Abstract
There has been an increased interest in studying other factors that affect asthma pathogenesis and cause heterogeneity in prevalence and incidence of asthma. The reason there are such varied expression patterns of disease in asthmatics is because of multiple variables that affect the pathogenesis of asthma. As an exemplar of an epidemiologic variable, we will discuss geographical location, obesity and vitamin D status of the individual, and their effects on asthma burden in humans. There is varying data regarding the prevalence or severity of asthma in urban versus rural setting which is likely related to the difference of the populations studied, complexity of causal variables involved, and local geographic factors. In addition to cross-sectional and cohort studies in humans, animal models and studies have established a link between asthma and obesity by investigating the mechanisms affecting both disease processes. The complicated interrelationship between obesity and asthma is an active area of epidemiological and experimental research with new insights being discovered at a rapid pace. Finally, vitamin D, an important immunomodulator thought to be important in pathogenesis of asthma, has both mechanistic and therapeutic implications in treatment of asthma. The influences of these factors on the clinical expression of asthma are discussed below.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Rural
- Urbanization
- Obesity
- Population differences in asthma burden
- Vitamin D
- Public health and asthma
- Heterogeneous expression of asthma in population
1 Key Epidemiologic Factors and Influences on Asthma
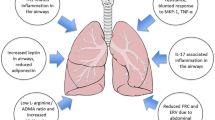
The prevalence of asthma is diverse and the penetrance of the disease in the population is variable. Since there are multiple pathogenetic factors (Fig. 3.1) that are responsible for evolution of the disease process including but not limited to environmental factors (air pollution, pollens, mold and other aeroallergens, and weather), host factors (geographical location, BMI, smoking status, nutritional factors), agent factors (infectious agents like respiratory syncytial virus RSV, rhinovirus, etc.), and genetic factors (asthma susceptibility loci on genes involved in pathogenesis of the disease), the burden of asthma in the society is also mixed. Some factors have a direct input on the expression of the disease state like exposure to infectious asthmogenic viruses or exposure to allergens, while other factors act in more subtle ways. In the section that follows, we will discuss the effect of three factors as an exemplar of direct or indirect effect on the expression of the disease state in the population and will cover urban–rural differences in asthma prevalence, obesity and its link to asthma and vitamin D status, and its role in pathogenesis of asthma. Examples of triggers that act indirectly include obesity and nutritional state of the individual and have a broad influence on immunological or local (respiratory epithelium or bronchial smooth muscle) physiological mechanisms that lead to development of asthma. Some factors like geographical location act indirectly or directly because there are many things that influence the asthmogenic triggers like local weather, environmental pollutants, prevalent aeroallergens, and effects of local ecosystems.
2 Role of Urbanization
Asthma has a genetic component in pathogenesis, but the effect of the environment is also extremely important, and the increased exposure to air pollutants and inhalants may intuitively suggest an increase in respiratory-related disease in adults and children. Outdoors air pollution including ozone, diesel fumes, smoke, and small particulate matter is thought to contribute to the pathogenesis of airway hyperresponsiveness and has been reviewed in Chap. 3. ISAAC study has demonstrated that globally for affluent nations, the combined odds ratio for association of atopic sensitization (positive skin prick test) and asthma symptoms (wheezing) was approximately twice compared to nonaffluent nations (Weinmayr et al. 2007). The authors observed a link between improving economic development and atopic sensitization and asthma symptoms.
