Abstract
Understanding the molecular basis of plant responses to the major abiotic stresses such as drought and salinity is very important for the biotechnological application of stress adaptation for crop improvement. In this context, thousands of stress-responsive genes have been identified and a few of them have been functionally characterized. Some of them have been proposed as suitable targets for genetic engineering in order to impart stress tolerance in plants. Amidst numerous genes analyzed, transcription factors are considered to be very good targets for studying the molecular mechanisms of abiotic stress response as they singularly or in conjunction regulate the expression of many downstream target genes. Among the various transcription factor encoding genes, homeobox genes, which are well known to be involved in diverse aspects of development, have also recently been implicated in abiotic stress responses. Through various overexpression and mutant studies, the versatility of homeobox genes in plants has been revealed. There are evidences where these genes have been found to confer stress tolerance in plants. This review highlights the importance of homeobox genes in abiotic stress responses and their potential for engineering stress tolerance for crop improvement.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
1 Introduction
Various abiotic stresses such as drought, salinity, and extremes of temperature pose a major challenge for survival of plants and have a great impact on crop productivity. The world population is increasing at an alarming rate and on the contrary, availability of food resources is decreasing due to the abiotic stress factors. Thus, there is a great need to generate stress-tolerant crop plants with improved sustenance and better yield. Plants elicit several responses to combat the adverse effects of various abiotic stresses, including production and accumulation of osmolytes, maintenance of intracellular ion homeostasis, and scavenging of reactive oxygen species. Understanding plant responses to abiotic stresses at the molecular level provides an essential foundation for future breeding and genetic engineering programs. Abiotic stress response is a complex trait, which involves interplay of numerous regulatory molecules at the cellular level. Various approaches have provided a holistic view of the ongoing cellular activities in response to abiotic stresses.
Numerous genes which are induced in response to abiotic stresses have been identified and the products of these genes are supposed to enhance stress tolerance in plants. The role of several stress-inducible genes has been explored, which regulate gene expression via various signal transduction pathways. Among these genes, transcription factors represent master switches controlling several target genes and are considered most important for regulation of gene expression. Although several transcription factors have been implicated in abiotic stress responses, only a few master switches/regulons have been identified and characterized in detail so far. The identification of master switches, which control stress-inducible genes, seems to be the most challenging task. Serious endeavor needs to be made in this direction to identify promising master regulator genes and eventually gain insights into the complex gene regulatory network, which leads to abiotic stress responses.
Several excellent comprehensive reviews are available on various aspects of complex signal transduction pathways controlling abiotic stress responses, emerging trends in genomics of abiotic stress responses and gene regulatory networks involved in abiotic stress responses and tolerance (Bartels and Sunkar 2005; Chinnusamy et al. 2004; Hirayama and Shinozaki 2010; Mahajan and Tuteja 2005; Nakashima et al. 2009; Urano et al. 2010; Vij and Tyagi 2007; Yamaguchi-Shinozaki and Shinozaki 2006). In this review, we provide a brief overview of the role of transcription factors in abiotic stress responses and majorly focus on the emerging role of homeobox transcription factors in abiotic stress responses and their potential as target genes for engineering stress tolerance.
2 Overview of Stress-Responsive Genes
Several studies in Arabidopsis and rice have revealed that quite a large proportion of the genome is involved in abiotic stress responses. The global expression profiling in various plant species has revealed that expression of thousands of genes is altered in response to various abiotic stress conditions (Hadiarto and Tran 2011; Hirayama and Shinozaki 2010; Nakashima et al. 2009; Urano et al. 2010; Vij and Tyagi 2007). These stress-responsive genes have been broadly categorized in two groups (Yamaguchi-Shinozaki and Shinozaki 2006). The first group is comprised of genes involved directly in abiotic stress tolerance, including those encoding for metabolic proteins like water channel proteins, enzymes required for synthesis of various osmoprotectants (like sugars, glycine-betaine, and proline), proteins that aid in protecting macromolecules and membranes (for example, LEA proteins, osmotin, chaperones), proteases for protein turnover and detoxification enzymes like glutathione-S-transferases. The second group is comprised of genes encoding for regulatory proteins like protein kinases, transcription factors, protein phosphatases, and other signaling molecules. Overall, the expression of genes involved in diverse cellular processes is altered in response to abiotic stresses. The comparative analysis has revealed that a larger fraction of stress-responsive genes are common among various plant species, indicating the conserved mechanism of abiotic stress response. Further, many of these genes have been analyzed for their exact function and ability to provide stress tolerance in transgenic plants (Vij and Tyagi 2007).
