Abstract
Early life stress is a term used to describe adversities which may occur during childhood and adolescence. The majority of human studies focus on negative outcomes resulting from early life stress. There is an emerging research field which aims to investigate possible positive outcomes of early life stress. In this chapter, human studies are reviewed with regard to both maladaptive and adaptive consequences of early life stress. Recent findings suggest that early life stress is associated with central nervous alterations, altered physiological stress responses, impaired cognitive functioning as well as an increased risk of developing behavior problems, and somatic and psychiatric illnesses. Early life stress may also result in posttraumatic growth (PTG) which describes changes in self-perception, interpersonal relationships, and worldview. A psychobiological stress model is proposed to integrate the findings.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
1 Introduction
There is a growing body of literature reporting that early life stress during childhood and adolescence appear to have a negative impact on development and numerous health outcomes later in life. Periods of heightened brain plasticity mark developmental phases during which the brain is most vulnerable to stress. It is well known that exposure to adverse conditions during these critical sensitive phases in development may lead to long-term alterations in brain structures, various bodily functions (e.g., immunological or endocrinological), and behavior, which may facilitate the manifestation of disease.
There is sufficient evidence that stress-responsive systems1 play a mediating role in the association between stress exposure and various developmental or health consequences. Stress processing originates in specific brain regions that are responsible for the early perception and evaluation of potentially threatening or stressful features of the current situation. The outcome of such an evaluation may lead to the activation of the stress axes, the sympatho-adrenal-medullary (SAM) system and the hypothalamus–pituitary–adrenal (HPA) axis (for details, see Sect. 2 of Chap. 6). Briefly, the SAM system elicits the release of norepinephrine and epinephrine, both of which are involved in the “flight-or-fight” response. The HPA axis includes the paraventricular nucleus (PVN) in the hypothalamus which releases the corticotrophin-releasing hormone or factor (CRH or CRF, respectively) which in turn induces the secretion of the adrenocorticotropic hormone (ACTH) from the anterior lobe of the pituitary gland into the blood stream. Finally, cortisol is secreted from the adrenal cortex, exerting its effects throughout the body (e.g., on metabolism, immune system, behavior, or cognition). Cortisol has counter-regulating effects on the HPA axis by inhibiting the further release of CRH and ACTH and thus acts as a negative feedback factor. The pulsatile secretion of cortisol follows a diurnal rhythm characterized by high morning levels and a decrease of cortisol levels during the course of the day. The cortisol awakening response (CAR), an important marker for cortisol regulation, describes the morning peak of cortisol levels measured 30min after awakening and has been related to a number of negative health outcomes (Kudielka and Wust 2010). Thus, studying cortisol release during the morning and/or during the day constitutes a well-researched approach to study HPA axis activity. In human research, the most prominent and effective stressors to study HPA axis reactivity are (a) performing a cognitive task (e.g., giving a speech and/or solving a difficult arithmetic task) in the presence of an evaluating audience or (b) administration of pharmacological substances (pharmacological challenge test). The latter is used to study HPA axis regulation, particularly via activating the system’s inherent feedback mechanisms (Ditzen etal. 2013). Pharmacological challenge tests are used to investigate how HPA axis activity is affected by temporary suppression due to administration of synthetic glucocorticoids (e.g., dexamethasone suppression test, DST), by stimulation due to administration of CRH or ACTH (CRH stimulation test, ACTH1–24 stimulation test), or by a time-lagged combination of suppressing and stimulating substances (e.g., dexamethasone/corticotrophin-releasing hormone test, Dex/CRH test).
Considering the amount of literature and the limited space of a book chapter, a selective overview of the literature on stress experienced during childhood and adolescence will be presented.2 The overall goal of this chapter is to review the literature and give an overview of the current state of knowledge about possible short- and long-term consequences of stress occurring in childhood and adolescence (early lifestress).
2 Stress During Childhood and Adolescence: Early Life Stress
We introduce the term early life stress which refers to stress that occurs during childhood and adolescence. Early life stress describes here a relatively heterogeneous concept which encompasses various adverse events that most people perceive as stressful and thus possibly lead to the activation of the above-mentioned stress-responsive systems. One source of early life stress constitutes childhood maltreatment which in turn can be divided into emotional abuse, physical abuse, sexual abuse, emotional neglect, or physical neglect. Growing up in an orphanage or institutional care provides a setting with possibly increased risk for neglect. One of the most extreme cases of neglect became public after the fall of the Ceausescu regime in Romania. Living conditions in institutional care during that time were characterized by deprivation of basic needs as well as emotional and physical neglect (most of the cited studies in this chapter have been conducted in Romanian orphans). These adverse living circumstances in institutions seem to be more likely under conditions of social poverty or under strict political regimes. Another source of early life stress may be the separation or loss of a parent or someone close. Finally, traumatic events such as natural disasters or war experiences are considered here as early life stress.
Children and adolescents might be particularly prone to stress since certain developmental stages (e.g., puberty) are characterized by various biological and hormonal changes. Early life stress during such sensitive periods may have consequences for biological outcomes, temperament and behavior, mental development, or general health later in life.
In this chapter we present findings organized along the following outcomes:
-
Effects on the central nervous system (CNS). This includes alterations in brain structure or brain functions (indicated by volume or activity of certain brain regions) using neuroimaging methods. Since recent research has revealed important central nervous effects of the neuropeptide oxytocin in humans (e.g., interpersonal bonding, affiliation behavior, and stress reduction), we present preliminary findings for this hormone here.
-
Effects on the HPA axis. Findings are separately presented for basal HPA axis activity (e.g., cortisol diurnal profile or CAR) and HPA axis reactivity in response to a psychological stressor or a pharmacological challenge test.
-
Effects on temperament and behavior. Measurement of personality or temperament is often achieved through assessing the infant’s attachment style, which describes how secure the bonding between infant and primary caregiver is. Negative behavior is usually operationalized as internalizing problems (withdrawal, somatic complaints, anxiety/depression) or externalizing problems (aggressive, delinquent behavior) which are thought to be disadvantageous manifestations of behavior.
-
Effects on cognitive functioning and academic performance. Developmental tests and intelligent tests are used to measure executive functions (e.g., decision making, response control), attention, memory, verbal skills, reasoning, and general intellectual/academic performance.
-
Effects on somatic health. Somatic health is measured either as perceived general health or as the occurrence of specific somatic illnesses (e.g., infectious disease).
-
Effects on psychological health. Psychological health may be measured via assessing symptoms such as depression, anxiety, or suicidal thoughts, and via diagnosing clinical disorders, such as personality disorders, major depressive disorder, anxiety disorders, and substance abuse.
In general, the traditional research focus in this field lies on negative outcomes of stress. Therefore, the majority of findings present maladaptive consequences in this chapter. In the next paragraphs we will present a selection of findings related to these effects, with maladaptive outcomes measured during childhood and adolescence (2.1.), maladaptive outcomes measured during adulthood (2.2.), and adaptive outcomes in children, adolescents, and adults (2.3.).
2.1 Maladaptive Outcomes Measured During Childhood and Adolescence
Studying outcomes of early life stress in children and adolescents may provide valuable information about short-term effects, since adverse events may have taken place only recently. In the following, possible effects on the CNS, the HPA axis, temperament and behavior, cognitive functioning and academic performance, somatic health, and psychological health are discussed.
Effects on the CNS. Several neuroimaging studies suggest that maltreated children and adolescents have small volumes of selected temporal regions (De Bellis etal. 2002) and the orbitofrontal cortex (Hanson etal. 2010), both of which are involved in executive and cognitive functions (e.g., reasoning, memory) and emotion regulation, as well as a small corpus callosum (Teicher etal. 2004), which connects both hemispheres. In another study, adopted orphans showed decreased activity in some of the aforementioned brain regions (specifically in the orbitofrontal cortex, temporal cortex, and brain stem) compared to same-aged children (7-11 years) with medically intractable partial epilepsy or to healthy adults (Chugani etal. 2001). De Bellis and colleagues, using both a cross-sectional (De Bellis etal. 2002) and a longitudinal approach (De Bellis etal. 2001), failed to show that childhood maltreatment was related to altered hippocampus or amygdala volumes, i.e., brain regions which are thought to be associated with emotion regulation and memory. In summary, there are some findings suggesting that early life stress may be associated with structural and functional alterations of brain regions that are related to cognitive and emotional functioning.