2.1 Increased Asthma in Urban Setting: Case for Local Factors
It may be inferred that since economic progress shares some basis in urban-industrial development, urbanization directly or indirectly may be responsible for higher rates of asthma. This has been demonstrated to be true at least in few instances where prevalence of asthma increased with increasing level of urbanization (Rodriguez et al. 2011). Specific epidemiologic studies to address the question of urban–rural disparity in asthma prevalence have been conducted using the National Health Survey (1988), which showed significant increase in asthma reported from urban areas (7.1 %) as compared to rural areas (5.7 %) (Gergen et al. 1988). The finding that urban areas have higher prevalence of asthma than rural areas was also documented in Canada, where investigators looked at age-defined cross-sectional data and found that asthma had lower prevalence in rural areas (Lawson et al. 2011). These differences have been validated by direct bronchoprovocation challenge studies between rural and urban residents with higher hyperreactivity in urban children with asthma (Turner et al. 1986). Whether increase in asthma secondary to urbanization was a direct consequence of human development or due to increase in atopy from urbanization in the population is still being investigated. In a study by Bibi et al. of the 448 asthmatic children tested, those living in urban areas demonstrated more allergic response to indoor and outdoor allergens (Bibi et al. 2002). There are also clues indicating that urban environment affects asthma prevalence without affecting atopy. Environmental air pollution is thought to have a direct influence on asthma, especially in previously sensitized individuals, where it made preexisting symptoms worse (Corbo et al. 1993). GABRIEL advance studies have shown a protective effect of farm exposure in asthma independent of atopic status (Fuchs et al. 2012).
2.2 Conflicting Results: Rural Versus Urban Asthma Debate
As early as 1978, Bouhuys et al. published that there was no difference in lung function tests between lifetime residents in a rural area and those in a small industrial town in Connecticut, USA (Bouhuys et al. 1978). The incongruence between air pollution and airway responsiveness was mirrored when bronchial hyperresponsiveness and asthma in young students in Guangzhou, China, were studied, and there was no significant difference between urban and rural population (Zhong et al. 1990). This lack of difference in lung function between long-standing residents of urban versus rural residents was surprising and may reflect the local environmental effects or protective genetic factors in populations studied. The importance of location-specific results is demonstrated by studies that show that overall prevalence of asthma is not different between urban and rural residence areas (Morrison et al. 2009) or that the difference is in increased morbidity from asthma in rural areas as opposed to prevalence (Pesek et al. 2010).
NHANES II found that residents of urban areas reported reduced prevalence of asthma as compared to residents of rural areas (Turkeltaub and Gergen 1991). In a study based in Tennessee, USA, rural children had increased asthma prevalence but similar asthma morbidity compared to urban children (Valet et al. 2011). The conflicting data regarding the different results of prevalence of asthma in urban setting is likely related to the heterogeneity of the populations studied, complexity of causal variables involved, and local geographic factors. Each defined geographic location has a distinct and unique set of factors that may be contributing to pathogenesis of asthma.
3 Obesity
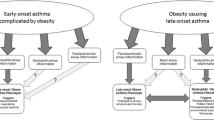
Asthma manifests with dyspnea and wheezing which can also be seen in obese individuals. There has been a tremendous interest in obesity-related diseases including asthma in the recent time. In addition to the cross-sectional and cohort studies in humans showing a modest increase in asthma with increasing prevalence of obesity, animal models and studies have also established a link between asthma and obesity by investigating the mechanisms affecting both disease processes (Beuther et al. 2006).
3.1 Correlative Studies of Obesity and Asthma
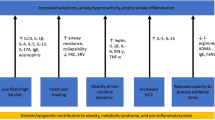
In children with single diagnosis of asthma, the prevalence of being overweight was significantly higher in those with moderate to severe asthma (Luder et al. 1998). Though the population here was predominantly black and Hispanic children from inner cities, this was also reported to be an issue in women. In a prospective cohort after controlling for the confounding factors, relative risk for asthma increased as BMI increased. Moreover, women who gained weight after age 18 were at increased risk of developing asthma (Camargo et al. 1999). This propensity to develop asthma after weight gain was not limited to adult-onset obesity but was also documented in preadolescent girls who became obese between 6 and 11 years of age (Castro-Rodríguez et al. 2001). Thus, the risk for asthma shows an increase with increase in BMI since childhood. While data seem definitive in women (Guerra et al. 2002), a meta-analysis showed a similar increase in odds ratio of incident asthma due to increasing weight in men as well (Beuther and Sutherland 2007). Correlation with asthma is not universal with all morphometric types of obesity; central obesity was significantly associated with increased risk of nonatopic asthma (Appleton et al. 2006).
