Abstract
Mycoheterotrophic plants lack chlorophyll and depend on an intimate association between their underground organs and fungi connected to photosynthetic plants for carbon compounds. The diverse fungi involved may also increase access to soil-derived nutrients. Examples of mycoheterotrophic species occur within nonvascular plants, the gametophytes of several seedless vascular plant genera, and the roots/rhizomes of many angiosperms. This chapter focuses on the structural diversity of the underground organs of mycoheterotrophic plants and the complex mycorrhizal colonization patterns observed using a variety of microscopic methods. Evolutionary trends of mycoheterotrophic plants towards compact root/rhizome systems combined with colonization patterns that result in a sustained benefit from the fungus occur in most angiosperm lineages and are textbook examples of convergent evolution.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others

Keywords
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
4.1 Introduction
For most, if not all plants, subterranean parts are less known than their aerial counterparts, due in part to the difficulty in extracting a complete root system (see Kutschera and Lichtenegger 1982, 1992; Kutschera et al. 2009) and the lack of morphological information in floras and taxonomic descriptions because many herbarium specimens do not include underground parts such as roots and rhizomes. Likewise, information from the fossil record is biased towards aerial structures (Peterson 1992) although there have been discoveries of fossils showing fungal associations with underground organs (e.g., Kidston and Lang 1921, Remy et al. 1994; Taylor et al. 1995; LePage et al. 1997; Stockey et al. 2001). To date, fossils of root-fungal associations of mycoheterotrophic plants are unknown.
(a–c) Lycopodiaceae, (d–f) Ophioglossaceae, (g–j) Psilotaceae. (a) Lycopodium obscurum sporophyte showing strobili. (b) Lycopodium clavatum mycoheterotrophic gametophytes with shoots (arrowheads). (c) Section of L. obscurum gametophyte showing zone of arbuscular mycorrhizal fungi (arrowheads) and antheridia (arrows).Fig. 4.1 (continued) (d) Shoot of Botrychium virginianum with fertile segment of leaf with sporangia (arrowhead). (e) Mycoheterotrophic gametophyte (arrow) of B. virginianum with a root (arrowhead) and base of a shoot (double arrowhead). (f) Intracellular hyphal coils of Glomus-Group A in a Botrychium crenulatum mycoheterotrophic gametophyte. Photo courtesy of Jennifer Winther. (g) Shoots of Psilotum nudum with synangia (arrowheads). (h) Branched mycoheterotrophic gametophyte of P. nudum. (i) Intracellular hyphal coils of an arbuscular mycorrhizal fungus in a sectioned P. nudum mycoheterotrophic gametophyte stained with Toluidine blue O. (j) Transmission electron micrograph of hyphae within a mycoheterotrophic gametophyte of P. nudum showing the interface consisting of host plasma membrane (perifungal membrane) (arrowheads) and host-derived intracellular matrix (arrows). (k) Buxbaumia aphylla sporophyte, 1.5 cm high
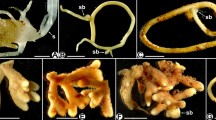
(a–h) Afrothismia hydra, (i, j) Afrothismia winkleri (Thismiaceae). (a) Seed (left, 0.6 mm long) and an early germination stage (right) of A. hydra with disrupted seed coat (sc), giving rise to a first root tubercle. (b–d) More tubercles develop successively at the base of the initial one and the root extensions elongate. The seed coat (sc) Fig. 4.2 (continued) is still attached. The whitish content is the fungal colonization. (e) The rhizome has developed into a shoot terminated by a 1 cm long flower. (f) A. hydra in its natural habitat with the filiform root elongations superficially clinging to the substrate. (g) A. hydra showing the strictly sympodial flowering mode with clusters of root tubercles at the base of each pedicel. Three basal flowers (A, B, C) have already detached, the following are in dissemination stage with placentophore developed (D), in fruit (E) and in bud (F). Note early tubercle (tb) development at the base of the flower bud. (h) Close up of the placentophore (pl). (i) Flower of A. winkleri, measuring 1.5 cm from the subtending scale leaf to the bending of the tube. (j) Root/rhizome system of A. winkleri
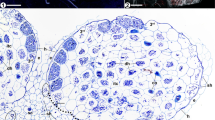
Afrothismia saingei (Thismiaceae). (a) Rhizome tip with many root tubercles; some tubercles have been detached. The characteristic hyphal loops (green rectangle), developed in spiral lines within the tubercles, are Fig. 4.3 (continued) visible from the outside. (b) Close up of the green rectangle in (a). Tangential section through two hypodermal cells colonized by looped hyphae that are connected to each other. (c) Specimen (Wilks No. 1179) of A. saingei under investigation from the Herbarium in Utrecht, labeled as Afrothismia winkleri. (d) Cleared preparation of a filiform root extension showing straight growing hyphae and vesicles. (e) Transverse section through a rhizome of A. saingei with coils of enlarged hyphae (vh) in the cortex. Mostly only once per tubercle these hyphae transit (green circle) over an inconspicuous separating layer (sl) into the hypodermis of a tubercle to start the spiral line of hyphal loops (see (a, b)). (f) Transverse section through a shoot/pedicel of A. saingei. The enlarged hyphae in the rhizome cortex (e) are continuous with the straight growing, also quite large hyphae (sh) in the outer shoot cortex, thus connecting the spatially separated tubercle clusters along the plant (c). The inner cortex (ic) is free of hyphae. (g) Longitudinal section through a young root tubercle showing the looped coils in an alternating pattern in the hypodermis as to be expected from its spiral arrangement (1–5) whereas all other cortex cells contain degenerated hyphal coils (hc−). (h) Longitudinal section through an old root tubercle where the digestion of hyphal coils has advanced but the epidermis now contains straight growing, nondegenerated hyphae (sh) linking those in the filiform root extension (she, see (d)) with the enlarged hyphae in the rhizome cortex (see (e)). (i) Schematic view of the mycorrhizal colonization pattern in A. saingei: Straight hyphae grow through root extension and tubercle epidermis, enter the rhizome cortex becoming enlarged and coiled, transit once per tubercle into the hypodermis of the tubercle starting a spiral line of looped interconnected hyphae around it (hl), and from there send hyphal branches into the rest of the cortex parenchyma for digestion (dh). The red line signifies an impenetrable barrier to the fungus. The spatially separated clusters of tubercles share the fungus via straight hyphae growing along the shoot axis in its outer cortex parenchyma
Afrothismia gesnerioides (Thismiaceae). (a) A. gesnerioides emerging about 1.5 cm above the soil. In contrast to many other Afrothismia spp., roots and rhizome are subterranean. (b) Root/rhizome system of Fig. 4.4 (continued) A. gesnerioides, about 1 cm wide. (c) Transverse section through a rhizome (rh) and longitudinal sections of root tubercles (tb) showing starch grains (st) in the inner rhizome cortex and uncolonized root cortex, straight hyphae (sh) in the outer rhizome cortex, nondegenerated dense hyphal coils in the third cell layer of the root tubercle (hc+) and degenerated hyphal coils in the tubercle parenchyma (hc−). The hypodermis (hd) is largely collapsed. (d) Longitudinal section through a young tubercle of A. gesnerioides where the hypodermis (hd) is still visible. Fungal colonization has just started from the tubercle base in the third cell layer (hc+) and starch depositions (st) are still present in the parenchyma, which will disappear when fungal colonization proceeds. First degenerated hyphal coils (hc−) are also present in the inner parenchyma. The root extension has not yet developed. (e) Longitudinal section through a root tip of an older tubercle showing the root extension (ex) partly in transverse view, colonized by straight hyphae (sh) which proceed into the root epidermis (ep, see (f)). (f) Root tubercle epidermis (ep) only contains straight growing hyphae (sh) which never penetrate the hypodermis (hd, collapsed) but are continuous with those in the outer rhizome cortex. (g) Four neighboring tissues of the root tubercle hosting distinct morphotypes of fungal colonization: epidermis (ep) with straight hyphae (sh), hypodermis (hd) as a barrier to the fungus, the third root layer with nondegenerated dense hyphal coils (hc+) and the multilayered root parenchyma containing degenerated hyphal coils (hc−). (h) Transition of colonization (green oval) between the straight hyphae (sh) in the outer rhizome cortex and the nondegenerating hyphal coils (hc+) in the third root layer of the tubercle across a layer continuous with the otherwise impenetrable hypodermis (hd). Uncolonized parenchyma cells and the inner rhizome cortex contain starch grains (st). (i) Schematic view of the mycorrhizal colonization pattern in A. gesnerioides: straight hyphae grow through root extension, tubercle epidermis and outer rhizome cortex, transit at the base of the tubercle into its third layer to form dense coils (dc, green texture), and branches from there colonize the inner tubercle parenchyma to become digested (dh). The red marked hypodermis is impenetrable for the fungus
Dictyostega orobanchoides (Burmanniaceae). (a) Inflorescence of D. orobanchoides, composed of a bifurcate cincinnus. (b) Single preserved flower of D. orobanchoides, 2.5 mm long. (c) Rhizome (rh) of D. orobanchoides about 1.5 mm thick with a tuft of filiform roots (r). Apically the rhizome turns into a shoot (s). (d) Root of D. orobanchoides, cortex (rc) partly detached, exposing the thickened endodermis (en) which encloses the central cylinder (cc). The whitish cell contents are fungal coils. (e) Longitudinal section through a root epidermis of D. orobanchoides. Fungal colonization consists of coiled hyphae (hc+), partly decomposed arbusculate Fig. 4.5 (continued) structures (a), and vesicles (v). (f) Transverse section through a root of D. orobanchoides showing the central cylinder (cc) consisting of a central tracheary element surrounded by one ring of smaller tracheary elements and the pericycle, a thickened endodermis (en), two layers of small parenchyma layers, and epidermal cells (ep). The epidermal cells contain hyphal coils (hc+) and arbuscules (a), the latter partly degraded. (g) SEM micrograph of a rhizome of D. orobanchoides imbricately covered by peltate scale leaves with fringed margins. The leaf interstitials contain fungal hyphae (hy). (h) Transverse section through a rhizome covered with imbricate scale leaves (le) showing fungal colonization including vesicles (v) within (ihy) and in between (ohy) the scale leaves. The rhizome axis (rh) is not colonized but contains starch grains (st). (i) Tangential section through a rhizome (rh) including the imbricate scale leaves (le) showing dense hyphal masses in the leaf interstitials (li). (j) Schematic view of the mycorrhizal colonization pattern of D. orobanchoides: the peltate scale leaves and their interstitials are colonized by hyphal coils and vesicles. The root is colonized only in the epidermis by hyphal coils, arbuscules, and vesicles; the arbuscules are the first to degenerate
(a–c) Apteria aphylla, (d–e) Gymnosiphon divaricatus, (f–h) Hexapterella gentianoides, (i–k) Campylosiphon congestus (Burmanniaceae). (a) Preserved flower (9 mm long) and fruit of A. aphylla. (b) Top view of a flower of A. aphylla (courtesy of H and PJM Maas). (c) Subterranean system of A. aphylla. The shoot (s) is continuous with the short (3 mm) orthotropous rhizome (rh) bearing numerous filiform roots. The root cortex parenchyma is often disrupted, leaving the thickened endodermis (en) with central cylinder enclosed as the only connection with the rhizome. Fig. 4.6 (continued) (d) Subterranean system of G. divaricatus, very similar to that of A. aphylla (see (c)) with short rhizome (rh) and exposed endodermis (en). (e) Top view of a flower of G. divaricatus (courtesy of H and PJM Maas). (f) Flower of H. gentianoides (courtesy of H and PJM Maas). (g) Subterranean system of H. gentianoides, also with a short rhizome (rh, 4 mm long) continuous with the shoot (s). The rhizome bears filiform roots (r); light coloration indicates fungal colonization. (h) Top view of a flower of H. gentianoides (courtesy of H and PJM Maas). (i) Preserved inflorescence of C. congestus. (j) Preserved single flower (9 mm long) of C. congestus. (k) Subterranean system of C. congestus with the shoot (s) continuous with a slightly tuberous rhizome (rh), 9 mm long and 2.5 mm thick, bearing filiform roots (r)
(a–f) Burmannia tenella, (g–i) Burmannia hexaptera (Burmanniaceae). (a) Preserved flower (6 mm long) of B. tenella. (b) Root system of B. tenella with several star-like radiating vermiform roots (r), about 1.2 mm thick, at the base of the shoot (s). (c) Inflorescence of B. tenella, the bifurcate cincinnus usually consists of a few flowers. (d) Transverse section through a root of B. tenella with extensive fungal colonization of the multilayered cortex parenchyma. The central cylinder is reduced and surrounded by a tertiary endodermis (en). (e, f) The fungus in the root parenchyma cells of B. tenella forms heteromorphic coils of hyphae of various width (hc+), arbusculate structures (a) as well as vesicles (v), often within one cortex cell. Degeneration begins with the arbusculate structures (a−); the thicker hyphae tend to persist longer. (g) Flowers of B. hexaptera emerging only a few centimeters above the soil surface. (h) Preserved flower (1 cm long) of B. hexaptera. (i) Root system of B. hexaptera comprised of vermiform roots (r), about 1.2 mm thick, also with the tendency to radiate at the base of the shoot (s), resulting in a coralloid appearance
(a–i) Triuris hyalina, (j–k) Sciaphila ledermannii (Triuridaceae). (a) Subterranean shoot (s) of T. hyalina with two nodes (n) bearing paired roots (r) in the axils of the scale leaves. The female inflorescence has one mature flower and one bud (b). (b) Female flower of T. hyalina with the characteristic tail-like tepal appendages. Fig. 4.8 (continued) The apocarpous gynoecium is about 1.5 mm wide. (c) Subterranean shoots of T. hyalina with spaced nodes (n) where paired roots (r) arise from each scale leaf axil giving it a ladder-like appearance. The roots are uniformly 0.4 mm thick. (d) Each node seen in (c) may develop aggregations of paired roots (agr) as explained in text. (e) An aggregation of roots seen in (d) results in a star-like root system. At this stage, it may have already borne several flowering shoots (detached). A new shoot (s), 2 cm long, bearing a flower bud has developed. (f) Transverse section through a T. hyalina root measuring 0.4 mm in diameter. The epidermis (ep) is mostly free of hyphae and the exodermis (ex) is a barrier to the fungus except for the short cells (shc) with thickend outer tangential walls serving as passage cells. The outer cortex parenchyma layer bears dense hyphal coils (hc+) which do not become digested but may collapse when older. The middle parenchyma layer consists of enlarged cells containing mostly amorphous clumps of hyphal masses (hc−). The inner cortex layer of much smaller cells is free of hyphae. The endodermis (en) is only slightly suberized. (g) Longitudinal section through a T. hyalina root showing epidermis (ep), exodermis (ex), the dense hyphal coils (hc+) in the outer and the degenerated ones (hc−) in the middle cortex parenchyma layer. Occasionally vesicles (v) may occur in both layers. (h) Schematic view of the colonization pattern in T. hyalina: after penetration of epidermis and a short cell of the exodermis the hyphae start to coil and decrease their diameter while spread longitudinally and tangentially within the outer cortex parenchyma. The dense coils of narrow hyphae (see (g)) send branches into the middle parenchyma layer where they degenerate to amorphous clumps. Vesicles may occur in both layers. The red marked cells are impenetrable to the fungus. (i) Inflorescence of S. ledermannii showing female flowers. (j) Subterranean system of S. ledermannii consisting of a short rhizome (rh) continuous with the epiterrestrial shoot (s). The rhizome bears filiform roots (r) in this specimen up to 9 cm long and 0.8 mm thick
Sciaphila polygyna (Triuridaceae). (a) Top view of a female flower of S. polygyna with its numerous carpels in fruiting stage (about 3 mm wide). (b) Same flower from the lower side showing the tepals (tp) with hair tufts. (c) Apex of an inflorescence of S. polygyna with numerous flower buds. (d) Subterranean system of S. polygyna with Fig. 4.9 (continued) thick roots (r, several have detached) radiating from the base of two shoots (s, one is detached). For the architecture of this root system, see Imhof (2004). (e) Transverse section through a central part of a 1.2 mm thick root of S. polygyna surrounded by epidermis (ep) and exodermis (ex) with short cells (shc) serving as the only passage cells for fungal penetrations (p). Root anatomy and mycorrhizal pattern are highly heteromorphic with cells of the fourth root layer being much larger (“giant cells,” blue border) than others dislocating the central cylinder (cc) out of its central position to create a dorsiventral architecture of the root. Fungal material degenerates (hc−) only in the fourth layer. The third layer has loose hyphal coils with swellings (yellow border), coils without swellings (not marked) and very dense coils of thin hyphae (green border, ventral side). Colonization by dense coils follows a v-shaped pattern leaving a gap of colonization (g) in some parts of the roots (see right hand side of (h)). (f) Tangential section through the dorsal side of a S. polygyna root showing areas colonized by coils with many (yellow border) and with less swellings. (g) Tangential section through the ventral side of the third root layer at the same magnification as (f) indicating the differences of the three types of coils in the third layer. (h) Schematic view of the mycorrhizal colonization pattern in S. polygyna in transverse (left) and longitudinal view (right). The coloration corresponds to those in (e, f). Note that the different colonization morphotypes, the appearance of giant cells, the digestion of hyphae therein is also heteromorphic in the longitudinal view as it is in transverse view (see details in Imhof 2003)
(a–c) Seychellaria madagascariensis, (b–f) Kupea martinetugei, (Triuridaceae) (g, h) Geosiris aphylla, (Iridaceae) (i, j) Thismia panamensis (Thismiaceae). (a) Female flowers of S. madagascariensis about 2 mm wide with the basal filiform styles projecting above the carpels. (b) Young male flower of S. madagascariensis. Fig. 4.10Fig. 4.10 (continued) (c) Root aggregation of S. madagascariensis (compare Fig. 4.8e) showing four shoots (s) and numerous pilose roots (r) up to 10 cm long and 0.6 mm thick. (d) Female inflorescence of K. martinetugei. (e) Male inflorescence of K. martinetugei. (f) Root system of K. martinetugei with several radiating, 1.5 mm thick and up to 7 mm long, roots (r) at the base of the shoot (s). (g) Subterranean parts of G. aphylla with a rhizomatous tuber (t) and filiform roots (r) at its base. (h) Preserved capitulum of G. aphylla. (i) Subterranean tuber (t) of T. panamensis with filiform roots (r) radiating from it. (j) Flower of T. panamensis (courtesy of H and PJM Maas)
Arachnitis uniflora (Corsiaceae). (a) Flowering stems of Arachnitis uniflora, each with a single flower. Image courtesy of Laura Domínguez. (b) Propagules (arrowheads) on a fleshy root (asterisk) of A. uniflora. (c) Confocal microscopy of intracellular branched hyphal structures of Glomus-Group A in a root of A. uniflora. (d) Longitudinal section of resin-embedded root of A. uniflora stained with toluidine blue O showing the apical meristem (asterisk), intracellular hyphae of Glomus-Group A (arrowheads), and intracellular branched hyphal structures (arrow). (e) Enlarged portion of a similar section of a A. uniflora root showing intracellular branched hyphal structures of Glomus-Group A (arrows). (f) Clearings of root cells of A. uniflora stained with acid fuchsin showing numerous intracellular branched hyphal structures of Glomus-Group A (arrows). (g) Clearings of root cells of A. uniflora stained with acid fuchsin showing intracellular hyphae (arrowheads) and a vesicle (arrow) of Glomus-Group A
Orchidaceae. (a) Flowering stems of Corallorhiza trifida. (b) Diagram of a developing orchid protocorm with intracellular fungal hyphal coils (pelotons) (arrowheads). (c) Section of an orchid root showing intact pelotons (arrowheads) and degraded hyphae (arrows). Image courtesy of Carla Zelmer. (d) C. trifida root cells (arrow) showing peloton stained with chlorazol black E (arrowheads). Scale bar = 10 μm. Image courtesy of Carla Fig. 4.12 (continued) Zelmer and Randy Currah. (e) The same fungus isolated from C. trifida root cells and inoculated on Pinus contorta roots formed typical ectomycorrhizas with a mantle (arrow) and Hartig net (arrowheads). Scale bar = 25 μm. Image courtesy of Carla Zelmer and Randy Currah. (f) Subterranean system of Neottia nidus-avis, consisting of numerous roots, 1–2.5 cm long and 2 mm thick, emerging from a short orthotropous rhizome. (g) Subterranenean system of Wullschlaegelia calcarata, with spindle-shaped root tubers (max. 2 cm long and 2 mm thick) at a short rhizome
Epirixanthes spp. (Polygalaceae). (a) Subterranean system of E. papuana with approximately 0.6 mm thick roots (r) arising from a short rhizome (rh) continuous with the epiterrestrial shoot (s). Additional roots (ar) may develop along the shoot where it is connected to the soil. (b) Isolated flower of E. papuana (spirit material), little more than 2 mm long, with three tepals (te). Not all of the five sepals (se) are visible. Fig. 4.13 (continued) (c) Inflorescence of E. papuana. (d) Subterranean system of E. elongata similar to that of E. papuana seen in (a). This specimen has basal shoot ramifications. Labels as in (a). (e) Longitudinal section through the cortex of an E. papuana root showing a part of the straight, cascading hyphae (ch) in the outer parenchyma, coiled hyphae (hc+) in layer 2 (l2) and degenerated coils (hc−) in layer 1 (layers counted from the endodermis). (f) Longitudinal section through parenchyma layers 1 (l1) and 2 (l2) of a E. elongata root. Layer 2 contains initially straight hyphae sending hyphal branches (hb1) into layer 1 where they immediately degenerate to amorphous clumps (hc−). (g) Tangential section external to the central cylinder through an E. papuana root showing 2 cell rows of each layer 1 (l1), 2 (l2) and 3 (l3). Hyphae in layer 2 remain functional (hc+) and send branches centripetally into layer 1 (hb1) as well as centrifugally into layer 3 (hb3), both of which digest the fungal material (hc−). A part of the cascading hyphae coming from the outer cortex layers is also visible (ch). (h) Transverse section through an E. papuana root. The epidermis (ep) as well as the outer three cortex parenchyma layers are not colonized by hyphal coils, layer 1 (l1) and 3 (l3) contain degenerated coils whereas layer 2 (l2 and dotted line) contains functional hyphae. The central cylinder inside the endodermis (en) is largely composed of lignified fibers. (i) Schematic view of the mycorrhizal colonization pattern in Epirixanthes spp.. After penetration, the hyphae grow straight in a cascading manner through the outer cortex (1), retain the straight growth when reaching layer 2 but send branches into layer 1 for digestion (2), start to coil hyphae in layer 2 when the mycorrhization proceeds (3) and then also send branches in layer 3 for digestion (4)
Monotropa uniflora (Ericaceae/Monotropoideae). (a) Cluster of flowering stems in a hardwood forest in southern Ontario, Canada. (b) Young shoots and associated root ball. (c) Mycorrhizal root tip showing compact mantle with cystidia (arrowheads). Photo courtesy of Brent Young. (d) Scanning electron micrograph of a root tip showing cystidia. (e) Higher magnification scanning electron micrograph of portion of a mycorrhiza with a calcium-oxalate crystal (arrowhead) among cystidia. (f) Scanning electron micrograph of a fracture of a Fig. 4.14 (continued) mycorrhiza with mantle (asterisk), fungal peg (arrow) and flask-shaped cystidia (arrowheads). (g) Scanning electron micrograph of large calcium oxalate crystals (arrows) among flask-shaped cystidia. (h) Freehand transverse section of root showing mantle (asterisk) and labyrinthine branching of Hartig net (arrowheads). (i) Longitudinal section of resin-embedded root stained with Toluidine blue O showing the apical meristem (asterisk), and the mantle covering the root apex. (j) Paradermal section of resin-embedded root stained with Toluidine blue O showing labyrinthine branching of Hartig net hyphae and fungal pegs in transverse section (arrows). (k) Higher magnification of a longitudinal section of resin-embedded root stained with Toluidine blue O showing mantle (asterisk), Hartig net (arrowheads) and fungal peg (arrow). (l) Transmission electron micrograph showing detail of the fungal peg with finger-like wall depositions (arrows)
Pterospora andromedea (Ericaceae/Monotropoideae). (a) Two flowering stems in the boreal forest in British Columbia, Canada. (b) Root ball showing mycorrhizal root tips. (c) Branched root tip showing colored mantle characteristic of a Rhizopogon sp. (d) Scanning electron micrograph of a portion of mantle showing compact hyphae. (e) Higher magnification scanning electron micrograph showing irregular hyphae and abundant small crystals (arrowheads). (f) Higher magnification scanning electron micrograph showing details of various crystals (arrowheads). (g) Longitudinal section of a root showing the thick mantle (asterisk) and Hartig net (arrows). (h) Longitudinal section of a root showing the inner mantle (asterisk), Hartig net (arrowhead) and fungal peg (arrow) penetrating the radial epidermal cell wall