Effects on the HPA axis. There are numerous studies that investigated HPA axis activity (using basal cortisol levels) with regard to early life stress. One study showed that adoptees who spent more than 9 months in an orphanage displayed augmented daily cortisol levels, suggesting a generally enhanced HPA axis activity, compared to early adopted children and non-adopted children (Gunnar etal. 2001). Other studies focused on cortisol levels at a specific time of the day, mostly morning cortisol levels. One study found that neglected children (aged 3–31 months), still living with their birth parents, showed lower waking cortisol levels and significantly higher cortisol levels at bedtime compared to maltreated children who had been placed in foster care and compared to non-maltreated children (Bernard etal. 2010). In line with this finding, another study has found that sexually abused 5–7-year-old girls with posttraumatic stress disorder (PTSD) showed lower morning cortisol levels than control girls (King etal. 2001). In contrast to the aforementioned findings, sexually and physically abused children (mean age, 9.25 years) displayed higher morning cortisol levels than non-maltreated, emotionally maltreated, neglected, or physically abused subgroups who were studied during a day camp research program (Cicchetti and Rogosch 2001). A possible explanation for the inconsistent results in morning cortisol levels may be differences in age (range, 3 months–9 years) and time of morning sampling (range, 5–12 am) among these studies.
In addition to basal measurement, studying HPA axis reactivity specifically contributes to the understanding of underlying mechanisms of HPA axis (dys-)regulation. Healthy non-abused adolescents, aged 12–16 years, showed increased cortisol levels and a gradual flattening over time following an acute stress test, while nondepressed maltreated adolescents did not show such a pattern in cortisol levels (MacMillan etal. 2009). Another study found that the presence of any anxiety disorder, early life stress, or chronic stress during adolescence predicted peak cortisol response to a psychological stress test, whereby the interaction of early life stress and chronic stress during adolescence was the best predictor of this stress response (Rao etal. 2008). Another study reported that intravenous CRH administration led to greater ACTH peaks in depressed abused children (age range, 7–13 years) than in depressed, non-abused children or children with no history of either depression or abuse, whereas no group differences were apparent for cortisol responses (Kaufman etal. 1997). These studies hint at an altered HPA axis response under challenge; however, the direction of this response appears to be unclear. It is conceivable that anxious or depressive symptoms partly determine the direction and extent of the stress response, leading to either hyper- or hyporeactivity of the HPA axis.
Effects on temperament and behavior. In institutions such as orphanages, caregivers have to provide care and supervise several children at the same time, which may make it difficult to meet the child’s individual needs and to form selective interpersonal bonding between a primary caregiver and the child (Drury etal. 2011). Asforming a child-caregiver bond may thus be limited in such an environment, several studies have examined attachment styles in children with a history of institutional care. One study in adoptees found insecure attachment styles and high occurrence of indiscriminately friendly behavior, as determined by parents’ reports (Chisholm 1998). In another study, adoptees showed barely any attachment behavior in a strange situation procedure, which often serves as a tool to assess an infant’s attachment style to a primary caregiver (Zeanah etal. 2005). These findings suggest that experiences of institutional care may be a risk factor for maladaptive temperament, as measured via attachment styles.
There is also some evidence that early life stress may be related to behavioral problems. For example, one study found that adolescents, who had been physically abused in early childhood, were more likely to have been arrested for violent and nonviolent offenses than non-abused adolescents (Lansford etal. 2007). Another study found gender differences among sexually abused adolescents, with boys showing more sexual risk behavior and delinquent behavior than girls (Chandy etal. 1996). A summer camp study, using a longitudinal design, found that maltreated children exhibited significantly greater externalizing behavior problems and more impaired affect and behavior regulation (lower levels of ego-resiliency and higher levels of ego-undercontrol3) than non-maltreated children (Kim etal. 2009). In another study, children with a history of institutional care were more likely to show externalized or internalized disorders than never-institutionalized children (Zeanah etal. 2009). Similarly, sexually abused children and adolescents had more externalizing and internalizing symptoms than those without a history of abuse in another study (Maikovich-Fong and Jaffee 2010). In line with these findings, maltreatment-related PTSD was associated with internalizing and externalizing symptoms in children and adolescents (De Bellis etal. 2002). Finally, parent–child aggression was found to be associated with conduct problems, attention problems, anxiety-withdrawal, and motor excess in boys, whereas girls only showed increased anxiety-withdrawal (Jouriles etal. 1987). Considering the available literature, early life stress seems to raise the risk for attachment disturbance and behavioral problems.
Effects on cognitive functioning and academic performance. Evidence for possible cognitive alterations in affective children or adolescents comes from studies examining academic performance or performance in standardized tests. One study found that physically abused adolescents were less likely to have graduated from high school than non-abused peers (Lansford etal. 2007). Studying gender differences in maltreated adolescents revealed that male adolescents with a history of sexual abuse tended to report performing below average and had a higher dropout risk than sexually abused female adolescents (Chandy etal. 1996). In another study, maltreated adolescents were found to score lower on tests assessing reading performance and reasoning compared to non-maltreated adolescents (14 years) (Mills etal. 2011). A longitudinal study found that children living in an orphanage (measured at the age of 42 and 54 months) showed more cognitive impairment (e.g., verbal abilities) than orphans who were placed in a foster family before the age of 31 months (Nelson etal. 2007). This same study suggested an age-associated beneficial effect of foster care, i.e., the younger the child at the time of entry in foster care, the more cognitive improvement due to foster care was found. In another study, 8-year-old children with any orphanage experiences in early life were found to perform worse on tests requiring visual memory and executive function (e.g., decision making or response control) compared to peers without a history of institutional care (Bos etal. 2009). Another study partly confirmed these findings, i.e., adoptees at the age of 8 years showed deficits in visual memory, attention, and associative learning measures but not in executive functions (Pollak etal. 2010). Mild impairment in verbal skills, impulsivity, and attention were reported for adoptees compared to never-institutionalized children with medically intractable partial epilepsy (Chugani etal. 2001). In a longitudinal study, non-maltreated children showed a pronounced decline in attention problems over the course of time, while in maltreated children attention problems slightly increased between age 4 and age 6 and then remained consistently on a high level until age 10 (Thompson and Tabone 2010). These findings clearly indicate that early life stress has negative effects on a child’s cognitive development, as indicated by early school leaving and poor academic performance, as well as poor results in tests assessing memory, verbal skills, or attention.
Effects on somatic health. Several findings suggest that early life stress may negatively affect a child’s health. One study showed that parents of maltreated 4–6-year-old children rated their child’s health as poor more often and reported more serious illness when the child suffered from repeated adverse events than children without such adversities (Flaherty etal. 2006). In another study, childhood maltreatment occurring before the age of 11 years was found to increase the risk of asthma by 73% and even double the risk of non-asthma cardiorespiratory and nonsexually transmitted infectious diseases in adolescents (Lanier etal. 2010). These studies indicate a heightened risk for diseases later in life due to early life stress.
Effects on psychological health. Apart from detrimental effects of early life stress on somatic health, the psyche may be affected as well. One study found that single and multiple negative early life events (e.g., loss of someone close, parental separation, serious conflicts) were associated with depression disorder in adolescents (Patton etal. 2003). In another study, the report of repeated childhood sexual abuse was associated with personality disorder in a dose–response manner, going together with higher scores on neuroticism and lower agreeableness (Moran etal. 2011). Ina longitudinal study of children at the age between 4 and 10 years, maltreated children were found to show a steeper increase of anxiety/depression over time (Thompson and Tabone 2010). Other studies have confirmed this finding for high levels of depressive symptoms (Schilling etal. 2007), whereby girls seemed to be more affected (Chandy etal. 1996). High depression scores have been found in female adolescents who experienced a relatively mild life event in the past year (i.e.,new stepbrother or stepsister; (step)brother or (step)sister leaving home and father losing his job) and who had at least one parent having a history of depression or anxiety (Silberg etal. 2001). These studies suggest that early life stress may enhance the risk for showing high levels of depression and personality disorders.
To summarize these findings, early life stress appears to be associated with decreased volumes of certain brain regions which are known to regulate memory and emotion (e.g., corpus callosum, orbitofrontal cortex, and temporal regions). Inconsistent results were reported for HPA axis activity and reactivity, a finding that may be partly explained by different study designs. Externalizing and internalizing problems, as well as antisocial behavior were predominately present in children and adolescents with early life stress. Also, early life stress was associated with early school leaving and impaired cognitive performance (e.g., memory, verbal skills, or attention). Children or adolescents, who experienced early life stress, seem to be at high risk for somatic and psychiatric conditions.
2.2 Maladaptive Outcomes Measured During Adulthood
Numerous studies investigated adults who retrospectively reported having experienced early life stress. These studies provide valuable information whether early life stress may have long-lasting effects on health later in life. In the following paragraphs, a selection of findings for the effect on the CNS, the HPA axis, temperament and behavior, cognitive functioning and academic performance, somatic health, and psychological health are presented.
