Abstract
This chapter explores metabolic basis of cardiac decompensation through maladaptative changes in metabolic enzyme expression that leads to dysregulation of lipid and carbohydrate metabolism. The consequences of this metabolic dysregulation are examined in detail with respect to inefficiencies in energy production in the myocardium that contribute to the energy starved heart and dysregulation of lipid metabolism that contributes to the potential for lipotoxicity. The initial section considers the link between early metabolic changes and early manifestations of impaired contractility in the decompensating, myopathic heart. Changes in the metabolic fate of the primary fuel for ATP in the heart, long chain fatty acids, occur in the decompensated and failing heart at the level of gene expression. As discussed below, long chain fatty acid oxidation and storage are both impaired in failing hearts, leading to a general dysregulation of lipid dynamics in the cardiomyocyte that contributes to the energy starved condition of the heart. The consequential metabolic adaptations in the cardiomyocyte invoke changes in the cytosolic and mitochondrial metabolism of not only lipids but also carbohydrates, as glucose is inefficiently metabolized for the production ATP. With changes in glycolytic activity and the reduction/oxidation state in the cytosolic space, transport of metabolic intermediates across the mitochondrial membrane serves to transduce the pathophysiological state of the cytosol to the mitochondrial matrix, while reciprocal exchange of intermediates from oxidative pathways links mitochondrial activity to the reduction/oxidation state of the cytosol. The latter sections of this chapter examine the details of altered mitochondrial transporter activity in response to the bioenergetic and biophysical state of the cell.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Fatty Acid Oxidation
- Diabetic Cardiomyopathy
- Hypertrophied Myocardium
- Transverse Aortic Constriction
- Hypertrophied Heart
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
This chapter explores metabolic basis of cardiac decompensation through maladaptative changes in metabolic enzyme expression that leads to dysregulation of lipid and carbohydrate metabolism. The consequences of this metabolic dysregulation are examined in detail with respect to inefficiencies in energy production in the myocardium that contribute to the energy starved heart and dysregulation of lipid metabolism that contributes to the potential for lipotoxicity. The initial section considers the link between early metabolic changes and early manifestations of impaired contractility in the decompensating, myopathic heart. Changes in the metabolic fate of the primary fuel for ATP in the heart, long chain fatty acids, occur in the decompensated and failing heart at the level of gene expression. As discussed below, long chain fatty acid oxidation and storage are both impaired in failing hearts, leading to a general dysregulation of lipid dynamics in the cardiomyocyte that contributes to the energy starved condition of the heart. The consequential metabolic adaptations in the cardiomyocyte invoke changes in the cytosolic and mitochondrial metabolism of not only lipids but also carbohydrates, as glucose is inefficiently metabolized for the production ATP. With changes in glycolytic activity and the reduction/oxidation state in the cytosolic space, transport of metabolic intermediates across the mitochondrial membrane serves to transduce the pathophysiological state of the cytosol to the mitochondrial matrix, while reciprocal exchange of intermediates from oxidative pathways links mitochondrial activity to the reduction/oxidation state of the cytosol. The latter sections of this chapter examine the details of altered mitochondrial transporter activity in response to the bioenergetic and biophysical state of the cell.
Early Signatures of Cardiomyopathy
Examinations of left ventricular (LV) wall contractility link early changes in metabolism to functional deficits in the heart that can predate both overt cardiomyopathy and the onset of complex disease [1, 2]. Indeed, proton magnetic resonance spectroscopy (MRS) detection of triglyceride content in the human myocardium demonstrates a close association of cardiac steatosis to glucose intolerance in patients, development of type II diabetes, and the ultimate development of diabetic cardiomyopathy [1]. As discussed in this section, studies in animal models lead to the recognition that indices of LV function derived from magnetic resonance imaging (MRI) tagging experiments provide sensitive indicators of early functional impairment of disease as well as early indicators of therapeutic value. Practical application of this realization has led to an increasing volume of clinical and preclinical studies to evaluate the efficacy of treatments to improve LV function in diabetic cardiomyopathy [3, 4]. Such changes in the pathophysiology of the heart are also distinguished by a signature reduction in the bioenergetic potential of the cell. Ample evidence for the notion that the failing heart is energy starved exists in the current literature, and examinations of phosphorylation potential and alterations in phosphoryl group transfer also serve as prognostic indicators of patient survival rates and help define the early signatures of cardiomyopathy. This section presents the early changes in LV wall mechanics, metabolic indices, and energetic changes that characterize the early development of heart failure.
Two-Dimensional Strains and Early Decompensation
Biophysical parameters for assessing ventricular wall mechanics with MRI of the heart have evolved to provide noninvasive indices of transmural and regional contractility through the examination of two-dimensional Lagrangian strains, strain rates, circumferential and radial strains, and ventricular torsion, via cardiac tagging methods. The process of MRI detection involves superimposing a grid pattern across the image of the heart, through selective saturation of proton nuclear spins, and tracking the deformation of the grid at systole and/or diastole (Fig. 1). This approach of cardiac tagging with MRI has contributed greatly to characterizing and understanding the functional impact and manifestation of early, subcellular alterations in the contractile apparatus from early contractile defects to decompensated cardiomyopathy to over heart failure.
Cardiac MRI tagging of in vivo mouse heart. Tagged images shown are short-axis tagged, with a 0.33 × 0.33 mm grid and a 0.1 mm grid line. (a) Tagged mouse heart image at end diastole with septal segment demarcated by white lines; (b) image at end systole; (c) zoomed image of septum at end diastole; (d) zoomed image of septum at end systole. (c, d) Display triangulated tagging elements for homogeneous strain measurement with endocardium shown in yellow tagging elements and epicardium tagging elements shown in white. Centroids of the triangulated tagging sections are marked red. From Hankiewicz et al., Circulation: Cardiovascular Imaging 2010;3:710–717
Interestingly, the two-dimensional values termed E1 and E2, associated with the positive component and negative component, respectively, of the Lagrangian strains reflect very early changes in regional ventricular function [5]. For example, in a study by Hankeiwicz et al., utilizing very high resolution tagging grids of the order of 0.3 mm, the positive strain component, E1, during systole was compromised in a murine mouse model of dilated cardiomyopathy 2 months prior to any significant wall thinning in the LV [6]. The initially surprising findings suggest that such biophysical parameters are more closely associated with fundamental changes at the level of regulation of sarcomere activity and responsiveness than are other traditional indices of ventricular performance. Thus, such approaches in examining strain in the ventricular wall appear to reflect fundamental alterations in contractility of the finite elements within the myocardium, such as the sarcomere [6, 7].
In a later study, Li et al. further demonstrated that strain measurements from cardiac-tagged MRI studies showed impaired regional function in the mdx mouse model of muscular dystrophy in the absence of any changes in temporally matched indices of global ventricular function [8]. The notion of strain measurements detecting fundamental changes in the sarcomere is further confirmed by subsequent findings of early reduction in torsion and strain in the LV of a mouse model of decreased expression of cardiac myosin binding protein C that corresponded to altered stretch-activated, cross-bridge kinetics in skinned fibers. Altered torsion and strains also occur in the left ventricles of hypothyroid rats that only express the slow β-myosin heavy chain compared to rats predominantly expressing the α-MHC isoform, as a likely consequence of slowed cross-bridge performance [9].
Contractility and Metabolic Dysregulation
The close association of MRI-based measures of strain in the LV to fundamental changes in sarcomere function, that is not as evident in gross measures of LV function, may also account for the sensitivity of MRI-detected strains to metabolic disruptions that lead to the development of cardiomyopathy. As discussed in greater detail below, the metabolic signaling processes that are invoked by impaired fat and carbohydrate oxidation and storage in the cardiomyocyte may serve as mediators of myocyte contractility and thereby changes in the generation of LV strains.
While the overstorage of cardiac triglyceride is associated with the eventual development of type II diabetes and the occurrence of diabetic cardiomyopathy [1], whether a more direct relationship between myocardial triglyceride and contractile dysfunction exists is addressed by a study of a cardiac-specific mouse model of low, overexpression of the nuclear hormone receptor, peroxisome proliferator-activated receptor-α (PPARα, strain 404-4) [2]. The intramyocellular content of triglyceride has been determined in vivo by proton MRS in hearts of the low overexpressing PPARα mice and non-transgenic littermates over the course of a short-term high fat diet, composed of 60 % calories from fat (Fig. 2).
Localized 1H MRS (septum) of endogenous cardiac lipid content (1 × 1 × 1 mm volume of interest). (a) Axial scout image of in vivo mouse heart with indicated localized volume (white square). (b) Water suppressed 1H spectrum showing triglyceride signals in pre- and post-high fat diet hearts of non-transgenic littermate mice and MHC-PPARα transgenic mice. From Hankiewicz et al., Circulation: Cardiovascular Imaging 2010;3:710–717
Using a very high resolution tagging method that enabled resolution of the tagging grid in endocardial and epicardial layers of the LV, increased myocardial lipid after only 2 weeks of a high fat diet was associated with concurrent reductions in E1 and E2 strains in the PPARα overexpressing mice, with the most pronounced reduction occurring in the endocardium (Fig. 3). Myocardial triglyceride was consistently lower in the non-transgenic littermates than in the MHC-PPARα mice and the strains were only attenuated at the highest levels of lipid accumulation, suggesting a threshold response. The importance of elucidating these relationships is that 2D strains are impaired early and without left ventricular diastolic dysfunction, owing to cardiac steatosis, and that only in hearts predisposed to cardiac steatosis did a short-term high fat diet affect contractility. This study was the first evidence that cardiac steatosis is not only prognostic but also directly involved in the development of cardiac dysfunction. The evidence for the role of lipid metabolism in the pathogenesis of cardiomyopathy presented above is supported by a wealth of recent data in the literature to indicate the direct role that disruption of metabolic balance, including energy yielding pathways that influence bioenergetic state, plays in the pathogenesis of the failing heart.