(a, b) Allotropa virgata, (e–h) Pleuricospora fimbriolata, (Ericaceae/Monotropoideae). (a) Flowering shoots of Allotropa virgata. (b) Longitudinal section of a root showing a small apical meristem (asterisk) and some fungal colonization (arrowheads). (c) Higher magnification showing Hartig net (arrowhead) and fungal peg (arrow). (d) Transmission electron micrograph showing Hartig net (arrowhead) and detail of the fungal peg with finger-like wall depositions (arrows). (e) Emerged shoot with flowers of Pleuricospora fimbriolata. Photo courtesy of Dan Luoma. (f) Longitudinal section of a root showing the apical meristem (asterisk) and well-developed mantle (arrowheads). (g) Mantle (asterisk) and fungal peg (arrow) penetrating the outer tangential wall of an epidermal cell. (h) Transmission electron micrograph showing a fungal peg with finger-like wall depositions (arrowheads)
Pityopus californicus (Ericaceae/Monotropoideae). (a) Flowering stems of Pityopus californicus. Photo courtesy of Barry Rice. (b) Developing embryo with multilayered mantle (arrows) on developing root. Remnants of seed coat are obvious (arrowheads). (c) Longitudinal section of an older root with mantle covering the apex. An emerging lateral root (arrow) is evident. (d) Higher magnification showing mantle (arrows) and Hartig net (arrowheads). (e) Detail of mantle (asterisk), Hartig net (arrowheads), and fungal peg (arrow) penetrating the tangential wall of an epidermal cell. (f) Scanning electron micrograph of fractured root showing a thick mantle (asterisk). (g) Transmission electron micrograph showing a fungal peg with finger-like wall depositions (arrows)
(a–c) Voyria tenuiflora, (d, e) Voyria obconica, (f–h) Voyria spruceana, (i–k) Exochaenium oliganthum (Gentianaceae). (a) V. tenuiflora in its natural habitat. (b) Coralloid shaped root system of V. tenuiflora with branched roots (r) clumped at the base of a shoot (s). (c) Subterranean organs of V. tenuiflora showing the tendency to radiating roots (r) at the base of a shoot (s). The roots can be up to 1 mm thick and several centimeters long. (d) V. obconica in its natural habitat (courtesy of H and PJM Maas). (e) Subterranean system of V. obconica with stout, up to 1.5 mm thick and 1 cm long roots (r) at the base of a shoot (s). (f) Characteristic fringed, tail-like thecae appendages (ap) of V. spruceana. (g) Coralloid shaped root system of V. spruceana, also having the tendency for a star-like structure (this specimen measuring 14 mm in maximal extension). (h) Preserved flower (1.2 cm long) of V. spruceana. (i) Vermiform to filiform root of E. oliganthum with thickenings up to 0.8 mm where a light brown coloration indicates fungal colonization (fc). (j) E. oliganthum tends to develop radiating roots (r) at the base of a shoot (s). (k) Two preserved flowers (7 mm long) of E. oliganthum
(a–g) Voyria truncata, (h–n) Voyria aphylla (Gentianaceae). (a) Epiterrestrial part of V. truncata. (b) Subterranean shoot (s) of V. truncata, spirally bent due to soil obstructions. The shoot arises from the axil between a main root (r) and a side root (sr). (c) Complete specimen of V. truncata extracted from the soil, basally arising from Fig. 4.19 (continued) a plagiotropous root (detached). Only the upper, reddish branches were epiterrestrial and, hence, appeared superficially as clustered but distinct individuals. (d) Subterranean shoot (s) of V. truncata arising from a runner-like, plagiotropic root (r), intermingled with roots of neighboring plants (rnp). (e) Runner-like root (7 cm long and up to 2 mm thick) of V. truncata extracted from the soil with several side roots (sr) in the axils of which one or at most two root-borne shoots (s) develop. (f) Longitudinal section through a V. truncata root showing the epidermis (ep) and multilayered cortex with intracellular hyphal coils in various stages of degradation. The green dotted line indicates a course of colonization from penetration to the inner cortex. The passage through a short cell (shc) of the exodermis (ex) is not visible on this section but present on the subsequent one (not shown). The pattern of newly inserted cell walls (arrows) indicate an ongoing primary thickening. (g) Schematic view of the mycorrhizal colonization pattern in V. truncata. After penetration of the epidermis and a short cell (shc) of the exodermis as the only passage cells, the hyphae grow in a coiling manner from cell to cell deeper into the cortex. Extent of hyphal degradation increases with cortex depth. (h) Flower of V. aphylla. (i) Shoot (s) of V. aphylla arising from a net of runner-like, up to 0.5 mm thick roots (r), much smaller than in V. truncata. (j) Radiating roots (r) at the base of a shoot (s) of V. aphylla. (k) Tangential section through the cortex of a V. aphylla root showing the epidermis (ep) with straight, nondegenerated hyphae (sh), the exodermis (ex) with only the short cells (shc, anatomically not particular distinct) being used as passage cells for the hyphae, and the cortex parenchyma with often degenerated hyphal coils (hc−). Root hairs (h) only occur where organic material is attached to the root. Fungal penetrations (p) mostly happen via the root hairs. (l) Tangential section just external to the central cylinder through a V. aphylla root, showing the epidermis (ep) with straight hyphae (sh) and mostly degenerated hyphal coils (hc−) in the cortex parenchyma. However, straight hyphae (sh) also occur in the innermost parenchyma layers (ic), linked to the hyphae in the epidermis by nondegenerated coiled hyphae (not shown). (m) Root of V. aphylla (r) attached to a root of a neighboring plant (rnp). Root hairs (h) develop only at such root to root connections. (n) Schematic view of the mycorrhizal colonization pattern in V. aphylla. After penetration of root hairs, the hyphae grow straight in the epidermis, cross the exodermis via passage cells (miss the red coloration), build coils in the outer cortex parenchyma which partly degenerate but also reach the innermost cortex parenchyma layers, where they again grow in a straight manner along the root axis. From these inner straight hyphae which do not become digested, branches grow back into the outer cortex to build coils for digestion
Voyria tenella (Gentianaceae). (a) Three specimens of V. tenella in various stages of development. The younger specimens still show the nodding flower bud. (b) Youngest specimen of V. tenella found, measuring 2 mm in length. The primary root formed during germination (the arched part on the left hand side) has initiated three root primordia (on the right hand side). A shoot bud has not formed at this stage. (c) The first shoot primordium (s) Fig. 4.20 (continued) appears only after the development of a characteristically radiating root system (this specimen is 4 mm wide in its maximal extension). (d) Young specimen of V. tenella, the shoot is 1 mm thick. (e) Flower of V. tenella. (f) Longitudinal section through a young root of V. tenella showing the penetration and the subsequent direct growth of the straight hypha (sh) towards the inner root cortex where it proceeds along the central cylinder. (g) Longitudinal section through a mature root of V. tenella showing the straight hyphae (sh) in the inner cortex parenchyma around the central cylinder (cc) and the degenerated coils of hyphae (hc−) in the outer cortex. (h) Transverse section through a root of V. tenella (0.8 mm in diameter). The straight hyphae (sh) in the inner cortex parenchyma are visible as small circles. The epidermal tissue (et) consist of 2–3 layers of smaller cells never colonized by the fungus except for penetration points. Their outermost cells slough off (so) and are replaced by derivates of the layers underneath. The obvious digestion of hyphal coils (hc−) takes place in the majority of the cortex parenchyma. (i) Schematic view of the mycorrhizal colonization pattern in V. tenella. After penetration, the hyphae grows straighly towards the inner cortex layers which are longitudinally elongated and proceed therein along the central cylinder. Branches of these inner hyphae grow back into the outer cortex and degenerate (dh)
In autotrophic plants, many scientific questions can be dealt with using generalized concepts of root structure and function (e.g., Kutschera and Lichtenegger 1992; Polomski and Kuhn 1998; Gregory 2006). However, this certainly does not hold for mycoheterotrophic (MH) plants. The structure of roots, rhizomes, or subterranean scale leaves of MH plants intimately linked to the association with soil fungi is of critical ecological relevance because these plants essentially depend upon fungi for their carbon and perhaps other nutrient needs. Hence, the subterranean organs of MH plants often show remarkable morphological and anatomical adaptations to meet their specific requirements. This chapter, therefore, addresses the importance of morphology and anatomy to complement modern methods for understanding the fungal colonization patterns in MH plants and their relationships to function.
In the following, we summarize the current knowledge of structural aspects of the underground parts (for a peculiar exception, see Afrothismia) of MH plants ranging from bryophytes to angiosperms, the latter in systematical order following the Angiosperm Phylogeny Group (APG 2009), which has been regularly updated by Stevens (2001 onwards). We are aware of the gradual differences between species in terms of mycorrhizal dependence, however, due to space limitations, we focus on the visibly achlorophyllous species, and only include the partially mycoheterotrophs where they add to the common picture.
The final section interprets the available information in terms of detecting phylogenetic trends of MH plants, in order to understand their evolutionary history, a subject that is receiving considerable attention in the mycorrhizal literature (see Brundrett 2002).
4.2 Nonvascular Plants
4.2.1 Aneura
Aneura mirabilis (Aneuraceae/Hepaticae) was described as Cryptothallus mirabilis by von Malmborg (1933, 1934), although it was noted earlier around 1914 (Schiffner 1934) and suggested to be either an Aneura or Riccardia species (e.g., Denis 1919; Schiffner 1934). Recently, Cryptothallus was formally transferred to Aneura by Wickett and Goffinet (2008) based on molecular and morphological characteristics. This decision is supported by the observation that the endophyte in Aneura mirabilis belongs to the same genus (Tulasnella) as that in Aneura pinguis (Bidartondo et al. 2003), and the mycorrhizal pattern in both species is very similar (Ligrone et al. 1993).
Aneura mirabilis mostly occurs in maritime climates (e.g., Sjörs 1949; Williams 1950; Petersen 1972; Wiehle et al. 1989; Sergio and Seneca 1997; Sergio and Garcia 1999; Boudier et al. 1999) in cool, humid, mostly peaty environments with large mats of bryophytes (Wiehle et al. 1989). Only a part of the seta and the sporangium is elevated above the surrounding mats consisting of several moss species (von Malmborg 1933; Wiehle et al. 1989; Sergio and Garcia 1999; Boudier et al. 1999). The whitish, vermiform to lobular-coralloid, brittle gametophytes are only a few centimeters in length and remain embedded within the mosses or litter. Male and female gametophytes differ in lobe shape (Williams 1950; Benson-Evans 1952; Wiehle et al. 1989).
The first structural work on the mycorrhiza in A. mirabilis by Denis (1919) revealed intracellular fungal colonization with hyphal coils in the ventral (lower) part of the thallus, although he considered the specimen as an albino of another chlorophyllous Aneura species. Von Malmborg (1933) observed hyphae growing through the seta into the sporangium and assumed that the fungus is distributed together with the spores. Williams (1950), unable to confirm this statement of von Malmborg (1933), published the first detailed investigations and provided drawings of the full life cycle, including the pattern of mycorrhizal colonization. The thallus lobes bearing antheridia or archegonia are devoid of hyphae; starch is deposited in the upper part of the thallus and around the gametangia. In an ultrastructural comparison of green hepatics and Aneura mirabilis (still called Cryptothallus), Pocock and Duckett (1984) confirmed the concentration of fungal colonization in the lower half of the thallus, but more recently, Ligrone et al. (1993) showed that the upper parts of the thallus can also become colonized in later stages. Rhizoids are also colonized, albeit in an uncoiled manner (Duckett et al. 1990). Most likely, these straight hyphae within rhizoids represent the connection to the external substrate. The carbon of this liverwort probably comes from surrounding beech (Fagus sylvatica) trees (Read et al. 2000; Bidartondo et al. 2003), with which it is connected via the mutual Tulasnella mycorrhizal fungus, although Ligrone et al. (1993) found dissimilar dolipore structures in the endophytes of birch (Betula spp.) and Cryptothallus. The fungal coils within the thallus cells eventually degenerate to dark masses (von Malmborg 1933; Williams 1950; Pocock and Duckett 1984), interpreted as digestion, and the cells can be reinfected (Ligrone et al. 1993). Williams (1950) and Pocock and Duckett (1984) stressed the difference in fungal identity between Aneuraceae hosting basidiomycetes and resembling orchid mycorrhiza, in contrast to other liverworts hosting “phycomycetous” (today considered as Glomeromycota, Schüßler et al. 2001) endophytes forming arbuscular mycorrhiza (AM) in higher plants. This fact, together with the identification of the fungi in A. mirabilis as Tulasnella spp. (Read et al. 2000; Bidartondo et al. 2003), has led to the hypothesis of a novel acquisition of Tulasnella spp. as associates in Aneuraceae. By attaining an epiphytic habit during phylogeny, liverworts may have lost the original symbiotic relationship with Glomeromycota. Secondarily terrestrial Aneuraceae then could have associated with new fungal partners (Kottke and Nebel 2005; Bidartondo and Duckett 2009).
4.3 Seedless Vascular Plants
Mycoheterotrophy in the seedless vascular plants is restricted to their gametophytic phase (Read et al. 2000; Smith and Read 2008). Genera possessing acholorophyllous gametophytes (and photosynthetic sporophytes) belong to Lycopodiaceae (e.g., Lycopodium, Huperzia, Fig. 4.1a, b), Ophioglossaceae (Ophioglosssum, Botrychium, Helminthostachys, Mankyua Fig. 4.1d–f), Psilotaceae (Psilotum, Tmesipteris, Fig. 4.1g, h), some species of Schizaea and Actinostachys in the Schizaeaceae, and the monotypic species Stromatopteris moniliformis in the Gleicheniaceae.
4.3.1 Lycopodiaceae (Fig. 4.1a–c)
It was recognized very early that subterranean gametophytes of several Lycopodium species are associated with endophytic fungi (Treub 1885, 1890; Bruchmann 1885, 1910; Lang 1899; Burgeff 1938). Illustrations in Burgeff (1938) and Boullard (1979) clearly show that fungi colonize the basal region of gametophytes shortly after spore germination. Mature subterranean gametophytes show variations in form from disc-shaped with convoluted margins (L. clavatum, Fig. 4.1b, L. obscurum) to elongated, cylindrical structures (L. complanatum = Diphasiastrum complanatum, Bierhorst 1971; Gifford and Foster 1996). Gametophytes of all species have fungal colonization restricted to a zone underlying more surficial cells that give rise to antheridia and archegonia (Bierhorst 1971, Fig. 4.1c).
Although the identity of the fungus was unknown in these early studies, it was described as being aseptate and forming intracellular hyphal coils. An ultrastructural investigation of the fungal endophyte in association with achlorophyllous gametophytes of L. clavatum showed that complex hyphal coils and vesicles formed but arbuscules were absent (Schmid and Oberwinkler 1993a). Entrance of the fungus occurred either through rhizoids, degenerated epidermal cells, or between epidermal cells. Once within parenchyma cells of the gametophyte, host-derived plasma membrane and wall material was deposited around invading hyphae. Hyphae were multi-nucleate and contained bacterium-like organelles (BLOs). Hyphae became progressively more vacuolated and ultimately degenerated. The authors came to the conclusion, based on a number of unusual structural features, that this fungus-gametophyte interaction was unlike anything described in the literature and could not be attributed to a known mycorrhizal association. They therefore proposed a new term “lycopodioid mycothallus interaction” to describe the association.
More recently, based on structural features of the fungi within cells, the fungal symbionts in the gametophytes of all seedless vascular plants were suspected to be members of the Glomeromycota and to have the Paris-type arbuscular mycorrhiza association (Read et al. 2000). Molecular studies have confirmed this for the fungus associated with two subterranean gametophytes of Huperzia hypogaea collected in Ecuador: the fungus was identified as belonging to a specific clade of Glomus-Group A (Winther and Friedman 2008). Observations of these sectioned gametophytes confirmed earlier reports that hyphal coils are restricted to the basal region and that arbuscules are not formed.
4.3.2 Ophioglossaceae (Fig. 4.1d–f)
Gametophytes of Ophioglossum may be cylindrical (O. nudicaule, O. vulgatum, Boullard 1957; Gifford and Foster 1996), globose (O. crotalophoroides, Mesler 1976), or highly branched (O. palmatum, Mesler 1975). Fungal hyphae may be evenly distributed but avoiding the meristematic area and gametangia (Bierhorst 1971). Fungal colonization occurs immediately after spore germination (Campbell 1908) and gametophytes do not develop unless they are associated with the appropriate fungus. Hyphal coils, some of which have undergone degeneration, are illustrated in gametophyte cells of O. pendulum (Burgeff 1938). Mesler (1975) described the endophytic hyphae in gametophytes of O. palmatum as being non-septate and multi-nucleate. He also showed what he interpreted as vesicles in some gametophyte cells. Mesler (1976) gave a similar description of the fungal endophyte in O. crotalophoroides. Details at the ultrastructural level are lacking for gametophytes of Ophioglossum spp. and the identity of the fungus remains unknown.
Gametophytes of Botrychium also vary in their morphology from being tuber-like to disc-shaped (Bruchmann 1906; Burgeff 1943; Gifford and Foster 1996; Winther and Friedman 2007); endophytic fungi are restricted to a basal zone of parenchymatous cells (Bruchmann 1906; Bierhorst 1971). The fungus in B. lunaria has been described as forming aseptate intracellular coils and irregular vesicles (Bruchmann 1906). An ultrastructural study of the fungus-gametophyte interaction of this species (Schmid and Oberwinkler 1993b) has provided additional details. The intracellular hyphae contain vacuoles, endoplasmic reticulum, mitochondria, and lipid-like bodies. They are enclosed by host-derived plasma membrane and wall material that shows irregular outgrowths. Vesicles, some very irregular in shape, contain BLOs, and lipids; they can become very enlarged and then undergo degeneration. The identity of the fungal endophyte has been determined for the subterranean gametophytes of B. crenulatum (Fig. 4.1f) and B. lanceolatum based on DNA sequence data (Winther and Friedman 2007). The endophytes in both species belong to a major clade of glomalean fungi, Glomus-group A.
A third genus in the Ophioglossaceae, Helminthostachys, is monotypic (H. zeylanica) and native to the Indo-Malayan region (Gifford and Foster 1996). It also forms achlorophyllous mycoheterotrophic gametophytes (Lang 1902) but little is known of the fungal association.
A new genus and species (Mankyua chejuense) has been described from Cheju Island, Korea (Sun et al. 2001) based on differences in sporophyte morphological characters from the other genera in the family. Gametophytes have not been described but are presumed to be subterranean.
4.3.3 Psilotaceae (Fig. 4.1g–j)
The two genera, Psilotum, with two species and Tmesipteris, with ten species, have historically been of considerable interest because of the belief that they represented some of the most primitive extant seedless vascular plants (Bierhorst 1971; Gifford and Foster 1996). The lack of roots and the presence of much reduced leaf-like structures of the sporophyte strengthened this view. However, based on molecular evidence, Smith et al. (2006) include this family within the Psilotales, an order belonging to the extant ferns.
Subterranean gametophytes of Psilotum are highly variable cylindrical structures (Fig. 4.1h) sometimes showing repeated branching (Bierhorst 1971). Asexual reproductive propagules (gemmae) are frequently developed (Bierhorst 1971). Darnell-Smith (1917) was the first to succeed in achieving spore germination and to monitor early stages in gametophyte development. He reported that endophytic fungi appeared as dense “skeins” within interior cells of gametophytes and that hyphae entered rhizoids. Other authors have described an aseptate intracellular fungus thought to be a phycomycete in either field-collected gametophytes (Burgeff 1938; Boullard 1957) or gametophytes growing in greenhouse pots containing various angiosperm species (Bierhorst 1953). Aspects of the ultrastructure of the gametophyte-fungus interaction have been described from gametophytes collected from greenhouse pots (Davis 1976; Peterson et al. 1981). The fungus in these gametophytes is aseptate and forms complex coils (Fig. 4.1i) that undergo degeneration; arbuscules are not formed. Intracellular hyphae are separated from the gametophyte cell cytoplasm by host-derived plasma membrane (perifungal membrane) and interfacial matrix material (Peterson et al. 1981, Fig. 4.1j), characteristics of arbuscular mycorrhizal associations (Bonfante and Perotto 1995). To date, the fungus has not been identified but the structural characteristics are typical of a Paris-type arbuscular mycorrhiza.
The fungal endophyte in subterranean gametophytes of Tmesipteris tannensis was described by Lawson (1917) and Holloay (1921) as consisting of intracellular fungal coils (pelotons). As with Psilotum gametophytes, the identity of the fungus has not been determined.
4.3.4 Schizaeaceae
The gametophytes of the genus (Schizaea) in this leptosporangiate fern family may either be surficial and green, subterranean and achlorophyllous, or a combination of both, depending on species and habitat (Bierhorst 1968, 1971). Gametophytes of all species are associated with endophytic fungi that have been described as aseptate and frequently associated with rhizoids (Bierhorst 1971; Swatzell et al. 1996).
Gametophytes of all species in the genus Actinostachys are axial structures that are subterranean with fungal hyphae confined to a distinctive zone (Bierhorst 1968). The identity of the fungi associated with achlorophyllous gametophytes in these two genera is unknown.
4.3.5 Gleicheniaceae
The monotypic genus Stromatopteris moniliformis (subfamily Stromatopteridaeae), has axial subterranean gametophytes with coiled fungal hyphae (Bierhorst 1971), reminiscent of Paris-type arbuscular mycorrhizas. Although Bierhorst (1971) concluded that the fungus present in the gametophyte is the same as that in the photosynthetic sporophyte, this needs to be confirmed with molecular methods.
Experimental evidence confirming transfer of nutrients from fungi to the subterranean gametophytes of all seedless vascular plants is lacking.
4.4 Gymnosperms
4.4.1 Podocarpaceae
The New Caledonian endemic Parasitaxus usta (not P. ustus, as many authors repeated the linguistically incorrect transfer from Podocarpus to the feminine genus Parasitaxus by de Laubenfels 1972) is a succulent shrub or small tree (up to 2 m high) with wine-red scale leaves (Cherrier et al. 1992; Schneckenburger 1999), unable to photosynthesize (Feild and Brodribb 2005) and only occurring closely associated with Falcatifolium taxoides (also Podocarpaceae, Sinclair et al. 2002). Root graft-like subterranean connections between the two species have led to the notion of parasitism in P. usta (de Laubenfels 1959; Köpke et al. 1981). However, Cherrier et al. (1992) and an English version of that paper adding a SEM micrograph (Woltz et al. 1994) found an endophytic mycelium (called “ectendomycelium”) in both species, together with haustorial-like connections apparent at the cellular level developing in tissues up to the cambium of F. taxoides. The authors assume a symbiotic association of the three partners but, based on their anatomical observations, are convinced of parasitism in this case. The latest investigation on P. usta confirms the intimate vascular association of both species, but results from stable carbon isotope investigations suggest that most carbon is provided by the fungus (Feild and Brodribb 2005). With respect to water physiology however, P. usta has higher stomatal conductance and lower water potential values relative to its host, which is typical for parasitic angiosperms (Feild and Brodribb 2005). Hence, apart from being a gymnosperm, woody, and relatively large, Parasitaxus is even more unique among heterotrophic organisms in possibly being a parasitic and mycoheterotrophic plant at the same time.