Effects on CNS. Several neurocognitive studies have found small left hippocampus volumes, a brain structure which is associated with memory, in abused females with depression (Vythilingam etal. 2002) or anxiety disorder (Stein etal. 1997). Adults with a history of early life stress were found to have small anterior cingulated cortex and caudate nuclei (located within basal ganglia), brain regions that are associated with attention and motor control, respectively (Cohen etal. 2006). Inanother study, PTSD women with childhood abuse showed increased affective responses to a trauma script as well as decreased activity in the anterior cingulated cortex and right hippocampus, both of which are related to memory (Bremner etal. 1999). These studies suggest that childhood maltreatment seems to be associated with decreased volume and activation of memory-related brain regions. Oxytocin is suggested to play an important role in bonding and attachment and thus may influence interpersonal relationships and interactions. One study found that early life stress was related to reduced plasma oxytocin levels (Opacka-Juffry and Mohiyeddini 2011). Similarly, another study found that reduced cerebrospinal fluid oxytocin levels were associated with childhood maltreatment, especially in females with severe childhood maltreatment (Heim etal. 2009). These two studies indicate that early life stress is related to decreased oxytocin levels later in life, which may be associated with maladaptive bonding and attachment in personal relationships.
Effects on the HPA axis. Measuring basal cortisol levels enables to study alterations in HPA axis activity in adults who have experienced early life stress. One study found that adults with a current anxiety disorder, who also had been severely maltreated before being adopted, demonstrated lower morning cortisol levels and a flattened diurnal cortisol secretion (van der Vegt etal. 2010). Another study found positive associations between severe childhood maltreatment and elevated cortisol levels throughout the day in women with either fibromyalgia or osteoarthritis (Nicolson etal. 2010). However, physically and psychologically healthy women, who experienced childhood maltreatment, did not differ in their diurnal cortisol pattern from those without such adverse experiences (Klaassens etal. 2009). The inconsistency in these findings for diurnal cortisol levels might be partly ascribed to the different samples (i.e., patient sample with an anxiety disorder, pain patients, and healthy subjects without a current somatic or psychiatric disorder). In contrast, a relatively consistent picture appears for the effect of early life stress on the CAR, which describes a rapid increase of cortisol concentrations within 30min after awakening and is usually considered a valid index of an intact HPA axis regulation. A decreased CAR has been found in adults with chronic fatigue who have been maltreated during childhood (Heim etal. 2009), in adults who were severely maltreated before adoption (van der Vegt etal. 2009), or in adults who lost a parent before the age of 14 (Meinlschmidt and Heim 2005). Based on these findings, a preliminary conclusion can be drawn in that adults with childhood maltreatment experiences may show attenuated basal morning cortisol levels.
In addition, there is a variety of studies which investigated the impact of early life stress on HPA axis reactivity using various approaches. Studying the HPA axis response to psychological stress, females with a history of childhood abuse and current PTSD showed higher cortisol levels before, during, and immediately after exposure to a trauma script than abused women without current PTSD (Elzinga etal. 2003). Giving a speech about a controversial topic was used as a stressor in another study, with the finding that adults with parental loss experiences before the age 16 showed increases of cortisol concentrations in response to a stress test in contrast to a control group (Luecken 1998). In another study, both abused women with and without current major depression showed increased ACTH responses to a psychological stress test compared to non-abused women (Heim etal. 2000). In the same study, only depressed women with a history of childhood abuse showed higher cortisol responses to the stressor than depressed women without childhood abuse and nondepressed women with or without childhood abuse experiences. In the same sample both the maximum ACTH and cortisol responses to a psychological stress test were predicted by the history of childhood abuse and the number of distinct types of abuse events (Heim etal. 2002). Another study found that adults, who retrospectively reported multiple adverse events, exhibited a smaller cortisol response to a psychological stress test than control subject; interestingly, this effect was more pronounced in male subjects (Elzinga etal. 2008). Lovallo etal. (2012) investigated whether early adverse life events (e.g., being mugged, threatened with a weapon, experiencing a robbery, sexual assault, or parental separation before age 15) have an impact on endocrine stress reactivity in adults without a psychiatric disorder. Adults who retrospectively reported more such adversities showed smaller cortisol responses to a psychological stressor. These findings provide some evidence that early life stress may be associated with altered HPA axis responses to psychological stressors.
Another approach to study HPA axis reactivity is using pharmacological challenge tests. Using the low-dose dexamethasone suppression test (DST), a test which is used to examine HPA axis negative feedback sensitivity, depressed women with childhood trauma were found to have lower cortisol and ACTH afternoon levels (super-suppression) compared to abused nondepressed women or non-abused healthy controls, whereas no difference was found when the test was conducted using the standard dose (Newport etal. 2004). The administration of CRH or ACTH stimulates the HPA axis with results in secretion of subsequent hormones of the HPA axis and thus is used to study the integrity of top-down HPA axis regulation. In one study, abused nondepressed women showed higher mean stimulated ACTH concentrations 5 and 30min after the CRH stimulation test compared to healthy non-abused controls; in contrast, depressed women (regardless of the presence of history of childhood) showed lower CRH-stimulated ACTH levels than controls. In the same study both abused women with and without major depression demonstrated lower baseline and stimulated cortisol levels than control subjects (Heim etal. 2001). Further, the same sample underwent an ACTH1–24 stimulation test, with the finding that abused females without depressive symptoms demonstrated decreased basal and stimulated cortisol concentrations, whereas abused depressed females had only lower baseline cortisol values compared to controls (Heim etal. 2001). The combined dexamethasone/corticotrophin-releasing hormone (Dex/CRH) test is a useful tool to study HPA axis sensitivity. One study found that abused men with current major depression demonstrated augmented ACTH and cortisol responses to the Dex/CRH test (Heim etal. 2008). Another study corroborated these increased cortisol levels in response to the Dex/CRH test in adults who had lost a parent before the age of 18 years. However,no effect of parental loss was present for ACTH responses (Tyrka etal.2008). In another study, adults with a history of childhood maltreatment exhibited nonsignificant decreased ACTH and cortisol responses to a Dex/CRH test after adjusting for potential confounders (age, menopausal stage, body mass index, use of contraceptives) (Klaassens etal. 2009). In contrast to the aforementioned findings for cortisol levels, Carpenter etal. (2009) found attenuated cortisol responses to the Dex/CRH test in adults who had been emotionally abused as a child. These contradicting results might be partly explained by the type or severity of early life stress, as well as by specific genetic factors. As for the latter, a protective CRHR1 receptor polymorphism in males has been found, i.e., maltreated men with this polymorphism showed low cortisol responses to the Dex/CRH test (Heim etal. 2009). These preliminary findings indicate generally low basal HPA axis activity, accompanied by altered HPA axis reactivity. Whether hyper- or hyporeactivity is predominant appears to be partly determined by the type of HPA axis stimulation, genetic factors, or comorbidity with a psychological disorder.
Effects on temperament and behavior. Using a longitudinal design, childhood maltreatment was found to be associated with internal and external problems in early adulthood (Silverman etal. 1996). One cross-sectional study found that adults, who were maltreated as child, were at high risk of showing antisocial behavior and conduct disorders (Nelson etal. 2002). This was corroborated in two longitudinal studies which found that young adults reporting early life stress, such as parental separation or childhood maltreatment, had an increased rate of antisocial behavior (Schilling etal. 2007; Thornberry etal. 2010). There is some evidence for genetic contributions that seem to moderate the association between childhood maltreatment and antisocial behavior. In particular, several studies found a moderating role of the polymorphism in the monoamine oxidase (MAO)-A gene, which encodes the activity of the enzyme MAO-A, which in turn metabolizes monoamines, i.e., neurotransmitters such as norepinephrine, serotonin, and dopamine. It was found that men who had a history of childhood maltreatment and a polymorphism associated with low MAO-A activity showed an increased risk for antisocial behavior compared to maltreated men with high MAO-A activity (Beach etal. 2010a, b; Caspi etal. 2002). These studies suggest that early life stress may increase the risk for behavioral problems in adulthood.
Effects on cognitive functioning and academic performance. One study showed that adults with a history of childhood abuse had greater verbal short-term memory deficits than controls but no difference was present in intelligence scores (Bremner etal. 1995). In accordance to this finding, two other studies found impaired memory, particularly affecting the verbal domain, in adults who reported childhood maltreatment, whereas no negative effect on attention or executive function (e.g., reasoning or planning abilities) was observed (Majer etal. 2010; Navalta etal. 2006). These studies indicate memory impairment in adults who experienced early life stress.
Effects on somatic health. Emotional abuse was found to be related to obesity in men but not in women (Gunstad etal. 2006). A representative survey found that adults, who retrospectively reported childhood physical abuse, showed a higher risk for having a heart disease than non-abused adults (Fuller-Thomson etal. 2010). Inanother study, adults with a history of multiple adverse childhood experiences (such as abuse, domestic violence, criminal familial environment) were more likely to be hospitalized due to autoimmune disease compared to adults without such a history (Dube etal. 2009). One study found that women with a history of childhood sexual abuse were more likely to experience chronic fatigue, asthma, or cardiovascular problems than female controls, whereas physically abused women suffered more from chronic pain compared to controls (Romans etal. 2002). In accordance with the aforementioned finding, physically abused women were found to suffer more from chronic pelvic pain and chronic low-back pain, and women with severe sexual abuse during childhood reported chronic pelvic pain more often compared to controls (Lampe etal. 2003). Two studies provide some evidence for positive associations between childhood maltreatment experiences and heightened risk of chronic fatigue syndrome (Heim etal. 2009; Heim etal. 2006). Another study reported that emotional abuse or physical neglect raised the risk for chronic fatigue syndrome, irritable bowel syndrome, fibromyalgia, and arthritis (Tietjen etal. 2010). In summary, early life stress appears to increase the risk for negative health outcomes later in life.