Effect of high fat diet on values for myocardial 2D principal strains, E1 and E2. (a) E1 and E2 in epicardium; (b) E1 and E2 in endocardium. Note reduced endocardial strains in MHC-PPARα hearts after 2-week high fat diet. * P < 0.05, post-diet MHC-PPARα vs. post-diet NTG; # P < 0.05, pre-diet MHC-PPARα vs. post-diet MHC-PPARα. From Hankiewicz et al., Circulation: Cardiovascular Imaging 2010;3:710–717
Bioenergetic Markers of Cardiomyopathy
The pathogenesis of cardiomyopathy has long been associated with reduced cellular energy potential of the cardiomyocytes. The myocardium that progresses to decompensation and overt failure is characterized by impaired availability and production of chemical energy in the form of ATP to support cell maintenance and contractile function [10–13]. In studies in animal models, 31P NMR measurements have clearly demonstrated a general reduction in free energy charge in the hypertrophied or dilated myocardium, as evidenced by reduced ratios of phosphocreatine (PCr) to ATP that trend below 1.8 PCr:ATP [14–20]. Similar observations have been made in human subjects across a range of cardiomyopathies, including both hypertrophic and dilated hearts [21–26]. This PCr/ATP ratio serves as an index of the phosphorylation potential within the cell:
where ΔG p is the phosphorylation potential and P i represents inorganic phosphate. The equation is determined from the free energy of ATP hydrolysis in the cell which is given by:
where ΔG ATP is ATP hydrolysis under standard conditions at −30.5 kJ/mol, R is the gas constant (9.314 J/mol K), and T is temperature in Kelvin, and which can be assessed by the relationship of the high energy phosphates to the other reactants and products across the creatine kinase (CK) equilibrium reaction:
where K eq is a known constant of 1.66 × 109 at pH 7.1 and 37 °C.
In accordance with the CK equilibrium kinase reaction, 31P NMR measurements of whole tissues, as in the heart, have made major contributions to the ability to assess phosphorylation potential because NMR spectroscopy provides measures of ATP, PCr, and intracellular pH (from the pH sensitive chemical shift of inorganic phosphate (P i)). Importantly, destructive tissue assays for ADP content include both the bound and unbound fractions, when the unbound ADP participates in the CK reaction. Thus, when a known assay result or literature value for creatine content is applied, NMR measurements of ATP, PCr, and pH enable the determination of the actual free ADP content of the cell from the CK equilibrium reaction shown above [27]. Consequently, determined changes in ADP/ATP of an intact tissue reflect a change in the free energy of ATP hydrolysis at steady state conditions.
This PCr/ATP ratio, as an index of the phosphorylation potential, drops in the decompensated, cardiomyopathic heart and has been suggested as a prognostic indicator of mortality in patients with dilated cardiomyopathy [28]. Limitations occur in bioenergetic state and phosphoryl group transfer among high energy phosphates, and both are implicated in the energetic inefficiency of the hypertrophied myocardium [12, 29] (Fig. 4).
Phosphocreatine (PCr) and ATP contents are reduced in failing hearts as shown by 31P NMR spectroscopy of intact, beating hearts. 31P spectra enable detection of extra- and intracellular contents of inorganic phosphate (Pi), PCr, and the three phosphate groups of ATP, γ, α, and β. Due to chemical shift effects induced by local electromagnetic environments, the β phosphate group is the only phosphate group that is purely from ATP, while the other two have signal contributions from other phosphates. Top panel, 31P spectrum from normal rat heart. Bottom panel, 31P spectrum from failing rat heart. Note reduced PCr and elevated intracellular Pi content relative to ATP, represented by the β phosphate signal that indicates low energy potential in the failing heart
One limiting factor in the energetic state of the failing heart is the consequence of reduced cellular content of both ATP and PCr along with reductions in myocardial creatine content due to reduced creatine transporter proteins on the sarcolemma [30]. However, restoration of creatine levels by increasing expression of the creatine transporter in mouse hearts proved ineffective in supporting normal PCr levels [31]. Instead, the induced increases in intracellular creatine produce an abnormally low fraction of phosphorylated creatine that drives down the chemical driving force for the ATPase reactions, with a deleterious effect on contractile function.
More recently the unidirectional rate of ATP synthesis across the creatine kinase reaction has been found to decline in left ventricular hypertrophy (LVH) due to pressure overload, with further, distinguishing reductions in this rate in congestive heart failure (CHF) [12, 29]. These bioenergetic distinctions of LVH and CHF are not only early signatures of the presence of disease but also contribute to the underlying metabolic basis of cardiomyopathy via the consequence of the failing heart being an energy starved heart. Indeed, a mouse model of myofibrillar CK overexpression showed improved ATP flux through CK and contractile function during pressure overload induced by transverse aortic constriction (TAC) [32].
Based on the observed deficiencies in the energetic state of the decompensated myocardium, clinical efforts have focused on restoring both PCr and ATP contents in failing human hearts. For example, blocking the progressive catabolism of the purine moiety from ATP breakdown, through chronic allopurinol administration to inhibit the breakdown of hypoxanthine, has met with some success in maintaining cardiac PCr/ATP ratios, PCr concentration, and CK flux in the failing human heart [33].
Metabolic Dichotomy of Heart Failure
Early changes in metabolic activity, as consequence of altered metabolic enzyme expression, are now well recognized to contribute to the pathogenesis of heart failure [20, 34–40]. As discussed above, the myopathic heart is often characterized as “energy starved”; by virtue of a reduced bioenergetic potential and altered free energy of ATP hydrolysis. An increasing awareness and focus on the defects in metabolic pathways that generate the reducing equivalents, that serve as the currency for oxidative ATP synthesis, has yielded important new findings in the metabolic basis of heart failure. However, the metabolic distinctions associated with the pathogenesis of different cardiomyopathies are not always clearly understood, and often confused. For example, the metabolic changes that contribute to the development of cardiomyopathy in diabetes or metabolic syndrome are different, and at times converse to changes associated with chronic pressure overload. This section examines the underlying changes and restrictions in intermediary metabolism that are associated with the spectrum of cardiomyopathies leading to heart failure.
Distinctions in Fuel Supplies
As discussed in greater detail in later section, the blood-borne, carbon-based fuels that support energy synthesis in the cardiomyocyte include primarily long chain fatty acids, to a somewhat lesser extent carbohydrates, and under variable circumstances ketones. Of these, long chain fatty acids are the most energy rich, yielding more ATP per mole than can be produced from the complete metabolism of either glucose or ketones. Indeed, the normal heart has been characterized as an “omnivore,” able to utilize a variety of substrates and adjust to transient changes in either the availability of substrates for energy production or demand, as during workload jumps. Nonetheless, the preferential and predominant fuel for supporting ATP synthesis in the myocardium is the energy rich long chain fatty acid supply.
A well-recognized metabolic response to chronic pressure overload in the heart is the reduced oxidation of long chain fatty acids. In the pressure overloaded heart, glycolytic flux increases but does not lead to a commensurate increase in flux through pyruvate dehydrogenase (PDH) on the mitochondrial membrane for concerted oxidative production of mitochondrial NADH from glycolytic end products. Thus, a clear reduction in the potential energy provided by the carbon-based substrates that fuel the oxidative intermediary metabolism of the mitochondria occurs. The principles behind the shift away from fat oxidation and the inefficiencies in carbohydrate metabolism that occur in cardiac hypertrophy are discussed in detail below, but from the discussion presented above, the state of low energy charge in the failing heart is consistent with this model of low fat oxidation. However, in addressing the dichotomous nature of metabolism of the failing heart, this section contrasts the low fat metabolism in the pressure overloaded heart to the high fat oxidation state of the diabetic heart, which also develops cardiomyopathy progressing to overt failure.
Consequently, the notion of an energy starved heart may seem paradoxical to the diabetic heart, in which long chain fatty acid metabolism is disproportionately elevated in the presence of limited glucose uptake. Yet, the myocardial PCr/ATP ratio in diabetic cardiomyopathy is also reduced, as in other forms of cardiomyopathy, while improving glucose uptake into the heart with rosiglitazone does elevate PCr/ATP in hearts of patients with type 2 diabetes [41]. Thus, the diabetic heart develops cardiomyopathy and energy deficits amidst an abundance of high energy fuel availability.
Restricted Metabolic Flexibility
Clearly, different pathophysiological states, all leading to heart failure, can present opposing metabolic deficits that challenge a simplistic understanding of fueling cardiac energetics and infer distinctions in metabolic signaling. Caution is indicated in generalizing the metabolic shifts of the failing heart, and investigators must recognize that the pathophysiological condition introduces different metabolic variables. This dichotomy leads to the notion of restricted metabolic plasticity as a deleterious consequence in the myopathic heart. The common metabolic feature in cardiomyopathies is the lack of flexibility in the ability of the cardiomyocyte to adjust substrate utilization for energy production to either changes in substrate supply in the blood or energy demand in response to chronic changes in workload [42, 43].
An example of an inability to adjust to transient changes in work and metabolic demand is the simple model of workload jumps by the heart. During increased work, as can be induced by adrenergic challenge to the beating heart, the additional energy requirement has been found to be supplied by the recruitment of additional carbohydrate metabolism, on top of a high baseline level of long chain fatty acid oxidation [44, 45]. The inability to accommodate this recruitment due to metabolic restrictions, through either impaired glucose uptake in diabetic hearts or reduced oxidation through PDH in hypertrophied hearts, eventually leads to a chronic state of energetic deficiencies in the cardiomyocyte, which are now believed to eventually contribute to the development of impaired energy balance and cardiomyopathy [40, 42, 43]. More recently the role of leptin in regulating myocardial metabolism has been recognized and impaired leptin levels or deficient leptin signaling results in restricted glucose metabolism leading to impaired functional responses to stress [46].
Inefficiencies in Fuel Oxidation and Energy Production in the Hypertrophied and Failing Heart
The hypertrophied myocardium that progresses to decompensation and overt failure is characterized by impaired availability and production of chemical energy in the form of ATP to support cell maintenance and contractile function [10–13]. Changes in the energy yielding intermediary pathways are implicated in the hypertrophic gene expression program, and can be related to specific inefficiencies in ATP synthesis [14, 20, 38, 40, 47–50]. But a generalized description of this metabolic remodeling as a simple reduction in fatty acid oxidation with increased carbohydrate use neglects the balance of carbon flux in and out of the tricarboxylic acid (TCA) cycle and changes in the overall dynamics of lipid utilization/storage that produce physiological effects [20, 40, 51, 52].
With direct relevance to the efficiency of ATP production by the heart, the reduced contribution of fatty acid oxidation represents a net loss of ATP production per mole of substrate in comparison to that of carbohydrate. Although the complete oxidation of glucose requires less oxygen consumption than does that of long chain fatty acids, such as palmitate or oleate, these long chain fats yield more ATP per mole of substrate oxidized than glucose. Thus, in the absence of impaired tissue oxygen tension (PO2), such as in the flow-limited myocardium, oxygen use is not a critical factor in the absence of limited tissue oxygenation, and the most efficient fuels for ATP production by the mitochondria are the long chain fatty acids. Indeed, while energy balance is impaired in CHF, oxygen availability and tissue PO2 have been demonstrated to be adequate in remodeled myocardium [53]. The generic extrapolation of the reduced use of oxygen by glucose oxidation as a benefit to all forms of heart disease is not always well considered. Therefore, the efficiency of metabolic pathways that support ATP synthesis and adaptive shifts in the metabolic balance of the hypertrophied myocardium can be argued as more critical concerns than oxygen sparing.