4.5 Monocots
4.5.1 Petrosaviaceae (Petrosavia)
The three species of Petrosavia are distributed from Japan to Java. The external morphology of the underground structures does not differ much among the species. Their subterranean rhizomes can be branched and thus, may bear several 10–15 cm high scapes with terminal racemes or corymbs of white flowers. Rhizomes measure up to 1.5 mm in diameter and are densely covered by sheathing scale leaves (Groom 1895a; Makino 1903; Stant 1970; Jessop 1979; Chen and Tamura 2000; Cameron et al. 2003). The filiform, hairless, approximately 0.5 mm thick and sparsely branched adventitious roots, are initiated from the rhizome, especially close to the base of the scape. They most likely originate from the axils of the scale leaves, as do the rhizome branches (Groom 1895a). In Petrosavia sakuraii, the roots predominantly grow horizontally through the substrate and can be up to 20 cm long (Watanabe 1944). This author also reports hyphae penetrating into the roots 2–5 mm proximal from the root tip.
The epidermis is either ephemeral (Groom 1895a) or partly persistent (Stant 1970). The cortex consists of a suberized exodermis, 4–6 layers of parenchyma cells, and an endodermis with particularly strong u-shaped tertiary thickenings surrounding the tetrarch central cylinder (Groom 1895a; Watanabe 1944; Stant 1970). This is similar to many mycoheterotrophic Burmanniaceae (Johow 1889; Uphof 1929) with Dictyostega orobanchoides as an extreme example (Imhof 2001, Fig. 4.4f). Watanabe (1944) mentions that segments of older roots lose the cortex parenchyma but remain connected to the rhizome by the central cylinder that is surrounded by the fortified endodermis. The maintenance of connectivity between roots and rhizomes bearing inflorescences is particularly important for MH plants having filiform roots, since not only water and nutrients but also carbohydrates must be transported through these comparatively long structures. A tertiary endodermis, a synapomorphy of monocots (Esau 1965), seems to be less costly than the production of layers of lignified tissue, which is the equivalent option for non-monocots in order to protect the connectivity. This economical advantage of monocots may be part of the explanation as to why monocots include disproportionately so many MH plants (Imhof 2010).
Previous investigations on Petrosavia (Groom 1895a; Watanabe 1944; Stant 1970) report coiled mycorrhizal hyphae within the cortex parenchyma cells. The figures and descriptions of Watanabe (1944) resemble a Paris-type AM but without the typical lateral arbuscules, which is similar to the mycorrhiza found in Voyria truncata (Gentianaceae, Imhof and Weber 1997). The advantage of the frequent feature of MH plants of having a specialized mycorrhizal colonization pattern allowing a selective digestion of hyphae while keeping the fungus alive (see further), is not apparent. Petrosaviaceae are a rather basal clade of the monocots (Cameron et al. 2003; APG 2009), which might explain its plesiomorphic, i.e., basic mycorrhizal pattern. Most recently, Yamato et al. (2011a) confirmed the structural descriptions of Watanabe (1944), and revealed this mycorrhiza as an association with a highly specific clade of Glomus-group A.
4.5.2 Thismiaceae (Figs. 4.2 –4.4 and 4.10)
Thismiaceae are either considered to be a tribe, Thismieae, in the Burmanniaceae (e.g., Jonker 1938, Cronquist 1988) or a separate family (e.g., Agardh 1858; Thorne 1992; Takhtajan 1997; Stevens 2001 onwards). APG (2009) is still reluctant to separate them from Burmanniaceae but acknowledge the arguments for separation given by Merckx et al. (2006). We regard them as a family based on floral morphology (e.g., Maas et al. 1986; Caddick et al. 2000) and molecular evidence (Merckx et al. 2006).
4.5.2.1 Haplothismia, Oxygyne, Tiputinia
The extremely rare Haplothismia exannulata from India has vermiform to tuberous, up to 3.5 cm long, roots radiating from the shoot base (Airy Shaw 1952; Sasidharan and Sujanapal 2000). For Oxygyne triandra from Cameroon (probably extinct, Yokoyama et al. 2008), the subterranean organs are unknown. The Japanese species, O. shinzatoi and O. yamashitae, only known from their type localities, have vermiform roots (Yokoyama et al. 2008; Yahara and Tsukaya 2008), and the original description of O. hyodoi, also from Japan, states “rhizoma repens” (Abe and Akasawa 1989). Tiputinia foetida is represented by a single specimen from Ecuador (Woodward et al. 2007), measuring about 9 cm in length. The largest part of it is an orthotropous, vermiform, 4 mm thick root, giving rise to two subterranean shoots, with only the terminal flower being epiterrestrial. The root cortex contains “intracellular, looped, septate” hyphae (Woodward et al. 2007).
4.5.2.2 Thismia (Fig. 4.10g, h)
This genus is by far the largest of the family, with a worldwide, although mostly tropical, distribution. The underground structures are quite variable. Most species have horizontal runner-like, vermiform roots of 1–2 mm in diameter which bear root-borne shoots (e.g., Groom 1895b; Warming 1901; Pfeiffer 1914; Chantanaorrapint 2008; Chiang and Hsieh 2011) and give rise to additional similar roots where the shoots emerge, thus forming star-like clusters (e.g., Groom 1895b; Bernard and Ernst 1910; Pfeiffer 1914; Larsen 1965; Saunders 1996; Yang et al. 2002; Wapstra et al. 2005). This indicates the trend towards a star-like radiating root system, typical for MH plants. The runner-like parts of the roots can be short (e.g., Thismia appendiculata, Schlechter 1919), and in this case, the shoots emerge in nest-like tufts above the soil surface. In other species, the root system is reduced to a coralloid structure, e.g., Thismia yorkensis (Cribb 1995), T. goodii (Kiew 1999), or T. clandestina and T. versteegii (Bernard and Ernst 1911). Thismia versteegii shows similarities to the unique fan-shaped roots of Thismia clavigera (Stone 1980), which probably develop through short, dichotomously branched and congenitally merged roots. Thismia annamensis and T. tentaculata have short rhizomes bearing a dense covering of vermiform roots (Larsen and Averyanov 2007), also resulting in a star-like root system. This is morphologically similar but ontogenetically quite different from the other species mentioned above. The decision whether a condensed root system is developed by root-borne shoots or shoot-borne roots is often difficult to make and sometimes requires anatomical investigations (see Imhof 2004). Finally, some neotropical species have globose tubers (see Fig. 4.10g), from which a shoot as well as numerous filiform roots arise (e.g., Thismia hyalina, Miers 1866, T. glaziovii, Poulsen 1890a, T. janeirensis, Warming 1901, T. panamensis, Maas et al. 1986, Fig. 4.8j, T. saülensis, Maas and Maas 1987). Inferred from T. luetzelburgii, these tubers are roots, giving rise to up to four endogenous flowering shoots. The filiform roots can develop new tubers at their apices (Goebel and Süssenguth 1924).
The fungal colonization of Thismia spp. has been investigated quite early and in great detail. Like many other MH plants, Thismia also shows different fungal morphologies in distinct tissue compartments, which sometimes are anatomically different. In T. clandestina (coralloid root system) and T. aseroe (vermiform roots), the outer cortex parenchyma layers contain straight hyphae with only a few coils; in the middle cortex layers, the hyphae are coiled but not digested; the inner layers show amorphous fungal material (Groom 1895b; Janse 1896; Meyer 1909; Bernard and Ernst 1911). In T. americana, T. rodwayi, and T. javanica (vermiform roots), straight hyphae are missing, instead, the outer cortex layer is occupied by coiled hyphae which do not degenerate. Digestion takes place in the inner cortex (Bernard and Ernst 1910; Pfeiffer 1914; Coleman 1936; McLennan 1958; Campbell 1968). Of the species having root tubers, T. luetzelburgii (Goebel and Süssenguth 1924) and T. glaziovii (Poulsen 1890a) have been investigated. They also show compartmentation of digested and undigested fungal material, whereas the digestion is more prominent in the proximal and central part of the tuber. The filiform roots connecting the mother tuber with smaller daughter tubers bear straight undigested hyphae linking the two tubers (Goebel and Süssenguth 1924). This is partly reminiscent of structures found in Afrothismia spp. (see next paragraph).
Due to the structural characteristics typical of a Paris-type AM, the fungus colonizing Thismia spp. almost certainly belongs to the Glomeromycota. This has recently been confirmed for T. rodwayi using molecular identification methods (Merckx et al. 2012).
4.5.2.3 Afrothismia (Figs. 4.2–4.4)
The genus Afrothismia from tropical Africa is characterized by its dense aggregates of small tuberous roots elongated by a filiform extension of various lengths between the species (Figs. 4.2g + j, 4.3a + c, and 4.4b). Although our chapter deals with subterranean organs, this is not entirely correct for some Afrothismia spp. In fact, the peculiar root/rhizome/shoot systems often grow entirely epiterrestrially (Fig. 4.2f), the filiform part of the roots clinging to rotten wood, leaf litter, or bare soil (e.g., A. foertheriana, Franke et al. 2004, A. hydra, Sainge and Franke 2005, A. winkleri, Imhof pers. observ., Fig. 4.2i–j). Only A. baerae (Cheek 2003a) and A. gesnerioides (Imhof pers. observ., Fig. 4.4a) are known to be rooted in the soil. The latter two species also differ by their conspicuously short filiform parts of the roots (Fig. 4.4b, Cheek 2003a; Maas-van de Kamer 2003). Afrothismia zambesiaca, described from a herbarium specimen collected in 1955, is only inferred to have an underground stem with bulbils (Cheek 2009). The ontogeny of Afrothismia hydra from seed to the open fruit has been described (Imhof and Sainge 2008), and since structurally the genus is quite consistent, this example will be detailed here to represent the whole genus. With germination, a root tubercle without a filiform extension is generated (Fig. 4.2a). Successively, more tubercles develop on a rhizome that is gradually increasing in size and the root extensions elongate (Fig. 4.2b–d). This creates a globose to ovate, coarsely echinate structure due to the characteristic roots. At some point, the rhizome proceeds to grow without root development. This axis, now more accurately called a shoot, will terminate with a flower (Fig. 4.2e), and a side shoot appears in the uppermost scale leaf of the shoot. The base of this side shoot also bears a cluster of tubercles with extensions, and this shoot will also end in a flower. This sympodial pattern is repeated several times (Fig. 4.2g). The fruit is a pyxidium, opening by means of a placentophore (Fig. 4.2h, see details in Imhof and Sainge 2008).
The fungal colonization of Afrothismia saingei is an extreme example of mycorrhizal complexity (Imhof 1999a, treated as A. winkleri Footnote 1). Briefly, the pattern of colonization is as follows (schematic view on Fig. 4.3i). The filiform root extension bears straight hyphae, continuous with those in the epidermis of the tubercle (Fig. 4.3d + h). These hyphae never pass from the epidermis into the cortex of the tubercle but proceed around it towards the rhizome. As soon as the fungus reaches the rhizome at the tubercle base, it colonizes the rhizome cortex tissue with coiled, swollen, vesicle-like structures, but still does not show signs of degeneration (Fig. 4.3e). From there, few hyphae re-enter the tubercle from the rhizome cortex, and grow towards the subepidermal layer of the tubercle (Fig. 4.3e). Characteristic loops of hyphae are developed in the subepidermal cells (Fig. 4.3b + g), and an upward spiral line of cells containing such looped hyphae proceed around the tubercle (Fig. 4.3a). No digestion of hyphae occurs to this stage. Side branches from these hyphal loops enter the other cells of the tubercle cortex, where they degenerate to amorphous clumps (Fig. 4.3g, h). Connections to more distant tubercle clusters along the plant are provided by straight growing hyphae in the outer cortex of the shoot internodes (Fig. 4.3f, see details in Imhof 1999a). This complicated plant structure and colonization pattern represent a sophisticated and ecologically functional system. The filiform root extensions increase the surface for contact with and invasion by hyphae, the root tubercle increases the number of cells for colonization by hyphae and eventual digestion, representing the locations of the beginning and end of the mycorrhizal colonization pattern. Between these events, the different hyphal forms serve three fundamental functions: (1) transportation and distribution of carbohydrates and nutrients within the root-rhizome-complex, (2) storage, and finally (3) as a carbon source for the plant following digestion. The straight hyphae in the filiform root extension and the epidermis allow for rapid transport of nutrients and carbohydrates towards the rhizome. The swollen hyphae in the rhizome cortex store these substances, eventually for the benefit of the plant. The spiral line of hyphal loops is the geometrically and economically optimal distribution mode around the parenchyma of the tubercle. With a minimum of living hyphae, this provides short distances and limits the number of cell passages for side branches to penetrate into all parenchyma cells, necessary due to the quick degeneration process therein. The fungus in Afrothismia gesnerioides shows a similar colonization pattern with straight hyphae in the short root extension (Fig. 4.4e) and the root epidermis (Fig. 4.4f, g), as well as digestive tissue in the inner root parenchyma (Fig. 4.4c, see details in Imhof 2006). However, it does not develop a spiral line of hyphae around the tubercle parenchyma. Instead, dense coils of living irregular hyphae develop in the third root layer, encompassing the parenchyma in a collar-like pattern (Fig. 4.4c + f–h). Economically speaking, this pattern is less efficient than that in A. saingei, considering the amount of living fungal biomass necessary to supply the digesting cells with hyphal branches. Moreover, the rhizome of A. gesnerioides contains straight growing hyphae in its outer cortex, whereas the inner cortex cells contain starch deposits (Fig. 4.4c), as does the uncolonized tubercle cortex (Fig. 4.4d + h, schematic view on Fig. 4.4i). This means that A. gesnerioides, in contrast to A. saingei, converts the carbon delivered by the fungus into starch grains. In the case of Afrothismia spp. however, this appears as a unnecessary metabolic step, since the carbon source is permanently present. Therefore, although the mycorrhizal patterns in Afrothismia spp. are highly complex, they still show signs for an ongoing evolutionary progression of mycorrhizal structures within the genus, whereas A. gesnerioides can be considered to be less advanced than A. saingei. More of the 12 Afrothismia species described so far should be investigated to determine if intermediate structures exist (see 4.8 Trends, Conclusions, and Future Directions).
The fungal species associated with Afrothismia spp., as identified by molecular methods, all belong to Glomus-group A (Franke et al. 2006), and are species-specific (Merckx and Bidartondo 2008).
4.5.3 Burmanniaceae (Figs. 4.5–4.7)
Of the ten genera in this family, only Burmannia contains green representatives. Burmannia tenella is the only entirely achlorophyllous neotropical species, others occur in Africa (e.g., B. hexapterella) and Asia (e.g., B. championii, B. candida). However, many species with intermediate mycoheterotrophic status, between leafy Burmannias and achlorophyllous, scale-leaved species exist (Jonker 1938; Maas et al. 1986; Leake 1994), suggesting an evolutionary trend towards mycoheterotrophy. All other genera are fully mycoheterotrophic. The monotypic genus Desmogymnosiphon (Guinea Lopez 1946) is most probably a Gymnosiphon species (compare to Maas et al. 1986).
4.5.3.1 Apteria, Campylosiphon, Dictyostega, Gymnosiphon, Hexapterella, Marthella, Miersiella (Figs. 4.5 and 4.6)
Except for Campylosiphon congestus and the pantropical Gymnosiphon, all these genera are exclusively neotropical (Jonker 1938; Maas et al. 1986). All species have the same basic architecture for their underground parts. The aerial shoots are continuous with rhizomes, densely covered by scale leaves. These scale leaves are conspicuously fringed in Dictyostega (Imhof 2001, Fig. 4.5g), which has led to the hypothesis they might ecologically replace the missing root hairs (Goebel and Süssenguth 1924; Maas et al. 1986). The rhizomes can be longer (up to 7.5 cm in e.g., Miersiella umbellata, Maas et al. 1986, up to 4 cm in Dictyostega orobanchoides, Imhof 2001, Fig. 4.5c) or rather short (e.g., Apteria aphylla (Fig. 4.6c), Uphof 1929, Gymnosiphon longistylus, Hepper 1968, G. divaricatus (Fig. 4.6d), Maas et al. 1986, Hexapterella gentianoides (Fig. 4.6g)), and can be slightly tuberous (e.g., Campylosiphon purpurascens, Maas et al. 1986, Campylosiphon congestus, Fig. 4.6i–k). Many filiform, less than 0.5 mm thick, sparsely branched roots arise from the axils of the scales. Species with short rhizomes, therefore, have a star-like root system (Fig. 4.6c, d + g), but roots also emerge as tufts on longer rhizomes (Imhof 2001, Fig. 4.5c). As a peculiar exception in this group of species, Gymnosiphon afro-orientalis develops little tubers of unknown origin beside scale leaves and filiform roots at the short rhizome (Cheek 2009), superficially reminiscent of those found in Afrothismia (e.g., Fig. 4.4b), but fundamentally differing in being distinct from the filiform roots.
Anatomically, these roots are characterized by a much reduced central cylinder with one central enlarged tracheary element surrounded by a ring of much smaller tracheary elements, and a pericycle (e.g., Fig. 4.5f). The tertiary endodermis is conspicuously reinforced (e.g., Marthella trinitatis, erroneously called Burmannia capitata by Johow 1885, Gymnosiphon refractus (formerly Cymbocarpa refracta, Merckx 2008), treated under two different synonyms by Johow 1889 and Goebel and Süssenguth 1924, Apteria aphylla, Uphof 1929). In transverse sections of a Dictyostega orobanchoides root, the fortification of a single endodermal cell may even be wider than the entire central cylinder (Imhof 2001, Fig. 4.5f). This reinforcement protects the essential connection to the shoot. In fact, the thin-walled cortex tissue is often found to be disrupted (Figs. 4.5d and 4.6c, d + g) whereas the central strand is even hard to disconnect using forceps (Imhof 2001, see section on Petrosavia for interpretation).
The two to three parenchyma layers and, in particular, the often large-celled persistent epidermis (Johow 1889; Imhof 2001 and unpublished observations) are colonized by coils of hyphae (Uphof 1929), vesicles, as well as arbuscular-like structures, often all together within a single cell (Imhof 2001). The fungal material often appears amorphous, suggesting a digestion process (Fig. 4.5e, f).
Dictyostega orobanchoides also has fungal colonization in the scale leaves (Fig. 4.5h) as well as in the interstitials of their imbricate arangement along the rhizome (Fig. 4.5g–i), but not in the rhizome axis. These hyphae and vesicles do not show signs of degeneration, and it has been hypothesized that they serve as a refugium for the fungus, which in turn enhances the rhizomosphere with the appropriate mycobiont (Imhof 2001, see Fig. 4.5j for a schematic view). It can be interpreted as a strategy for a sustained use from the fungus, analogous to the often complex colonization pattern in other MH plants (e.g., Voyria, Afrothismia, Triuris, Sciaphila). More investigations might clarify the possible general relevance of rhizomes and their scale leaves for the mycorrhiza in other Burmanniaceae.
Franke et al. (2006) found several Glomus-group A species and an Acaulosporaceae in Campylosiphon congestus (treated as Burmannia congesta). Also, Dictyostega orobanchoides is associated with Glomus-group A species (Merckx et al. 2010), as are Apteria (Courty et al. 2011) and Gymnosiphon spp. (Dirk Redecker, pers. comm. cited in Leake 2005; Courty et al. 2011).
4.5.3.2 Burmannia (Fig. 4.7)
Burmannia species are more diverse with respect to their subterranean structures than their sister genera. Although they are sometimes similar to the latter (e.g., B. championii, Ernst and Bernard 1911), they also can have thicker roots also arising from rhizomes (e.g., B. larseniana, Zhang and Saunders 1999) or even vermiform, up to 2.6 mm thick roots and no (visible) rhizomes (e.g., Burmannia candida, Smith 1911, B. liukiuensis, Terashita and Kawakami 1991, B. tenella, Imhof 1999b, Fig. 4.7b, B. hexaptera, Imhof unpublished, Fig. 4.7i). Others have tuberous organs of uncertain nature (Burmannia hunanensis, Liu et al. 2001), with filiform roots. However, more taxonomic investigation may result in new classifications resolving some of this subterranean diversity, as in fact, Burmannia congesta, having a tuberous rhizome, only recently was attributed to Campylosiphon (Fig. 4.6i–k) by molecular and morphological data (Merckx 2008; Maas-van de Kamer and Maas 2010).
Root anatomy is also diverse. Epidermal cells may be conspicuously enlarged (Johow 1889; Ernst and Bernard 1911, 1912; Bernard and Ernst 1914) or not (Colozza 1910; Ernst and Bernard 1911; Imhof 1999b, Fig. 4.7d). Depending on the variability of root thickness, the cortex parenchyma layers can be from three to many (Janse 1896; Ernst and Bernard 1911; Larsen 1963; Terashita and Kawakami 1991; Imhof 1999b, Fig. 4.7d), and can be uniform (Imhof 1999b) or heteromorphic (Ernst and Bernard 1911) or with lacunae (Johow 1889; Malme 1896a; Colozza 1910). Similar to the other genera of the family, the endodermis has obvious tertiary reinforcements and the central cylinder is much reduced (e.g., Malme 1896a; Ernst and Bernard 1911; Terashita and Kawakami 1991; Imhof 1999b, Fig. 4.7d).
In species with filiform roots, the mycorrhizal fungus colonizes epidermal cells (Johow 1889; Ernst and Bernard 1911), whereas in the species with thick roots, the cortex parenchyma cells are colonized (Meyer 1909; Ernst and Bernard 1911; Terashita and Kawakami 1991; Imhof 1999b, Fig. 4.7d). In the thick roots of Burmannia tenella, hyphal coils, vesicles and arbuscular-like structures may occur together in a single cell (Fig. 4.7e, f ). A colonization pattern with compartmentation of root tissue similar to other MH plants is not obvious (Fig. 4.7d). However, a selective digestion of the thinner, arbusculate hyphae but not the thicker hyphae within cells (Imhof 1999b, Fig. 4.7e, f ), seems to allow a sufficient spread of the colonization within the cortex parenchyma by the latter, while carbon and nutrients are obtained through digestion of the former.
The only Burmannia species which has been investigated for the identity of its mycorrhizal fungus is B. hexaptera (Fig. 4.7g–i). It is mycorrhizal with Glomus-group A species (Franke et al. 2006; Merckx and Bidartondo 2008).
4.5.4 Triuridaceae (Figs. 4.8–4.10)
Fossil specimens of this exclusively achlorophyllous family from the Upper Cretaceous (ca. 90 mya) are the oldest unequivocal monocotyledonous remnants known (Gandolfo et al. 2002). Eleven genera are grouped in three tribes, the Sciaphilae are pantropical, Triurideae neotropical (Maas-van de Kamer and Weustenfeld 1998), and Kupeaeae only occur in tropical Africa (Cheek 2003b). All genera except for Sciaphila and Andruris (included in Sciaphila by van de Meerendonk 1984) contain only one to three species. The affiliation of the family was long uncertain (Rübsamen-Weustenfeld 1991; Maas-van de Kamer 1995; Maas-van de Kamer and Weustenfeld 1998). Today, molecular methods have assigned them to the Pandanales, which is supported by structural features (Furness et al. 2002; Rudall and Bateman 2006).