Effects on psychological health. There are several studies which investigated the association between early life stress and the occurrence of psychological disorders based on clinical diagnoses according to international classification systems, such as the ICD-10 (WHO 2004) or the DSM-IV (APA 2000). For example, one epidemiological survey found that early life stress increased the risk for psychological disorders in adulthood, including affective, anxiety, or substance disorders (Fujiwara etal. 2011). Another study found that both childhood sexual or physical abuse was related to an increased risk for depression, PTSD, alcohol, or drug dependence (Silverman etal. 1996). Another study reported similar findings, with self-reported childhood sexual abuse being associated with depression, general anxiety disorder, panic disorder, alcohol, or drug dependence (Kendler etal. 2000). Children and adolescents (age range, 6–18 years) who had lost their father during the Kosovo War were more likely to show major depression, posttraumatic disorder, or panic disorder 10 years later compared to peers with similar war experiences but no father loss (Morina etal. 2011). Two other studies found more symptoms of personality disorders in adults who retrospectively reported childhood maltreatment compared to non-maltreated adults (Johnson etal. 1999; Tyrka etal. 2009). One study found that adults, who retrospectively reported childhood sexual abuse, were at high risk to develop depression (Kendler etal. 2004). Another study showed that depressive patients reported the loss of the mother before the age of 17 years more often than controls; this negative effect was not found for the loss of the father (Kunugi etal. 1995). Another study found that men with alcohol dependence did not differ from controls with regard to parental loss before the age of 16 (Furukawa etal. 1998).
Numerous studies operationalized psychological health by assessing psychological symptoms (e.g., depression or anxiety levels measured by questionnaires) or high-risk behavior (e.g., regular use of drugs), which do not meet fully the criteria of psychological disorders. However, these psychological symptoms and risk behavior may give indications for psychological well-being or risk to develop a psychological disorder in the future. One cross-sectional study (Nelson etal. 2002) and two longitudinal studies (Schilling etal. 2007; Thornberry etal. 2010) found an increased rate of drug use in young adults when they had experienced early life stress. Thoughts and attempts of suicide can be seen as indicators for depression. Several studies found that the risk for suicide thoughts or attempts was dramatically increased in adults with a history of early life stress (Dube etal. 2001; Thornberry etal. 2010). A great number of studies give evidence that early life stress, in particular childhood maltreatment or parental loss, may be associated with increased levels of depression (Kiecolt-Glaser etal. 2011; Lampe etal. 2003; Nelson etal. 2002; Wingo etal. 2010), or both anxiety and depression (Handa etal. 2008; Heim etal. 2009; Heim etal. 2006; Nicolson etal. 2010; Tyrka etal. 2008). Summarizing these findings, early life stress seems to be associated with an increased risk for psychiatric disorders in adulthood.
To summarize the findings, early life stress may be associated with reduced volumes of memory-related brain regions (e.g., hippocampus). Adults, who retrospectively reported early life stress, appear to have decreased levels of oxytocin, which is known to be related to bonding. Low morning cortisol levels seem to be characteristic for adults with a history of early life stress. Early life stress may be associated with altered HPA axis responses to psychological stressors and to pharmacological challenge tests. Further, early life stress may be related to behavioral problems, such as antisocial behavior. The findings suggest that early life stress may influence negatively memory, particularly of verbal material. Adults with a history of early life stress may be more likely to suffer from negative somatic health outcomes (e.g., pain disorder, chronic fatigue) or negative psychological health outcomes (e.g., depression, anxiety, drug abuse, personality disorders) later in life.
2.3 Adaptive Outcomes Measured in Children, Adolescents, and Adults
The previous sections have delineated maladaptive outcomes of early life stress, which is the traditional focus in human research. Adaptive consequences of early life stress are often disregarded in human research. In the last years, however, researchers have started to investigate adaptive outcomes of developmental stress, which will be briefly summarized in the next paragraphs.
There are three concepts that may provide approaches to study adaptive consequences of early life stress: (a) resilience, (b) compensating factors to reduce negative effects of stress, and (c) posttraumatic growth (PTG); we present findings particularly for the latter concept, since PTG comes most closely to the idea of adaptive consequences.
Resilience in this context describes the individual’s ability to develop in a normal range despite experienced early life stress (see also review, Feder etal. 2009). Aresilient individual may have genetic dispositions (protective polymorphism; see also conclusion) which protect the individual from negative consequences of adverse conditions. Also adaptive coping styles (e.g., cognitive reappraisal) and resilient personality factors (e.g., dispositional optimism) may characterize resilience in an individual.
Individuals who are vulnerable to stress and consequently are at high risk for maladaptive consequences due to early life stress may benefit from compensating factors. These include various types of social support (e.g., instrumental, emotional support) and a supportive social network which may buffer the stress impact (Ozbay etal. 2007). Interventions such as educational support, assistance in academic performance (tutoring, coaching), and public health programs may diminish developmental deficits and enable a “normal development” of the infant.
Finally, the concept of PTG proposes possible positive effects resulting from experiencing and overcoming an adverse event (see review, Meyerson etal. 2011). According to the theory of PTG, an adverse event (trauma) may change the individuals’ concept of themselves, relationship to others, and life in general. The Posttraumatic Growth Inventory (PTGI) has been developed to assess PTG; there are separate versions for both children and adults (Kilmer etal. 2009; Tedeschi and Calhoun 1996). This scale comprises 5 domains, which may be changed due to trauma experience: (1) perception of self/personal strength (e.g., being stronger than individual thought), (2) relationship to others (e.g., closer relationship to family and friends), (3) appreciation of life (e.g., changing priorities), (4) new possibilities (e.g., new interests and goals), and (5) spiritual change (e.g., strengthening faith). There is some evidence that children who have experienced a traumatic event (e.g., injury or death of a loved one, natural disaster, or accident) report higher PTG scores than children without such trauma experiences (Alisic etal. 2008). The PTGI has been successfully administered in children and adults of various types of early life stress: cancer during childhood or adolescence (Barakat etal. 2006; Kamibeppu etal. 2010; Turner-Sack etal. 2012), serious or chronic illness in childhood (Devine etal. 2010), death of a close one (Ho etal. 2008; Wolchik etal. 2008), traffic accidents (Salter and Stallard 2004), childhood sexual abuse (Shakespeare-Finch and de Dassel 2009), terror exposure (Laufer etal. 2010; Levine etal. 2008; Levine etal. 2009), or exposure to natural disasters (Cryder etal. 2006; Hafstad etal. 2010; Kilmer and Gil-Rivas 2010; Yu etal. 2010).
This research trend contributes to the understanding of a comprehensive picture of early life stress and its consequences on the individual and may further provide applicable interventions and treatments for individuals who suffered from early life stress.
3 Integration and Interpretation of the Findings
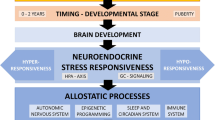
The findings presented in this book chapter suggest that stress experienced during childhood or adolescence may have long-lasting consequences long after exposure to adversity has stopped. We propose a psychobiological stress model that is used to integrate and interpret some of these findings (Fig.7.1).
It is established that the human brain is particularly sensitive to stress in early life due to the heightened brain plasticity during this vulnerable period. The HPA axis appears to be involved in such functional and structural brain alterations. Specifically, there are several brain regions with a particularly high density of glucocorticoid receptors (GR), e.g., hippocampus and prefrontal cortex, both of which regulate HPA axis activity by negative feedback mechanisms. Consequently, excessive cortisol secretion due to early life stress may result in persistent altered GR sensitivity in these brain regions. This in turn may lead to functional impairments, particularly when the individual is exposed to stress and thus to glucocorticoids. Impaired memory, which was found in both children and adults with a history of early life stress, may be related to such altered GR sensitivity of the memory-associated brain regions (e.g., hippocampus and prefrontal cortex). Animal models showed that glucocorticoid overexposure may have neurotoxic effects, particularly in the CA3 region of the hippocampus, which may explain decreased hippocampal volumes as the result of excessive stress.
Specifically, the findings summarized above suggest that early life stress may negatively affect HPA axis activity and reactivity. Heim and colleagues (Heim etal. 2008) hypothesized that early life stress may lead to the development of highly sensitive stress-responsive systems which is indicated by increased psychological and biological responses to adulthood stress, accompanied by alterations in HPA axis regulation. In particular, the authors suggest that increased ACTH secretion, as it has been observed in the literature, may be mediated by downregulation of pituitary CRH receptors and impaired negative feedback regulation of the HPA axis under stimulation.