When considering the metabolic efficiency of the heart, in the absence of limited oxygen availability, the more concerning parameter becomes the efficiency of ATP production per unit of carbon-based fuel oxidized. In the of the decompensated, pressure-overloaded myocardium that appears “energy starved,” the shift away from fuels, such as long chain fatty acids, that can yield the greatest amount of ATP per mole, may be maladaptive. Several key inefficiencies in the metabolism of carbon-based fuels for ATP synthesis during the previous funding period are: (1) β-oxidation: reduced fatty acid oxidation rate and ATP synthesis from energy rich fuel; (2) lipid storage dynamics: elimination of the endogenous triacylglyceride (TAG) pool as an oxidative fuel source in hypertrophied hearts and reduced incorporation of palmitate into the TAG pool with potential implications for lipotoxicity; (3) carbohydrate metabolism: increased glucose uptake and glycolysis, but a shift in carbohydrate oxidation away from entry into the TCA cycle via PDH and toward anaplerosis via malic enzyme (ME), bypassing NADH producing steps for ATP synthesis. These three inefficiencies are examined in the subsequent sections.
Reduced β-Oxidation in Hypertrophied and Failing Hearts
As introduced above, a classic metabolic distinction of the hypertrophied myocardium is a reduction in long chain fatty acid oxidation in the support of mitochondrial oxidative ATP production. Changes in the expression of enzymes associated with long chain fatty acid oxidation mediate this shift. The mitochondrial membrane is impermeable to long chain free fatty acids (LCFA) which are oxidized via β-oxidation and the TCA cycle in the inner matrix (Fig. 5) and long chain fatty acids enter the mitochondrial matrix via a shuttle system. Carnitine palmitoyltransferase 1 (CPT1) serves as a translocase for fatty acyl esters, by spanning the outer mitochondrial membrane and catalyzing acyl group transfer from Coenzyme A to carnitine in the intermembrane space. CPT2 converts fatty acyl-carnitine ester back into fatty acyl CoA in the matrix. Generally considered the rate-determining process in fatty acid oxidation by heart, CPT1 is regulated by malonyl CoA levels. Malonyl CoA is produced from acetyl CoA, via the action of acetyl CoA carboxylase 1 (ACC-1) at the mitochondrial membrane, which in turn is formed via β-oxidation and other sources of oxidative fuels within the matrix [54]. An ACC-2 isoform also exists in the cytosolic fraction, though in much lower abundance in cardiomyocytes. ACC-2 is associated with supplying malonyl CoA for fatty acid synthesis and chain elongation. The level of malonyl is also determined by its catabolism by malonyl CoA decarboxylase (MCD) [55]. Thus, the extent of long chain fatty acid oxidation by mitochondria is regulated by the activity of CPT1, which in the normal adult heart is primarily the muscle isoform.
Regulation of the rate-limiting activity of CPT1 on long chain fatty acid entry into mitochondria for β-oxidation. CPT1 and ACC-2 are integrated into the outer mitochondrial membrane. The muscle isoform of CPT1 (M or β) is inhibited by malonyl CoA levels produced by the carboxylation of acetyl CoA by ACC-2. Questions remain as to whether other factors regulate the activity of the liver CPT1 isoform (L or α) that is expressed in greater abundance in fetal and hypertrophied cardiomyocytes
In general, the fetal heart, and thus neonatal cardiomyocytes, does not utilize fatty acids as a major fuel for ATP production, as the fetus is provided a constant supply of glucose. Thus, enzymes of fatty acid oxidation, including CPT1, are expressed at very low levels until the immediate postnatal period, when they are rapidly induced [56, 57]. There are two structural genes that encode CPT1, the α or L (liver) gene and the β or M (muscle) gene [57–60]. These genes are differentially expressed among tissues that utilize fatty acids as fuel, and they are co-expressed in heart. The major cognate CPT1 enzymes encoded by these genes have different kinetic properties, such that differing relative expression levels among tissues are reflected in different kinetics [56, 60, 61]. Specifically, L-CPT1 is less sensitive to inhibition by malonyl CoA, and has a lower Km for carnitine. In neonates, the L form has been suggested to be 25 % of the total CPT1 activity in heart, while activity from the M form constitutes ~75 % of the activity [57, 62]. During the progression to maturity, this ratio changes such that only ~2 % of enzyme activity is from the L isoform of CPT1.
A teleological argument has been offered that the relatively high proportion of the L-type enzyme in early life permits activity at the low carnitine levels characteristic of neonatal myocardium and may be an adaptive response to the low fatty acid oxidation rates of the hypertrophied heart [36, 56, 57]. However, such suggestions are based on cell culture studies of unloaded cells, and usually neonatal cardiomyocytes that are predisposed to carbohydrate metabolism and not fat oxidation. However, electrical stimulation of neonatal cardiomyocytes does result in a redistribution of CPT1 isoforms [36]. Later investigation using Northern blot analysis showed no significant differences in transcript level of L-CPT1 between normal control human myocardium and in ten failing human heart samples ranging from idiopathic, peripartum, and ischemic cardiomyopathies [63]. However, with the development of specific antibodies, coupled to flux measurements, the first actual demonstration of elevated L-CPT1 enzyme protein content in hypertrophied myocardium of the pressure overloaded rat heart, with online measures of reduced flux through CPT1 in the mitochondria of the same beating hearts, was reported in 2007 [20].
Recently, a study in the ACC-1 deficient mouse heart indicated that a resulting elevation in fatty acid oxidation coincided with an attenuated hypertrophic response to left ventricular pressure overload [64]. However, baseline levels of malonyl CoA were only reduced by about 50 % despite the absence of ACC-2 and, in that particular study, baseline contributions of long chain fatty acids in the control animals were already a good deal lower than reported previously, and the ACC-2 KO hearts displayed only moderate levels of fatty acid oxidation in comparison to other reports for the contribution of long chain fatty acids to oxidative ATP production [20, 37, 40, 44, 47]. Other studies of the intact and in vivo heart indicate no direct relationship between malonyl CoA content and LCFA oxidation in hearts [65–67]. Studies demonstrating the absence of a link between malonyl CoA content and LCFA oxidation were performed on hearts with a predominantly normal distribution of M-CPT1 and L-CPT1 contents, and these findings suggest that L-CPT1 may be subject to additional levels of regulation that have yet to be fully identified. Indeed, Kim et al. report on a malonyl CoA-resistant level of palmitate oxidation in red versus white skeletal muscle preparations [68]. Therefore, posttranslational modifications and as yet undetected levels of regulation beyond malonyl CoA must be considered in the intact functioning myocardium that may limit LCFA oxidation through L-CPT1.
Reductions in fatty acid oxidizing enzymes have been reported in hypertrophied myocardium and are associated with reduced activation of PPARα [35, 38]. However, a shift in CPT1 isoform distribution toward increased content of the liver (L-CPT1) isoform occurs concurrent with reduced flux of free fatty acid into oxidative metabolism [20]. The link between increased L-CPT1 and reduced palmitate oxidation in cardiac hypertrophy is consistent with the reduced fatty acid oxidation rates under conditions of limited carnitine availability in fetal and neonatal hearts and thus the reversion to a general programmatic shift toward fetal isoform expression [20, 36, 38, 50, 57, 60, 63]. However, from recent understanding of inducible gene expression and the realization that the metabolic changes in response to altered workload occur relatively early, preceding anatomical changes in the left ventricle, an early programmatic change in gene expression that induces the L isoform of CPT1 appears to be a more likely scenario.
Interestingly, in a rat model of acute L-CPT1 overexpression, induced by adenoviral-mediated delivery of an exogenous gene, a fourfold increase in L-CPT1 expression over control hearts was associated with a significant drop in fatty acid oxidation [69]. Despite the consideration that L-CPT1 is less sensitive to inhibition by malonyl CoA, the expression of L-CPT1 in the fetal heart, the hypertrophied heart, and hearts overexpressing L-CPT1 is consistently associated with lower oxidation of long chain fatty acids. The findings in the otherwise normal, L-CPT1 expressing rat heart strongly suggest that other factors in the intact myocardium regulate fatty acid oxidation in addition to, or instead of malonyl CoA. In fact, CPT1 activity, in tissues in which the L-CPT1 isoform predominates, is not directly proportional to protein content, and thus both hypertrophied hearts and L-CPT1 overexpressing hearts may also lose this proportionality [20, 68–70].
Reduced Triacylglyceride Content and Metabolism in Cardiac Hypertrophy
The content and turnover of the endogenous form of stored lipid in the cardiomyocyte, TAG, are reduced in hypertrophied hearts, as reported in animal model of pressure overload following TAC [47]. Interestingly, in a rat model of cardiac hypertrophy, TAG turnover is reduced by 40 %, a finding not obvious from TAG content alone [47]. While myocardial TAG turnover is a difficult parameter to monitor in humans, reduction in TAG content in the myocardium of heart failure patients has been shown to match these findings of prior animal studies [71], and has been related to potential deleterious elevations of physiologically active and potential lipotoxic acyl derivatives, such as ceramides and sphingosines [72–74]. However, the presence or absence of altered TAG content in heart failure patients does not indicate the extent of potential changes in TAG dynamics that are evident with 13C NMR of experimental models [47, 75, 76].
Along with this finding of reduced TAG turnover, the contribution of TAG to β-oxidation, by supplying long chain fatty acids produced during TAG lipolysis, is also severely reduced in the hypertrophied heart [47]. Thus, a portion of the reduced fatty acid oxidation rates in hypertrophied myocardium is accountable from a severe reduction in the oxidation of endogenous TAG, which cannot be recruited by adrenergic challenge [47]. As an energy rich source, TAG becomes essentially unavailable to support oxidative ATP production in the mitochondria of the hypertrophied and failing heart. However, potential factors that could affect TAG pool size and synthesis, such as long chain fatty acid uptake into the cell and the different affinities of different fatty acid chain lengths for TAG formation, are not yet completely understood [51]. The potential also exists for any limitations in long chain fatty acid incorporation into the TAG pool to contribute to lipotoxicity, with implications for the pathogenesis of cardiomyopathy [47, 51, 71].
While activation of PPARα has long been associated with target gene expression for the enzymes catalyzing long chain fatty acid oxidation, PPARα is now also known to regulate expression of TAG synthases and lipase that contribute to the rates of TAG turnover in the cardiomyocyte [75]. The reduced activation level of PPAR in the hypertrophied heart is therefore implicated in this downregulation of TAG dynamics and subsequent contributions to β-oxidation.