With only few exceptions, the subterranean organs of Triuridaceae are rather uniform. The epiterrestrial shoots are continuous plagiotropically to orthotropically with subterranean shoot segments with various internode lengths without increasing in diameter (Fig. 4.8a + c, d). In addition to occasional side shoots, the axils of the nodal scale leaves bear pairs (sometimes solitary) of long filiform roots about as thick as the shoots (e.g., van de Meerendonk 1984; Maas and Rübsamen 1986; Maas and Maas-van de Kamer 1989), which can be glabrous (e.g., Triuris hyalina, Imhof 1998, Triuridopsis intermedia, Franke et al. 2000, Fig. 4.8a + c–e), sparsely hairy (e.g., several Sciaphila spp., Schlechter 1913, Lacandonia schismatica, Martinez and Ramos 1989) to conspicuously pilose (e.g., Soridium spruceanum, Miers 1852. several Sciaphila spp., Johow 1889; Hemsley 1907; Larsen 1972, Andruris spp., Schlechter 1913, Seychellaria madagascariensis, Fig. 4.10c). Usually the scale leaves and, consequently, the root pairs are spaced along the subterranean shoot (Fig. 4.8a + c), but there can also be dense clumps of filiform roots seemingly radiating from a single origin (e.g., several Sciaphila, Triuris, and Peltophyllum spp., Larsen 1972; Maas and Rübsamen 1986, Fig. 4.8d, e + j, Seychellaria madagascariensis, Fig. 4.10c), sometimes occurring in two or three tiers along the subterranean shoot (Fig. 4.8d). There are also a few species with more stout roots but also showing a star-like arrangement at the base of the shoot, namely the three species of the Kupeaeae (Cheek et al. 2003; Cheek 2003b, Fig. 4.10d–f), but also Sciaphila polygyna (Imhof 2004, Fig. 4.9a–d). Sciaphila ledermannii (Fig. 4.8i) has an intermediate root thickness (Fig. 4.8j). The star-like root aggregations by filiform or stout roots, even if they appear superficially very different, all follow the same developmental pattern, that is maximally one pair of shoot-borne roots per node, but are formed by the initiation of a side shoot from the scale leaf axil directly bearing a next node with scale leaf, giving rise to another pair of roots and a side shoot and so forth. The side shoots often do not elongate, which explains the abundance of roots (see details in Imhof 1998, 2004).
The tendency towards aggregations of thick and short roots seems to be characteristic for mycoheterotrophic plant families (Leake 1994, Imhof 2010, this chapter). Hence, the quite recent discovery of this feature in the Triuridaceae (Cheek et al. 2003; Imhof 2003, 2004, see Figs. 4.9d and 4.10f) was not too surprising.
The root anatomy of Triuridaceae is quite uniform also. Internal to the epidermis, there is a suberized exodermis (Fiebrig 1921; Imhof 1998, 2003) and two (Johow 1889; Tomlinson 1982), three (Fiebrig 1921; Imhof 1998), to several (Imhof 2003) cortical parenchyma layers. The endodermis and/or pericycle may be reinforced (Poulsen 1886, 1890b; Johow 1889, Milanez and Meira 1943; Larsen 1963; Tomlinson 1982) or not (Malme 1896b; Imhof 1998, 2003). The central cylinder is much reduced. Very characteristic is the second cortex parenchyma layer, which mostly consists of conspicuously enlarged cells (Poulsen 1886, 1890b; Johow 1889; Fiebrig 1921; Tomlinson 1982; Imhof 1998, 2003), with the exception of Sciaphila thaidanica according to Larsen (1963, Figs. 4.8f, g and 4.9e).
Mycorrhizal colonization was recognized very early (e.g., Poulsen 1886, 1890b; Johow 1889; Janse 1896), with additional information added later (Fiebrig 1921; Ohga and Sinoto 1932; Milanez and Meira 1943; Palacios-Mayorga and Pérez-Silva 1993), but details of the colonization pattern were described rather recently (Imhof 1998, 2003; Franke 1999). Triuris hyalina attains a sustained benefit from the endophytic fungus by maintaining the hyphae in a functional state in the first cortical parenchyma layer and digesting them only in the enlarged cells of the second parenchyma layer (Fig. 4.8f–h, see details in Imhof 1998). An unpublished diploma thesis on Sciaphila purpurea (Franke 1999) not only yielded detailed information on morphology, anatomy, and ecology of the reproductive parts, but also confirmed the distinction of undigested hyphae in the outer vs. the digestion of hyphae in the inner enlarged cells of the root cortex. Beyond that, the structural diversity of the mycorrhizal colonization pattern in Sciaphila polygyna (Fig. 4.9a–d) is much more complicated and certainly belongs to the most complex mycorrhizas known (Fig. 4.9e–h). It includes four different morphologies of hyphae occurring in four distinct root tissue compartments. Moreover, it shows a disparate colonization at the tip compared to the base as well as the dorsal vs. the ventral side of the root, creating a monosymmetrical (only one plane of symmetry) root in transverse and longitudinal sections (Fig. 4.9h, see details in Imhof 2003). The purpose of these complex structures, except for the strictly localized digestion in the “giant cells” for a sustained carbon influx (Imhof 2003), is not yet understood.
The fungus in Sciaphila secundiflora (Yamato 2001, treated as S. tosaensis, the two treated as being synonymous by Ohashi 2000) was determined by DNA sequencing to be a Glomus species (Glomeromycota). More recently, S. secundiflora (still called S. tosaensis) and Andruris japonica (treated as Sciaphila japonica) were described to associate with Glomus-group A fungi (Yamato et al. 2011b), the phylotypes extracted from each species being closely related to another but quite distant when compared between the two species. Sciaphila ledermannii was also found to be colonized by a species from Glomus-group A as well as by an Acaulospora sp. (Franke et al. 2006), whereas Merckx and Bidartondo (2008) detected only a Glomus-group A fungi in a specimen of S. ledermannii from Mount Cameroon. Kupea martinetugei was associated with two closely related fungi of Glomus-group A (Franke et al. 2006), confirmed by Merckx and Bidartondo (2008).
4.5.5 Corsiaceae (Fig. 4.11)
4.5.5.1 Arachnitis
Roots of the inconspicuous plant Arachnitis uniflora (Corsiaceae, Fig. 4.11a), one of two unusual mycoheterotrophs in the genus confined to a few locations in the southern hemisphere (Dimitri 1972; Cribb et al. 1994; Ibisch et al. 1996; Domínguez and Sérsic 2004), are short and fleshy, radiating from the shoot base, and lack root hairs. Reiche (1907) described colonization of peripheral parenchyma cells in roots by endotrophic mycorrhizal fungi whereas Colozza (1910) called the plant “parassita,” adding “fors’anche saprofita?” (= perhaps saprophytic?) in brackets. More recently, Minoletti (1986) referred to the colonization pattern as an ectendomycorrhiza because both intercellular and intracellular hyphae were present in the outer cortex of roots. Molecular methods have proven that roots are colonized by an AM fungus belonging to Glomus-Group A (Bidartondo et al. 2002). However, details of the structural characteristics of the plant-fungus interaction are unlike other plant associations with Glomus spp. (Domínguez et al. 2006, 2009). Unusual branched structures with inflated ends (Fig. 4.11c–g) form in addition to hyphal coils (Fig. 4.11d) in the cortical cells of the plant’s fleshy roots. Arbuscules do not form and vesicles (Fig. 4.11g) rarely occur. The function of the branched structures is unknown but they, along with the hyphal coils, may be involved in the transfer of sugars from fungus to root cells (Domínguez et al. 2009). Arachnitis uniflora also develops unusual asexual propagules (Fig. 4.11b) on its fleshy roots (Domínguez et al. 2006) that are colonized by fungi from the parent root before they detach. The propagules develop a shoot apical meristem and adventitious roots and ultimately new plants that presumably link to neighboring photosynthetic plants for their source of carbon (Domínguez et al. 2006, 2009)
4.5.5.2 Corsia
Only a general description of roots in Corsia species is given by van Royen (1972); no further specific information can be found in the taxonomic section of his monograph. The roots are filiform, unbranched, white to cream-colored, growing horizontally through the humus layer. Compared to the entire plant, they are “quite sizable” and “extend over considerable distance in many directions” (van Royen 1972). They arise from short, creeping rhizomes, with sheathing scale leaves (Williams 1946; van Royen 1972). Beccari’s (1877) and Schlechter’s (1905) drawings, however, show some root branches in Corsia ornata and C. unguiculata, respectively. Similarly, Cribb (1985), without discussing them, depicts branching roots in C. pyramidata, also arising from branching rhizomes a few centimeters below the soil surface. Jones and Gray (2008), describing the only Australian species C. dispar, explained this discrepancy as an oversight by van Royen, since many herbarium sheets of Corsia spp. in the herbarium of Canberra have branched roots as well. According to Jones and Gray (2008), the rhizome in C. dispar is about 4 mm thick and grows in annual increments. In contrast to the information given by van Royen (1972), Cribb (1985) and Jones and Gray (2008), a new variety C. purpurata var. wiakabui (Takeuchi and Pipoly 1998), later considered to be a separate species (Jones and Gray 2008), has a conspicuously tuberous rhizome bearing the roots (interpreted from the drawing in Takeuchi and Pipoly 1998). This is partly reminiscent of the third genus of the family, Corsiopsis, discussed below. No anatomical studies exist on this genus which could elucidate its mycorrhiza.
4.5.5.3 Corsiopsis
The monotypic Corsiopsis chinensis is only known from a single herbarium specimen (Zhang et al. 1999). The original description is of an ellipsoid rhizome 12–15 mm long and 5 mm in width, the drawing showing it in an orthotropous orientation. Roots were not seen.
4.5.6 Orchidaceae (Fig. 4.12)
The family Orchidaceae has the largest number of mycoheterotrophic genera of any plant family, with approximately 35 % of more than 500 fully mycoheterotrophic angiosperm species recognized (Leake 1994; Merckx et al. 2009; Imhof 2010). It is impossible to characterize the subterranean structures of all mycoheterotrophic orchid species (see Rasmussen 1995 for a thorough discussion) but a few examples will demonstrate the variability. Some species (e.g., Cyrtosia javanica) have rhizomes bearing fleshy adventitious roots, others (e.g., Epipogium aphyllum; Corallorhiza spp., Rhizanthella garderi) with rhizomes only, and others (e.g., Wullschlaegelia calcarata) with roots, some of which are modified as tubers.
Regardless of the nature of the underground structures, the majority of achlorophyllous orchid species are associated with fungi that form intracellular hyphal coils (pelotons) similar to those in photosynthetic orchids. These can develop within the majority of root or rhizome cortical cells and sometimes even in scale leaf tissue (Groom 1895c) and have an ephemeral existence since they undergo digestion by host cells (Smith and Read 2008). This process, termed tolypophagy (Burgeff 1932), can be repeated with recolonization by pelotons of cells containing hyphal remnants and subsequent digestion of these. Often, the cortex parenchyma is divided into an outer “Pilzwirtsschicht” (fungus host layer), where the coils do not degenerate, and an inner “Pilzverdauungsschicht” (fungus digestion layer), where digestion takes place (Magnus 1900; Burgeff 1932). Moreover, in some mycoheterotrophic species (e.g., Gastrodia spp.), a process called ptyophagy occurs (Burgeff 1932; Wang et al. 1997; Rasmussen 2002). While keeping the fungus host cell layers, this is characterized by only short hyphae penetrating the single-layered and particularly voluminous digestion cells and releasing their contents into it without coiling (Janse 1896; Burgeff 1932; Campbell 1962, 1963, 1964). As such, it very much resembles the “hyphal pegs” in monotropoid mycorrhizas (Lutz and Sjolund 1973; Duddridge and Read 1982). It is open to speculation if this can be interpreted as an evolutionary progression within orchid mycorrhiza, from non-differentiated colonization pattern (see e.g., Peterson et al. 2004), over the tissue compartmentation in host and digestion layers, to the ptyophagy as a special type of the latter in few MH orchids. More structural work is needed to elucidate this, but since arbuscular mycorrhizas and ectomycorrhizas seem to have undergone evolutionary progression (Imhof 2009), it would be surprising if this is not the case in orchid mycorrhizas.
Because of their “dust seeds,” consisting of a rudimentary embryo and limited storage reserves, all orchid species (Fig. 4.12a) growing in native habitats require a suitable fungal partner to germinate and for the subsequent development of the protocorm (Peterson et al. 1998, 2004). The intracellular fungal hyphal coils (pelotons) are essential features for metabolite transfer into developing protocorms (Fig. 4.12b) and roots (Fig. 4.12c). All orchid species can, therefore, be considered to be mycoheterotrophic during this early stage of their life cycle (Leake 2004). The fungi involved are basidiomycete anamorphs such as Ceratorhiza, Epulorhiza and Moniliopsis which are capable of enzymatically reducing complex carbohydrates to simple sugars that are not only used for fungal growth but are also transferred to developing protocorms to enable seedling establishment to occur.
Although the majority of orchid species develop photosynthetic adult plants, a considerable number of genera remain dependent on mycorrhizal fungi for carbon compounds throughout their life cycle and therefore continue to be mycoheterotrophs. In these situations, developing seedlings link to photosynthetic plant species via fungal mycelium, mostly belonging to members of the Basidiomycota (Taylor and Bruns 1997). Zelmer and Currah (1995) demonstrated that the fungus isolated from roots of Corallorhiza trifida, although not identified, formed pelotons in Corallorhiza trifida root cells (Fig. 4.12d) and typical ectomycorrhizas with lodgepole pine (Pinus contorta, Fig. 4.12e). It was recently demonstrated by Zimmer et al. (2008) that the fungal symbiont associated with C. trifida is a Tomentella sp. (Thelephoraceae). Another example in which seeds of Neottia nidus-avis (also a non-photosynthetic orchid) were enclosed in seed packets and placed either near adult plants or at some distance from them in a beech (Fagus sylvatica) woodland, McKendrick et al. (2002) were able to show that the genus Sebacina (anamorph, Epulorhiza) was the symbiont involved in the stimulation of seed germination. Adult plants of the mycoheterotroph Neottia nidus-avis remain associated primarily with the basidiomycete family Sebacinaceae (Selosse et al. 2002) whereas Cephalanthera austinae (another mycoheterotroph) associates with members of the Thelephoraceae (Taylor and Bruns 1997). Recently, Ogura-Tsujita and Yukawa (2008) reported the extreme specificity of the mycoheterotrophic orchid Eulophia zollingeri with the fungal symbiont, Psathyrella candolleana in the Agaricales (Basidiomycetes). In contrast, mycoheterotrophic species within the genus Epipactis have been reported to associate not only with members of the Basidiomycota but also with members of the Ascomycota, including Tuber (truffle) species (Selosse et al. 2004).
4.5.7 Iridaceae (Geosiris, Fig. 4.10g, h)
Unlike the other larger families comprising both autotrophic and mycoheterotrophic species (Orchidaceae, Burmanniaceae, Gentianaceae, Polygalaceae, Ericaceae), Iridaceae do not comprise morphologically intermediate species with reduced photosynthetic surface or amount of chlorophyll. Geosiris aphylla (Fig. 4.10g, h) and the recently described G. albiflora (Goldblatt and Mannings 2010) are the only mycoheterotrophic exceptions in the entire family. Systematically, Geosiris has been treated as a member of Iridaceae, Burmanniaceae or a family of its own (see Rübsamen-Weustenfeld et al. 1994). Within the Iridaceae, it has been considered as Nivenioideae (Goldblatt 1990; Goldblatt et al. 1987, 1998), but recently a position in its own subfamily Geosiridoideae, as suggested earlier (e.g., Thorne 1983), has been confirmed (Goldblatt et al. 2008).
Geosiris aphylla has an orthotropous, corm-like, oval to elongate rhizome with numerous scale leaves. The flowering shoots arise from the apical tip of this tuber-like organ, whereas at its base numerous filiform roots develop (Fig. 4.10g), similar to the base of onion bulbs. Anatomically, the rhizome consists of a wide cortex parenchyma surrounding a vascular cylinder with occasional gaps due to leaf and bud traces. A thin-walled epidermis and a fortified endodermis around the vascular cylinder are present and some isolated amphivasal (xylem around phloem) bundles are found in the ground tissue of the central pith. The parenchyma cells, particularly in the pith, contain starch grains (Goldblatt et al. 1987). The roots also have a thin-walled epidermis, the cortex has four layers of parenchyma cells and a strongly fortified tertiary endodermis (Goldblatt et al. 1987). As pointed out previously (see Petrosavia), this again corroborates the view of Imhof (2010), who considers a strong tertiary endodermis as one of the common adaptations of monocotyledonous MH plants in order to secure the essential linkage of roots and shoots.
The only anatomical work on G. aphylla (Goldblatt et al. 1987), aside from Rübsamen-Weustenfeld et al. (1994) studying embryology, does not mention any fungal colonization of roots or rhizomes. This is rather curious, since a carbon source for this non-photosynthetic plant is mandatory. A parasitic mode of life (sensu Weber 1993) is highly unlikely because, in contrast to the roughly 4,500 eudicotyledonous parasitic plants, monocots have never been found to be parasitic (Raynal-Roques and Paré 1998; Heide-Jørgensen 2008). Possibly, Goldblatt et al. (1987) may have overlooked the mycorrhizal structures. Therefore, further anatomical investigations focusing on the putative mycorrhiza of this species are necessary.
4.6 Eudicots
4.6.1 Polygalaceae (Epirixanthes, Fig. 4.13)
Epirixanthes from Southeast Asia is the only genus in the Polygalaceae entirely devoid of chlorophyll, although there are other species in Polygala and Salomonia (e.g., Polygala setacea (southeast USA) and Salomonia ciliata (Southeast Asia and northern Australia) that also show reductions in photosynthetic surface). The taxonomic accounts (e.g., Smith 1912; Ridley 1922; Backer and van den Brink 1963; van der Meijden 1988; Hsieh et al. 1995; Pendry 2010) of the six species of Epirixanthes do not yield information on the subterranean organs. However, there are two older and one contemporary study on the mycorrhizal roots of E. papuana, E. elongata, and E. cylindrica (Penzig 1901; van der Pijl 1934; Imhof 2007).
The rhizome of E. papuana and E. elongata is only a few millimeters long and continuous with the aerial shoot. The scale leaf axils give rise to sparsely branched filiform roots that are up to 12 cm long and have a maximum diameter of 0.65 mm (Imhof 2007, Fig. 4.13a + d). A primary root was never found. Since the rhizome is short, the roots seem to be radiating from the shoot base, but additional roots may develop along the shoot when it is still covered by soil or litter substrate (Imhof 2007, Fig. 4.13a). Epirixanthes cylindrica has thicker roots (up to 0.75 mm diameter) and the rhizome bearing the roots is longer (Penzig 1901). Roots of Epirixanthes have a triarch central cylinder with many lignified fibers, a pericycle, a suberized endodermis, up to seven cell layers of cortex parenchyma, and an epidermis (Fig. 4.13h). The cells of the innermost cortex parenchyma layer are larger in radial but shorter in longitudinal direction than the other cells (Penzig 1901; van der Pijl 1934; Imhof 2007, Fig. 4.13e, f + h).
Penzig (1901) recognized the coiled intracellular hyphae, their degeneration stages, especially in parenchyma layers 1 and 3 when counted outwards from the endodermis, and the nearly fungus-free outer cortex layers. Van der Pijl (1934) added the observation that the hyphae in layer two grow in longitudinal direction and send hyphal branches into the inner layer for digestion. The whole colonization pattern, however, is more complicated and only perceivable when considering sequential sections. After penetration, the hyphae grow straight through the cells of the outer cortex, branch repeatedly and spread coarsely in this root segment until they reach layer 2 (Fig. 4.13e, layer 1 being the innermost cortex parenchyma layer). There the hyphae keep growing straight (Fig. 4.13f), but later develop hyphal branches that coil within the cells (Fig. 4.13f, g). Directly after having reached layer 2, lateral hyphae enter the anatomically distinct layer 1 where they immediately swell and degenerate (Fig. 4.13e, f). In a later stage, layer 3 is also colonized from coiled hyphae in layer 2, again with immediate degeneration in layer 3 (Fig. 4.13g, h). In contrast, the hyphae in layer 2 as well as the straight hyphae in the outer cortex, remain alive for nearly the lifetime of the root (Fig. 4.13g, h). This rather complicated colonization pattern is interpreted as a reasonable strategy in order to have maximum as well as sustained benefit from the few fungal penetration events. It includes a coarse (outer cortex colonization) as well as a fine scale distribution mode (colonization in layer 2), specialized cells for digestion, and tissue to keep the fungus alive (schematic view on Fig. 4.13i, details see Imhof 2007).
4.6.2 Ericaceae (Monotropoideae, Figs. 4.14–4.17)
Eleven genera of mycoheterotrophic species are now recognized in the Monotropoideae: Allotropa, Cheilotheca, Hemitomes, Hypopitys, Monotropa, Monotropastrum, Monotropsis, Pityopus, Pleuricospora, Pterospora and Sarcodes, with several species endemic to a particular continent (Wallace 1975). Molecular phylogeny work has revealed that Monotropa uniflora is more closely related to Monotropastrum humile, whereas Monotropa hypopitys seem to be sister of Pityopus californicus (Bidartondo and Bruns 2001; Tsukaya et al. 2008). Therefore, some standard taxonomies (Stevens et al. 2004; Seybold 2011) have erected Hypopitys as a separate genus from Monotropa, and now use Hypopitys monotropa coined by Crantz (1766).
The minute seeds of members of the Monotropoideae have underdeveloped embryos and minimal nutritive tissue and therefore depend on the presence of a suitable fungus to provide sugars and perhaps other nutrients needed for germination and seedling establishment (Bruns and Read 2000; Leake et al. 2004; Smith and Read 2008). Massicotte et al. (2007) have shown that, in Pityopus californicus, fungal hyphae become associated with germinating seeds and form a mantle as the embryo begins to elongate. Later, a mantle, Hartig net, and fungal pegs form in the developing root. Mycoheterotrophy is therefore established very early in the life cycle of these plant species. Their root systems vary among species, ranging from large root balls comprised of numerous mycorrhizal roots (e.g., Monotropa, Pterospora), to more diffuse root systems with mycorrhizal roots distributed more randomly (e.g., Pleuricospora, Monotropsis, Allotropa). Hirce and Finocchio (1972) described in detail the remarkably compact root system and anatomy of M. uniflora and concluded that it represents a variation of the normal dicotyledonous condition. They documented a decrease in anatomical complexity of first order (hexarch stelar configuration of vascular tissue, several centimeters long and up to 1.4 mm thick, linking adjacent plants) over second order (up to 8 mm long and 0.85 mm thick) to the third order roots (protostelic arrangement, max. 4 mm long and 0.5 mm thick), even if all roots are densely covered with a mantle (and are presumably active). At the other extreme, Allotropa exhibits a more loose system of elongated rhizomes with first and second order adventitious roots (Massicotte et al. 2010), and likewise in Monotropsis odorata (treated as Cryptophila pudica) “the root system resembles a slender, mostly repeated many-branched, creeping rhizome” (Wolf 1922). Compared to Allotropa, Pleuricospora seems to have a slightly more condensed subterranean system (Massicotte et al. 2010).
In the Monotropoideae (formerly Pyrolaceae), a progressive compaction of the root system, from fibrous roots (e.g., Allotropa) to coralloid roots (e.g., Pleuricospora) to tight rootballs (e.g., Monotropa) has been hypothesized as reflecting a progressive dependence on epiparasitic mycotrophy (Furman and Trappe 1971), although this remains to be tested physiologically.