Early life stress was found to negatively affect both somatic and psychological health which has been also largely demonstrated in studies using animal model (Cirulli etal. 2009; O’Mahony etal. 2009; Schmidt etal. 2011). There is sufficient evidence that diseases or psychological disorders are related to hypo- or hyperactivity of the HPA axis as a result of stress (Chrousos 2009). Since the stress axes and the immune system interact (e.g., cortisol has suppressive or stimulating effects on immune processes), dysregulation of the HPA axis may contribute to the development and maintenance of conditions in which the immune system plays an important pathophysiological role. There are a handful of studies which investigated the association between early life stress and inflammation, which is an approach to study immune activity. These studies found increased levels of inflammatory markers. For example, adults who had been maltreated as child showed higher basal interleukin-6 levels and higher tumor necrosis factor (TNF)-alpha levels than controls (Kiecolt-Glaser etal. 2011) as well as increased IL-6 responses to a psychological stressor (Carpenter etal. 2010), indicating increased inflammation. Alongitudinal study found that childhood maltreatment was associated with increased C-reactive protein (CRP) levels in adults, indicating systemic inflammation (Danese etal. 2009). A persistent inflammation may increase the risk for developing diseases later in life; for example, high CRP levels appear to be related to cardiovascular diseases and high TNF-alpha levels to rheumatoid arthritis.
Besides the aforementioned negative outcomes of early life stress, there is also some evidence for adaptive consequences of stress. Some individuals report that the evaluation of themselves, relationships to others, and life in general has changed in a positive direction after the exposure of a traumatic event, which in essence describes the idea of PTG. Early life stress may thus force individuals to reappraise and reorganize general concept of themselves, their world, and the future. There is some evidence that expected maladaptive consequences of early life stress may be alleviated or even inhibited by an individual’s protective factors (resilience) or compensating factors ( e.g., social support), which have biological correlates and pathways (Feder etal. 2009). These protective and compensating factors may act as a moderating role in the association between early life stress and (mal)adaptive outcomes.
This psychobiological stress model proposes a biological link between stress and (mal)adaptive consequences. The brain plays a central role in this model. Both maladaptive and adaptive consequences may result from brain plasticity and active cognitive reappraisals.
4 Conclusion
This book chapter summarized the main findings of the last few decades of research on the impact of early life stress in humans. However, despite the relative abundance of studies, some questions which should be addressed in future studies remain. Inthe following, we want to give an outlook on current research and future directions by addressing still insufficiently studied research questions.
4.1 Underlying Biological Mechanisms for Associations Between Early Life Stress and Negative Outcomes
There is great evidence that early life stress has extensive negative effects on mind and body. These negative effects are probably partly mediated by alterations of the HPA axis, one of the most prominent and most studied stress-responsive systems. However, this mediation cannot explain all findings; thus, other stress-responsive systems and factors should be taken into account in order to understand the underlying mechanisms of how early life stress may lead to negative outcomes. To the best of our knowledge, the association between early life stress and the SAM system has been scarcely investigated. However, it is known that alteration of both the HPA axis and the SAM system may increase the risk for diseases (McEwen and Seeman 1999). Thus, further research is needed incorporating assessment of both the HPA axis and the SAM system. Another stress-responsive system, which is scarcely investigated with regard to early life stress, is the above-mentioned immune system. First studies, however, suggest that early life stress may be associated with enhanced inflammation, indicating an enhanced activity of the immune system. Clearly, the intricate relationships between stress axis and the immune system should be considered in future studies in order to further elucidate the detrimental effects of early life stress on both psychological and somatic health.
4.2 Risk and Protective Factors (Vulnerability and Resilience)
In general, the presented studies in this chapter suggest that early life stress may increase the likelihood or risk for negative outcomes, but not that early life stress per se inevitably leads to negative outcomes. It is conceivable that stress may play a triggering role, and that stress and other potential risk factors (vulnerability), but also protective factors (resilience) may be relevant in whether negative health outcomes manifest or not. There is some evidence from the literature for polymorphisms that might act as risk or protective factors. However, these alone cannot explain the variance of stress-related morbidity. We should not neglect the role of environmental risk and protective factors, e.g., social support may have a buffering effect and can be used as basis for intervention programs. Future studies are required to investigate interindividual differences by considering possible risk and protective factors. These vulnerability factors may help to identify high-risk individuals, who may benefit from prevention and intervention programs.
4.3 Epigenetics
There is some evidence that early life stress may interact with genetic predispositions for psychological conditions. For example, one study found that certain glucocorticoid receptor gene polymorphisms in interaction with occurrence of early life stress may increase the risk of (recurrent) depression symptoms (Bet etal. 2009). The severity of depression was found to be influenced by an interaction of childhood maltreatment with a CRHR1 polymorphism (Bradley etal. 2008; Heim etal.2009) or with a polymorphism associated with high MAO-A activity (Beach etal. 2010). A similar gene x environment interaction was found for PTSD symptoms, with the severity of PTSD symptoms being predicted by the FKBP5 polymorphism and childhood maltreatment (Binder etal. 2008). These polymorphisms are related to monoamine metabolism (e.g., catecholamine, serotonin) or glucocorticoid receptors. There is some evidence that these neurotransmitters (e.g., serotonin) and hormones (e.g., cortisol) may be at least partly involved in the development and manifestation of psychological conditions.
It is well established that the environment can alter gene activity; this can be seen by the extent of DNA methylation. The extent of DNA methylation in the promoter regions of genes regulates gene expression and thus the transcription activity of genes, i.e., the synthesis of gene products, mostly proteins. Protein synthesis is fundamental and essential for body functioning. Studies have shown that childhood abuse was associated with overall methylation at the SLC6A4 region (Beach etal.2010) and methylation at the 5HTT promoter region (Beach etal. 2011); in these studies, the extent of methylation was suggested to influence the synthesis of the serotonin transporter, which in turn plays an important role in the pathophysiology of depression.
Another genetic marker, which appears to be influenced by environmental conditions, is the shortening of telomere length. As a marker of cellular aging, shortening of telomere length is suggested to be indicative for biological aging, and to be a risk factor for early onset of a variety of age-related diseases. Telomeres, i.e., repetitive DNA sequences at the end of chromosomes, shorten with age due to repetitive cell divisions. The shortening of telomeres appears to be promoted by unhealthy lifestyle (e.g., smoking) or stress and thus may constitute a possible stress-sensitive marker (Tyrka etal. 2010). One study found that the time reared in institutional care before adoption was associated with shorter telomere length in children aged 4–10 years, indicating telomere length shortening due to early life stress (Drury etal. 2011). Further evidence for telomere shortening due to early life stress was provided by several cross-sectional studies which were conducted in adult samples (Kananen etal. 2010; Kiecolt-Glaser etal. 2011; O’Donovan etal. 2011; Tyrka etal. 2010).
Clearly, these studies, still small in number, hold great promise of elucidating mechanisms that translate early life stress into negative outcomes later in life by studying biological systems on a molecular level.
4.4 The Type of Early Life Stress
In this book chapter we introduced early life stress as a concept that encompasses various stressful events, such as childhood maltreatment (i.e., forms of abuse and neglect), parental loss, and traumatic events (e.g., war). Thus, we cannot give any differentiated conclusion about the impact of a single type of early life stress. Itseems plausible that the type of early life stress may be a determining factor. Toour knowledge, there is a lack of comparison studies, e.g., comparison among the type of childhood maltreatment (emotional neglect, physical neglect, emotional abuse, physical abuse, and sexual abuse). One reason for this lack of such studies might be that individuals often experienced several types (physical and sexual abuse); thus, a more precise analysis may not be possible. Other factors, such as the number, severity, and duration of such adverse events, possibly have additional effects and thus should be assessed in future studies.
In summary, stress throughout the entire life span seems to be associated with negative consequences for health and well-being. Future research is needed to explore possible risk factors, protective factors, and the underlying biological mechanisms between the association of stress and health. This research will hopefully advance the development of appropriate interventions.