In contrast to pressure overload hypertrophy, diabetic cardiomyopathy occurs under conditions of high mitochondrial oxidation of LCFA [2, 24–27]. This metabolic phenotype is implicated in the pathogenesis of diabetic cardiomyopathy, but also involves increased TAG storage and accelerated TAG turnover rates [77–81]. Thus, the imbalances in lipid utilization and storage persist in the diabetic heart, promoting formation of lipotoxic intermediates. Such potential contributions of altered lipid dynamics to lipotoxicity in the heart are considered further in a subsequent section.
Considerations of Lipotoxicity as Consequence of Altered Lipid Dynamics in the Heart
Palmitate is a common dietary LCFA that is readily oxidized by the heart, but in minimized cell culture models has been related to cardiotoxic effects and the induction of apoptosis [51, 82–84]. Studies supporting this notion of a direct, palmitate-induced apoptosis are limited to the extreme experimental conditions of unloaded, essentially metabolically inactive neonatal cardiomyocytes that are already predisposed to limited ability to oxidize long chain fatty acids, following incubation with high palmitate concentrations for time periods as extreme as 20–48 h [82, 83]. However, the activation of palmitate to palmitoyl CoA does provide substrate for ceramide production via the activity of the serine palmytoyltransferase. While induction of apoptosis directly by palmitate has never been demonstrated in the intact or in vivo heart, a diet high in saturated fats has been found to induce coincident elevations in ceramides and apoptotic cardiomyocytes in rat hearts, along with changes in PPAR-regulated gene expression [85]. But these static changes in ceramide content and apoptotic cell count occurred independent of function, and the complex relationship between fatty acid metabolism and cardiac performance in vivo remains poorly resolved. Non-ceramide-dependent apoptosis has also been reported in neonatal cardiomyocytes through the formation of reactive oxygen species [86]. On the other hand, the incidence of cardiomyopathy subsequent to the dysregulation of lipid dynamics, and not simple static metrics, in the heart remains as an undeniable link. Therefore, any dysregulation of the balance between LCFA oxidation in the mitochondria and storage into the TAG pool can then result in the accumulation of palmitate and palmitoyl-esters with potential deleterious, or at least physiological effects on the cardiomyocyte, as discussed below.
Consequently, TAG production from activated LCFA has been considered a protective mechanism to reduce the intracellular content of lipotoxic acyl-intermediates, like activated palmitate and their derivatives. However, disruption in the dynamics between lipogenesis and lipolysis of the TAG pool, relative to LCFA oxidation rates, can result in changes in TAG content. Static measures of TAG, particularly changes in TAG content, may then serve as an indicator of intracellular conditions that are conducive to the production of lipotoxic intermediates. Metabolites of these acyl-intermediates that are known to exert lipotoxic affects on the cardiomyocyte include diacylglyceride, which has been associated with cardiomyopathies, ceramide and its metabolite via the enzyme ceramidase, sphingosine [72, 87–90] (Fig. 6). These compounds influence myofilament phosphorylation with resulting increases in myofilament sensitivity to Ca2+ [91]. Ceramide is the product of serine palmitoyltransferase activity on palmitoyl CoA. The product of ceramidase, sphingosine, activates p21-activated kinase (PAK1) [92, 93] and recent studies have shown that PAK1 affects myofilament sensitivity to Ca2+, possibly through PAK1 pathway-activated dephosphorylation of cTnI and cTnT [94, 95].
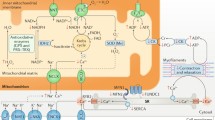
Metabolic fates of long chain fatty acids in the cardiomyocyte. Asterisk indicates active intermediates that induce metabolic signaling pathways with potential downstream effects on nuclear receptor activation, lipotoxicity, apoptosis, Ca++ sensitivity at the sarcomere, PKC signaling, and insulin sensitivity. LCFA long chain fatty acids, PPAR peroxisome proliferator-activated receptor, DAG diacylglyceride, TAG triacylglyceride, S-1-P sphingosine-1-phosphate, ACS acyl CoA synthase, DAGT1 diacylglyceride transferase-1, SPT serine palmitoyltransferase, CPT1 carnitine palmitoyltransferase 1, ATGL adipose triglyceride lipase, SphK sphingosine kinase
Inefficient Carbohydrate Metabolism in Heart Failure
While glycolytic activity is increased in the hypertrophied heart, oxidation of glycolytic end products through PDH does not keep pace with the increased glycolytic activity [34, 96–98]. However, some of this mismatch between glycolytic flux and pyruvate oxidation via PDH in hypertrophied myocardium is accommodated by the entry of pyruvate into the second span of the TCA cycle, as malate from the carboxylation of pyruvate through the cytosolic, NADP+-dependent malic enzyme-1 (ME-1) (Fig. 7) [20, 40]. Stable isotope studies have demonstrated entry of isotopically enriched pyruvate from [1,6-13C2] glucose into oxidative metabolism in the absence of direct enrichment of acetyl CoA via PDH with corresponding increases in myocardial malate content [20, 40]. Yet radiotracer approaches that would rely on the trapping of 14CO2 gas, released from oxidation of 14C-1 labeled glucose, as an indicator of 14C labeled pyruvate, are insensitive to both 14CO2 fixation and any measure of anaplerosis. Indeed, an increase in CO2 fixation by such carboxylation reactions could be mistakenly attributed to further reductions in pyruvate oxidation. Thus, the fate of glycolytic end products is associated with limited oxidation of pyruvate through PDH, with increased carboxylation of pyruvate due to an increase in ME-1 expression and protein content.
Carbon unit entry into the TCA cycle from carbohydrates and fatty acids is altered in hypertrophied and failing hearts. Flux through CPT1 is reduced (red arrow), resulting in lower fatty acid oxidation rates and less efficient ATP production in hypertrophied hearts. Oxidation and decarboxylation of pyruvate from glycolysis through pyruvate dehydrogenase (PDH) are slowed and do not keep pace with increased glycolytic activity in hypertrophied myocardium. Baseline production of oxaloacetate is generated through the transamination reaction involving aspartate. Carboxylation of pyruvate via malic enzyme-1 (ME-1) is increased, with the production of malate bypassing three of the four reactions of the TCA cycle that produce reducing equivalents to form NADH and the production of FADH2. The carboxylation of pyruvate by ME-1 also consumes NADPH. Conversion of pyruvate to oxaloacetate via pyruvate carboxylase (PC) is relatively inactive in heart muscle and is not increased in hypertrophy. Increased expression and activity of ME-1 in hypertrophied hearts catalyzes inefficient utilization of carbohydrate for ATP production and consumes NADPH required for glutathione reduction and maintenance of redox balance in the cell
The energetic consequence of pyruvate carboxylation and production of malate, which then enters the second span of the TCA cycle, as opposed to the decarboxylation of pyruvate via PDH is the consumption of pyruvate in the absence of NADH generation. Oxidation of pyruvate through PDH in the normal heart produces the two-carbon acetyl CoA that enters the TCA cycle via citrate synthase. The process of converting pyruvate into acetyl CoA directs this carbon mass from pyruvate metabolism through downstream reactions in the TCA cycle that produce NADH, thereby providing reducing equivalents for electron transport and coupling to oxidative phosphorylation for mitochondrial ATP production. However, in hypertrophied myocardium, the conversion of pyruvate to malate via ME-1 transfers these carbon units to the second span of the TCA cycle, bypassing the NADH and FADH2 production that would otherwise be supported by pyruvate decarboxylation via PDH to produce acetyl CoA and the downstream dehydrogenase reactions of the TCA cycle (Fig. 7). The energetic consequence of this increase in ME-1 catalyzed pyruvate carboxylation is the inefficient utilization and entry of glucose metabolites into the oxidative metabolism of the mitochondria and loss of potential NADH production [40, 48]. Reducing flux through ME-1 and restoring pyruvate decarboxylation through PDH result in improved contractility in the hypertrophied heart, perhaps as a consequence of countering the maladaptive influence of ME-1 expression and activity on ATP synthesis and redox state [40, 48].
The anabolic formation of four-carbon intermediates of the second span of the TCA cycle is referred to as anaplerosis [99]. Normal anaplerosis provides necessary carbon mass within the second span of the TCA cycle [100, 101], and becomes critical for providing substrate for the condensation reaction of citrate synthase in the setting of high acetyl CoA levels that can potentially limit CoA availability for the rate-limiting reaction catalyzed by α-ketoglutarate (αKG) dehydrogenase [102–104]. Thus, under conditions such as ketosis, anaplerosis is critical to maintaining TCA cycle flux [102, 105, 106]. Indeed, elevated pyruvate carboxylation has been reported to be beneficial to the heart during cardio-pulmonary bypass and reperfusion [107].
In hypertrophied hearts displaying reduced in fatty acid oxidation and lack of coupling between glycolytic flux and PDH activity, increased anaplerosis then becomes a compensatory mechanism for both pyruvate oxidation and maintaining carbon mass within the TCA cycle [20, 40]. However, the compensatory recruitment of anaplerosis may come at the cost of other less beneficial adjustments in metabolism. In fact, reducing pyruvate carboxylation via ME for anaplerosis, via increased pyruvate oxidation through PDH, can restore TAG levels and improved contractility [40].
While relatively inactive in the normal heart, in hypertrophied hearts elevated ME-1 activity converts pyruvate to malate, in the process consuming NADPH:
The consumption of increased ME flux may limit other NADPH-dependent reactions [48]. For example, regulation of redox state is affected by the ratio of oxidized to reduced glutathione (GSSG/GSH) [108–110]. NADPH maintains glutathione in the reduced state. Thus, redox balance may be adversely affected by upregulated ME expression in hearts, which can produce contractile dysfunction due to oxidant stress [108]. While sources of mitochondrial and cytosolic NADPH remain somewhat controversial, work by Jain et al. would indicate that anaplerotic activity through the cytosolic NADPH-dependent malic enzyme is maladaptive in limiting cytosolic NADPH for the reduction of glutathione [109, 110] (Fig. 7). Consistent with the notion that ME-1 upregulation in cardiac hypertrophy is a metabolic maladaptation, pharmacologic activation of PDH in the hypertrophied heart, to compete against ME-1-catalyzed pyruvate carboxylation, improved contractility, as determined by the rate of pressure development in the left ventricle, dP/dt [40].