Structurally, monotropoid mycorrhizas resemble ectomycorrhizas in that a mantle and a Hartig net in this case confined to the epidermis, form (Peterson et al. 2004). However, they possess a unique feature, the invasion of epidermal cells by short hyphae originating from the Hartig net or inner mantle. These structures, referred to as fungal pegs (Lutz and Sjolund 1973; Duddridge and Read 1982; Robertson and Robertson 1982; Peterson and Massicotte 2004; Peterson et al. 2004) form either along the outer tangential wall of epidermal cells, or at the base of the radial wall of epidermal cells. Host cells respond by depositing additional cell wall material, in finger-like projections, around each peg. It has been hypothesized (Lutz and Sjolund 1973; Duddridge and Read 1982; Massicotte et al. 2005) that these structures, resembling “transfer cells” in other plant species, may be involved in nutrient transfer between the fungus and root cells although there is no experimental evidence to support this. In these systems, the Hartig net likely also plays a role in nutrient transfer but this needs to be confirmed. Kuga-Uetake et al. (2004) have shown the close association of microtubules with the fungal pegs in M. uniflora.
All of these species form monotropoid mycorrhizas with various fungal genera. The vast majority of fungi colonizing monotropoid roots are basidiomycetes and most of them have been identified using molecular approaches (Cullings et al. 1996; Lefevre 2002; Kretzer et al. 2000; Bidartondo and Bruns 2001, 2002). In the following paragraphs, we explore these critical features for five genera of Monotropoideae.
4.6.2.1 Monotropa uniflora (Fig. 4.14)
Monotropa uniflora, a northern hemisphere species (Fig. 4.14a), along with the Asian Monotropastrum humile (Yokoyama et al. 2005; Yamada et al. 2008; Matsuda et al. 2011) have a strong affinity for fungi in the family Russulaceae, including many species of Russula such as R. brevipes, R. decolorans, R. nitida, as well as Lactarius spp. (Young et al. 2002, Bidartondo 2005; Bidartondo and Bruns 2005; Yang and Pfister 2006). Hypopitys monotropa (=Monotropa hypopitys), in contrast, forms mycorrhizas mostly with fungal species in the Tricholomataceae. Excavated plants of M. uniflora reveal well-developed root balls (Fig. 4.14b), packed with mycorrhizal tips of Russulaceae (Fig. 4.14c), most forming numerous cystidia in the outer mantle (Fig. 4.14d). Scanning electron microscopy of the mantle surface has shown that cystidia can take various forms including, awl-shaped (Fig. 4.14e) and flask-shaped (Fig. 4.14f). As well, frequent calcium-oxalate crystals may be present (Fig. 4.14g). The mantle (Fig. 4.14h, i) and Hartig net (Fig. 4.14h) are easily observed but sectioning of mycorrhizal roots is required to show the presence and structure of fungal pegs (Fig. 4.14j–l).
4.6.2.2 Pterospora andromedea (Fig. 4.15)
Pterospora andromedea (Fig. 4.15a) and Sarcodes sanguinea (not shown), are confined to western North America and appear to associate almost exclusively with the section of Rhizopogon (Rhizopogonaceae) encompassing R. ellenae, R. salebrosus and R. arctostaphyli (Kretzer et al. 2000; Bidartondo and Bruns 2001, 2002; Taylor et al. 2002; Dowie et al., 2011). Large root balls (Fig. 4.15b) dominated with Rhizopogon mycorrhizas (Fig. 4.15c) are evident on excavated plants. A compact mantle with crystal inclusions of variable dimensions and shapes (Fig. 4.15d–f) is characteristic of the mycorrhizas of this species when viewed by scanning electron microscopy. Light microscopy shows a thick mantle, a well-developed Hartig net (Fig. 4.15g) and a fungal peg apparatus penetrating the radial epidermal cell wall (Fig. 4.15h).
4.6.2.3 Allotropa virgata and Pleuricospora fimbriolata (Fig. 4.16)
Allotropa virgata (Fig. 4.16a) forms mycorrhizas exclusively with Tricholoma magnivelare (Lefevre 2002; Taylor et al. 2002), and is one of the most specific host-fungal monotropoid symbiosis documented so far. Light microscopy reveals sporadic colonization at the root surface with a thin mantle (Fig. 4.16b) and a fungal peg penetrating radial walls of epidermal cells (Fig. 4.16c, d). Pleuricospora fimbriolata (Fig. 4.16e) parasitizes the fungal species Gautieria monticola (Bidartondo and Bruns 2001, 2002), a truffle forming species belonging to the Gomphaceae (Humpert et al. 2001). Typically, a well-developed mantle envelops the root (Fig. 4.16f) and fungal pegs, penetrating the outer tangential walls of the epidermis (Fig. 4.16g), are obvious. Characteristic finger-like wall depositions are found on the fungal peg (Fig. 4.16h).
4.6.2.4 Pityopus californicus (Fig. 4.17)
Pityopus californicus (Fig. 4.17a) also forms mycorrhizas mostly with fungal species in the Tricholomataceae, in this case Tricholoma myomyces (Bidartondo and Bruns 2005). However, a developmental study on young mycorrhizal embryos of P. californicus suggests other fungi are present in earlier stages (Fig. 4.17b) that are presumably replaced at later stages by T. myomyces (Fig. 4.17c, Massicotte et al. 2007). Mature mycorrhizal roots typically show a thick mantle (Fig. 4.17d + f), a well-developed Hartig net and a fungal peg, penetrating the outer tangential wall of epidermal cells (Fig. 4.17e). Small finger-like projections can be seen on the fungal peg (Fig. 4.17g).
4.6.3 Gentianaceae (Figs. 4.18–4.20)
In the Gentianaceae, 25 species in four genera are mycoheterotrophic. Additional to the genera covered here, two others are also considered to be at least partially “saprophytic” (Johow 1889; Knoblauch 1894; Gilg 1895; Holm 1897, 1906; Perrot 1898; Wood and Weaver 1982). The monotypic Obolaria virginica has scale-like leaves along the lower stem with larger spatulate-obdeltoid leaves towards the inflorescence. The fleshy stem and leaves are purplish-green. The roots are coralloid and mycorrhizal (Holm 1897; Gillett 1959; Wood and Weaver 1982), like many of the Voyria species described below. Bartonia comprises four species, B. virginica, B. verna, B. paniculata, the latter of which has two subspecies (Gillett 1959), and B. texana (Correll 1966). All species have only scale leaves but an overall greenish appearance. Compared to Obolaria virginica, the Bartonia species are more delicate, but also have fleshy, sparsely branched mycorrhizal root systems (Holm 1906). Recent physiological investigations using a stable isotope distribution approach found strong indications for a partial mycoheterotrophy in Obolaria virginica and Bartonia virginica (Cameron and Bolin 2010).
4.6.3.1 Exacum
Cotylanthera was originally a genus comprising four achlorophyllous species, which was suspected to be closely related to the large genus Exacum (e.g., Raynal 1967a, Klackenberg 1985, 2002, Yuan et al. 2003); the genus has now been formally transferred into Exacum by Klackenberg (2006). The taxonomic accounts (e.g., Miquel 1856; Gray 1871; Clarke 1885; Gilg 1895; Lace 1914; Hara 1975) mostly do not refer to the subterranean parts, but there are two rather old but quite detailed morphological descriptions of the roots for Exacum tenue (Janse 1896; Figdor 1897, treated as Cotylanthera tenuis). Janse (1896) described the roots as tufted around the stem base. Such star-like root systems are a common feature of most MH plants and interpreted as a strategy to decentralize carbohydrate and nutrient transport: a root system with many but hierarchically equivalent roots can better compensate for the failure of some of its elements than few but high-capacity roots, as is the case in most allorhizic root systems (Imhof 2010). Combining the information given by Figdor (1897) and Imhof et al. (1994), the former showing a seedling of E. tenue, the latter describing the ontogeny of another gentian (Voyria tenella), the star-like root system of E. tenue is probably generated by a primary root, developing ray-like lateral roots, and only then does this structure give rise to a root-borne shoot. Later, further root-borne shoots are a means of vegetative propagation. Root hairs, if present, are much reduced (Figdor 1897). A considerable portion of the stem, up to half its length, may also be subterranean, often bent due to obstacles in the soil (Figdor 1897). Similar observations were made in Voyria truncata (Imhof and Weber 1997) and Triuris hyalina (Imhof 1998). The roots are several centimeters long and irregularly thickened, with the thicker parts three (Figdor 1897) to four times (Janse 1896) wider than the thinner ones. Tangential cell divisions in the outer cortex increase the number of parenchyma cell layers in those parts where fungal hyphae are found within the inner cortex cells. Uncolonized root segments, thus, have only four parenchyma layers whereas the mycorrhizal parts can have up to eight (Janse 1896). Cells in the course of division do not become colonized, neither does the epidermis, exodermis nor the first cortical parenchyma layer. The hyphae within the cells are coiled (Figdor 1897) and show local swellings. They form “sporangioles,” a term used by Janse (1896) for degenerating hyphae. Figdor (1897) also speaks of clumped masses of dead hyphae. A prolonged primary thickening of the root as described by Janse (1896) has also been seen in Voyria truncata, although there the cell divisions happen throughout the cortex, irrespective of being colonized or not (Imhof and Weber 1997).
No information is given as to whether the subterranean part of the stem is colonized by the fungus. For comparison, more recent studies in Voyria species did not find hyphae in shoot tissues (Imhof and Weber 1997, 2000; Imhof 1997, 1999c), although Svedelius (1902) reports hyphae in aerial stems of V. tenella (treated as Leiphaimos azurea).
4.6.3.2 Exochaenium (Fig. 4.18i–k)
Exochaenium oliganthum (previously Sebaea oligantha Kissling et al. 2009; Kissling 2012) from Central Africa is the only species in this genus that is achlorophyllous (Raynal 1967a, Kissling et al. 2009). However, representatives with little photosynthetic surface such as E. debilis, E. rara and E. pulsilla (Marais and Verdoorn 1963) suggest that partial mycoheterotrophy may also be present in other species of the genus. As is often the case, the subterranean organs are neglected in taxonomic accounts (Gilg 1899; Robyns 1962). Raynal (1967a), recognizing this lack, described the roots as radiating from the stem base, being few (her drawing shows five at the shoot base), sparsely branched, terete, carnose, and up to 0.5 mm thick (Fig. 4.18i, j). This description is very similar to that for achlorophyllous Exacum species discussed above. More exciting is Raynal’s (1967a) finding of additional, almost subterranean cleistogamous flowers covered by the leaf litter of the soil surface. In contrast to the straight epiterrestrial shoots, the stems and pedicels of these “subterranean” flowers are mostly positively geotropic, coiled, and intermingled with each other, but basically develop the same flowers and fruits as the aerial counterparts, except for some reductions in floral structures. The fruits, due to positive geotropism, are geocarpic (Raynal 1967a).
Information on the roots additional to that mentioned above is very scarce. Professor Mangenot did investigate the mycorrhiza of Exochaenium oliganthum, but never published it. Raynal (1967a) reports his findings communicated to her, which revealed fungal coils within the cells very similar to the conditions in Neottia nidus-avis (Orchidaceae). However, we know today that orchid mycorrhizas (e.g., Smith and Read 2008) and the AMs in gentians (e.g., Imhof 1999c) are only superficially alike. Recently, molecular methods have detected a Glomus-group A endophyte in Exochaenium oliganthum, which seems to be highly specific for this species (Franke et al. 2006).
4.6.3.3 Voyriella
There are roughly 30 publications mentioning the monotypic Voyriella, many of them only as part of an enumeration of gentianaceous genera. The approximately dozen of taxonomic or geographic accounts mostly lack information on subterranean organs. In fact, there are only five statements on the roots of Voyriella parviflora: “radice fibrosa” (Miquel 1851), “roots on the transition from filiform to coralloid shape” (Johow 1889, translated from German), “filiform” (Jonker 1936), and “30 mm long and 0.3 mm thick” (Maas and Ruyters 1986). The latter statement is simply restated by Pires O’Brien (1997) in a plant checklist of the Jari river in Brazil. There is no figure showing the roots of V. parviflora, but its mycorrhizal fungus has been identified by 18S rDNA sequencing. V. parviflora seem to be highly specific to a basal clade of Glomus-group A of the Glomeromycota (Bidartondo et al. 2002).
4.6.3.4 Voyria (Figs. 4.18–4.20)
There is considerable information available for the roots of several of the 19 Voyria species. The earliest observations by Aublet (1775) on Voyria rosea indicated that it has irregularly tuberous roots of the size of a fist and become eaten by the indigenous people (Garipons of the Guianas) after being cooked in a coal fire, tasting similar to potatoes. However, although V. rosea has roots up to 40 mm long and 15 mm (!) thick, it appears more as a loosely coralloid root system rather than a tuberculous one (Maas and Ruyters 1986). Either the specimen seen by Aublet had densely intermingled roots only appearing like a tuber of that size, or Aublet confused it with another plant. Aublet (1775) called the achlorophyllous genus after the Garipon name of that edible plant, Voyria. There is still some confusion on the nature of the subterranean parts of Voyria spp. as they are sometimes erroneously called rhizomes (e.g., Süssenguth 1937; Jonker 1936; Fukarek et al. 1994; Pringle 1995). Possibly, since the root-borne shoots (Imhof et al. 1994; Imhof and Weber 1997; Imhof 1997) often have to grow through a considerable layer of soil before they reach the surface (Fig. 4.19b–d, Imhof and Weber 1997, see before under Exacum), they might have been misinterpreted as orthotropous rhizomes.
Considerable differences, most probably representing evolutionary steps, occur within this genus. Paralleled by the reduction of floral (Oehler 1927; Maas and Ruyters 1986) and shoot anatomical features (Johow 1885; Solereder 1908; Oehler 1927; ter Welle 1986), the root systems can also be arranged according to reduction particularly in root length. Roots of Voyria truncata (a primitive member of the genus), can presumably be several meters long, growing horizontally and runner-like (Fig. 4.19e) as deep as 20 cm beneath the soil surface (Fig. 4.19c, d), and up to 2 mm thick, frequently branched, and give rise to two root-borne shoots in the axils of side roots (Imhof et al. 1994; Imhof and Weber 1997, Fig. 4.19e). Hence, seemingly distinct specimens above soil can well belong to the same individual, either by shoot ramifications already in the soil (Fig. 4.19c), or because of different root sprouts originating from the same root (Fig. 4.19e). Field observations recognized V. truncata shoots (Fig. 4.19a–d) emerging like a chain of beads for several meters, denoting the root course in the soil (Imhof et al. 1994). Aublet (1775) also reports on roots of V. rosea being a foot deep in the soil.
In contrast, the most advanced representative of the genus, Voyria tenella, has a small, star-like root system, shallowly rooted or only loosely connected to the litter substrate (Imhof et al. 1994, Fig. 4.20a–e). Ontogenetic studies revealed a root to appear first after germination (Fig. 4.19b) and only after a small star-like root system form does a first shoot arise (Imhof et al. 1994, Fig. 4.20c). In spite of the ray-like appearance, the roots do not grow evenly in all directions. In fact, there is a single pole of growth representing the primary root which develops side roots at very short distances creating the globose structure (Imhof 1997).
In conclusion, although the two root systems of V. truncata and V. tenella seem to be very different, the root system of V. tenella is easily interpreted as an extremely abbreviated root system of V. truncata. This notion is supported by intermediate root systems linking V. truncata and V. tenella, e.g., V. aphylla (Imhof 1999c, Fig. 4.19h–j), V. rosea (Maas and Ruyters 1986), V. chionea (Progel 1865), and V. obconica (Imhof and Weber 2000, Fig. 4.18d, e). The only African representative, V. primuloides, which is considered to be sister to V. chionea (Albert and Struwe 1997), is the only species in the genus with prominent root hairs (Raynal 1967b).
The root anatomy and mycorrhizal colonization patterns in Voyria also support the progression proposed above. Voyria truncata has an almost identical root anatomy to a young Gentiana lutea (Perrot 1898), including a distinct dimorphic exodermis with short and long cells (Fig. 4.19f, g) and a substantial central cylinder with lignified tracheary elements, but lacking secondary thickening. Instead, a prolonged primary thickening is established (Fig. 4.19f), which constantly enhances the essential tissue for fungal colonization and is interpreted as an important adaptation to its mycoheterotrophy (Imhof and Weber 1997). The type of arbuscular mycorrhiza is also very similar to other gentians (Neumann 1934; Jacquelinet-Jeanmougin and Gianinazzi-Pearson 1983), except for the lack of lateral arbuscules formed from the coiled intracellular hyphae, typical for a Paris-type AM. The degradation of hyphal coils in the cells, best explained as a digesting process of the plant to absorb carbon and nutrients from the hyphae, happens after about 15 cell passages (Imhof and Weber 1997, Fig. 4.19f).
The roots of V. aphylla (Fig. 4.19h, i) have all anatomical elements of V. truncata but are reduced in their size and there is a tendency for a radiating formation of side roots at the shoot bases (Fig. 4.19i, j). The mycorrhizal associations are also similar. However, some new features have been acquired: (1) a longitudinal spread of straight hyphae within the epidermis as well as the innermost cortex layers (Fig. 4.19k, l) and (2) the development of root hairs only where roots of neighboring plants are attached (Imhof 1999c, Fig. 4.19m). Whereas the hyphae in roots of V. truncata are largely restricted regarding the cortical spread, the nondegenerating hyphae in the epidermis of V. aphylla are able to reach more distant segments of the root as well. Direct hyphal bridges from attached roots of neighboring plants are frequent sources of fungal root penetrations in several Voyria species. The locally developed root hairs of V. aphylla increase this contact zone and, in fact, receive most of the external fungal penetrations (Imhof 1999c; Fig. 4.19k + m). Hyphal passage through the exodermis still is exclusively via the short cells, although those are not as anatomically distinct as they are in V. truncata (Imhof and Weber 1997). Digestion of fungal material takes place in the cortex parenchyma except for its innermost layers, where a still imperfect internal spread along the central cylinder can be seen. The latter feature foreshadows the highly efficient colonization pattern of the further derived Voyria tenella, V. obconica and V. flavescens (Fig. 4.20i).
The root anatomy of the most advanced V. tenella had been investigated by Johow (1885) and Vigodsky-de Philippis (1938, under the synonym Leiphaimos brachyloba). Both stress the reduced character of the vascular system, the lack of suberization of the endodermis in V. tenella, and the voluminous root cortices (more details in Imhof 1997, Fig. 4.20a–e). Johow (1885) also recognized the coiled fungal mycelium within the cortex cells, but called it “parasitic” and, in accepting Drude’s hypothesis (1873), assigned its presence to the attraction through a “particularly rich flow of organic nutrients” (translated from German) he assumed to be a compulsory attribute of the roots of this “saprophytic” plant. He was aware of Kamienski’s (1882) new notion of a symbiotic association between fungus and plant, yet with cautious criticism. Vigodsky-de Philippis (1938) called the hyphae a “micelio micorrhizico.” However, these classical papers did not recognize the specialized colonization pattern. Root penetrating hyphae initially grow straight towards the innermost cortex layers and proceed in longitudinal direction along the reduced central cylinder. From there, hyphal branches grow back into the outer cortex parenchyma where they begin to coil, quickly inflate, and finally collapse into amorphous clumps (see details in Imhof 1997; Imhof and Weber 2000; Franke 2002, Fig. 4.20f–h). By this means, a sustained use of only few external penetrations of the fungus is attained, maintaining the hyphae alive in the inner cortex and only digesting branches of them in the outer cortex: an intraradical fungus garden (Imhof 1997, Fig. 4.19i). In summary, within Voyria, we can retrace not only morphological and anatomical reductions but also the evolutionary progression of a mycorrhiza (compare Figs. 4.19g, n and 4.20i), resulting in a highly efficient system to benefit from a fungus.
The mycorrhizal fungi of several Voyria species have been determined by molecular identification methods. Almost all of the endophytes belong to Glomus-group A of the Glomeromycota. However, Voyria spp. seem to not be as specific regarding their mycorrhizal associates as other mycoheterotrophic species (Bidartondo et al. 2002; Merckx et al. 2010).
Since mycoheterotrophic plants are very difficult to cultivate, the unexpected emergence of a Voyria species in the Botanical Garden in Hamburg (Germany) shall be briefly reported here. As an epiphytic stowaway on the trunk of a tree fern (Alsophila salvinii) imported in the mid seventies, a yellow Voyria (perhaps V. aphylla, which is known to grow also epiphytically, Groenendijk et al. 1997) was discovered around 1980. Unfortunately, it died in 1983, when its host fern tree was placed outside during summer (Poppendieck 1997).
4.7 Selected Species of Questionable Trophic Status
4.7.1 Buxbaumia spp. (Bryophyta, Fig. 4.1k)
The genus Buxbaumia is comprised of 12 species in the northern hemisphere (Crosby et al. 2000; Goffinet et al. 2008). In contrast to the majority of mosses, the up to 3 cm high sporophyte is the prominent phase of this genus and consists of a bulky, oblique-oval capsule on top of the seta (Eastwood 1939). This appearance has led to its enchanting vernacular names, e.g., Elfcap Moss, Humpbacked Elves, Bug-on-a-stick (Fig. 4.1k). While most species have greenish sporophytes (Udar et al. 1971; Ligrone et al. 1982; Stone 1983; Düll and Düll-Wunder 2008), the sporophytes of Buxbaumia aphylla and B. minakatae (Okamura 1911; Iwatsuki and Sharp 1967) seem to be largely devoid of chlorophyll. The gametophyte of Buxbaumia aphylla is minute and achlorophyllous (Goebel 1892; Dening 1928; Mueller 1972; Hancock and Brassard 1974). The possibly perennial protonema (Steven and Long 1989), consisting of single-lined threads of thick walled cells (Mueller 1972), which may form velvety mats (McClymont 1950), is green but of questionable trophic relevance (Haberlandt 1886; Eastwood 1939). Early stages of the sporophyte may show some green color (Goebel 1892; Dening 1928; Eastwood 1936; Hancock and Brassard 1974; Schoepe and Philippi 2000; van Rompu and Stieperaere 2002), but many recent as well as older publications consider it to have a heterotrophic mode of life (e.g., Haberlandt 1886; Eastwood 1936; Mueller 1972; Watson and Dallwitz 2005 onwards; Düll and Düll-Wunder 2008). It is clear therefore that the relation of auto- vs. heterotrophy in the whole genus is completely unknown and should be studied.
In contrast to Eastwood (1939), who believed in direct absorption by Buxbaumia of organic substances from humus or neighboring green mosses, we know today that plants are unable to do that directly, but are either parasitic (Kuijt 1969; Weber 1993) or mycoheterotrophic when lacking chlorophyll (Leake 1994, 2005). However, no information is yet available on mycorrhizal, endophytic, or parasitic associations, which could possibly explain the strange habit of these mosses. The ultrastructural investigation on the green B. piperi did not find any association with fungi (Ligrone et al. 1982). Neither did the detailed description (Stone 1983) on the foot and vaginula of B. novae-zelandiae mention fungal hyphae, but explained the dense indumentum with anastomosing elements as “rhizoidal outgrowth from the epidermal cells of the fertile axis.” Udar et al. (1971) also show drawings of longitudinal and transverse sections of the tomentose and slightly tuberous basis of the sporophyte of B. himalayensis, which superficially resembles an ectomycorrhiza. Unfortunately, the authors did not comment on this feature at all. However, Haberlandt (1886) has shown drawings of what he interpreted to be rhizoids. These “rhizoids” could well be fungal hyphae, and Haberlandt (1886) even explicitly remarked on their striking similarity to hyphae, particularly because of their frequent anastomoses, which, as far as we know, is not characteristic for moss rhizoids. Similarly, Goebel (1892) reports of anastomosing as well as achlorophyllous protonemata, which even show clamp-like connections (line drawing in Goebel 1892). In any case, contemporary studies providing photographic micrographs instead of drawings on the micromorphology and anatomy of Buxbaumia aphylla as well as stable isotope investigations (see Chap. 8) in order to elucidate the trophic mode, are urgently needed.