References
Alisic E, van der Schoot TA, van Ginkel JR, Kleber RJ (2008) Looking beyond posttraumatic stress disorder in children: posttraumatic stress reactions, posttraumatic growth, and quality of life in a general population sample. J Clin Psychiatry 69(9):1455–1461
APA (2000) Diagnostic statistical manual of mental disorders (Revised 4th edition). Author, Washington, DC
Barakat LP, Alderfer MA, Kazak AE (2006) Posttraumatic growth in adolescent survivors of cancer and their mothers and fathers. J Pediatr Psychol 31(4):413–419
Beach SR, Brody GH, Gunter TD, Packer H, Wernett P, Philibert RA (2010a) Child maltreatment moderates the association of MAOA with symptoms of depression and antisocial personality disorder. J Fam Psychol 24(1):12–20
Beach SR, Brody GH, Todorov AA, Gunter TD, Philibert RA (2010b) Methylation at SLC6A4 is linked to family history of child abuse: an examination of the Iowa adoptee sample. Am J Med Genet B Neuropsychiatr Genet 153B(2):710–713
Beach SR, Brody GH, Todorov AA, Gunter TD, Philibert RA (2011) Methylation at 5HTT mediates the impact of child sex abuse on women's antisocial behavior: an examination of the Iowa adoptee sample. Psychosom Med 73(1):83–87
Bernard K, Butzin-Dozier Z, Rittenhouse J, Dozier M (2010) Cortisol production patterns in young children living with birth parents vs children placed in foster care following involvement of Child Protective Services. Arch Pediatr Adolesc Med 164(5):438–443
Bet PM, Penninx BWJH, Bochdanovits Z, Uitterlinden AG, Beekman ATF, van Schoor NM etal (2009) Glucocorticoid receptor gene polymorphisms and childhood adversity are associated with depression: new evidence for a gene-environment interaction. Am J Med Genet B Neuropsychiatr Genet 150B(5):660–669
Binder EB, Bradley RG, Liu W, Epstein MP, Deveau TC, Mercer KB etal (2008) Association of FKBP5 polymorphisms and childhood abuse with risk of posttraumatic stress disorder symptoms in adults. JAMA 299(11):1291–1305
Bos KJ, Fox N, Zeanah CH, Nelson CA (2009) Effects of early psychosocial deprivation on the development of memory and executive function. Front Behav Neurosci 3:16
Bradley RG, Binder EB, Epstein MP, Tang Y, Nair HP, Liu W etal (2008) Influence of child abuse on adult depression: moderation by the corticotropin-releasing hormone receptor gene. Arch Gen Psychiatry 65(2):190–200
Bremner JD, Randall P, Scott TM, Capelli S, Delaney R, McCarthy G etal (1995) Deficits in short-term memory in adult survivors of childhood abuse. Psychiatry Res 59(1–2):97–107
Bremner JD, Narayan M, Staib LH, Southwick SM, McGlashan T, Charney DS (1999) Neural correlates of memories of childhood sexual abuse in women with and without posttraumatic stress disorder. Am J Psychiatry 156(11):1787–1795
Carpenter LL, Tyrka AR, Ross NS, Khoury L, Anderson GM, Price LH (2009) Effect of childhood emotional abuse and age on cortisol responsivity in adulthood. Biol Psychiatry 66(1):69–75
Carpenter LL, Gawuga CE, Tyrka AR, Lee JK, Anderson GM, Price LH (2010) Association between plasma IL-6 response to acute stress and early-life adversity in healthy adults. Neuropsychopharmacology 35(13):2617–2623
Caspi A, McClay J, Moffitt TE, Mill J, Martin J, Craig IW etal (2002) Role of genotype in the cycle of violence in maltreated children. Science 297(5582):851–854
Chandy JM, Blum RW, Resnick MD (1996) Gender-specific outcomes for sexually abused adolescents. Child Abuse Negl 20(12):1219–1231
Chisholm K (1998) A three year follow-up of attachment and indiscriminate friendliness in children adopted from Romanian orphanages. Child Dev 69(4):1092–1106
Chrousos GP (2009) Stress and disorders of the stress system. Nat Rev Endocrinol 5(7):374–381
Chugani HT, Behen ME, Muzik O, Juhasz C, Nagy F, Chugani DC (2001) Local brain functional activity following early deprivation: a study of postinstitutionalized Romanian orphans. Neuroimage 14(6):1290–1301
Cicchetti D, Rogosch FA (2001) Diverse patterns of neuroendocrine activity in maltreated children. Dev Psychopathol 13(3):677–693
Cirulli F, Francia N, Berry A, Aloe L, Alleva E, Suomi SJ (2009) Early life stress as a risk factor for mental health: role of neurotrophins from rodents to non-human primates. Neurosci Biobehav Rev 33(4):573–585
Cohen RA, Grieve S, Hoth KF, Paul RH, Sweet L, Tate D etal (2006) Early life stress and morphometry of the adult anterior cingulate cortex and caudate nuclei. Biol Psychiatry 59(10):975–982
Cryder CH, Kilmer RP, Tedeschi RG, Calhoun LG (2006) An exploratory study of posttraumatic growth in children following a natural disaster. Am J Orthopsychiatry 76(1):65–69
Danese A, Moffitt TE, Harrington H, Milne BJ, Polanczyk G, Pariante CM etal (2009) Adverse childhood experiences and adult risk factors for age-related disease: depression, inflammation, and clustering of metabolic risk markers. Arch Pediatr Adolesc Med 163(12):1135–1143
De Bellis MD, Hall J, Boring AM, Frustaci K, Moritz G (2001) A pilot longitudinal study of hippocampal volumes in pediatric maltreatment-related posttraumatic stress disorder. Biol Psychiatry 50(4):305–309
De Bellis MD, Keshavan MS, Shifflett H, Iyengar S, Beers SR, Hall J etal (2002) Brain structures in pediatric maltreatment-related posttraumatic stress disorder: a sociodemographically matched study. Biol Psychiatry 52(11):1066–1078
Devine KA, Reed-Knight B, Loiselle KA, Fenton N, Blount RL (2010) Posttraumatic growth in young adults who experienced serious childhood illness: a mixed-methods approach. J Clin Psychol Med Settings 17(4):340–348
Ditzen B, Nater UM, Heim C (2013) HPA axis challenge tests. In: Gellman MD, Turner JR (eds) Encyclopedia of behavioral medicine, Springer, Berlin pp. 1468–1471
Drury SS, Theall K, Gleason MM, Smyke AT, De Vivo I, Wong JY etal (2011) Telomere length and early severe social deprivation: linking early adversity and cellular aging. Mol Psychiatry 17(7):719–27
Dube SR, Anda RF, Felitti VJ, Chapman DP, Williamson DF, Giles WH (2001) Childhood abuse, household dysfunction, and the risk of attempted suicide throughout the life span: findings from the Adverse Childhood Experiences Study. JAMA 286(24):3089–3096
Dube SR, Fairweather D, Pearson WS, Felitti VJ, Anda RF, Croft JB (2009) Cumulative childhood stress and autoimmune diseases in adults. Psychosom Med 71(2):243–250
Elzinga BM, Schmahl CG, Vermetten E, van Dyck R, Bremner JD (2003) Higher cortisol levels following exposure to traumatic reminders in abuse-related PTSD. Neuropsychopharmacology 28(9):1656–1665
Elzinga BM, Roelofs K, Tollenaar MS, Bakvis P, van Pelt J, Spinhoven P (2008) Diminished cortisol responses to psychosocial stress associated with lifetime adverse events a study among healthy young subjects. Psychoneuroendocrinology 33(2):227–237
Feder A, Nestler EJ, Charney DS (2009) Psychobiology and molecular genetics of resilience. Nat Rev Neurosci 10(6):446–457
Flaherty EG, Thompson R, Litrownik AJ, Theodore A, English DJ, Black MM etal (2006) Effect of early childhood adversity on child health. Arch Pediatr Adolesc Med 160(12):1232–1238
Fujiwara T, Kawakami N, World Mental Health Japan Survey Group (2011) Association of childhood adversities with the first onset of mental disorders in Japan: results from the World Mental Health Japan, 2002–2004. J Psychiatr Res 45(4):481–487
Fuller-Thomson E, Brennenstuhl S, Frank J (2010) The association between childhood physical abuse and heart disease in adulthood: findings from a representative community sample. Child Abuse Negl 34(9):689–698
Furukawa T, Harai H, Hirai T, Fujihara S, Kitamura T, Takahashi K (1998) Childhood parental loss and alcohol dependence among Japanese men: a case–control study. Group for Longitudinal Affective Disorders Study (GLADS). Acta Psychiatr Scand 97(6):403–407
Gunnar MR, Morison SJ, Chisholm K, Schuder M (2001) Salivary cortisol levels in children adopted from Romanian orphanages. Dev Psychopathol 13(3):611–628
Gunstad J, Paul RH, Spitznagel MB, Cohen RA, Williams LM, Kohn M etal (2006) Exposure to early life trauma is associated with adult obesity. Psychiatry Res 142(1):31–37
Hafstad GS, Gil-Rivas V, Kilmer RP, Raeder S (2010) Parental adjustment, family functioning, and posttraumatic growth among Norwegian children and adolescents following a natural disaster. Am J Orthopsychiatry 80(2):248–257