In other tissues, ME-1 is characterized as a lipogenic enzyme, via NADPH production through the reverse reaction converting malate to pyruvate. ME expression in adipocytes is induced by high fat diet [111]. Since NADPH is also required for the reduction of dihydroxyacetone phosphate in the cytosol for TAG synthesis, NADPH consumption by the activity of ME-1 may also contribute to the observed reduction in endogenous triglyceride stores in hypertrophied myocardium [40, 47]. Interestingly, though the exact mechanism has yet to be fully elucidated, the same study of pharmacologic activation of PDH to compete with ME-1 activity for pyruvate also normalized TAG levels in hypertrophied hearts [40].
Role of the Mitochondrial Membrane in Metabolic Communication and Transduction of Pathophysiological State
Shuttling of Reducing Equivalents for Mitochondrial Oxidative Energy Production
The principal outcome of the intermediary metabolic pathways in fueling oxidative ATP production is the generation of reducing equivalents, which are carried by the positively charged, acceptor molecule, nicotinamide adenine dinucleotide (NAD+). In the mitochondrial matrix, carbon-based fuels are progressively oxidized, being stripped of electrons, and the resulting reduction of the acceptor, NAD+, forms NADH and a proton (+H). Other acceptor molecules, such as flavin adenine dinucleotide (FAD) can be linked to oxidative reactions, as well. These reducing equivalents are then carried to the electron transport chain to supply the proton-motive force that then drives the F1-ATPase in the direction of ATP synthesis.
However, nonoxidative, glycolytic metabolism also produces NADH in the cytosolic compartment. Although glycolysis produces a relatively small fraction of ATP in the myocyte compared to the mitochondria, the production of NADH can have profound influence on not only the balance between the metabolic state of the mitochondrial and cytosolic compartments, but the pathophysiological state of the myocyte is also affected. As the rate-limiting glycolytic enzymes require NAD+ as a cofactor, oxidation of NADH must occur to maintain glycolytic flux. This regeneration of cytosolic NAD+ can occur either nonoxidatively, through the production of lactate from pyruvate via the lactate dehydrogenase reaction, or oxidatively, by shuttling the reducing equivalents from cytosolic NADH to the mitochondrial matrix via the malate–aspartate shuttle (see Fig. 8).
Transfer of reducing equivalents from the cytosol to mitochondria via intermediate exchange across the malate–aspartate shuttle. High cytosolic NADH drives coordinated flux through the two transporters in the net forward direction. I, denotes the reversible α-ketoglutarate (2-oxoglutarate)–malate carrier (OMC). II, denotes the unidirectional aspartate–glutamate carrier
Of the shuttle mechanisms for cytosolic NADH, the malate–aspartate shuttle predominates in the heart with minimal activity from the glycerophosphate shuttle [112–115]. After birth, the myocardium becomes less reliant on glycolytic metabolism and expression of the proteins for these shuttles is reduced [114–116]. The function of the 2-oxoglutarate (α-ketoglutarate)–malate carrier protein (OMC) on the mitochondrial membrane is important to the maintenance of cytosolic NADH handling and holds particular importance under the conditions of increased glucose metabolism in hypertrophied myocardium [20, 50, 115].
While it is generally acknowledged that cytosolic NADH content of the myocyte is difficult to accurately measure due to the high NADH content of the mitochondria, Scholz et al. demonstrated that the distinctions in cytosolic redox state can be assessed using glycerol-3-phosphate levels relative to dihydroxyacetone phosphate as an indicator of the cytosolic NADH/NAD+ [117]. In this manner, Scholz et al. [117] were able to confirm the distinctly different cytosolic redox state condition that is to be expected between isolated hearts supplied pyruvate versus hearts supplied lactate. Briefly, the increase in cytosolic lactate concentration establishes a metabolic equilibrium with the reverse flux through lactate dehydrogenase producing a higher baseline level of NADH/NAD+ in the cytosol. This lactate-induced shift in cytosolic redox state can stimulate malate–aspartate shuttle activity, due to elevated NADH/NAD+ in the cytosol [118].
Intermediate Exchange Across the Mitochondrial Membrane and Subcellular Communication
Another important consideration is the competition between the exchange of α-ketoglutarate and malate across the mitochondrial membrane and the α-ketoglutarate dehydrogenase enzyme of the TCA cycle for α-ketoglutarate (also referred to as 2-oxoglutarate) as a substrate [118, 119]. The balance between these two competing processes is central to linking energy metabolism to the metabolic state of the cytosol (Fig. 9). The exchange of α-ketoglutarate and malate is involved in transferring reducing equivalents generated in the cytosol to the mitochondria for oxidative energy production, while the α-ketoglutarate dehydrogenase is a calcium-activated, mitochondrial enzyme of the TCA cycle. This exchange occurs via the OMC on the mitochondrial membrane.
Importantly, α-ketoglutarate dehydrogenase is also very sensitive to the mitochondrial NADH/NAD+, as the enzyme-catalyzed reaction utilizes NAD+ as a cofactor and produces NADH. The mitochondrial dehydrogenases are also Ca++ activated, and thus the activity of α-ketoglutarate dehydrogenase competes with OMC activity for its substrate, α-ketoglutarate. In this manner, the rates of the mitochondrial dehydrogenases, in response to mitochondrial NADH/NAD+ and Ca++ levels, and OMC activity, are coordinated to the cytosolic NADH/NAD+ which is also linked to cytosolic Ca++ levels, providing a homeostatic mechanism for coordinating the contractile state of the cardiomyocyte with flux through the TCA cycle for NADH production leading to oxidative ATP synthesis [118–121]. Thus, the rate of exchange of metabolic intermediates between the mitochondrial matrix and cytosolic compartment of the cell enables the transduction of pathophysiological state between compartments to coordinate oxidative activity in the mitochondrial matrix with the functional state of the cell.
13C NMR detection of flux through the oxidative intermediary pathways in the intact heart relies on the observation of the large pool of glutamate that becomes labeled through exchange with the TCA cycle. The enzymes for the TCA cycle are inside the mitochondria, while at least 90 % of the glutamate is cytosolic [122–124]. Exchange of 13C enriched TCA cycle intermediates with the NMR-observed glutamate pool in the cytosol is achieved by the coordinated activity of two types of carrier proteins that span the mitochondrial membrane. One carrier is responsible for the reversible exchange of malate and αKG. The other carrier is unidirectional by virtue of being electrogenic, and exchanges cytosolic glutamate for mitochondrial aspartate. This transport system is the malate–aspartate shuttle, and, as discussed above, transports reducing equivalents cytosol to the mitochondrial matrix. At high cytosolic redox state (NADH/NAD+), the two carriers are driven in the forward direction, increasing exchange between αKG and glutamate [50, 118].
NADH Transfer into Mitochondria in Hypertrophied Hearts
The increased reliance on glycolytic metabolism by hypertrophied hearts implies that the heart also becomes increasingly dependent on the oxidation of reducing equivalents produced in the cytosol. The neonatal heart, as it develops to adult, shows reduced expression of both the malate–aspartate shuttle and glycerophosphate shuttle proteins, with minimal contributions from the glycerophosphate shuttle [114]. As discussed above, both shuttles carry reducing equivalents, produced by glycolysis in the cytosol and accepted by NAD+ across the NADH-impermeable membrane of the mitochondria for oxidative production of chemical energy. This link between the more glycolytic metabolism of the neonatal heart and the expression and activity of these transporters is clear, as the immature myocardium relies heavily on carbohydrates for fueling ATP production. With development, the myocardium becomes less reliant on glycolysis and the expression and activity of the exchange proteins is reduced [114–116].
However, in the hypertrophied myocardium, the activity but not the expression of the OMC, also referred to as the αKG–malate exchanger, is increased in the intact heart [50, 115]. In a study of the intact, hypertrophied rat heart, the activity of OMC was dramatically increased upon pressure overload to the heart, prior to any initial cardiac hypertrophy, and returned to baseline upon evidence of significant LVH [117]. The findings of that study suggest that the immediate stress of pressure overload elevates cytosolic NADH, and upon the initial development of hypertrophy, prior to functional decline, the metabolic stress is transiently alleviated by hypertrophy with OMC activity returned to baseline levels. Subsequently, as the pressure overloaded heart decompensates, the OMC activity rises again. This response of OMC to pressure overload is quite likely induced by an elevated cytosolic NADH/NAD+ ratio in the cytosol due to increased glycolytic activity in cardiac hypertrophy, as discussed in the previous sections. However, the response is purely activity-driven by metabolic regulation, inducing intermediate exchange across the mitochondrial members, because OMC content over the course of the hypertrophic either remains unchanged from the initial induction of the hypertrophic stimulus to the point of compensatory hypertrophy or displays a modest, initial drop [50, 115].
Metabolite exchange across OMC also enables the influx and/or efflux of carbon mass through the first span of the TCA cycle, serving to maintain equilibrium in the carbon mass balance between the first and the second spans of the cycle. The ability to exchange carbon mass, through metabolic intermediate transfer across the mitochondrial membrane, maintains the equilibrium condition required for net forward flux through the TCA, especially in response to the elevated influx of carbon mass in the hypertrophied heart due to increased malate production via ME-1, as discussed in previous sections of this chapter [20, 40].
References
McGavock, J. M., Lingvay, I., Zib, I., Tillery, T., Salas, N., Unger, R., et al. (2007). Cardiac steatosis in diabetes mellitus: A 1H-magnetic resonance spectroscopy study. Circulation, 116, 1170–1175.
Hankiewicz, H. J., Banke, N. H., Farjah, M., & Lewandowski, E. D. (2010). Early impairment of transmural principal strains in the left ventricle wall following short-term, high fat feeding of mice predisposed to cardiac steatosis. Circulation. Cardiovascular Imaging, 3, 710–717.
Chung, J., Abraszewski, P., Yu, X., Liu, W., Krainik, A. J., Ashford, M., et al. (2006). Paradoxical increase in ventricular torsion and systolic torsion rate in type I diabetic patients under tight glycemic control. Journal of the American College of Cardiology, 47, 384–390.
Giannetta, E., Isidori, A. M., Galea, N., Cabone, I., Mandosi, E., Vizza, C. D., et al. (2012). Chronic inhibition of cGMP phosphodiesterase 5A improves diabetic cardiomyopathy. Circulation, 125, 2323–2333.
Hankiewicz, J. H., & Lewandowski, E. D. (2007). Improved cardiac tagging resolution at high field elucidates transmural differences in principle strain measurements in the mouse heart and reduced stretch in dilated cardiomyopathy. Journal of Cardiovascular Magnetic Resonance, 9, 8838–8890.