4.7.2 Pyrola picta (Pyrola aphylla, Ericaceae)
Pyrola aphylla was first described by James Edward Smith 1814 in Abraham Rees’ Cyclopaedia (Vol. 29, No. 7), calling it “leafless.” Other authors as well, such as de Candolle (1838) and Hooker (1840), described this species as having no leaves. However, Nuttall (1843, cited by Holm 1898) detected leaves of this species, and Holm (1898) ascertained subterranean shoots connecting apparently leafless specimens with rosettes of green leaves, proving that they belong to the same individual. Also Andres (1914) noted that P. picta can have few to no leaves. The same observation was made by Camp (1940), who revisited herbarium sheets and argued for a close relationship, if not identity, of Pyrola aphylla, P. picta and P. dentata. More recently, Haber (1987) eventually merged the three species within the highly variable Pyrola picta Sm.
In addition to the subterranean shoots (stolons) already mentioned, P. picta also has horizontally growing, runner-like, branching roots. Root-borne shoots as well as adventitious roots from the stolons can develop (Holm 1898). The only specific investigations on the mycorrhiza of P. picta is by Largent et al. (1980), calling it arbutoid and ericoid (both seen in specimens of P. picta var. picta) and ericoid (in P. picta var. aphylla). Other authors have called the mycorrhizas of Pyrola spp. arbutoid (Robertson and Robertson 1985; Massicotte et al. 2008; Vincenot et al. 2008), ectendomycorrhizal (Wang and Qiu 2006), or pyroloid (Cullings 1996). It also has been considered as a linking mycorrhizal type between arbutoid and ericoid mycorrhiza in a new classification of mycorrhizas (Imhof 2009).
Because P. picta has green leaves, and the extreme, leafless variant of P. picta, P. aphylla, still has chlorophyll in the shoot bark (Holm 1898), it actually should not be fully mycoheterotrophic. However, Hynson et al. (2009) found characteristic stable isotope signatures typical for mycoheterotrophic plants in Pyrola aphylla specimen. Interestingly, Pyrola picta with leaves, although being the same species taxonomically, did not show signs of mycoheterotrophy according to carbon stable isotope signatures (Hynson et al. 2009). Hence, the trophic status of this species is ambiguous. Possibly, dependency on the fungal carbon is not determined by the species in the taxonomical sense but on the actual ability for assimilation in a particular specimen.
4.8 Trends, Conclusions, and Future Directions
MH plants have distinctive structural necessities in contrast to autotrophic species due to their mycorrhizal dependence for carbon supply. Secondary growth of roots, for example, is deleterious since it sheds the primary tissue, which alone can host the indispensable mycobiont. Moreover, the primary tissue, less important in autotrophic plants, must be present in sufficient quantity. Most importantly, intracellular (in contrast to intercellular) mycorrhizal colonization is a major prerequisite. In fact, there is no mycoheterotroph having an Arum-type AM or an ectomycorrhiza, both of which are characterized by predominantly intercellular hyphal growth. Obviously, the transfer of nutrients and carbohydrates provided by those mycorrhizal types is not sufficient to support achlorophylly. There also must be a high probability to become colonized by an appropriate fungus, keeping in mind that MH plants are often quite specific with respect to their endophyte (e.g., Kretzer et al. 2000; Taylor et al. 2002; Bidartondo et al. 2002; Franke et al. 2004; Ogura-Tsujita and Yukawa 2008). A widely branched allorhizic root system seems to be suitable for this, but in turn, is susceptible to functional failure of large parts by only a single blocking, collapsing or disconnection event in a proximal segment. This is particularly critical when secondary growth for securing the connection is impossible. In any case, the transfer of carbon to the reproductive parts must be either short or reliably assured. These challenges are reasons for the following convergent evolutionary trends concerning subterranean organs of MH plants in unrelated plant families:
-
1.
Star-like root systems consisting of many roots radiating from the base of the shoot, either created by root-borne shoots or shoot-borne roots, reduce the risk of becoming disconnected to a major part of the root system (e.g., Fig. 4.2g, 4.4b, 4.6c + g + k, 4.7b, 4.8e + j, 4.10c + f + g + i, 4.12f + g, 4.13a + d, 4.14b, 4.15b, 4.18b + e + g + j, 4.19j, 4.20a).
-
2.
Short and thick roots shorten the transport distance of carbon to the shoot while retaining the tissue volume of long and thin roots (e.g., Figs. 4.3a, 4.4b, 4.7b, 4.9d, 4.10f, 4.12f + g, 4.14b, 4.15b, 4.17e + g, 4.19c).
-
3.
Specialized colonization pattern that enables a sustained use of a few fungal penetrations counterbalance the reduced probability to become colonized in short and thick roots compared to filiform roots (e.g., Figs. 4.3i, 4.4i, 4.5j, 4.8h, 4.9h, 4.13h, 4.14k, 4.16d, 4.19n, 4.20i).
-
4.
Strong reinforcement of thin roots, either by tertiary endodermae (in monocots) or the development of multicellular fibrous tissue, protect the carbon supply of the shoot (e.g., Figs. 4.5d + f, 4.6c + d, 4.13h).
The contradicting needs for a large root surface for high infection probability and short distances for carbon transport, has been discussed as the “mycoheterotroph’s dilemma” (Imhof 2010) and supposedly has shaped much of the subterranean organs in MH plants during evolution. As an effect, advanced MH plants within a family have stout, clumped roots and (in orchids) rhizomes mostly with a specialized fungal colonization pattern. This trend is best exemplified in Voyria (Gentianaceae, Imhof 1999c) and in Ericaceae (Furman and Trappe 1971). Gentianales and Ericaceae especially, having two fundamentally distinct groups of mycorrhiza (AM group vs. ECM group, Imhof 2009) but both show evolutionary reductions from trees to achlorophyllous herbs (Henderson 1919; Imhof 1999c) including changes in mycorrhizal pattern, turn out to be a textbook example for convergent evolution. In Triuridaceae (Imhof 2003), Burmanniaceae (Imhof 2001), Thismia (this chapter) and Afrothismia (both Thismiaceae, Imhof 2006), and Orchidaceae (Furman and Trappe 1971), this trend is partly detectable, but further investigations are necessary for more support. Research on taxa-like Geosiris (Iridaceae), Corsia (Corsiaceae), Kupea (Triuridaceae), Haplothismia (Thismiaceae) and others for which nothing is known concerning the fungal structures, will also help to understand the evolution of mycoheterotrophy. Moreover, given that 15 investigated vascular MH plants associated with AM fungi (i.e., Monotropoideae and orchids excluded) revealed 13 different mycorrhizal colonization patterns, there is a considerable chance for more fascinating novelties. In orchid mycorrhizas, although belonging to the oldest fields of mycorrhizal research (e.g., Schleiden 1845), comparatively little is known on the two existing types: tolypophagy and ptyophagy (Burgeff 1932; Wang et al. 1997; Rasmussen 2002; Imhof 2009). Since the latter type was found exclusively in achlorophyllous orchids so far (e.g., Janse 1896; Campbell 1963, 1964; Wang et al. 1997), an examination of the mycorrhizal structures of other MH orchids is highly desired.
In conclusion, mycoheterotrophy is based on a number of specializations with respect to morphology and anatomy of the underground parts, and, most importantly, on the evolution of sophisticated mycorrhizal pattern.
References
Abe C, Akasawa Y (1989) A new species of Oxygyne (Burmanniaceae) found in Shikoku, Japan. J Jap Bot 64:161–164
Agardh JG (1858) Theoria systematis plantarum, vol 99. Gleerup, Lundae
Airy Shaw HK (1952) A new genus and species of Burmanniaceae from South India. Kew Bull 2:277–279
Albert VA, Struwe L (1997) Phylogeny and classification of Voyria (saprophytic Gentianaceae). Brittonia 49:466–479
Andres H (1914) Studien zur speziellen Systematik der Pirolaceen II. Allg Bot Zeitschr 20:109–117
APG (Angiosperm Phylogeny Group) (2009) An update of the angiosperm phylogeny group classification for the orders and families of flowering plants: APG III. Bot J Linn Soc 161:105–121
Aublet JBCF (1775) Voyria. In: Histoire des Plantes de la Guiane Françoise, vol 1. P.-F. Didot jeune, London, Paris, p 208–211
Backer CA, Bakhuizen van den Brink RC (1963) Polygalaceae. In: Flora of Java. Noordhoff, Groningen, p 196–201
Beccari O (1877) Descrizione di una nuova e singolare pianta parassita: Gen. Corsia Becc. Malesia 1:238–240
Benson-Evans K (1952) The distribution of sex in Cryptothallus. Nature 169:39
Bernard C, Ernst A (1910) Beiträge zur Kenntnis der Saprophyten Javas. II. Äussere und innere Morphologie von Thismia javanica J. J. S. Ann Jard Bot Buitenzorg 23:36–46
Bernard C, Ernst A (1911) Beiträge zur Kenntnis der Saprophyten Javas. V. Anatomie von Thismia clandestina Miq. und Thismia versteegii Sm. Ann Jard Bot Buitenzorg 24:61–69
Bernard C, Ernst A (1914) Beiträge zur Kenntnis der Saprophyten Javas. XIV. Äussere und innere Morphologie von Burmannia tuberosa Becc. Ann Jard Bot Buitenzorg 28:102–118
Bidartondo MI, Bruns TD (2001) Extreme specificity in epiparasitic Monotropoideae (Ericaceae): widespread phylogenetic and geographical structure. Mol Ecol 10:2285–2295
Bidartondo MI, Bruns TD (2002) Fine-level mycorrhizal specificity in the Monotropoideae (Ericaceae): specificity for fungal species groups. Mol Ecol 11: 557–569
Bidartondo MI, Redecker D, Hijri I, Wiemken A, Bruns TD, Dominguez L, Sérsic A, Leake J, Read DJ (2002) Epiparasitic plants specialized on arbuscular mycorrhizal fungi. Nature 419:389–392
Bidartondo MI, Bruns TD, Weiß M, Sérgio C, Read DJ (2003) Specialized cheating of the ectomycorrhizal symbiosis by an epiparasitic liverwort. Proc Roy Soc Lond B 270:835–842
Bidartondo MI (2005) The evolutionary ecology of myco-heterotrophy. New Phytol 167:335–352
Bidartondo MI, Bruns TD (2005) On the origins of extreme mycorrhizal specificity in the Monotropoideae (Ericaceae): performance trade-offs during seed germination and seedling development. Mol Ecol 14: 1549–1560
Bidartondo MI, Duckett JG (2009) Conservative ecological and evolutionary patterns in liverwort-fungal symbioses. Proc Roy Soc Lond B 277:485–492
Bierhorst DW (1953) Structure and development of the gametophyte of Psilotum nudum. Am J Bot 40: 649–658
Bierhorst DW (1968) Observations on Schizaea and Actinostachys spp., including A. oligostachys, sp. nov. Am J Bot 55:87–108
Bierhorst DW (1971) Morphology of vascular plants. Macmillan, New York
Bonfante P, Perotto S (1995) Strategies of arbuscular mycorrhizal fungi when infecting host plants. New Phytol 130:3–21
Boudier P, Bardat J, Perrera S (1999) Cryptothallus mirabilis v. Malmborg (Aneuraceae, Hepaticopsida) dans le Perche d’Eve-et-Loire (France). Cryptogamie Bryol 20:189–196
Boullard B (1957) La mycotrophie chez les Ptéridophytes. Sa fréquence, ses caractères, sa signification. Le Botaniste 41:5–185
Boullard B (1979) Considérations sur la symbiose fongique chez les Ptéridophytes. Syllogeus 19:1–58. National Museum of Natural Sciences, Ottawa
Bruchmann H (1885) Das Prothallium von Lycopodium. Bot Centralbl 21:23–28 + 309–313
Bruchmann H (1906) Über das Prothallium und die Sporenpflanze von Botrychium lunaria Sw. Flora 96:203–230
Bruchmann H (1910) Die Keimung der Sporen und die Entwicklung der Prothallien vor Lycopodium clavatum L., L. annotinum L. und L. selago L. Flora 101:220–267
Brundrett MC (2002) Coevolution of roots and mycorrhizas of land plants. New Phytol 154:275–304
Bruns TD, Read DJ (2000) In vitro germination of nonphotosynthetic, myco-heterotrophic plants stimulated by fungi isolated from the adult plants. New Phytol 148:335–342
Burgeff H (1932) Saprophytismus und Symbiose. Studien an tropischen Orchideen. G. Fischer, Jena
Burgeff H (1938) Mycorrhiza. In: Verdoom F (ed) Manual of pteridology. M. Nijhoff, The Hague, pp 159–196
Burgeff H (1943) Problematik der Mycorhiza. Naturwissenschaften 31:558–567
Caddick LR, Rudall PJ, Wilkin P (2000) Floral morphology and development in Dioscoreales. Fedd Repert 111:189–230
Cameron DD, Bolin JF (2010) Isotopic evidence of partial mycoheterotrophy in the Gentianaceae: Bartonia virginica and Obolaria virginica as case studies. Am J Bot 97:1272–1277
Cameron KM, Chase MW, Rudall PJ (2003) Recircumscription of the monocotyledonous family Petrosaviaceae to include Japonolirion. Brittonia 55:214–225
Camp WH (1940) Aphyllous forms in Pyrola. Bull Torrey Bot Club 67:453–465
Campbell DH (1908) Symbiosis in fern prothallia. Am Nat 42:154–165
Campbell EO (1962) The mycorrhiza of Gastrodia cunninghamii Hook. f. Trans Roy Soc New Zealand Bot 1:289–296
Campbell EO (1963) Gastrodia minor Petrie, an epiparasite of Manuka. Trans Roy Soc New Zealand 2:73–81
Campbell EO (1964) The fungal association in a colony of Gastrodia sesamoides R. Br. Trans Roy Soc New Zealand 2:237–246
Campbell EO (1968) An investigation of Thismia rodwayi F. Muell. and its associated fungus. Trans Roy Soc New Zealand 3:209–219
Chantanaorrapint S (2008) Thismia angustimitra (Thismiaceae), a new species from Thailand. Blumea 53:524–526
Cheek M (2003a) A new species of Afrothismia (Burmanniaceae) from Kenya. Kew Bull 58:951–955
Cheek M (2003b) Kupeaeae, a new tribe of Triuridaceae from Africa. Kew Bull 58:939–949
Cheek M (2009) Burmanniaceae. In: Timberlake JR, Marins ES (eds) Flora Zambesiaca, vol 12(2). Flora Zambesiaca Managing Committee, London, pp 141–148
Cheek M, Williams S, Etuge M (2003) Kupea martinetugei, a new genus and species of Triuridaceae from western Cameroon. Kew Bull 58:225–228
Chen X, Tamura MN (2000) Petrosavia. In: Flora of China, vol 24. Science Press, Beijing, p 77
Cherrier J-F, Gondran M, Woltz P, Vogt G (1992) Parasitisme interspécifique chez les Gymnospermes. Données inédites chez deux Podocarpaceae endémiques Néo-Calédoniennes. Rev Cytol Biol Végét Bot 15:65–87
Chiang PY, Hsieh T-H (2011) Thismia huangii (Thismiaceae), a new species from Taiwan. Taiwania 56:138–142
Clarke CB (1885) Gentianaceae. In: Hooker JD (ed) Flora of British India, vol 4. Kent, L. Reeve, pp 93–132
Coleman DG (1936) Sarcosiphon rodwayi in Australia. Victorian Naturalist 52:163–166
Colozza A (1910) Contributo allo studio anatomico delle Burmanniaceae. Boll Soc Ital 1910:106–115
Correll DS (1966) Two new plants in Texas. Wrightia 3:188–191
Courty P-E, Walder F, Boller T, Ineichen K, Wiemken A, Rousteau A, Selosse M-A (2011) Carbon and nitrogen metabolism in mycorrhizal networks and mycoheterotrophic plants of tropical forests: a stable isotope analysis. Plant Physiol 156:952–961
Crantz HJN (1766) 1096. Hypopitys. In: Institutiones Rei Herbariae juxta nutum Naturae Digestae ex Habitu, Tomus II: 467. Ioannis Pauli Kraus, Viennensis (Vianden, Luxemburg)
Cribb AB (1995) The saprophytic flowering plant, Thismia yorkensis sp. nov., from Australia. Queensland Nat 33:51–56
Cribb P (1985) The saprophytic genus Corsia in the Solomon Islands. Kew Mag 2(3):320–323
Cribb PJ, Wilkin P, Clements M (1994) Corsiaceae: a new family for the Falkland Islands. Kew Bull 50:171–172
Cronquist A (1988) The evolution and classification of flowering plants, 2nd edn. New York Botanical Garden, New York
Crosby MR, Magill RE, Allen B, He S (2000) A checklist of the Mosses. Missouri Botanical Garden, St. Louis
Cullings KW, Szaro TM, Bruns TD (1996) Evolution of extreme specialization within a lineage of ectomycorrhizal epiparasites. Nature 379:63–66
Cullings KW (1996) Single phylogenetic origin of ericoid mycorrhizae within the Ericaceae. Can J Bot 74: 1896–1909
Darnell-Smith G (1917) The gametophyte of Psilotum. Trans Roy Soc Edinb 52:79–91
Dauby G, Parmentier I, Stévart T (2008) Afrothismia gabonensis sp. nov. (Burmanniaceae) from Gabon. Nord J Bot 25:268–271
Davis R (1976) A light and electron microscope investigation of the fungus-gametophyte association of Psilotum nudum (L.) Beauv. M.A. Thesis, Arcata, California/USA, Biological Sciences: Humboldt State University
de Candolle AP (1838) Ordo CXVI: Pyrolaceae. In: Prodromus systematis naturalis regni vegetabilis, vol 7. Treuttel et Würtz, Paris, p 772–776
de Laubenfels DJ (1959) Parasitic conifer found in New Caledonia. Science 130:97
de Laubenfels DJ (1972) Gymnospermes. In: Aubreville A, Leroy JF (eds) Flore de la Nouvelle-Calédonie et Dépendances. Museum d’Histoire Naturelle, Paris, p 4
Dening K (1928) Entwicklungsgeschichtliche Untersuchungen am Gametophyten von Buxbaumia aphylla (L.). Verh Naturhist Vereines Preuss Rheinl Westphalens 85:306–444
Denis MM (1919) Sur quelques thalles d’Aneura dépourvus de chlorophylle. Comptes rendus hebdomadaires des séances de l’Académie des. Sciences 168:64–66
Dimitri MJ (1972) Una nueva del genero Arachnitis Phil. (Corsiaceae). Rev Fac Agron Univ Nac La Plata 48:37–45
Domínguez L, Sérsic A, Melville L, Peterson RL (2006) ‘Prepackaged symbioses’: propagules on roots of the myco-heterotrophic plant Arachnitis uniflora. New Phytol 169:191–198
Domínguez LS, Sérsic A (2004) The southernmost myco-heterotrophic plant, Arachnitis uniflora: root morphology and anatomy. Mycologia 96:1143–1151
Domínguez LS, Melville L, Sérsic A, Faccio A, Peterson RL (2009) The mycoheterotroph Arachnitis uniflora has a unique association with arbuscular mycorrhizal fungi. Botany 87:1198–1208
Dowie NJ, Hemenway JJ, Trowbridge SM, Miller SL (2011) Mycobiont overlap between two mycoheterotrophic genera of Monotropoideae (Pterospora andromedea and Sarcodes sanguinea) found in the Greater Yellowstone Ecosystem. Symbiosis 54:29–36
Drude O (1873) Die Biologie von Monotropa hypopitys L. und Neottia nidus avis L. unter vergleichender Hinzuziehung anderer Orchideen. Dieterich’sche Univ. Buchdruckerei, Göttingen
Duckett JG, Renzaglia KS, Pell K (1990) Desiccation causes the proliferation of multicellular hairs, but not mucilage papillae, in Cryptothallus mirabilis (Hepatophyta): a correlated light and electron microscopy study. Can J Bot 68:697–706
Duddridge JA, Read DJ (1982) An ultrastructural analysis of the development of mycorrhizas in Monotropa hypopitys L. New Phytol 92:203–214
Düll R, Düll-Wunder B (2008) Moose einfach und sicher bestimmen: ein illustrierter Exkursionführer zu den Arten Deutschlands und angrenzender Länder. Quelle & Meyer, Wiebelsheim
Eastwood SK (1936) Notes on Buxbaumia aphylla (Linnaeus) Hedwig. Bryologist 39:127–129
Eastwood SK (1939) On the moss genus Buxbaumia. Trillia 10:28–35
Ernst A, Bernard C (1911) Beiträge zur Kenntnis der Saprophyten Javas. VIII. Äussere und innere Morphologie von Burmannia candida Engl. und Burmannia championii Thw. Ann Jard Bot Buitenzorg 24:84–95
Ernst A, Bernard C (1912) Beiträge zur Kenntnis der Saprophyten Javas. XI. Äussere und innere Morphologie von Burmannia coelestis Don. Ann Jard Bot Buitenzorg 26:224–232
Esau K (1965) Planzenanatomie. Wiley, New York
Feild TS, Brodribb TJ (2005) A unique mode of parasitism in the conifer coral tree Parasitaxus ustus (Podocarpaceae). Pl Cell Environ 28:1316–1325
Fiebrig C (1921) Fanerógamas saprófitas: Triuris mycoides sp. nov. Revista Jard Bot Mus Hist Nat Paraguay 1:164–165
Figdor W (1897) Ueber Cotylanthera Bl. Ein Beitrag zur Kenntnis tropischer Saprophyten. Ann Jard Bot Buitenzorg 14:213–239
Franke T (1999) Untersuchungen zur Biologie von Sciaphila purpurea Benth. (1855) (Triuridaceae, Monocotyledoneae). Diploma Thesis, München: Ludwig Maximilian Universität
Franke T, Beenken L, Hahn C (2000) Triuridopsis intermedia spec. nov. (Triuridaceae), a new myco-heterotrophic plant from Bolivia. Pl Syst Evol 225:141–144
Franke T (2002) The myco-heterotrophic Voyria flavescens (Gentianaceae) and its associated fungus. Mycol Prog 1:366–376