3.2 Mechanisms of Effect of Obesity on Asthma
While it is instinctive that increased weight may have a restrictive effect on lung function, there are, in addition, bronchospastic and non-bronchospastic mechanisms in place. While adult men with lowest and highest BMI were more likely to develop airway hyperresponsiveness (Litonjua et al. 2002) demonstrating a nonlinear relationship, each increase in BMI of five units in children was associated with a decrease in FEV1/FVC of over 1 % with mild-to-moderate asthma (Tantisira et al. 2003). The distinction between airflow limitation and dynamic airway hyperresponsiveness is important in obese individuals, and it may be that not all obese individuals have wheezing as a consequence of increase airway hyperreactivity.
Leptin and adiponectin are adipocyte-derived hormones, demonstrating immunomodulatory effects, and have been studied for their effect on asthma in human subjects. Adiponectin levels have been shown to be inversely related to asthma symptoms and positively with FEV1/FVC ratio in male subjects independent of body size (Kattan et al. 2010). Increased levels of leptin correlated with obesity status of children, and higher levels of leptin were associated with bronchial hyperresponsiveness induced by exercise challenge (Baek et al. 2011). Although associative studies do not offer insights into the causal role of the factors being studied, the direct role of adipokines in asthma has been elucidated in the laboratory using animal models of asthma. Leptin infusion augmented methacholine hyperresponsiveness and serum IgE response in mice alluding to a possible role of serum leptin in pathogenesis of asthma (Shore et al. 2005). In contrast to effect of leptin, adiponectin treatment has shown reduced OVA-induced allergic airway responses in mice indicating a protective effect of this hormone on pathogenesis of asthma (Shore et al. 2006).
In addition to airway hyperresponsiveness as a cause of increased asthma, other immune mechanisms have been investigated for their role in asthma with mixed results. While obese difficult-to-treat asthmatics demonstrated an inverse relationship to sputum eosinophils and FeNO with increasing body mass (Van Veen et al. 2008), there was no association between BMI and airway inflammation (including eosinophils) as measured by sputum cell counts (Todd et al. 2007). Results of studies attempting to find associations between systemic inflammation and asthma have been unclear as well. The question whether asthma develops in parallel to obesity or precedes or follows obesity is a difficult one to answer in human subjects. Using a cohort of patients from the NHEFS (nutrition evaluation survey epidemiologic follow-up study), increasing BMI was associated with increased asthma prevalence, but during observation period there was no increased incidence of asthma with increasing BMI (Stanley et al. 2005).
3.3 Treatment Considerations in Obese Asthmatics
Obese adults had poorer asthma control and higher likelihood of asthma-related hospitalizations (Mosen et al. 2008). In addition to the pathologic aspects of disease itself, there was also a synergistic influence of obesity on asthma. Compared to normal children as controls, quality of life was poorer in children with obesity and asthma, compared to children with asthma and normal weight and only overweight children (Van Gent et al. 2007). Besides physical impediments to asthma care and issues with symptom perception (Lessard et al. 2008), elevated BMI has effects on response to medications on a pharmacological level. Higher BMI was associated with blunted in vitro response to dexamethasone in obese patients with asthma (Sutherland et al. 2008). Obese asthmatics demonstrated less improvement with inhaled corticosteroids as measured by lung function and exhaled feNO than lean asthma patients (Sutherland et al. 2009). This reduction in benefit was also seen with combination controller therapy in obese asthmatics compared to normal asthmatics (Camargo et al. 2010). These differences could be related to the difference in distribution of the drug, metabolism, and receptor function in obese individuals resulting in a distinctive response to medications. This complicated interrelationship between obesity and asthma is an active area of epidemiological and experimental research with new results being discovered at a rapid pace.
4 Vitamin D
Airway inflammation in asthma plays an important role in disease progression. Early observations suggesting a pivotal role of vitamin D modulation of immune system and the immunological basis of airway inflammation alludes to effect of vitamin D on asthma (Mora et al. 2008).