Hankiewicz, J. H., Goldspink, G. H., Buttrick, P. M., & Lewandowski, E. D. (2008). Principal strain changes precede ventricular wall thinning during transition to heart failure in a mouse model of dilated cardiomyopathy. American Journal of Physiology. Heart and Circulatory Physiology, 294, H330–H336.
Desjardins, C. L., Chen, Y., Coulton, A. T., Hoit, B. D., Yu, X., & Stelzer, J. E. (2010). Cardiac myosin binding protein C insufficiency leads to early onset of mechanical dysfunction. Circulation. Cardiovascular Imaging, 5, 127–136.
Li, W., Liu, W., Zhong, J., & Yu, X. (2009). Early manifestation of alteration in cardiac function in dystrophin deficient mdx mouse using 3D CMR tagging. Journal of Cardiovascular Magnetic Resonance, 11, 40–51.
Desjardins, C. L., Chen, Y., Coulton, A. T., Hoit, B. D., Yu, X., & Stelzer, J. E. (2010). Altered in vivo left ventricular torsion and principal strains in hypothyroid rats. American Journal of Physiology. Heart and Circulatory Physiology, 299, H1577–H1587.
Ingwall, J. S., & Weiss, R. G. (2004). Is the failing heart energy starved? On using chemical energy to support cardiac function. Circulation Research, 95, 135–145.
Weiss, R. G., Gerstenblith, G., & Bottomley, P. A. (2005). ATP flux through creatine kinase in the normal, stressed, and failing human heart. Proceedings of the National Academy of Sciences of the United States of America, 102, 808–813.
Smith, C. S., Bottomley, P. A., Schulman, S. P., Gerstenblith, G., & Weiss, R. G. (2006). Altered creatine kinase adenosine triphosphate kinetics in failing hypertrophied human myocardium. Circulation, 114, 1151–1158.
Ingwall, J. S. (2006). On the hypothesis that the failing heart is energy starved: Lessons learned from the metabolism of ATP and creatine. Current Hypertension Reports, 8, 457–464.
Zhang, J., Merkle, H., Hendrich, K., Garwood, M., From, A. H., Ugurbil, K., et al. (1993). Bioenergetic abnormalities associated with severe left ventricular hypertrophy. The Journal of Clinical Investigation, 92, 993–1003.
Bache, R. J., Zhang, J., Path, G., Merkle, H., Hendrich, K., From, A. H., et al. (1994). High-energy phosphate responses to tachycardia and inotropic stimulation in left ventricular hypertrophy. American Journal of Physiology. Heart and Circulatory Physiology, 266, H1959–H1970.
Liao, R., Nascimben, L., Friedrich, J., Gwathmey, J. K., & Ingwall, J. S. (1996). Decreased energy reserve in an animal model of dilated cardiomyopathy. Relationship to contractile performance. Circulation Research, 78, 893–902.
Zhang, J., Wilke, N., Wang, Y., Zhang, Y., Wang, C., Eijgelshoven, M. H. J., et al. (1996). Functional and bioenergetic consequences of postinfarction left ventricular remodeling in a new porcine model. Circulation, 94, 1089–1100.
Tian, R., Nascimben, L., Ingwall, J. S., & Lorell, B. H. (1997). Failure to maintain a low ADP concentration impairs diastolic function in hypertrophied rat hearts. Circulation, 96, 1313–1319.
O’Donnell, J. M., Narayan, P., Bailey, M. Q., AbduljaliL, A. M., Altschuld, R. A., McCune, S. A., et al. (1998). 31P-NMR analysis of congestive heart failure in the SHHF/Mcc-facp rat heart. Journal of Molecular and Cellular Cardiology, 30, 235–241.
Sorokina, N., O’Donnell, J. M., McKinney, R. D., Pound, K. M., Woldegiorgis, G., LaNoue, K. F., et al. (2007). Recruitment of compensatory pathways to sustain oxidative flux with reduced CPT1 activity characterizes inefficiency in energy metabolism in hypertrophied hearts. Circulation, 115, 2033–2041.
Conway, M. A., Allis, J., Ouwerkerk, R., Niioka, T., Rajagopalan, B., & Radda, G. F. (1991). Detection of low phosphocreatine to ATP ratio in failing hypertrophied human myocardium by 31P magnetic resonance spectroscopy. Lancet, 338, 973–976.
Hardy, C. J., Weiss, R. G., Bottomley, P. A., & Gerstenblith, G. (1991). Altered myocardial high-energy phosphate metabolites in patients with dilated cardiomyopathy. American Heart Journal, 122, 795–801.
Masuda, Y., Tateno, Y., Ikehira, H., Hashimoto, T., Shishido, F., Sekiya, M., et al. (1992). High-energy phosphate metabolism of the myocardium in normal subjects and patients with various cardiomyopathies—The study using ECG gated MR spectroscopy with a localization technique. Japanese Circulation Journal, 56, 620–626.
de Roos, A., Doornbos, J., Luyten, P., Oosterwaal, L., van der Wall, E., & den Hollander, J. (1992). Cardiac metabolism in patients with dilated and hypertropic cardiomyopathy: Assessment with proton-decoupled P-31 MR spectroscopy. Journal of Magnetic Resonance Imaging, 2, 711–719.
Sieverding, L., Jung, W., Breuer, J., Widmaier, S., Staubert, A., van Erckelens, F., et al. (1997). Proton-decoupled myocardial 31P NMR spectroscopy reveals decreased PCr/Pi in patients with severe hypertrophic cardiomyopathy. The American Journal of Cardiology, 80, 34A–40A.
Neubauer, S., Horn, M., Pabst, T., Harre, K., Stromer, H., Bertsch, G., et al. (1997). Cardiac high-energy phosphate metabolism in patients with aortic valve disease assessed by 31P-magnetic resonance spectroscopy. Journal of Investigative Medicine, 45, 453–462.
Saupe, K. W., Eberli, F. R., Ingwall, J. S., & Apstein, C. S. (1999). Hypoperfusion-induced contractile failure does not require changes in cardiac energetics. American Journal of Physiology. Heart and Circulatory Physiology, 276, H1715–H1723.
Neubauer, S., Horn, M., Cramer, M., Harre, K., Newell, J. B., Peters, W., et al. (1997). Myocardial phosphocreatine-to-ATP ratio is a predictor of mortality in patients with dilated cardiomyopathy. Circulation, 96, 2190–2196.
Weiss, R. G., Gerstenblish, G., & Bottomley, P. A. (2005). ATP flux through creatine kinase in the normal, stressed, and failing human heart. Proceedings of the National Academy of Sciences, 102, 808–813.
Neubauer, S., Remkes, H., Spindler, M., Horn, M., Wiesmann, F., Prestle, J., et al. (1999). Downregulation of the Na(+)-creatine cotransporter in failing human myocardium and in experimental heart failure. Circulation, 100, 1847–1850.
Wallis, J., Lygate, C. A., Fischer, A., ten Hove, M., Schneider, J. E., Sebag-Montefiore, L., et al. (2005). Supranormal myocardial creatine and phosphocreatine concentrations lead to cardiac hypertrophy and heart failure: Insights from creatine transporter-overexpressing transgenic mice. Circulation, 112, 3131–3139.
Gupta, A., Akki, A., Wang, Y., Leppo, M. K., Chaco, V. P., Foster, D. B., et al. (2012). Creatine kinase-mediated improvement of function in failing mouse hearts provides causal evidence the failing heart is energy starved. The Journal of Clinical Investigation, 122, 291–302.
Hirsch, G. A., Bottomley, P. A., Gerstnblith, G., & Weiss, R. G. (2012). Allopurinol acutely increases adenosine triphosphate energy delivery in failing human hearts. Journal of the American College of Cardiology, 28, 802–808.
Allard, M. F., Schonekess, B. O., Henning, S. L., English, D. R., & Lopaschuk, G. D. (1994). Contribution of oxidative metabolism and glycolysis to ATP production in hypertrophied hearts. American Journal of Physiology. Heart and Circulatory Physiology, 267, H742–H750.
Sack, M. N., Rader, T. A., Park, S., Bastin, J., McCune, S. A., & Kelly, D. P. (1996). Fatty acid oxidation enzyme gene expression is downregulated in failing heart. Circulation, 94, 2837–2842.
Yang, X., Buja, M., & McMillin, J. B. (1996). Change in expression of heart carnitine palmitoyltransferase I isoforms with electrical stimulation of cultured rat neonatal cardiac myocytes. Journal of Biological Chemistry, 271, 12082–12087.
Doenst, T., Goodwin, G. W., Cedars, A. M., Wang, M., Stepkowski, S., & Taegtymeyer, H. (2001). Load-induced changes in vivo alter substrate fluxes and insulin responsiveness of rat heart in vitro. Metabolism, 50, 1083–1090.
Lehman, J. J., & Kelly, D. P. (2002). Gene regulatory mechanisms governing energy metabolism during cardiac hypertrophic growth. Heart Failure Reviews, 7, 175–185.
Finck, B. N., Han, X., Courtois, M., Aimond, F., Nerbonne, J. M., Kovacs, A., et al. (2003). A critical role for PPARalpha-mediated lipotoxicity in the pathogenesis of diabetic cardiomyopathy: Modulation by dietary fat content. Proceedings of the National Academy of Sciences of the United States of America, 100, 1226–1231.
Pound, K. M., Sorokina, N., Fasano, M., Berkich, D., LaNoue, K. F., O’Donnell, J. M., et al. (2009). Substrate-enzyme competition attenuates upregulated anaplerotic flux through malic enzyme in hypertrophied rat heart and restores triacylglyceride content. Circulation Research, 104, 805–812.
Scheuermann-Freestone, M., Madsen, P. L., Manners, D., Blamire, A. M., Buckingham, R. E., Styles, P., et al. (2003). Abnormal cardiac and skeletal muscle energy metabolism in patients with type 2 diabetes. Circulation, 107, 3040–3046.
Yan, J., Young, M. E., Cui, L., Lopaschuk, G. D., Liao, R., & Tian, R. (2009). Increased glucose uptake and oxidation in mouse hearts prevent high fatty acid oxidation but cause cardiac dysfunction in diet-induced obesity. Circulation, 119, 2818–2828.
Oakes, N. D., Thalen, P., Aasum, E., Edgley, A., Larsen, T., Furler, S. M., et al. (2006). Cardiac metabolism in mice: Tracer method developments and in vivo application revealing profound metabolic inflexibility in diabetes. American Journal of Physiology. Endocrinology and Metabolism, 290, E870–E881.
Goodwin, G. W., Ahmad, F., Doenst, T., & Taegtmeyer, H. (1998). Energy provision from glycogen, glucose, and fatty acids on adrenergic stimulation of isolated working rat hearts. American Journal of Physiology. Heart and Circulatory Physiology, 274, H1239–H1247.