Franke T, Sainge MN, Agerer R (2004) A new species of Afrothismia (Burmanniaceae; Tribe: Thismieae) from the western foothills of Mount Cameroon. Blumea 49:451–456
Franke T (2004) Afrothismia saingei (Burmanniaceae, Thismieae), a new myco-heterotrophic plant from Cameroon. Syst Geogr Pl 74:27–33
Franke T, Beenken L, Döring M, Kocyan A, Agerer R (2006) Arbuscular mycorrhizal fungi of the Glomus-group A lineage (Glomerales; Glomeromycota) detected in myco-heterotrophic plants from tropical Africa. Mycol Prog 5:24–31
Fukarek F, Hammer K, Hanelt P, Keller J, Kruse J, Gladis T (1994) Urania Pflanzenreich—Blütenpflanzen 2. Urania, Leipzig
Furman TE, Trappe JM (1971) Phylogeny and ecology of mycotrophic achlorophyllous angiosperms. Quart Rev Biol 46:219–225
Furness CA, Rudall PJ, Eastman A (2002) Contribution of pollen and tapetal characters to the systematics of Triuridaceae. Pl Syst Evol 235:209–218
Gandolfo MA, Nixon KC, Crepet WL (2002) Triuridaceae fossil flowers from the upper cretaceous of New Jersey. Am J Bot 89:1940–1957
Gifford EM, Foster AS (1996) Morphology and evolution of vascular plants, 3rd edn. W. H. Freeman, New York
Gilg E (1895) Gentianaceae. In: Engler A, Prantl K (eds) Die Natürlichen Pflanzenfamilien, IV. Teil, 2. Abteilung. W. Engelmann, Leipzig, pp 50–108
Gilg E (1899) Gentianaceae africanae—Beiträge zur Kenntnis der Gentianaceae II. Bot Jahrb 26:86–110
Gillett JM (1959) A revision of Bartonia and Obolaria (Gentianaceae). Rhodora 61:43–62
Goebel K (1892) Archegoniatenstudien. 1. Die einfachste Form der Moose. Flora 76:92–104
Goebel K, Süssenguth K (1924) Beiträge zur Kenntnis der südamerikanischen Burmanniaceen. Flora 117:55–90
Goffinet B, Buck WR, Shaw AJ (2008) Morphology, anatomy, and classification of the Bryophyta. In: Goffinet B, Shaw AJ (eds) Bryophyte biology, 2nd edn. Cambridge Univ. Press, Cambridge, pp 55–138
Goldblatt P, Rudall P, Cheadle VI, Dorr LJ, Wiliams CA (1987) Affinities of the madagascan endemic Geosiris, Iridaceae or Geosiridaceae. Bull Mus Nat Hist Paris 4 Serie Adansonia Section B 9; 3:239–248
Goldblatt P (1990) Phylogeny and classification of Iridaceae. Ann Missouri Bot Gard 77:607–627
Goldblatt P, Manning JC, Rudall P (1998) Iridaceae. In: Kubitzki K (ed) The families and genera of vascular plants, III flowering plants—monocotyledons, Lilianae (except Orchidaceae). Springer, Berlin, pp 295–333
Goldblatt P, Manning JC (2010) Iridaceae—Geosiris albiflora (Geosiridoideae), a new species from the Comoro Archipelago. Bothalia 40:169–171
Goldblatt P, Rodriguez A, Powell MP, Davies TJ, Manning JC, van der Bank M, Savolainen V (2008) Iridaceae ‘Out of Australasia’? Phylogeny, biogeography, and divergence time based on plastid DNA sequences. Syst Bot 33:495–508
Gray A (1871) Characters of a new genus consisting of two species of parasitic Gentianeae. J Linn Soc Bot 11:22–23
Gregory P (2006) Plant roots. Blackwell, Oxford
Groenendijk JP, van Dulmen ATJ, Bouman F (1997) The “forest floor” saprophytes Voyria spruceana and V. aphylla (Gentianaceae) growing as epiphytes in Colombian Amazonia. Ecotropica 3:129–131
Groom P (1895a) On a new saprophytic monocotyledon. Ann Bot 9:45–58
Groom P (1895b) On Thismia aseroe (Beccari) and its mycorhiza. Ann Bot 9:327–361
Groom P (1895c) Contribution to the knowledge of monocotyledonous saprophytes. J Linn Soc Bot 31: 149–215
Guinea Lopez E (1946) Novedades sistemáticas de mi primer viaje a la Guinea continental espanola (verano de 1945). Anales Jard Bot Madrid 6:463–485
Haber E (1987) Variability, distribution and systematics of Pyrola picta s.l. (Ericaceae) in Western North America. Syst Bot 12:324–335
Haberlandt G (1886) Beiträge zur Anatomie und Physiologie der Laubmoose. Jahrb Wiss Bot (Pringsheim) 17:359–498
Hancock JA, Brassard GR (1974) Phenology, sporophyte production, and life history of Buxbaumia aphylla in Newfoundland, Canada. Bryologist 77:501–513
Hara H (1975) A new species of Cotylanthera (Gentianaceae) from Philippines, with a conspectus of the genus. J Jap Bot 50:321–328
Heide-Jørgensen H (2008) Parasitic flowering plants. Brill, Leiden
Hemsley W (1907) Two new Triuridaceae, with some remarks on the genus Sciaphila, Blume. Ann Bot 21:71–77
Henderson MW (1919) A comparative study of the structure and saprophytism of the Pyrolaceae and Monotropaceae with reference to their derivation from the Ericaceae. Contr Bot Lab Morris Abor Univ Pennsylvania 5:42–109
Hepper FN (1968) Burmanniaceae. In: Flora of West Tropical Africa, vol 3, 2nd edn. The Crown Agents for Oversea Governments and Administration, London, p 176–180
Hirce EG, Finocchio AF (1972) Stem and root anatomy of Monotropa uniflora. Bull Torrey Bot Club 99:89–94
Holloay JE (1921) Further studies on the prothallus, embryo, and young sporophyte of Tmesipteris. Trans Proc N Z Inst 53:386–422
Holm T (1897) Obolaria virginica L.: a morphological and anatomical study. Ann Bot 11:369–383
Holm T (1898) Pyrola aphylla: a morphological study. Bot Gaz 25:246–254
Holm T (1906) Bartonia, Muehl. An anatomical study. Ann Bot 20:441–448
Hooker WJ (1840) Flora Boreali-Americana, vol 2. Henry G. Bohn, London
Hsieh C-F, Hsieh T-H, Lai I-L (1995) Epirixanthes elongata Bl.—a new record to the Flora of Taiwan. Taiwania 40:381–384
Humpert AJ, Muench EL, Giachini AJ, Castellano MA, Spatafora JW (2001) Molecular phylogenetics of Ramaria and related genera: evidence from nuclear large subunit and mitochondrial small subunit rDNA sequences. Mycologia 93:465–477
Hynson NA, Preiss K, Gebauer G, Bruns TD (2009) Isotopic evidence of full and partial myco-heterotrophy in the plant tribe Pyroleae (Ericaceae). New Phytol 182:719–726
Ibisch P, Neinhuis C, Rojas NP (1996) On the biology, biogeography, and taxonomy of Arachnitis Phil. nom. cons. (Corsiaceae) in respect to a new record from Bolivia. Willdenowia 26:321–332
Imhof S, Weber HC, Gómez LD (1994) Ein Beitrag zur Biologie von Voyria tenella Hook. und Voyria truncata (Standley) Standley & Steyermark (Gentianaceae). Beitr Biol Pflanzen 68:113–123
Imhof S, Weber HC (1997) Root anatomy and mycotrophy (AM) of the achlorophyllous Voyria truncata (Standley) Standley & Steyermark (Gentianaceae). Bot Acta 110:127–134
Imhof S (1997) Root anatomy and mycotrophy of the achlorophyllous Voyria tenella Hooker (Gentianaceae). Bot Acta 110:298–305
Imhof S (1998) Subterranean structures and mycotrophy of the achlorophyllous Triuris hyalina Miers (Triuridaceae). Can J Bot 76:2011–2019
Imhof S (1999a) Anatomy and mycotrophy of the achlorophyllous Afrothismia winkleri (Engl.) Schltr. (Burmanniaceae). New Phytol 144:533–540
Imhof S (1999b) Subterranean structures and mycorrhiza of the achlorophyllous Burmannia tenella Benth. (Burmanniaceae). Can J Bot 77:637–643
Imhof S (1999c) Root morphology, anatomy and mycotrophy of the achlorophyllous Voyria aphylla (Jacq.) Pers. (Gentianaceae). Mycorrhiza 9:33–39
Imhof S, Weber HC (2000) Root structures and mycorrhiza of the achlorophyllous Voyria obconica Progel (Gentianaceae). Symbiosis 29:201–211
Imhof S (2001) Subterranean structures and mycotrophy of the achlorophyllous Dictyostega orobanchoides (Hook.) Miers (Burmanniaceae). Rev Biol Trop 49:239–247
Imhof S (2003) A dorsiventral mycorrhizal root in the achlorophyllous Sciaphila polygyna (Triuridaceae). Mycorrhiza 13:327–332
Imhof S (2004) Morphology and development of the subterranean organs of the achlorophyllous Sciaphila polygyna (Triuridaceae). Bot J Linn Soc 146:295–301
Imhof S (2006) Two distinct fungi colonize roots and rhizomes of the myco-heterotrophic Afrothismia gesnerioides (Burmanniaceae). Can J Bot 84:852–861
Imhof S (2007) Specialized mycorrhizal colonization pattern in achlorophyllous Epirixanthes spp. (Polygalaceae). Plant Biol 9:786–792
Imhof S, Sainge MN (2008) Ontogeny of the mycoheterotrophic species Afrothismia hydra (Burmanniaceae). Bot J Linn Soc 157:31–36
Imhof S (2009) Arbuscular, ecto-related, orchid mycorrhizas—three independent structural lineages towards mycoheterotrophy: implications for classification? Mycorrhiza 19:357–363
Imhof S (2010) Are monocots particularly suited to develop mycoheterotrophy? In: Seberg O, Petersen G, Barfod A, Davis JI (eds) Diversity, phylogeny and evolution in the monocotyledons. Aarhus University Press, Kopenhagen, pp 11–23
Iwatsuki Z, Sharp AJ (1967) The bryogeographic relationships between eastern Asia and North America. J Hattori Bot Lab 30:152–170
Jacquelinet-Jeanmougin S, Gianinazzi-Pearson V (1983) Endomycorrhizas in the Gentianaceae. I. The fungi associated with Gentiana lutea L. New Phytol 95:663–666
Janse JM (1896) Les endophytes radicaux de quelques plantes javanaises. Ann Jard Bot Buitenzorg 14:53–212
Jessop JP (1979) Petrosavia. In: van Steenis CGGJ (ed) Flora Malesiana, Series 1, vol 9. Martinus Nijhoff, The Hague, pp 198–200
Johow F (1885) Die chlorophyllfreien Humusbewohner West-Indiens, biologisch-morphologisch dargestellt. Jahrb Wiss Bot 16:415–449
Johow F (1889) Die chlorophyllfreien Humuspflanzen nach ihren biologischen und anatomisch-entwicklungsgeschichtlichen Verhältnissen. Jahrb Wiss Bot 20:475–524
Jones DL, Gray B (2008) Corsia dispar D. L. Jones & B. Gray (Corsiaceae), a new species from Australia, and a new combination of Corsia for a New Guinea taxon. Austrobaileya 7:717–722
Jonker FP (1936) Gentianaceae. In: Pulle AA (ed) Flora of Suriname. Brill, Leiden, pp 400–430
Jonker FP (1938) A monograph of the Burmanniaceae. Meded Bot Mus Herb Rijks Univ Utrecht 51:1–279
Kamienski F (1882) Les organes végétatifs du Monotropa hypopithys L. Mém Soc Nat Sc Natur et Math Cherbourg 24:5–40
Kidston R, Lang WH (1921) On old red sandstone plants showing structure, from the Rhynie chert bed, Aberdeenshire. Trans Roy Soc Edinb 52:855–901
Kiew R (1999) Thismia goodii (Burmanniaceae), the blue-capped Thismia, a new species from Borneo. Gard Bull Singap 51:179–182
Kissling J (2012) Taxonomy of Exochaenium and Lagenias: two resurrected genera of tribe Exaceae (Gentianaceae). Syst Bot 37:238–253
Kissling J, Yuan Y-M, Küpfer PM, Mansion G (2009) The polyphyletic genus Sebaea (Gentianaceae): a step forward in understanding the morphological and karyological evolution of the Exaceae. Mol Phylogenet Evol 53:734–748
Klackenberg J (1985) The genus Exacum (Gentianaceae). Opera Bot 84:1–144
Klackenberg J (2002) Tribe Exaceae. In: Struwe L, Albert V (eds) Gentianaceae: systematics and natural history. Cambridge University Press, Cambridge, pp 66–108
Klackenberg J (2006) Cotylanthera transferred to Exacum (Gentianaceae). Bot Jahrb Syst 126:477–481
Knoblauch E (1894) Beiträge zur Kenntniss der Gentianaceae. Bot Centralbl 60:321–401
Köpke E, Musselman LJ, de Laubenfels DJ (1981) Studies on the anatomy of Parasitaxus ustus and its root connections. Phytomorph 31:85–92
Kottke I, Nebel M (2005) The evolution of mycorrhiza-like associations in liverworts: an update. New Phytol 167:330–334
Kretzer AM, Bidartondo MI, Grubisha LC, Spatafora JW, Szaro TM, Bruns TD (2000) Regional specialization of Sarcodes sanguinea (Ericaceae) on a single fungal symbiont from the Rhizopogon ellenae (Rhizopogonaceae) species complex. Am J Bot 87:1778–1782
Kuga-Uetake Y, Purich M, Massicotte HB, Peterson RL (2004) Host microtubules in the Hartig net region of ectomycorrhizas, ectendomycorrhizas, and monotropoid mycorrhizas. Can J Bot 82:938–946
Kuijt J (1969) The biology of parasitic flowering plants. Univ. Calif. Press, Berkeley
Kutschera L, Lichtenegger E (1982) Wurzelatlas mitteleuropäischer Grünlandpflanzen, Teil 1: Monocotyledonae. Fischer, Stuttgart
Kutschera L, Lichtenegger E (1992) Wurzelatlas mitteleuropäischer Grünlandpflanzen, Teil 2: Dicotyledonae. Fischer, Stuttgart
Kutschera L, Lichtenegger E, Sobotik M (2009) Wurzelatlas der Kulturpflanzen gemäßigter Gebiete mit Arten des Feldgemüsebaues. DLG Verlags-GmbH, Frankfurt
Lace JH (1914) Decades Kewenses—767. Cotylanthera caerulea. Bull Misc Inform 1914:150–156
Lang WH (1899) The prothallus of Lycopodium clavatum L. Ann Bot 13:279–317
Lang WH (1902) On the prothalli of Ophioglossum pendulum and Helminthostachys zeylanica. Ann Bot 16:23–56
Largent DL, Sugihara N, Wishner C (1980) Occurrence of mycorrhizae on ericaceous and pyrolaceous plants in northern California. Can J Bot 58:2274–2279
Larsen K (1963) Studies in the flora of Thailand. 14. Cytological studies in vascular plants of Thailand. Dansk Bot Ark 20:211–275
Larsen K (1965) Thismiaceae. Dansk Bot Ark 23:171–174
Larsen K (1972) Triuridaceae. In: Smitinand T, Larsen K (eds) Flora of Thailand, vol 2, Part 2. Asrct Press, Bangkok, pp 175–176
Larsen K, Averyanov LV (2007) Thismia annamensis and Thismia tentaculata, two new species of Thismiaceae from central Vietnam. Rheedea 17:13–19
Lawson AA (1917) The prothallus of Tmesipteris tannensis. Trans Roy Soc Edinb 51:785–794
Leake JR (1994) Tansley Review No. 69. The biology of myco-heterotrophic (‘saprophytic’) plants. New Phytol 127:171–216
Leake JR (2004) Myco-heterotroph/epiparasitic plant interactions with ectomycorrhizal and arbuscular mycorrhizal fungi. Curr Opin Plant Biol 7:422–428
Leake JR, McKendrick SL, Bidartondo M, Read DJ (2004) Symbiotic germination and development of the myco-heterotroph Monotropa hypopitys in nature and its requirement for locally distributed Tricholoma spp. New Phytol 163:405–423
Leake JR (2005) Plants parasitic on fungi: unearthing the fungi in myco-heterotrophs and debunking the ‘saprophytic’ plant myth. Mycologist 19:113–122
Lefevre CK (2002) Host associations of Tricholoma magnivelare, the American matsutake. Dissertation, Corvallis, Department of Forest Science: Oregon State University
LePage BA, Currah RS, Stockey RA, Rothwell GW (1997) Fossil ectomycorrhizae from the middle Eocene. Am J Bot 84:410–412
Ligrone R, Gambardella R, Castaldo R, Giordano S, de Lucia Sposito ML (1982) Gametophyte and sporophyte ultrastructure in Buxbaumia piperi Best (Buxbaumiales, Musci). J Hattori Bot Lab 52:465–499
Ligrone R, Pocock K, Duckett JG (1993) A comparative ultrastructural study of endophytic basidiomycetes in the parasitic achlorophyllous hepatic Cryptothallus mirabilis and the closely allied photosynthetic species Aneura pinguis (Metzgeriales). Can J Bot 71:666–679
Liu K-M, Long C-L, Liu Y-T (2001) Burmannia hunanensis (Burmanniaceae), a new species from Hunan, China. Ann Bot Fennici 38:211–214
Lutz RW, Sjolund RD (1973) Monotropa uniflora: ultrastructural details of its mycorrhizal habit. Am J Bot 60:339–345
Maas H, Maas PJM (1987) A new Thismia (Burmanniaceae) from French Guiana. Brittonia 39:376–378
Maas PJM, Rübsamen T (1986) Triuridaceae. Flora Neotropica Monogr 40:1–55
Maas PJM, Ruyters P (1986) Voyria and Voyriella (Saprophytic Gentianaceae). Flora Neotropica Monogr 41:1–93
Maas PJM, Maas-van de Kamer H, Benthem JV, Snelders HCM, Rübsamen T (1986) Burmanniaceae. Flora Neotropica Monogr 42:1–189
Maas PJM, Maas-van de Kamer H (1989) Triuridaceae. In: Görts-van Rijn ARA (ed) Flora of the Guianas, series A: Phanerogams, vol 5. Koeltz Scientific Books, Koenigstein, pp 3–16
Maas-van de Kamer H (1995) Triuridiflorae—Gardner’s delight? In: Rudall PJ, Cribb PJ, Cutler DF, Humphries CJ (eds) Monocotyledons: systematics and evolution. Royal Botanic Gardens, Kew, pp 287–301
Maas-van de Kamer H, Maas PJM (2010) Thismiaceae. In: Sosef MSM (ed) Flore du Gabon, vol 41. Margraf, Weikersheim, pp 60–62
Maas-van de Kamer H, Weustenfeld T (1998) Triuridaceae. In: Kubitzki K (ed) The families and genera of vascular plants, III flowering plants—monocotyledons, Lilianae (except Orchidaceae). Springer, Berlin, pp 452–458
Maas-van de Kamer H (2003) Afrothismia gesnerioides, another new species of Afrothismia (Burmanniaceae) from tropical Africa. Blumea 48:475–478
Magnus W (1900) Studien an der endotrophen Mycorrhiza von Neottia nidus avis L. Jahrb Wiss Bot 35:205–272
Makino T (1903) Observations of the flora of Japan. Bot Mag (Tokyo) 17:144–152
Malme GO (1896a) Die Burmannien der ersten Regnell’schen Expedition. Ein Beitrag zur Kenntnis der amerikanischen Arten dieser Gattung. Bih. Kongl. Svenska Vetensk.-Akad. Handl. 22 (Afd. III, No. 8):1–32
Malme GO (1896b) Über Triuris lutea (Gardn.) Benth. et Hook. Bih. Kongl. Svenska Vetensk.-Akad. Handl. 21 (Afd. III, No. 14):1–16
Marais W, Verdoorn IC (1963) Gentianaceae. In: Dyer RA, Codd LE, Rycroft HB (eds) Flora of Southern Africa, vol 26. Government Printer, Pretoria, pp 171–243
Martinez E, Ramos CH (1989) Lacandoniaceae (Triuriadales): una nueva familia de Mexico. Ann Missouri Bot Gard 76:128–135
Massicotte HB, Melville LH, Peterson RL (2005) Structural features of mycorrhizal associations in two members of the Monotropoideae, Monotropa uniflora and Pterospora andromedea. Mycorrhiza 15:101–110
Massicotte HB, Melville LH, Tackaberry LE, Peterson RL (2007) Pityopus californicus: structural characteristics of seed and seedling development in a myco-heterotrophic species. Mycorrhiza 17:647–653
Massicotte HB, Melville LH, Tackaberry LE, Peterson RL (2008) A comparative study of mycorrrhizas in several genera of Pyroleae (Ericaceae) from western Canada. Botany 86:610–622
Massicotte HB, Melville LH, Peterson RL, Tackaberry LE, Luoma DL (2010) Structural characteristics of root-fungus associations in two mycoheterotrophic species, Allotropa virgata and Pleuricospora fimbriolata (Monotropoideae), from southwest Oregon, USA. Mycorrhiza 20:391–397
Matsuda Y, Okochi S, Katayama T, Yamada A, Ito S-I (2011) Mycorrhizal fungi associated with Monotropastrum humile (Ericaceae) in central Japan. Mycorrhiza 21:569–576
McClymont JW (1950) Notes on Buxbaumia. Bryologist 53:176–277
McKendrick SL, Leake JR, Taylor DL, Read DJ (2002) Symbiotic germination and development of the myco-heterotrophic orchid Neottia nidus-avis in nature and its requirement for locally distributed Sebacina spp. New Phytol 154:233–247
McLennan EI (1958) Thismia rodwayi F. Muell. and its endophyte. Aust J Bot 6:25–37
Merckx V (2008) Myco-heterotrophy in Dioscoreales: systematics and evolution. Dissertation, Leuven, Faculteit Wetenschappen: K.U. Leuven
Merckx V, Schols P, Maas-van de Kamer H, Maas P, Huysmans S, Smets E (2006) Phylogeny and evolution of Burmanniaceae (Dioscoreales) based on nuclear and mitochondrial data. Am J Bot 93:1684–1698
Merckx V, Bidartondo MI (2008) Breakdown and delayed cospeciation in the arbuscular mycorrhizal mutualism. Proc Roy Soc Lond B 275:1029–1035
Merckx V, Bidartondo MI, Hynson NA (2009) Myco-heterotrophy: when fungi host plants. Ann Bot 104:1255–1261
Merckx VSFT, Janssens SB, Hynson NA, Specht CD, Bruns TD, Smets E (2012) Mycoheterotrophic interactions are not limited to a narrow phylogenetic range of arbuscular mycorrhizal fungi. Mol Ecol 21:1524–1532
Merckx V, Stöckel M, Fleischmann A, Bruns TD, Gebauer G (2010) 15N and 13C natural abundance of two mycoheterotrophic and a putative partially mycoheterotrophic species associated with arbuscular mycorrhizal fungi. New Phytol 188:590–596
Mesler MR (1975) The gametophytes of Ophioglossum palmatum L. Am J Bot 62:982–992
Mesler MR (1976) Gametophytes and young sporophytes of Ophioglossum crotalophoroides Walt. Am J Bot 63:443–448
Meyer K (1909) Untersuchungen über Thismia clandestina. Bull Soc Imp Naturalistes Moscou 1909:1–18
Miers J (1852) On the family of Triuridaceae. Trans Linn Soc Lond 21:43–59
Miers J (1866) On Myostoma, a new genus of the Burmanniaceae. Trans Linn Soc Lond 25:461–476
Milanez FR, Meira E (1943) Observacoes sobre ‘Triuris alata’ Brade. Arq Serv Florest 2:51–61
Minoletti OML (1986) Arachnitis uniflora Phil. una curiosa monocotiledonea de la Flora Chilena. Bol Soc Biol Concepcíon 57:7–20
Miquel FAW (1851) Gentianeae parasiticae. In: Stirpes surinamenses selectae. Arnz & Comp, Leiden, p 146–151