4.1 Relationship of Vitamin D and Pediatric Asthma
Insufficient vitamin D levels were associated with higher odds of exacerbation of asthma in children (Brehm et al. 2010). The magnitude of association of vitamin D deficiency and severity of asthma exacerbation was greater in children with nonatopic disease (Brehm et al. 2012). The relationship between vitamin D-deficient state and asthma was not only quantitative but also temporal because vitamin D levels at younger age were predictive of asthma–allergy phenotype at later ages (Hollams et al. 2011). Several studies have also looked at the role of maternal vitamin D levels and the outcome in offspring of those mothers. Surprisingly, both low and high levels of maternal vitamin D were associated with increased total and specific IgE through age 5 years, but this study did not find an association between vitamin D levels in cord blood and asthma in children (Rothers et al. 2011). While some studies found that higher maternal vitamin D levels were associated with decreased odds of asthma (Carroll et al. 2011), other investigators have not found a significant association between maternal late-pregnancy 25-hydroxyvitamin D status, and either asthma or wheeze at age 6 years (Pike et al. 2012) indicating that the role of maternal vitamin D levels in development of asthma in offspring is complex.
4.2 Adult Vitamin D and Asthma
There is increasing evidence regarding the role of vitamin D in adult asthma, and results from studies in non-pediatric segments of the population point towards an important role of vitamin D. Each 10 ng/ml decrease in vitamin D was associated with 8 % greater odds of asthma (Keet et al. 2011). A correlation between the latitude, sun exposure, and adult asthma prevalence has also been reported. Each 10-degree change in geographical latitude from southern to northern regions of eastern seaboard in the USA was associated with 2 % increase in adult asthma prevalence (Krstic 2011). This effect of vitamin D on the pathogenesis of asthma seems disease specific because baseline vitamin D levels had no relationship to rates of acute exacerbation of COPD or time to first exacerbation of COPD (Kunisaki et al. 2012).
4.3 Mechanisms of Vitamin D Modulation of Asthma
There is pathologic basis for poor asthma control and propensity to worsening in populations that have lower levels of vitamin D. Children with severe treatment-resistant asthma with lower vitamin D were associated with higher airway smooth muscle mass likely contributing to a worse lung function and poor asthma control (Gupta et al. 2011). Low vitamin D was associated with higher levels of serum IgE in pediatric asthmatic patients studied (Goleva et al. 2012). In offspring of vitamin D-deficient mice, increase in proliferation and cytokine production was seen in airway draining lymph nodes in response to OVA-induced asthma signifying an activated immune phenotype (Gorman et al. 2012). Vitamin D has a myriad of biologic effects, which have been shown to orchestrate essential roles in immune activation including downregulation of co-stimulators CD80/86, RANTES, CD40, MMPs, and PDGF while upregulating immunomodulators like IL-10 (Sandhu and Casale 2010). In addition to its effect in cell surface markers and biomolecules, levels of vitamin D were found to be positively correlated with Treg numbers. Regulatory T cells (Treg) are important in damping immune responses from effector T and help suppress inflammatory states. In addition to affecting immune system, vitamin D deficiency also has been shown to affect the physical properties of the lung in animal models including demonstration of reduction in lung volume in mice (Zosky et al. 2011). Thus, it is evident vitamin D has broad and overreaching influence on the genesis and evolution of asthma.
4.4 Treatment Considerations
The implications of vitamin D deficiency in asthma are not only mechanistic but also have therapeutic consequences. In animal studies, administration of vitamin D3 in ovalbumin (OVA)-induced asthma model of mice inhibited bronchial hyperresponsiveness, OVA-specific IgE, airway eosinophilia, and Th2-related cytokines in conjunction with OVA immunotherapy (Taher et al. 2008). Studies in humans have supported the role of vitamin D in asthma as well with reduced vitamin D being associated with impaired lung function, increased AHR, and reduced response to glucocorticoids in nonsmoking asthmatic patients (Sutherland et al. 2010). Levels of vitamin D have been demonstrated to have strong inverse correlation to the use of inhaled steroids or oral steroids in children with asthma (Searing et al. 2010), and children treated with inhaled budesonide and supplemented with vitamin D had fewer asthma exacerbations (Majak et al. 2011). These results point to an important role of vitamin D in treatment of asthma. At the present time, there is little evidence that treating vitamin D deficiency improves asthma, but there are several prospective clinical trials underway testing the hypothesis. While it may be premature to measure vitamin D levels in all asthmatics, it may be worthwhile considering levels of vitamin D in difficult-to-control asthmatics or those with severe asthma.
