.A.; Ferreira, D. Oligomeric flavanoids. Part 7. Novel base-catalyzed rearrangements of procyanidins. J. Chem. Soc., Perkin Trans. 1:203 (1990).
Morimoto, S.; Nonaka, G.; Nishioka, I. Tannins and related compounds. LIX. Aesculitannins, novel proanthocyanidins with doubly-bonded structures from Aesculus hippocastanum L. Chem. Pharm. Bull. 35:4717 (1987).
Hashimoto, F.; Nonaka, G.; Nishioka, I. Tannins and related compounds. XC. 8-C-Ascorbyl (-)-epigallocatechin 3-O-gallate and novel dimeric flavan-3-ols, oolonghomobisflavans A and B, from Oolong tea. (3). Chem. Pharm. Bull. 37:3255 (1989).
Cronjé, A.; Burger, J.F.W.; Brandt, E.V.; Kolodziej, H.; Ferreira, D. Assessment of 3,4-trans and 3,4-cis relative configurations in the A-series of (4,8)-linked proanthocyanidins. Tetrahedron Lett. 31:3789 (1990).
Musso, H. In: Oxidative coupling of phenols. Taylor, A.I and Battersby A. R. (eds.), Marcel Dekker, New York:75 (1967).
Jacques, D.; Haslam, E.; Bedford, G.R.; Greatbanks, D. Plant proanthocyanidins. Part II. Proanthocyanidin-A2 and its derivatives. J. Chem. Soc., Perkin Trans. 1:2663 (1974).
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1992 Springer Science+Business Media New York
About this chapter
Cite this chapter
Ferreira, D., Steynberg, J.P., Burger, J.F.W., Bezuidenhoudt, B.C.B. (1992). Oxidation and Rearrangement Reactions of Condensed Tannins. In: Hemingway, R.W., Laks, P.E. (eds) Plant Polyphenols. Basic Life Sciences, vol 59. Springer, Boston, MA. https://doi.org/10.1007/978-1-4615-3476-1_20
Download citation
DOI: https://doi.org/10.1007/978-1-4615-3476-1_20
Publisher Name: Springer, Boston, MA
Print ISBN: 978-1-4613-6540-2
Online ISBN: 978-1-4615-3476-1
eBook Packages: Springer Book Archive