Goodwin, G. W., Taylor, C. S., & Taegtmeyer, H. (1998). Regulation of energy metabolism of the heart during acute increase in heart work. Journal of Biological Chemistry, 273, 29530–29539.
Witham, W., Yester, K., O’Donnell, C. P., & McGaffin, K. R. (2012). Restoration of glucose metabolism in leptin-resistant mouse hearts after acute myocardial infarction through the activation of survival kinase pathways. Journal of Molecular and Cellular Cardiology, 53, 91–100.
O’Donnell, J. M., Fields, A. D., Sorokina, N., & Lewandowski, E. D. (2008). Absence of endogenous lipid oxidation in heart failure exposes limitations for triacylglycerol storage and turnover. Journal of Molecular and Cellular Cardiology, 44, 315–322.
Sack, M. N. (2009). Innate short-circuiting of mitochondrial metabolism in cardiac hypertrophy: Identification of novel consequences of enhanced anaplerosis. Circulation Research, 104, 717–719.
Ingwall, J. S. (2009). Energy metabolism in heart failure and remodeling. Cardiovascular Research, 81, 412–419.
Lewandowski, E. D., O’Donnnell, J. M., Scholz, T. D., Sorokina, N., & Buttrick, P. M. (2007). Recruitment of NADH shuttling in pressure overloaded and hypertrophic rat hearts. American Journal of Physiology. Cell Physiology, 292(5), C1880–C1886.
Listenberger, L. L., Han, X., Lewis, S. E., Cases, S., Farese, R. J., Jr., Ory, D. S., et al. (2003). Triglyceride accumulation protects against fatty acid-induced lipotoxicity. Proceedings of the National Academy of Sciences of the United States of America, 100, 3077–3082.
Chiu, H. C., Kovacs, A., Blanton, R. M., Han, X., Courtois, M., Weinheimer, C. J., et al. (2005). Transgenic expression of fatty acid transport protein 1 in the heart causes lipotoxic cardiomyopathy. Circulation Research, 96, 225–233.
Murakami, Y., Zhang, Y., Cho, Y. K., Mansoor, A. M., Chung, J. K., Chu, C., et al. (1999). Myocardial oxygenation during high workstates in hearts with postinfarction remodeling. Circulation, 99, 942–948.
Saddik, M., Gamble, J., Witters, L. A., & Lopaschuk, G. D. (1993). Acetyl-CoA carboxylase regulation of fatty acid oxidation in the heart. Journal of Biological Chemistry, 268, 25836–25845.
Dyck, J. R., Barr, A. J., Barr, R. L., Kolattukudy, P. E., & Lopaschuk, G. D. (1998). Characterization of cardiac malonyl-CoA decarboxylase and its putative role in regulating fatty acid oxidation. American Journal of Physiology. Heart and Circulatory Physiology, 275, H2122–H2129.
McGarry, J. D., & Brown, N. F. (1997). The mitochondrial carnitine palmitoyltransferase system. European Journal of Biochemistry, 244, 1–14.
Brown, N. F., Weis, B. C., Husti, J. E., Foster, D. W., & McGarry, J. D. (1995). Mitochondrial carnitine palmitoyltransferase I isoform switching in the developing rat heart. Journal of Biological Chemistry, 270, 8952–8957.
Yu, G. S., Lu, Y., & Gulick, T. (1998). Expression of novel isoforms of carnitine palmitoyltransferase I generated by alternative splicing of the CPT-Ib gene. Biochemical Journal, 334, 225–231.
Yu, G. S., Lu, Y., & Gulick, T. (1998). Rat carnitine palmitoyltransferase I b mRNA splicing isoforms. Biochimica et Biophysica Acta, 1393, 166–172.
Yamazaki, N., Shinohara, Y., Shima, A., & Terada, H. (1996). High expression of a novel carnitine palmitoyltransferase I like protein in rat brown adipose tissue and heart: Isolation and characterization of its cDNA clone. FEBS Letters, 363, 41–45.
McMillin, J. B., Wang, D., Witters, L. A., & Buja, L. M. (1995). Kinetic properties of carnitine palmitoyltransferase I in cultured neonatal rat cardiac myocytes. Archives of Biochemistry and Biophysics, 312, 375–384.
Weis, B. C., Esser, V., Foster, D. W., & McGarry, J. D. (1994). Rat heart expresses two forms of mitochondrial carnitine palmitoyltransferase I. Journal of Biological Chemistry, 269, 18712–18715.
Razeghi, P., Young, M. E., Alcorn, J. L., Moravec, C. S., Frazier, O. H., & Taegtmeyer, H. (2001). Metabolic gene expression in fetal and failing human heart. Circulation, 104, 2923–2931.
Kolwicz, S. C., Olson, D. P., Marney, L. C., Garcia-Menendez, L., Synovec, R. E., & Tian, R. (2012). Cardiac-specific deletion of acetyl CoA carboxylase 2 (ACC2) prevents metabolic remodeling during pressure-overload hypertrophy. Circulation Research, 22, 2012.
Zhou, L., Huang, H., Yuan, C. L., Keung, W., Lopaschuk, G. D., & Stanley, W. C. (2008). Metabolic response to an acute jump in cardiac workload: Effects on malonyl-CoA, mechanical efficiency, and fatty acid oxidation. American Journal of Physiology. Heart and Circulatory Physiology, 294, H954–H960.
Kudej, R. K., Fasano, M., Zhao, X., Lopaschuk, G. D., Fischer, S. K., Vatner, D. E., et al. (2011). Second window of preconditioning normalizes palmitate use for oxidation and improves function during low-flow ischaemia. Cardiovascular Research, 92, 394–400.
van der Vusse, G. J. (2002). The fascinating and elusive life of cardiac fatty acids. Cardiovascular Research, 92, 363–364.
Kim, J. Y., Koves, T. R., Yu, G. S., Gulick, T., Cortright, R. N., Dohm, G. L., et al. (2002). Evidence of a malonyl-CoA-insensitive carnitine palmitoyltransferase I activity in red skeletal muscle. American Journal of Physiology. Endocrinology and Metabolism, 282, E1014–E1022.
Lewandowski, E. D., Fischer, S. K., Fasano, M., Banke, N., Walker, L. A., Huqi, A., et al. (2013). Acute L-CPT1 overexpression recapitulates reduced palmitate oxidation of cardiac hypertrophy. Circulation Research, 112, 57–65.
Doh, K. O., Kim, Y. W., Park, S. Y., Lee, S. K., Park, J. S., & Kim, J. Y. (2005). Interrelation between long-chain fatty acid oxidation rate and carnitine palmitoyltransferase 1 activity with different isoforms in rat tissues. Life Sciences, 77, 435–443.
Chokshi, A., Drosatos, K., Cheema, F. H., Ji, R., Khawaja, T., Yu, S., et al. (2012). Circulation, 125, 2844–2853.
Park, T. S., Hu, Y., Noh, H. L., Drosatos, K., Okajima, K., Buchanan, J., et al. (2008). Ceramide is a cardiotoxin in lipotoxic cardiomyopathy. Journal of Lipid Research, 49, 2101–2112.
Sharma, S., Adrogue, J. V., Golfman, L., Uray, I., Lemm, J., Youker, K., et al. (2004). Intramyocardial lipid accumulation in the failing human heart resembles the lipotoxic rat heart. The FASEB Journal, 18, 1692–1700.
Goldberg, I. J., Trent, C. M., & Schulze, P. C. (2012). Lipid metabolism and toxicity in the heart. Cell Metabolism, 15, 805–812.
Banke, N. H., Wende, A. R., Leone, T. C., O’Donnell, J. M., Abel, E. D., Kelly, D. P., et al. (2010). Preferential oxidation of triacylglyceride-derived fatty acids in heart is augmented by the nuclear receptor PPARα. Circulation Research, 107, 233–241.
Banke, N. H., Pound, K. M., DeLorenzo, M., Yan, L., Reinhardt, H., Vatner, D. E., et al. (2012). Gender distinguishes myocardial triacylglyceride dynamics in response to long term caloric restriction in mice. Journal of Molecular and Cellular Cardiology, 52, 733–740.
Taegtmeyer, H., McNulty, P., & Young, M. E. (2002). Adaptation and maladaptation of the heart in diabetes: Part I general concepts. Circulation, 105, 1727–1733.
Young, M. E., McNulty, P., & Taegtmeyer, H. (2002). Adaptation and maladaptation of the heart in diabetes: Part II potential mechanisms. Circulation, 105, 1861–1870.
Belke, D. D., Larsen, T. S., Gibbs, E. M., & Severson, D. L. (2000). Altered metabolism causes cardiac dysfunction in perfused hearts from diabetic (db/db) mice. American Journal of Physiology. Endocrinology and Metabolism, 279, E1104–E1113.
Finck, B. N., Lehman, J. J., Leone, T. C., Welch, M. J., Bennet, M. J., Kovacs, A., et al. (2002). The cardiac phenotype induced by PPARα overexpression mimics that caused by diabetes mellitus. The Journal of Clinical Investigation, 109, 121–130.
O’Donnell, J. M., Alpert, N., Zampino, M., Geenen, D. L., & Lewandowski, E. D. (2006). Accelerated triacylglycerol turnover kinetics in hearts of diabetic rats include evidence for compartmented lipid storage. American Journal of Physiology. Endocrinology and Metabolism, 290, E448–E455.
de Vries, J. E., Vork, M. M., Roemen, T. H., de Jong, Y. F., Cleutjens, J. P., van der Vusse, G. H., et al. (1997). Saturated but not mono-unsaturated fatty acids induce apoptotic cell death in neonatal rat ventricular myocytes. Journal of Lipid Research, 38, 1384–1394.
Hickson-Bick, D. L., Buja, L. M., & McMillin, J. B. (2000). Palmitate-mediated alterations in the fatty acid metabolism of rat neonatal cardiac myocytes. Journal of Molecular and Cellular Cardiology, 32, 511–519.
Leroy, C., Tricot, S., Lacour, B., & Grynberg, A. (2008). Protective effect of eicosapentaenoic acid on palmitate-induced apoptosis in neonatal cardiomyocytes. Biochimica et Biophysica Acta, 1781, 685–693.
Listenberger, L. L., Ory, D. S., & Schaffer, J. E. (2001). Palmitate-induced apoptosis can occur through a ceramide-independent pathway. Journal of Biological Chemistry, 276, 14890–14895.