5 Summary
It is important to realize the variegated clinical presentation of asthma and the heterogeneous expression of the disease in the population share some of the causal factors. Recognition of these expression patterns of disease and appropriate identification of triggers will help clarify pathogenic mechanisms and allow for a patient-specific treatment regimen.
References
Appleton SL, Adams RJ, Wilson DH et al (2006) Central obesity is associated with nonatopic but not atopic asthma in a representative population sample. J Allergy Clin Immunol 118(6):1284–1291
Baek HS, Kim YD, Shin JH et al (2011) Serum leptin and adiponectin levels correlate with exercise-induced bronchoconstriction in children with asthma. Ann Allergy Asthma Immunol 107(1):14–21
Beuther DA, Sutherland ER (2007) Overweight, obesity, and incident asthma: a meta-analysis of prospective epidemiologic studies. Am J Respir Crit Care Med 175(7):661–666
Beuther DA, Weiss ST, Sutherland ER (2006) Obesity and asthma. Am J Respir Crit Care Med 174(2):112–119, Epub 2006 Apr 20
Bibi H, Shoseyov D, Feigenbaum D et al (2002) Comparison of positive allergy skin tests among asthmatic children from rural and urban areas living within small geographic area. Ann Allergy Asthma Immunol 88(4):416–420
Bouhuys A, Beck GJ, Schoenberg JB (1978) Do present levels of air pollution outdoors affect respiratory health? Nature 276(5687):466–471
Brehm JM, Schuemann B, Fuhlbrigge AL et al (2010) Serum vitamin D levels and severe asthma exacerbations in the Childhood Asthma Management Program Study. Childhood Asthma Management Program Research Group. J Allergy Clin Immunol 126(1):52–8.e5
Brehm JM, Acosta-Pérez E, Klei L et al (2012) Vitamin D insufficiency and severe asthma exacerbations in Puerto Rican children. Am J Respir Crit Care Med 186(2):140–146
Camargo CA Jr, Weiss ST, Zhang S et al (1999) Prospective study of body mass index, weight change, and risk of adult-onset asthma in women. Arch Intern Med 159(21):2582–2588
Camargo CA Jr, Boulet LP, Sutherland ER et al (2010) Body mass index and response to asthma therapy: fluticasone propionate/salmeterol versus montelukast. J Asthma 47(1):76–82
Carroll KN, Gebretsadik T, Larkin EK et al (2011) Relationship of maternal vitamin D level with maternal and infant respiratory disease. Am J Obstet Gynecol 205(3):215.e1–7
Castro-Rodríguez JA, Holberg CJ, Morgan WJ et al (2001) Increased incidence of asthma like symptoms in girls who become overweight or obese during the school years. Am J Respir Crit Care Med 163(6):1344–1349
Corbo GM, Forastiere F, Dell’Orco V et al (1993) Effects of environment on atopic status and respiratory disorders in children. J Allergy Clin Immunol 92(4):616–623
Fuchs O, Genuneit J, Latzin P et al (2012) Farming environments and childhood atopy, wheeze, lung function, and exhaled nitric oxide. J Allergy Clin Immunol 130(2):382–8.e6
Gergen PJ, Mullally DI, Evans R 3rd (1988) National survey of prevalence of asthma among children in the United States, 1976 to 1980. Pediatrics 81(1):1–7
Goleva E, Searing DA, Jackson LP et al (2012) Steroid requirements and immune associations with vitamin D are stronger in children than adults with asthma. J Allergy Clin Immunol 129(5):1243–1251
Gorman S, Tan DH, Lambert MJ et al (2012) Vitamin D(3) deficiency enhances allergen-induced lymphocyte responses in a mouse model of allergic airway disease. Pediatr Allergy Immunol 23(1):83–87
Guerra S, Sherrill DL, Bobadilla A et al (2002) The relation of body mass index to asthma, chronic bronchitis, and emphysema. Chest 122(4):1256–1263