Handa M, Nukina H, Hosoi M, Kubo C (2008) Childhood physical abuse in outpatients with psychosomatic symptoms. Biopsychosoc Med 2:8
Hanson JL, Chung MK, Avants BB, Shirtcliff EA, Gee JC, Davidson RJ etal (2010) Early stress is associated with alterations in the orbitofrontal cortex: a tensor-based morphometry investigation of brain structure and behavioral risk. J Neurosci 30(22):7466–7472
Heim C, Newport DJ, Heit S, Graham YP, Wilcox M, Bonsall R etal (2000) Pituitary-adrenal and autonomic responses to stress in women after sexual and physical abuse in childhood. JAMA 284(5):592–597
Heim C, Newport DJ, Bonsall R, Miller AH, Nemeroff CB (2001) Altered pituitary-adrenal axis responses to provocative challenge tests in adult survivors of childhood abuse. Am J Psychiatry 158(4):575–581
Heim C, Newport DJ, Wagner D, Wilcox MM, Miller AH, Nemeroff CB (2002) The role of early adverse experience and adulthood stress in the prediction of neuroendocrine stress reactivity in women: a multiple regression analysis. Depress Anxiety 15(3):117–125
Heim C, Wagner D, Maloney E, Papanicolaou DA, Solomon L, Jones JF etal (2006) Early adverse experience and risk for chronic fatigue syndrome: results from a population-based study. Arch Gen Psychiatry 63(11):1258–1266
Heim C, Mletzko T, Purselle D, Musselman DL, Nemeroff CB (2008a) The dexamethasone/corticotropin-releasing factor test in men with major depression: role of childhood trauma. Biol Psychiatry 63(4):398–405
Heim C, Newport DJ, Mletzko T, Miller AH, Nemeroff CB (2008b) The link between childhood trauma and depression: insights from HPA axis studies in humans. Psychoneuroendocrinology 33(6):693–710
Heim C, Bradley B, Mletzko TC, Deveau TC, Musselman DL, Nemeroff CB etal (2009a) Effect of childhood trauma on adult depression and neuroendocrine function: sex-specific moderation by CRH receptor 1 gene. Front Behav Neurosci 3:41
Heim C, Nater UM, Maloney E, Boneva R, Jones JF, Reeves WC (2009b) Childhood trauma and risk for chronic fatigue syndrome: association with neuroendocrine dysfunction. Arch Gen Psychiatry 66(1):72–80
Heim C, Young LJ, Newport DJ, Mletzko T, Miller AH, Nemeroff CB (2009c) Lower CSF oxytocin concentrations in women with a history of childhood abuse. Mol Psychiatry 14(10):954–958
Ho SM, Chu KW, Yiu J (2008) The relationship between explanatory style and posttraumatic growth after bereavement in a non-clinical sample. Death Stud 32(5):461–478
Huey SJ Jr, Weisz JR (1997) Ego control, ego resiliency, and the five-factor model as predictors of behavioral and emotional problems in clinic-referred children and adolescents. J Abnorm Psychol 106(3):404–415
Johnson JG, Cohen P, Brown J, Smailes EM, Bernstein DP (1999) Childhood maltreatment increases risk for personality disorders during early adulthood. Arch Gen Psychiatry 56(7):600–606
Jouriles EN, Barling J, Oleary KD (1987) Predicting child-behavior problems in maritally violent families. J Abnorm Child Psychol 15(2):165–173
Kamibeppu K, Sato I, Honda M, Ozono S, Sakamoto N, Iwai T etal (2010) Mental health among young adult survivors of childhood cancer and their siblings including posttraumatic growth. J Cancer Surviv 4(4):303–312
Kananen L, Surakka I, Pirkola S, Suvisaari J, Lonnqvist J, Peltonen L etal (2010) Childhood adversities are associated with shorter telomere length at adult age both in individuals with an anxiety disorder and controls. PLoS One 5(5):e10826
Kaufman J, Birmaher B, Perel J, Dahl RE, Moreci P, Nelson B etal (1997) The corticotropin-releasing hormone challenge in depressed abused, depressed nonabused, and normal control children. Biol Psychiatry 42(8):669–679
Kendler KS, Bulik CM, Silberg J, Hettema JM, Myers J, Prescott CA (2000) Childhood sexual abuse and adult psychiatric and substance use disorders in women: an epidemiological and cotwin control analysis. Arch Gen Psychiatry 57(10):953–959
Kendler KS, Kuhn JW, Prescott CA (2004) Childhood sexual abuse, stressful life events and risk for major depression in women. Psychol Med 34(8):1475–1482
Kiecolt-Glaser JK, Gouin JP, Weng NP, Malarkey WB, Beversdorf DQ, Glaser R (2011) Childhood adversity heightens the impact of later-life caregiving stress on telomere length and inflammation. Psychosom Med 73(1):16–22
Kilmer RP, Gil-Rivas V (2010) Exploring posttraumatic growth in children impacted by Hurricane Katrina: correlates of the phenomenon and developmental considerations. Child Dev 81(4):1211–1227
Kilmer RP, Gil-Rivas V, Tedeschi RG, Cann A, Calhoun LG, Buchanan T etal (2009) Use of the revised posttraumatic growth inventory for children. J Trauma Stress 22(3):248–253
Kim J, Cicchetti D, Rogosch FA, Manly JT (2009) Child maltreatment and trajectories of personality and behavioral functioning: implications for the development of personality disorder. Dev Psychopathol 21(3):889–912
King JA, Mandansky D, King S, Fletcher KE, Brewer J (2001) Early sexual abuse and low cortisol. Psychiatry Clin Neurosci 55(1):71–74
Klaassens ER, van Noorden MS, Giltay EJ, van Pelt J, van Veen T, Zitman FG (2009) Effects of childhood trauma on HPA-axis reactivity in women free of lifetime psychopathology. Prog Neuropsychopharmacol Biol Psychiatry 33(5):889–894
Kudielka BM, Wust S (2010) Human models in acute and chronic stress: assessing determinants of individual hypothalamus-pituitary-adrenal axis activity and reactivity. Stress 13(1):1–14
Kunugi H, Sugawara N, Aoki H, Nanko S, Hirose T, Kazamatsuri H (1995) Early parental loss and depressive disorder in Japan. Eur Arch Psychiatry Clin Neurosci 245(2):109–113
Lampe A, Doering S, Rumpold G, Solder E, Krismer M, Kantner-Rumplmair W etal (2003) Chronic pain syndromes and their relation to childhood abuse and stressful life events. J Psychosom Res 54(4):361–367
Lanier P, Jonson-Reid M, Stahlschmidt MJ, Drake B, Constantino J (2010) Child maltreatment and pediatric health outcomes: a longitudinal study of low-income children. J Pediatr Psychol 35(5):511–522
Lansford JE, Miller-Johnson S, Berlin LJ, Dodge KA, Bates JE, Pettit GS (2007) Early physical abuse and later violent delinquency: a prospective longitudinal study. Child Maltreat 12(3):233–245
Laufer A, Solomon Z, Levine SZ (2010) Elaboration on posttraumatic growth in youth exposed to terror: the role of religiosity and political ideology. Soc Psychiatry Psychiatr Epidemiol 45(6):647–653
Letzring TD, Block J, Funder DC (2005) Ego-control and ego-resiliency: generalization of self-report scales based on personality descriptions from acquaintances, clinicians, and the self. J Res Pers 39(4):395–422
Levine SZ, Laufer A, Hamama-Raz Y, Stein E, Solomon Z (2008) Posttraumatic growth in adolescence: examining its components and relationship with PTSD. J Trauma Stress 21(5):492–496
Levine SZ, Laufer A, Stein E, Hamama-Raz Y, Solomon Z (2009) Examining the relationship between resilience and posttraumatic growth. J Trauma Stress 22(4):282–286
Lovallo WR, Farag NH, Sorocco KH, Cohoon AJ, Vincent AS (2012) Lifetime adversity leads to blunted stress axis reactivity: studies from the oklahoma family health patterns project. Biol Psychiatry 71(4):344–349
Luecken LJ (1998) Childhood attachment and loss experiences affect adult cardiovascular and cortisol function. Psychosom Med 60(6):765–772
MacMillan HL, Georgiades K, Duku EK, Shea A, Steiner M, Niec A etal (2009) Cortisol response to stress in female youths exposed to childhood maltreatment: results of the youth mood project. Biol Psychiatry 66(1):62–68
Maikovich-Fong AK, Jaffee SR (2010) Sex differences in childhood sexual abuse characteristics and victims' emotional and behavioral problems: findings from a national sample of youth. Child Abuse Negl 34(6):429–437
Majer M, Nater UM, Lin JM, Capuron L, Reeves WC (2010) Association of childhood trauma with cognitive function in healthy adults: a pilot study. BMC Neurol 10:61
McEwen BS, Seeman T (1999) Protective and damaging effects of mediators of stress. Elaborating and testing the concepts of allostasis and allostatic load. Ann N Y Acad Sci 896:30–47
Meinlschmidt G, Heim C (2005) Decreased cortisol awakening response after early loss experience. Psychoneuroendocrinology 30(6):568–576