Okere, I. C., Chandler, M. P., McElfresh, T. A., Rennison, J. H., Sharov, V., Sabbah, H. N., et al. (2006). Differential effects of saturated and unsaturated fatty acid diets on cardiomyocyte apoptosis, adipose distribution, and serum leptin. American Journal of Physiology. Heart and Circulatory Physiology, 291, H38–H44.
Baranowski, M., Błachnio, A., Zabielski, P., & Górski, J. (2007). PPARalpha agonist induces the accumulation of ceramide in the heart of rats fed high-fat diet. Journal of Physiology and Pharmacology, 58, 57–72.
Hanada, K. (2003). Serine palmitoyltransferase, a key enzyme of sphingolipid metabolism. Biochimica et Biophysica Acta, 1632, 16–30.
Xia, P., Inoguchi, T., Kern, T. S., Engerman, R. L., Oates, P. J., & King, G. L. (1994). Characterization of the mechanism for the chronic activation of diacylglycerol-protein kinase C pathway in diabetes and hypergalactosemia. Diabetes, 43, 122–129.
Baranowski, M., Zabielski, P., Blachnio, A., & Gorski, J. (2008). Effect of exercise duration on ceramide metabolism in the rat heart. Acta Physiologica, 192, 519–529.
Ke, Y., Lei, M., & Solaro, R. J. (2008). Regulation of cardiac excitation and contraction by p21 activated kinase-1. Progress in Biophysics and Molecular Biology, 98, 238–250.
Bokoch, G. M., Reilly, A. M., Daniels, R. H., King, C. C., Olivera, A., Spiegel, S., et al. (1998). A GTPase-independent mechanism of p21-activated kinase activation. Regulation by sphingosine and other biologically active lipids. Journal of Biological Chemistry, 273, 8137–8144.
King, C. C., Gardiner, E. M. M., Zenke, F. T., Bohl, B. P., Newton, A. C., Hemmings, B. A., et al. (2000). p21-Activated kinase is phosphorylated and activated by 3-phosphoinositide-dependent kinase-1 (PDK1). Journal of Biological Chemistry, 275, 41201–41209.
Wu, S. C., & Solaro, R. J. (2007). Protein kinase C zeta. A novel regulator of both phosphorylation and de-phosphorylation of cardiac sarcomeric proteins. Journal of Biological Chemistry, 282, 30691–30698.
Sheehan, K. A., Ke, Y., Wolska, B. M., & Solaro, R. J. (2009). Expression of active p21-activated kinase-1 induces Ca2+ flux modification with altered regulatory protein phosphorylation in cardiac myocytes. American Journal of Physiology. Cell Physiology, 296, C47–C58.
Lydell, C. P., Chan, A., Wambolt, R. B., Sambandam, N., Parsons, H., Bondy, G. P., et al. (2002). Pyruvate dehydrogenase and the regulation of glucose oxidation in hypertrophied rat hearts. Cardiovascular Research, 53, 841–851.
Sambandam, N., Lopaschuk, G. D., Brownsey, R. W., & Allard, M. F. (2002). Energy metabolism in the hypertrophied heart. Heart Failure Reviews, 7, 161–173.
Wambolt, R. B., Lopaschuk, G. D., Brownsey, R. W., & Allard, M. F. (2000). Dichloroacetate improves postischemic function of hypertrophied rat hearts. Journal of the American College of Cardiology, 36, 1378–1385.
Ashworth, J. M., & Kornberg, H. L. (1966). The anaplerotic fixation of carbon dioxide by Escherichia coli. Proceedings of the Royal Society of London. Series B, 165, 179–188.
Peuhkurinen, K. F., Nuutinen, E. M., Pietilainen, E. P., Hiltunen, J. K., & Hassinen, I. E. (1982). Role of pyruvate carboxylation in the energy-linked regulation of pool sizes of tricarboxylic acid-cycle intermediates in the myocardium. Biochemical Journal, 208, 577–581.
Sundqvist, K. E., Heikkila, J., Hassinen, I. E., & Hiltunen, J. K. (1987). Role of NADP+-linked malic enzymes as regulators of pool size of tricarboxylic acid-cycle intermediates in the perfused heart. Biochemical Journal, 243, 853–857.
Russell, R. R., III, & Taegtmeyer, H. (1991). Changes in citric acid cycle flux and anaplerosis antedate the functional decline in isolated rat hearts utilizing acetoacetate. The Journal of Clinical Investigation, 87, 384–390.
Gibala, M. J., Young, M. E., & Taegtmeyer, H. (2000). Anaplerosis of the citric acid cycle: Role in energy metabolism of heart and skeletal muscle. Acta Physiologica Scandinavica, 168, 657–665.
Reszko, A. E., Kasumov, T., Pierce, B. A., David, F., Hoppel, C. L., Stanley, W. C., et al. (2003). Assessing the reversibility of the anaplerotic reactions of the propionyl-CoA pathway in heart and liver. Journal of Biological Chemistry, 278, 34959–34965.
Pisarenko, O. I., Solomatina, E. S., & Studfneva, I. M. (1986). The role of amino acid catabolism in the formation of the tricarboxylic acid cycle intermediates and ammonia in anoxic rat heart. Biochimica et Biophysica Acta, 885, 154–161.
Russell, R. R., III, & Taegtmeyer, H. (1991). Pyruvate carboxylation prevents the decline in contractile function of rat hearts oxidizing acetoacetate. American Journal of Physiology, 261, H1756–H1762.
Olson, A. K., Hyyti, O. M., Cohen, G. A., Ning, X. H., Sadilek, M., Isern, N., et al. (2008). Superior cardiac function via anaplerotic pyruvate in the immature swine heart after cardiopulmonary bypass and reperfusion. American Journal of Physiology. Heart and Circulatory Physiology, 295, H2315–H2320.
Takimoto, E., & Kass, D. A. (2007). Role of oxidative stress in cardiac hypertrophy and remodeling. Hypertension, 49, 241–248.
Jain, M., Brenner, D. A., Cui, L., Lim, C. C., Wang, B., Pimentel, D. R., et al. (2003). Glucose-6-phosphate dehydrogenase modulates cytosolic redox status and contractile phenotype in adult cardiomyocytes. Circulation Research, 93, e6–e9.
Jain, M., Cui, L., Brenner, D. A., Wang, B., Handy, D. E., Leopold, J. A., et al. (2004). Increased myocardial dysfunction after ischemia-reperfusion in mice lacking glucose-6-phosphate dehydrogenase. Circulation, 109, 898–903.
Zabala, A., Churruca, I., Fernandez-Quintela, A., Rodriguez, V. M., Macarulla, M. T., Martinez, J. A., et al. (2006). Trans-10, cis-122 conjugated linoleic acid inhibits lipoprotein lipase but increases the activity of lipogenic enzymes in adipose tissue from hamsters fed an atherogenic diet. British Journal of Nutrition, 95, 1112–1119.
Cederbaum, A. I., Lieber, C. S., Beattie, D. S., & Rubin, E. (1973). Characterization of shuttle mechanisms for the transport of reducing equivalents into mitochondria. Archives of Biochemistry and Biophysics, 158, 763–781.
Safer, B., & Williamson, J. R. (1973). Mitochondrial-cytosolic interactions in perfused rat heart. Role of coupled transamination in repletion of citric acid cycle intermediates. Journal of Biological Chemistry, 248, 2570–2579.
Scholz, T., & Koppenhafer, S. (1995). Reducing equivalent shuttles in developing myocardium: Enhanced capacity in the newborn heart. Pediatric Research, 38, 221–227.
Rupert, B. E., Segar, J. L., Schutte, B. C., & Scholz, T. D. (2000). Metabolic adaptation of the hypertrophied heart: Role of the malate/aspartate and alpha-glycerophosphate shuttles. Journal of Molecular and Cellular Cardiology, 32, 2287–2297.
Griffin, J., O’Donnell, J. M., White, L. T., Hajjar, R. J., & Lewandowski, E. D. (2000). Postnatal expression and activity of the 2-oxoglutarate malate carrier in intact hearts. American Journal of Physiology. Cell Physiology, 279, C1704–C1709.
Scholz, T. D., Laughlin, M. R., Balaban, R. S., Kupriyanov, V. V., & Heineman, F. W. (1995). Effect of substrate on mitochondrial NADH, cytosolic redox state, and phosphorylated compounds in isolated hearts. American Journal of Physiology, 268, H82–H91.
Yu, X., White, L. T., Alpert, N. M., & Lewandowski, E. D. (1996). Subcellular metabolite transport and carbon isotope kinetics in the intramyocardial glutamate pool. Biochemistry, 35, 6963–6968.
O’Donnell, J. M., Doumen, C., LaNoue, K. F., White, L. T., Yu, X., Alpert, N. M., et al. (1998). Dehydrogenase regulation of metabolite oxidation and efflux from mitochondria of intact hearts. American Journal of Physiology. Heart and Circulatory Physiology, 274, H467–H476.
Hansford, R. G. (1991). Dehydrogenase activation by Ca2+ in cells and tissues. Journal of Bioenergetics and Biomembranes, 23, 823–853.
Zima, A. V., Copello, J. A., & Blatter, L. A. (2004). Effects of cytosolic NADH/NAD(+) levels on sarcoplasmic reticulum Ca(2+) release in permeabilized rat ventricular myocytes. The Journal of Physiology, 555, 727–741.
Tischler, M., Pachence, J., Williamson, J. R., & LaNoue, K. F. (1976). Mechanism of glutamate-aspartate translocation across the mitochondrial membrane. Archives of Biochemistry and Biophysics, 173, 448–461.
LaNoue, K. F., & Schoolwerth, A. C. (1979). Metabolite transport in mitochondria. Annual Review of Biochemistry, 48, 871–922.
Yu, X., White, L. T., Doumen, C., Damico, L. A., LaNoue, K. F., Alpert, N. M., et al. (1995). Kinetic analysis of dynamic 13C NMR spectra: Metabolic flux, regulation, and compartmentation in hearts. Biophysical Journal, 69, 2090–2102.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2013 Springer Science+Business Media, LLC
About this chapter
Cite this chapter
Lewandowski, E.D. (2013). Biophysical Mechanisms for the Metabolic Component of Impaired Heart Function. In: Solaro, R., Tardiff, J. (eds) Biophysics of the Failing Heart. Biological and Medical Physics, Biomedical Engineering. Springer, New York, NY. https://doi.org/10.1007/978-1-4614-7678-8_5
Download citation
DOI: https://doi.org/10.1007/978-1-4614-7678-8_5
Published:
Publisher Name: Springer, New York, NY
Print ISBN: 978-1-4614-7677-1
Online ISBN: 978-1-4614-7678-8
eBook Packages: Physics and AstronomyPhysics and Astronomy (R0)