Gupta A, Sjoukes A, Richards D et al (2011) Relationship between serum vitamin D, disease severity, and airway remodeling in children with asthma. Am J Respir Crit Care Med 184(12):1342–1349
Hollams EM, Hart PH, Holt BJ et al (2011) Vitamin D and atopy and asthma phenotypes in children: a longitudinal cohort study. Eur Respir J 38(6):1320–1327
Kattan M, Kumar R, Bloomberg GR et al (2010) Asthma control, adiposity, and adipokines among inner-city adolescents. J Allergy Clin Immunol 125(3):584–592
Keet CA, McCormack MC, Peng RD et al (2011) Age- and atopy-dependent effects of vitamin D on wheeze and asthma. J Allergy Clin Immunol 128(2):414–16.e5
Krstic G (2011) Asthma prevalence associated with geographical latitude and regional insolation in the United States of America and Australia. PLoS One 6(4):e18492
Kunisaki KM, Niewoehner DE, Connett JE et al (2012) Vitamin D levels and risk of acute exacerbations of chronic obstructive pulmonary disease: a prospective cohort study. Am J Respir Crit Care Med 185(3):286–290
Lawson JA, Janssen I, Bruner MW et al (2011) Urban–rural differences in asthma prevalence among young people in Canada: the roles of health behaviors and obesity. Ann Allergy Asthma Immunol 107(3):220–228
Lessard A, Turcotte H, Cormier Y et al (2008) Obesity and asthma: a specific phenotype? Chest 134(2):317–323
Litonjua AA, Sparrow D, Celedon JC et al (2002) Association of body mass index with the development of methacholine airway hyperresponsiveness in men: the Normative Aging Study. Thorax 57(7):581–585
Luder E, Melnik TA, DiMaio M (1998) Association of being overweight with greater asthma symptoms in inner city black and Hispanic children. J Pediatr 132(4):699–703
Majak P, Olszowiec-Chlebna M, Smejda K et al (2011) Vitamin D supplementation in children may prevent asthma exacerbation triggered by acute respiratory infection. J Allergy Clin Immunol 127(5):1294–1296
Mora JR, Iwata M, von Andrian UH (2008) Vitamin effects on the immune system: vitamins A and D take centre stage. Nat Rev Immunol 8(9):685–698
Morrison T, Callahan D, Moorman J et al (2009) A national survey of adult asthma prevalence by urban–rural residence U.S. 2005. J Asthma 46(8):751–758
Mosen DM, Schatz M, Magid DJ et al (2008) The relationship between obesity and asthma severity and control in adults. J Allergy Clin Immunol 122(3):507–11.e6
Pesek RD, Vargas PA, Halterman JS et al (2010) A comparison of asthma prevalence and morbidity between rural and urban schoolchildren in Arkansas. Ann Allergy Asthma Immunol 104(2):125–131
Pike KC, Inskip HM, Robinson S et al (2012) Maternal late-pregnancy serum 25-hydroxyvitamin D in relation to childhood wheeze and atopic outcomes. Thorax 67(11):950–956
Rodriguez A, Vaca M, Oviedo G et al (2011) Urbanisation is associated with prevalence of childhood asthma in diverse, small rural communities in Ecuador. Thorax 66(12):1043–1050
Rothers J, Wright AL, Stern DA et al (2011) Cord blood 25-hydroxyvitamin D levels are associated with aeroallergen sensitization in children from Tucson, Arizona. J Allergy Clin Immunol 128(5):1093–9.e1-5
Sandhu MS, Casale TB (2010) The role of vitamin D in asthma. Ann Allergy Asthma Immunol 105(3):191–199
Searing DA, Zhang Y, Murphy JR et al (2010) Decreased serum vitamin D levels in children with asthma are associated with increased corticosteroid use. J Allergy Clin Immunol 125(5):995–1000
Shore SA, Schwartzman IN, Mellema MS et al (2005) Effect of leptin on allergic airway responses in mice. J Allergy Clin Immunol 115(1):103–109