Miquel FAW (1856) Gentianae. In: Flora van Nederlandsch Indie, vol 2. Fried. Fleischer, Leipzig, p 554–564
Mueller DMTJ (1972) Observations on the ultrastructure of Buxbaumia Protonema. Plasmodesmata in the cross walls. Bryologist 75:63–68
Neumann G (1934) Über die Mykorrhiza in der Gattung Gentiana. Zentralbl Bakteriol 2 Abt 89:433–458
Nuttall T (1843) Notices of new or rare plants in the natural orders Lobeliaceae, Campanulaceae, Vaccinieae, Ericaceae collected in a journey over the continent of North America. Trans Am Phil Soc 8:271
Oehler E (1927) Entwicklungsgeschichtlich-zytologische Untersuchungen an einigen saprophytischen Gentianaceen. Planta 3:641–729
Ogura-Tsujita Y, Yukawa T (2008) High mycorrhizal specificity in a widespread mycoheterotrophic plant, Eulophia zollingeri (Orchidaceae). Am J Bot 95:93–97
Ohashi H (2000) A new treatment in Sciaphila (Triuridaceae) in Taiwan and Japan. Taiwania 45:351–354
Ohga T, Sinoto Y (1932) Cytological studies on Sciaphila japonica Mak. II On pollen and embryo sac-development. III. On micorhiza. Bot Mag (Tokyo) 46:311–315
Okamura S (1911) Neue Beiträge zur Moosflora Japans. Bot Mag (Tokyo) 25:30–34
Palacios-Mayorga P, Pérez-Silva E (1993) Endotrophic mycorrhiza in Lacandonia schismatica, Lacandoniaceae, a new saprophytic Angiospermae family. Revista Latinoamer Microbiol 35:65–69
Pendry CA (2010) Epirixanthes compressa Pendry, a new mycoheterotrophic species of Polygalaceae from Thailand. Thai For Bull 38:184–186
Penzig O (1901) Beiträge zur Kenntniss der Gattung Epirrhizanthes Bl. Ann Jard Bot Buitenzorg 17:142–170
Perrot E (1898) Anatomie comparée des Gentianacées. Ann Sci Nat Bot Sér 7 16:105–292
Petersen PM (1972) Cryptothallus mirabilis Malmb. found on Disko Island, West Greenland (69°15′ N., 53°34′ W.). Lindbergia 1:189–190
Peterson RL, Howarth MJ, Whittier DP (1981) Interactions between a fungal endophyte and gametophyte cells in Psilotum nudum. Can J Bot 59:711–720
Peterson RL (1992) Adaptations of root structure in relation to biotic and abiotic factors. Can J Bot 70:661–675
Peterson RL, Uetake Y, Zelmer C (1998) Fungal symbioses with orchid protocorms. Symbiosis 25:29–55
Peterson RL, Massicotte HB (2004) Exploring structural definitions of mycorrhizas, with emphasis on nutrient-exchange interfaces. Can J Bot 82:1074–1088
Peterson RL, Massicotte HB, Melville LH (2004) Mycorrhizas: anatomy and cell biology. NRC Research Press, Ottawa
Pfeiffer NE (1914) Morphology of Thismia americana. Bot Gaz 57:122–135
Pires O’Brien MJ (1997) Annotated check-list of the flora in the Jari river region: I. Saprophytic parasites. Boletim-do-Museu-Paraense-Emilio-Goeldi-Serie-Botanica 13:49–67
Pocock K, Duckett JG (1984) A comparative ultrastructural analysis of the fungal endophytes in Cryptothallus mirabilis Malm. and other British thalloid hepatics. J Bryol 13:227–233
Polomski J, Kuhn N (1998) Wurzelsysteme. Paul Haupt, Bern
Poppendieck H-H (1997) Eine Voyria-Art als ‘blinder Passagier’ im Botanischen Garten Hamburg. Palmengarten 61:30
Poulsen VA (1886) Bidrag til Triuridaceernes Naturhistorie. Vidensk Meddel Dansk Naturhist Foren Kjøbenhavn 36–38:161–179
Poulsen VA (1890a) Thismia Glaziovii nov. sp. Overs Kongel Danske Vidensk Selesk Forh Medlemmers Arbeider 1890:18–38
Poulsen VA (1890b) Triuris major sp. nov. Et Bidrag til Triuridaceernes Naturhistorie. Bot Tidsskr 17:293–306
Pringle JS (1995) Gentianaceae. In: Harling G, Andersson L (eds) Flora of Ecuador, vol 53. Council for Nordic Publications in Botany, Copenhagen, pp 3–131
Progel A (1865) Gentianaceae: Voyria. In: Martius (ed) Flora Brasiliensis, vol 6, C.F.P.v. Frid. Fleischer, Leipzig, p 219–226
Rasmussen HN (1995) Terrestrial orchids. Froseed to mycotrophic plant. Cambridge Univ. Press, Cambridge
Rasmussen HN (2002) Recent developments in the study of orchid mycorrhiza. Pl Soil 244:149–163
Raynal A (1967a) Sur un Sebaea africain saprophyte (Gentianaceae). Adansonia 7:207–219
Raynal A (1967b) Étude critique des genres Voyria et Leiphaimos (Gentianaceae et révision des Voyria d`Afrique). Adansonia 7:53–71
Raynal-Roques A, Paré J (1998) Biodiversité des phanérogames parasites: leur place dans la classification systématique. Adansonia 20:313–322
Read DJ, Duckett JG, Francis R, Ligrone R, Russell A (2000) Symbiotic fungal associations in ‘lower’ land plants. Phil Trans R Soc Lond B 355:815–831
Reiche K (1907) Grundzüge der Pflanzenverbreitung in Chile. In: Engler A, Drude O (eds) Die Vegetation der Erde. Sammlung Pflanzengeographischer Monographien VIII. W. Engelmann, Leipzig
Remy W, Taylor TN, Hass H, Kerp H (1994) Four hundred-million-year-old vesicular arbuscular mycorrhizae. Proc Natl Acad Sci U S A 91:11841–11843
Ridley HN (1922) Polygalaceae. In: Flora of the Malay Peninsula, vol 1. Reeve & Co, London, p 137–149
Robertson DC, Robertson JA (1982) Ultrastructure of Pterospora andromedea Nuttal and Sarcodes sanguinea Torrey mycorrhizas. New Phytol 92:539–551
Robertson DC, Robertson JA (1985) Ultrastructural aspects of Pyrola mycorrhizae. Can J Bot 63(108):9–1098
Robyns A (1962) Deux Exochaenium nouveaux (Gentianaceae) d’Afrique tropicale. Bull Jard Bot Etat Bruxelles 32:471–475
Rudall PJ, Bateman RM (2006) Morphological phylogenetic analysis of Pandanales: testing contrasting hypotheses of floral evolution. Syst Bot 31:223–238
Rübsamen-Weustenfeld T (1991) Morphologische, embryologische und systematische Untersuchungen an Triuridaceae. Biblioth Bot 140:1–113
Rübsamen-Weustenfeld T, Mukielka V, Hamann U (1994) Zur Embryologie, Morphologie und systematischen Stellung von Geosiris aphylla Baillon (Monocotyledoneae—Geosiridaceae/Iridaceae). Mit einigen embryologischen Daten zur Samenanlage von Isophysis tasmanica (Hook.) T. Moore (Iridaceae). Bot Jahrb Syst 115:475–545
Sainge MN, Franke T (2005) A new species of Afrothismia (Burmanniaceae) from Cameroon. Nord J Bot 23:299–303
Sasidharan N, Sujanapal P (2000) Rediscovery of Haplothismia exannulata Airy Shaw (Burmanniaceae) from its type locality. Rheedea 10:131–134
Saunders RMK (1996) Typification of the name Thismia fumida Ridl. (Burmanniaceae). Taxon 45:107–110
Schiffner V (1934) Über Cryptothallus mirabilis Malmb. Ann Bryol 7:165–166
Schlechter R (1905) Heft 2: Microspermae. In: Schumann K, Lauterbach K (eds) Nachträge zu Flora der Deutschen Schutzgebiete in der Südsee. Gebr. Borntraeger, Leipzig, pp 71–234
Schlechter R (1913) Neue Triuridaceae Papuasiens. Bot Jahrb Syst 49:70–89
Schlechter R (1919) Eine neue papuasische Burmanniaceae. Bot Jahrb Syst 55:202–203
Schleiden MJ (1845) Grundzüge der Wissenschaftlichen Botanik, 2. Aufl. Leipzig, Wilhelm Engelmann
Schmid E, Oberwinkler F (1993a) Mycorrhiza-like interaction between the achlorophyllous gametophyte of Lycopodium clavatum L. and its fungal endophyte studied by light and electron microscopy. New Phytol 124:69–81
Schmid E, Oberwinkler F (1993b) Light and electron microscopy of the host-fungus interaction in the achlorophyllous gametophyte of Botrychium lunaria. Can J Bot 72:182–188
Schneckenburger S (1999) Einige bemerkenswerte Coniferen Neukaledoniens. Mitt Deutsch Dendrol Ges 84:5–21
Schoepe G, Philippi G (2000) Buxbaumiidae. In: Nebe M, Philippi G (eds) Die Moose Baden-Württembergs. Ulmer, Stuttgart, pp 90–97
Schüßler A, Schwarzott D, Walker C (2001) A new fungal phylum, the Glomeromycota: phylogeny and evolution. Mycol Res 105:1413–1421
Selosse M-A, Weiß M, Jany J-L, Tillier A (2002) Communities and populations of sebacinoid basidiomycetes associated with the achlorophyllous orchid Neottia nidus-avis (L.) L.C.M. Rich. and neighbouring tree ectomycorrhizae. Mol Ecol 11: 1831–1844
Selosse M-A, Faccio A, Scappaticci G, Bonfante P (2004) Chlorophyllous and achlorophyllous specimens of Epipactis microphylla (Neottieae, Orchidaceae) are associated with ectomycorrhizal Septomycetes, including Truffles. Microbial Ecol 47:416–426
Sergio C, Seneca A (1997) The first report of Cryptothallus mirabilis Malmborg (Hepaticae, Aneuraceae) in southern Europe (Portugal). Cryptogamie Bryologie Lichénologie 18:213–215
Sergio C, Garcia C (1999) Cryptothallus mirabilis Malmb. (Aneuraceae, Hepaticae) in Portugal. New ecological and phytogeographical data. Haussknechtia Beiheft 9:343–346
Seybold S (2011) Schmeil-Fitschen—Flora von Deutschland und der angrenzenden Länder, 95. Aufl. Quelle & Meyer, Wiebelsheim
Sinclair WT, Mill RR, Gardner MF, Woltz P, Jaffré T, Preston J, Hollingsworth ML, Ponge A, Möller M (2002) Evolutionary relationships of the New Caledonian heterotrophic conifer, Parasitaxus usta (Podocarpaceae), inferred from chloroplast trnL-F intron/spacer and nuclear rDNA ITS2 sequences. Pl Syst Evol 233:79–104
Sjörs H (1949) Nagra växter funna huvudsakligen ar 1948. Botaniska Notiser 1949:95–103
Smith AR, Pryer KM, Schuettpelz E, Korall P, Schneider H, Wolf PG (2006) A classification for extant ferns. Taxon 55:705–731
Smith JJ (1911) Beiträge zur Kenntnis der Saprophyten Javas. VII. Zur Systematik von Burmannia candida Engl. und Burmannia championii Thw. Ann Jard Bot Buitenzorg 24:79–82
Smith JJ (1912) Neue papuanische Pflanzen I. Fedd Repert 10:486–488
Smith SE, Read DJ (2008) Mycorrhizal symbiosis, 3rd edn. Acad. Press, San Diego
Solereder H (1908) Gentianae. In: Systematische Anatomie der Dicotyledonen. Ergänzungsband. F. Enke, Stuttgart, p 220–229
Stant MY (1970) Anatomy of Petrosavia stellaris Becc., a saprophytic monocotyledon. Bot J Linn Soc 63:147–161
Steven G, Long DG (1989) An update on the status of Buxbaumia aphylla on bings in central Scotland. Bot J Scotland 45:389–395
Stevens PF (2001) onwards. Angiosperm Phylogeny Website. http://www.mobot.org/MOBOT/research/APweb/
Stevens PF, Luteyn J, Oliver EGH, Bell TL, Brown EA, Crowden RK, George AS, Jordan GJ, Ladd P, Lemson K, McLean CB, Menadue Y, Pate JS, Stace HM, Weiller CM (2004) Ericaceae. In: Kubitzki K (ed) The families and genera of vascular plants, vol 6. Springer, Heidelberg, pp 145–194
Stockey RA, Rothwell GW, Accy HD, Currah RS (2001) Mycorrhizal association of the extinct conifer Metasequoia milleri. Mycol Res 105:202–205
Stone BC (1980) Rediscovery of Thismia clavigera (Becc.) F. v. M. (Burmanniaceae). Blumea 26:419–425
Stone G (1983) Buxbaumia in Australia, including one new species, Buxbaumia thorsborneae. J Bryol 12:541
Sun B-Y, Kim MH, Kim CH, Park C-W (2001) Mankyua (Ophioglossaceae): a new fern genus from Cheju Island, Korea. Taxon 50:1019–1024
Süssenguth K (1937) Einige bemerkenswerte Pflanzen des Münchner Staatsherbars. Repert Spec Nov Regni Veg 42:43–49
Svedelius N (1902) Zur Kenntnis der saprophytischen Gentianaceen. Kongl Svenska Vetenskapsakad Handl Bih 28(3) No. 4:3–16
Swatzell LJ, Powell MJ, Kiss JZ (1996) The relationship of endophytic fungi to the gametophyte of the fern Schizaea pusilla. Int J Plant Sci 157:53–62
Takeuchi W, Pipoly JJ III (1998) New flowering plants from southern New Ireland, Papua New Guinea. Sida 18:161–168
Takhtajan A (1997) Diversity and classification of flowering plants. Columbia Univ. Press, New York
Taylor DL, Bruns TD (1997) Independent, specialized invasions of ectomycorrhizal mutualism by two nonphotosynthetic orchids. Proc Natl Acad Sci U S A 94:4510–4515
Taylor DL, Bruns TD, Leake JR, Read DJ (2002) Mycorrhizal specificity and function in myco-heterotrophic plants. In: van der Heijden MGA, Sanders I (eds) Mycorrhizal ecology. Springer, Berlin, pp 375–413
Taylor TN, Remy W, Hass H, Kerp H (1995) Fossil arbuscular mycorrhizae from the early Devonian. Mycologia 87:560–573
ter Welle BJH (1986) Anatomy. In: Maas PJM, Ruyters P (eds) Voyria and Voyriella (Saprophytic Gentianaceae). Flora Neotropica Monograph 41:25–29
Terashita T, Kawakami Y (1991) An endomycorrhizal fungus of Burmannia liukiuensis. Trans Mycol Soc Japan 32:207–215
Thorne RF (1983) Proposed new realignments in the angiosperms. Nord J Bot 3:85–117
Thorne RF (1992) Classification and geography of the flowering plants. Bot Rev 58:225–348
Tomlinson PB (1982) Helobiae (Alismatidae). In: Metcalfe CR (ed) Anatomy of the monocotyledons, vol 7. Clarendon, Oxford, pp 1–559
Treub M (1885) Études sur les Lycopodiacées. Ann Jard Bot Buitenzorg 5:87–139
Treub M (1890) Études sur les Lycopodiacées. Ann Jard Bot Buitenzorg 8:1–37
Tsukaya H, Yokoyama J, Imaichi R, Ohba H (2008) Taxonomic status of Monotropastum humile, with special reference to M. humile var. glaberrimum (Ericaceae, Monotropoideae). J Plant Res 121:271–278
Udar R, Srivastava SC, Kumar D (1971) A new species of Buxbaumia Hedwig, B. himalayensis Udar, Srivastava et Kumar, from Deoban, Western Himalayas. Trans Brit Bryol Soc 6:266–269
Uphof JCT (1929) Beiträge zur Kenntnis der Burmanniacee Apteria aphylla (Nutt.). Barnh Österr Bot Z 78:71–80
van de Meerendonk JPM (1984) Triuridaceae. In: van Steenis CGGJ (ed) Flora Malesiana, Ser. I, vol 10. Kluwer Academic Publishers, Dordrecht, pp 109–121
van der Meijden R (1988) Polygalaceae. In: van Steenis CGGJ, de Wilde WJJO (eds) Flora Malesiana, vol 10. Kluwer Academic Publishers, Dordrecht, pp 455–539
van der Pijl L (1934) Die Mycorrhiza von Burmannia und Epirrhizanthes und die Fortpflanzung ihres Endophyten. Recueil Trav Bot Néerl 31:761–779
van Rompu W, Stieperaere H (2002) Fate of sporophytes in a Belgian population of Buxbaumia aphylla. J Bryol 24:83–85
van Royen P (1972) Sertulum papuanum 17. Corsiaceae of New Guinea and surrounding areas. Webbia 27:223–255
Vigodsky-de Philippis A (1938) Studio morfologico ed anatomica di Leipaimos brachyloba’ (Griseb.) Urb. var. ‘cumbrensis’ Urb. et Ekm. Nuovo Giorn Bot Ital 45:190–194
Vincenot L, Tedersoo L, Richard R, Horcine H, Kõljalg U, Selosse M-A (2008) Fungal associates of Pyrola rotundifolia, a mixotrophic Ericaceae, from two Estonian boreal forests. Mycorrhiza 19:15–25
von Malmborg S (1933) Cryptothallus nov. gen., ein saprophytisches Lebermoos. Ann Bryol 6:122–123
von Malmborg S (1934) Weiteres über die Gattung Cryptothallus. Ann Bryol 7:108–110
Wallace GD (1975) Studies of the Monotropoideae (Ericaceae). Taxonomy and distribution. Wasmann J Biol 33:1–88
Wang B, Qiu Y-L (2006) Phylogenetic distribution and evolution of mycorrhizas in land plants. Mycorrhiza 16:299–363
Wang H, Wang Z, Zhang F, Liu J, He X (1997) A cytological study on the nutrient-uptake mechanism of a saprophytic orchid Gastrodia elata. Acta Bot Sin 39:500–504
Wapstra M, French B, Davies N, O’Reilly-Wapstra J, Peters D (2005) A bright light on the dark forest floor: observations on fairy lanterns Thismia rodwayi F. Muel. (Burmanniaceae) in Tasmanian forests. Tasmanian Naturalist 127:2–18
Warming E (1901) Sur quelques Burmanniacées recueillies au Brésil par le Dr. A. Glaziou. Oversigt over Kgl. Danske Vidensk. Selsk. Forhand 1901:173–188
Watanabe K (1944) Morphologisch-biologische Studien über Miyoshia Sakuraii Makino. J Jap Bot 20:85–93
Watson L, Dallwitz MJ (2005) onwards. The moss families of the British Isles. Version: 21st June 2009. http://delta-intkey.com
Weber HC (1993) Parasitismus von Blütenpflanzen. Wissenschaftl. Buchges, Darmstadt
Wickett NJ, Goffinet B (2008) Origin and relationships of the myco-heterotrophic liverwort Cryptothallus mirabilis Malmb. (Metzgeriales, Marchantiophyta). Bot J Linn Soc 156:1–12
Wiehle W, Berg C, Grolle R (1989) Cryptothallus mirabilis Malmborg neu in Mitteleuropa. Herzogia 8:107–124
Williams LO (1946) Notes on the family Corsiaceae. Botanical Museum Leaflets Harvard University 12(5):179–182, Pl. 24
Williams S (1950) The occurrence of Cryptothallus mirabilis v. Malmb. in Scotland. Trans Brit Mycol Soc 1:357–366
Winther JL, Friedman WE (2007) Arbuscular mycorrhizal symbionts in Botrychium (Ophioglossaceae). Am J Bot 94:1248–1255
Winther JL, Friedman WE (2008) Arbuscular mycorrhizal associations in Lycopodiaceae. New Phytol 177:790–801
Wolf W (1922) Notes on Alabama plants. Am Midl Nat 8:104–127
Woltz P, Stockey RA, Gondran M, Cherrier J-F (1994) Interspecific parasitism in the Gymnosperms: unpublished data on two endemic New Caledonian Podocarpaceae using scanning electron microscopy. Acta Bot Gallica 141:731–746
Wood CE, Weaver RE (1982) The genera of the Gentianaceae in the Southeastern United States. J Arnold Arbor 63:441–487
Woodward CL, Berry PE, Maas-van de Kamer H, Swing K (2007) Tiputinia foetida, a new mycoheterotrophic genus of Thismiaceae from Amazonian Ecuador, and a likely case of deceit pollination. Taxon 56:157–162
Yahara T, Tsukaya H (2008) Oxygyne yamashitae, a new species of Thismiaceaefrom Yaku Island, Japan. Acta Phytotax Geobot 59:97–104
Yamada A, Kitamura D, Setoguchi M, Matsuda Y, Hashimoto Y, Matsushita N, Fukuda M (2008) Monotropastrum humile var. humile is associated with diverse ectomycorrhizal Russulaceae fungi in Japanese forests. Ecol Res 23:983–993
Yamato M (2001) Identification of a mycorrhizal fungus in the roots of achlorophyllous Sciaphila tosaensis Makino (Triuridaceae). Mycorrhiza 11:83–88
Yamato A, Yagame T, Shimomura N, Iwase K, Takahashi H, Ogura-Tsujita Y, Yukawa T (2011a) Specific arbuscular mycorrhizal fungi associated with non-photosynthetic Petrosavia sakuraii (Petrosaviaceae). Mycorrhiza 21:631–639
Yamato A, Yagame T, Iwase K (2011b) Arbuscular mycorrhizal fungi in roots of non-photsynthetic plants, Sciaphila japonica and Sciaphila tosaensis (Triuridaceae). Mycoscience 52:217–233
Yang S, Pfister DH (2006) Monotropa uniflora plants of eastern Massachusetts form mycorrhizae with a diversity of russulacean fungi. Mycologia 98:535–540
Yang S-Z, Saunders RMK, Hsu C-J (2002) Thismia taiwanensis sp. nov. (Burmanniaceae tribe Thismieae): first record of the tribe in China. Syst Bot 27:485–488
Yokoyama J, Fukuda T, Tsukaya H (2005) Molecular identification of the mycorrhizal fungi of the epiparasitic plant Monotropastrum humile var. glaberrimum (Ericaceae). J Plant Res 118:53–56
Yokoyama J, Koizumi Y, Yokota M, Tsukaya H (2008) Phylogenetic position of Oxygyne shinzatoi (Burmanniaceae) inferred from 18S rDNA sequences. J Plant Res 121:27–32
Young BW, Massicotte HB, Tackaberry LE, Baldwin QF, Egger KN (2002) Monotropa uniflora: morphological and molecular assessment of mycorrhizae retrieved from sites in the Sub-Boreal Spruce biogeoclimatic zone in central British Columbia. Mycorrhiza 12:75–82
Yuan Y-M, Wohlhauser S, Möller M, Chassot P, Mansion G, Grant J, Küpfer P, Klackenberg J (2003) Monophyly and relationships of the tribe Exaceae (Gentianaceae) inferred from nuclear ribosomal and chloroplast DNA sequences. Mol Phylogenet Evol 28:500–517
Zelmer CD, Currah RS (1995) Evidence for a fungal liaison between Corallorhiza trifida (Orchidaceae) and Pinus contorta (Pinaceae). Can J Bot 73:862–866
Zhang DX, Saunders RM (1999) Burmannia larseniana (Burmanniaceae): a new species from Thailand. Nord J Bot 19:241–244
Zhang DX, Saunders RMK, Hu CM (1999) Corsiopsis chinensis gen. et sp. nov. (Corsiaceae): first record of the family in Asia. Syst Bot 24(3):311–314
Zimmer K, Meyer C, Gebauer G (2008) The ectomycorrhizal specialist orchid Corallorhiza trifida is a partial myco-heterotroph. New Phytol 178:395–400
Acknowledgements
Thanks to Mori Thomann for his help with the Japanese paper of K. Watanabe, as well as Jesper Hansen for translating parts of the Danish articles by V. A. Poulsen.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2013 Springer Science+Business Media New York
About this chapter
Cite this chapter
Imhof, S., Massicotte, H.B., Melville, L.H., Peterson, R.L. (2013). Subterranean Morphology and Mycorrhizal Structures. In: Merckx, V. (eds) Mycoheterotrophy. Springer, New York, NY. https://doi.org/10.1007/978-1-4614-5209-6_4
Download citation
DOI: https://doi.org/10.1007/978-1-4614-5209-6_4
Published:
Publisher Name: Springer, New York, NY
Print ISBN: 978-1-4614-5208-9
Online ISBN: 978-1-4614-5209-6
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)