Meyerson DA, Grant KE, Carter JS, Kilmer RP (2011) Posttraumatic growth among children and adolescents: a systematic review. Clin Psychol Rev 31(6):949–964
Mills R, Alati R, O'Callaghan M, Najman JM, Williams GM, Bor W etal (2011) Child abuse and neglect and cognitive function at 14 years of age: findings from a birth cohort. Pediatrics 127(1):4–10
Moran P, Coffey C, Chanen A, Mann A, Carlin JB, Patton GC (2011) Childhood sexual abuse and abnormal personality: a population-based study. Psychol Med 41(06):1311–1318
Morina N, von Lersner U, Prigerson HG (2011) War and bereavement: consequences for mental and physical distress. PLoS One 6(7):e22140
Navalta CP, Polcari A, Webster DM, Boghossian A, Teicher MH (2006) Effects of childhood sexual abuse on neuropsychological and cognitive function in college women. J Neuropsychiatry Clin Neurosci 18(1):45–53
Nelson EC, Heath AC, Madden PA, Cooper ML, Dinwiddie SH, Bucholz KK etal (2002) Association between self-reported childhood sexual abuse and adverse psychosocial outcomes: results from a twin study. Arch Gen Psychiatry 59(2):139–145
Nelson CA, Zeanah CH, Fox NA, Marshall PJ, Smyke AT, Guthrie D (2007) Cognitive recovery in socially deprived young children: the Bucharest Early Intervention Project. Science 318(5858):1937–1940
Newport DJ, Heim C, Bonsall R, Miller AH, Nemeroff CB (2004) Pituitary-adrenal responses to standard and low-dose dexamethasone suppression tests in adult survivors of child abuse. Biol Psychiatry 55(1):10–20
Nicolson NA, Davis MC, Kruszewski D, Zautra AJ (2010) Childhood maltreatment and diurnal cortisol patterns in women with chronic pain. Psychosom Med 72(5):471–480
O'Donovan A, Epel E, Lin J, Wolkowitz O, Cohen B, Maguen S etal (2011) Childhood trauma associated with short leukocyte telomere length in posttraumatic stress disorder. Biol Psychiatry 70(5):465–71
O'Mahony SM, Marchesi JR, Scully P, Codling C, Ceolho AM, Quigley EM etal (2009) Early life stress alters behavior, immunity, and microbiota in rats: implications for irritable bowel syndrome and psychiatric illnesses. Biol Psychiatry 65(3):263–267
Opacka-Juffry J, Mohiyeddini C (2011) Experience of stress in childhood negatively correlates with plasma oxytocin concentration in adult men. Stress 15(1):1–10
Ozbay F, Johnson DC, Dimoulas E, Morgan CA, Charney D, Southwick S (2007) Social support and resilience to stress: from neurobiology to clinical practice. Psychiatry (Edgmont) 4(5):35–40
Patton GC, Coffey C, Posterino M, Carlin JB, Bowes G (2003) Life events and early onset depression: cause or consequence? Psychol Med 33(7):1203–1210
Pollak SD, Nelson CA, Schlaak MF, Roeber BJ, Wewerka SS, Wiik KL etal (2010) Neurodevelopmental effects of early deprivation in postinstitutionalized children. Child Dev 81(1):224–236
Rao U, Hammen C, Ortiz LR, Chen LA, Poland RE (2008) Effects of early and recent adverse experiences on adrenal response to psychosocial stress in depressed adolescents. Biol Psychiatry 64(6):521–526
Romans S, Belaise C, Martin J, Morris E, Raffi A (2002) Childhood abuse and later medical disorders in women. An epidemiological study. Psychother Psychosom 71(3):141–150
Salter E, Stallard P (2004) Posttraumatic growth in child survivors of a road traffic accident. J Trauma Stress 17(4):335–340
Schilling EA, Aseltine RH Jr, Gore S (2007) Adverse childhood experiences and mental health in young adults: a longitudinal survey. BMC Public Health 7:30
Schmidt MV, Wang XD, Meijer OC (2011) Early life stress paradigms in rodents: potential animal models of depression? Psychopharmacology (Berl) 214(1):131–140
Shakespeare-Finch J, de Dassel T (2009) Exploring posttraumatic outcomes as a function of childhood sexual abuse. J Child Sex Abus 18(6):623–640
Silberg J, Rutter M, Neale M, Eaves L (2001) Genetic moderation of environmental risk for depression and anxiety in adolescent girls. Br J Psychiatry 179:116–121
Silverman AB, Reinherz HZ, Giaconia RM (1996) The long-term sequelae of child and adolescent abuse: a longitudinal community study. Child Abuse Negl 20(8):709–723
Stein MB, Koverola C, Hanna C, Torchia MG, McClarty B (1997) Hippocampal volume in women victimized by childhood sexual abuse. Psychol Med 27(4):951–959
Tedeschi RG, Calhoun LG (1996) The Posttraumatic Growth Inventory: measuring the positive legacy of trauma. J Trauma Stress 9(3):455–471
Teicher MH, Dumont NL, Ito Y, Vaituzis C, Giedd JN, Andersen SL (2004) Childhood neglect is associated with reduced corpus callosum area. Biol Psychiatry 56(2):80–85
Thompson R, Tabone JK (2010) The impact of early alleged maltreatment on behavioral trajectories. Child Abuse Negl 34(12):907–916
Thornberry TP, Henry KL, Ireland TO, Smith CA (2010) The causal impact of childhood-limited maltreatment and adolescent maltreatment on early adult adjustment. J Adolesc Health 46(4):359–365
Tietjen GE, Brandes JL, Peterlin BL, Eloff A, Dafer RM, Stein MR etal (2010) Childhood maltreatment and migraine (part III). Association with comorbid pain conditions. Headache 50(1):42–51
Turner-Sack AM, Menna R, Setchell SR (2012) Posttraumatic growth, coping strategies, and psychological distress in adolescent survivors of cancer. J Pediatr Oncol Nurs 29(2):70–79
Tyrka AR, Wier L, Price LH, Ross N, Anderson GM, Wilkinson CW etal (2008) Childhood parental loss and adult hypothalamic-pituitary-adrenal function. Biol Psychiatry 63(12):1147–1154
Tyrka AR, Wyche MC, Kelly MM, Price LH, Carpenter LL (2009) Childhood maltreatment and adult personality disorder symptoms: influence of maltreatment type. Psychiatry Res 165(3):281–287
Tyrka AR, Price LH, Kao HT, Porton B, Marsella SA, Carpenter LL (2010) Childhood maltreatment and telomere shortening: preliminary support for an effect of early stress on cellular aging. Biol Psychiatry 67(6):531–534
van der Vegt EJ, van der Ende J, Kirschbaum C, Verhulst FC, Tiemeier H (2009) Early neglect and abuse predict diurnal cortisol patterns in adults A study of international adoptees. Psychoneuroendocrinology 34(5):660–669
van der Vegt EJ, van der Ende J, Huizink AC, Verhulst FC, Tiemeier H (2010) Childhood adversity modifies the relationship between anxiety disorders and cortisol secretion. Biol Psychiatry 68(11):1048–1054
Vythilingam M, Heim C, Newport J, Miller AH, Anderson E, Bronen R etal (2002) Childhood trauma associated with smaller hippocampal volume in women with major depression. Am J Psychiatry 159(12):2072–2080
WHO (2004) International Statistical Classification of Diseases and Health Related Problems. World Health Organization, Geneva
Wingo AP, Wrenn G, Pelletier T, Gutman AR, Bradley B, Ressler KJ (2010) Moderating effects of resilience on depression in individuals with a history of childhood abuse or trauma exposure. J Affect Disord 126(3):411–414
Wolchik SA, Coxe S, Tein JY, Sandler IN, Ayers TS (2008) Six-year longitudinal predictors of posttraumatic growth in parentally bereaved adolescents and young adults. Omega (Westport) 58(2):107–128
Yu XN, Lau JT, Zhang J, Mak WW, Choi KC, Lui WW etal (2010) Posttraumatic growth and reduced suicidal ideation among adolescents at month 1 after the Sichuan earthquake. J Affect Disord 123(1–3):327–331
Zeanah CH, Smyke AT, Koga SF, Carlson E (2005) Attachment in institutionalized and community children in Romania. Child Dev 76(5):1015–1028
Zeanah CH, Egger HL, Smyke AT, Nelson CA, Fox NA, Marshall PJ etal (2009) Institutional rearing and psychiatric disorders in Romanian preschool children. Am J Psychiatry 166(7):777–785
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2013 Springer Science+Business Media New York
About this chapter
Cite this chapter
Nater, U.M., Skoluda, N. (2013). Consequences of Developmental Stress in Humans: Adversity Experienced During Childhood and Adolescence. In: Laviola, G., Macrì, S. (eds) Adaptive and Maladaptive Aspects of Developmental Stress. Current Topics in Neurotoxicity, vol 3. Springer, New York, NY. https://doi.org/10.1007/978-1-4614-5605-6_7
Download citation
DOI: https://doi.org/10.1007/978-1-4614-5605-6_7
Published:
Publisher Name: Springer, New York, NY
Print ISBN: 978-1-4614-5604-9
Online ISBN: 978-1-4614-5605-6
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)