Shore SA, Terry RD, Flynt L et al (2006) Adiponectin attenuates allergen-induced airway inflammation and hyperresponsiveness in mice. J Allergy Clin Immunol 118(2):389–395
Stanley AH, Demissie K, Rhoads GG (2005) Asthma development with obesity exposure: observations from the cohort of the National Health and Nutrition Evaluation Survey Epidemiologic Follow-up Study (NHEFS). J Asthma 42(2):97–99
Sutherland ER, Goleva E, Strand M et al (2008) Body mass and glucocorticoid response in asthma. Am J Respir Crit Care Med 178(7):682–687
Sutherland ER, Lehman EB, Teodorescu M et al (2009) Body mass index and phenotype in subjects with mild-to-moderate persistent asthma. J Allergy Clin Immunol 123(6):1328–34.e1
Sutherland ER, Goleva E, Jackson LP et al (2010) Vitamin D levels, lung function, and steroid response in adult asthma. Am J Respir Crit Care Med 181(7):699–704
Taher YA, van Esch BC, Hofman GA et al (2008) 1 alpha,25-dihydroxyvitamin D3 potentiates the beneficial effects of allergen immunotherapy in a mouse model of allergic asthma: role for IL-10 and TGF-beta. J Immunol 180(8):5211–5221
Tantisira KG, Litonjua AA, Weiss S et al (2003) Association of body mass with pulmonary function in the Childhood Asthma Management Program (CAMP). Thorax 58(12):1036–1041
Todd DC, Armstrong S, D’Silva L et al (2007) Effect of obesity on airway inflammation: a cross-sectional analysis of body mass index and sputum cell counts. Clin Exp Allergy 37(7):1049–1054
Turkeltaub PC, Gergen PJ (1991) Prevalence of upper and lower respiratory conditions in the US population by social and environmental factors: data from the second National Health and Nutrition Examination Survey, 1976 to 1980 (NHANES II). Ann Allergy 67(2 Pt 1):147–154
Turner KJ, Dowse GK, Stewart GA et al (1986) Studies on bronchial hyperreactivity, allergic responsiveness, and asthma in rural and urban children of the highlands of Papua New Guinea. J Allergy Clin Immunol 77(4):558–566
Valet RS, Gebretsadik T, Carroll KN et al (2011) High asthma prevalence and increased morbidity among rural children in a Medicaid cohort. Ann Allergy Asthma Immunol 106(6):467–473
Van Gent R, Van der Ent CK, Rovers MM et al (2007) Excessive body weight is associated with additional loss of quality of life in children with asthma. J Allergy Clin Immunol 119(3):591–596
Van Veen IH, Ten Brinke A, Sterk PJ et al (2008) Airway inflammation in obese and nonobese patients with difficult-to-treat asthma. Allergy 63(5):570–574
Weinmayr G, Weiland SK, Björkstén B, ISAAC Phase Two Study Group et al (2007) Atopic sensitization and the international variation of asthma symptom prevalence in children. Am J Respir Crit Care Med 176(6):565–574
Zhong NS, Chen RC, O-yang M et al (1990) Bronchial hyperresponsiveness in young students of southern China: relation to respiratory symptoms, diagnosed asthma, and risk factors. Thorax 45(11):860–865
Zosky GR, Berry LJ, Elliot JG et al (2011) Vitamin D deficiency causes deficits in lung function and alters lung structure. Am J Respir Crit Care Med 183(10):1336–1343
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer Science+Business Media New York
About this chapter
Cite this chapter
Divekar, R., Calhoun, W.J. (2014). Heterogeneity of Asthma in Society. In: Brasier, A. (eds) Heterogeneity in Asthma. Advances in Experimental Medicine and Biology, vol 795. Humana Press, Boston, MA. https://doi.org/10.1007/978-1-4614-8603-9_3
Download citation
DOI: https://doi.org/10.1007/978-1-4614-8603-9_3
Published:
Publisher Name: Humana Press, Boston, MA
Print ISBN: 978-1-4614-8602-2
Online ISBN: 978-1-4614-8603-9
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)