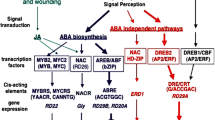
The emerging role of various plant hormones has also been explored in the context of abiotic stress responses. Abscisic acid (ABA) is found to be the key hormone produced in plants under stress conditions and is crucial for abiotic stress responses (Hirayama and Shinozaki 2007). The application of exogenous ABA often mimics the abiotic stress responses. The expression of several stress-responsive genes is also induced by ABA (Shinozaki et al. 2003; Zhu 2002). However, several studies report that many stress-responsive genes do not show response to ABA, suggesting the existence of two signal transduction pathways, namely ABA-dependent and ABA-independent pathways, in abiotic stress responses (Nakashima et al. 2009; Zhu 2002). Other phytohormones, such as salicylic acid, ethylene, and jasmonic acid have also been shown to play important roles in abiotic stress responses directly or via interplay with ABA (Fujita et al. 2006; Grant and Jones 2009; Pieterse et al. 2009). Recently, auxin has also been implicated in abiotic stress responses. Quite a large number of auxin-responsive genes have been shown to be differentially expressed under various abiotic stress conditions (Jain and Khurana 2009).
Although a comprehensive knowledge has accumulated about the stress-responsive genes, the biggest challenge is to decipher their functions and logically integrate the available knowledge to understand the mechanism underlying abiotic stress response and selection of most suitable target gene(s) for improving stress tolerance.
3 Overview of Gene Regulatory Network Involved in Abiotic Stress Response
The differential expression of a large number of genes indicates that the gene regulatory network operative during abiotic stress is very complex in plants. The expression of several transcription factor encoding genes is also induced in response to various abiotic stresses. The transcription factors (TFs) are considered as master regulators of the gene expression. An individual TF can govern the expression of numerous target genes by binding to specific cis-regulatory motifs present in their promoters either independently or in coordination with other proteins constituting gene regulatory network (Nakashima et al. 2009; Urano et al. 2010). Some of the stress-associated TFs are themselves regulated at the transcriptional level. This type of transcriptional regulatory system is called regulon and is required for fine-tuned gene expression in response to abiotic stresses.
TFs and their regulons involved in both ABA-dependent and ABA-independent pathways have been identified and characterized in plants (Nakashima et al. 2009). The regulons involving dehydration-responsive element (DRE) binding protein 1 (DREB1)/C-repeat binding factor (CBF) and DREB2 TFs regulate stress response via ABA-independent pathway. These TFs bind to the conserved DRE motif A/GCCGACNT sequence in the promoter region of their target genes. The regulons involving ABA-responsive element (ABRE) binding protein (AREB)/ABRE binding factor (ABF) TFs act via ABA-dependent pathway (Nakashima et al. 2009). AREB/ABF TFs harboring a bZIP type DNA-binding domain binds to the ABRE (PyACGTGG/TC) and plays a pivotal role in ABA-dependent gene activation (Choi et al. 2000). Other regulons comprised of NAC and MYB/MYC TFs are also supposed to regulate the abiotic stress response via ABA-independent pathway. NAC proteins recognize a MYC-like target sequence and activate downstream gene expression (Tran et al. 2006). The components of these regulons and their functions have been found to be conserved in dicots and monocots indicating the common regulatory mechanisms of gene expression among them in response to stress (Nakashima et al. 2009). Recently, another regulon involving the transcriptional regulator, multiprotein bridging factor 1 (MBF1), has been identified in Arabidopsis, which regulates heat-response (Suzuki et al. 2011).
Although most of the TF regulons are functional and have overlapping roles in response to multiple stresses, some of them are specific to particular abiotic stress condition(s) only. DREB1/CBF regulon responds to cold stress and controls the expression of several downstream genes. The overexpression of DREB1/CBF of Arabidopsis and homologous genes from other plants in transgenics resulted in strong tolerance to abiotic stresses, including drought, high salinity, and freezing (Dubouzet et al. 2003; Kasuga et al. 1999; Qin et al. 2004). DREB2 regulon functions in both osmotic and heat stress responses in Arabidopsis, whereas in dehydration and high salinity in grasses (Nakashima et al. 2009). Arabidopsis AREB/ABFs are induced in response to ABA, dehydration, and high salinity. The overexpression of AREB1 resulted in ABA hypersensitivity and drought tolerance (Fujita et al. 2005). It has also been suggested that phosphorylation may be responsible for the activation of AREB/ABFs (Fujii et al. 2007; Furihata et al. 2006; Uno et al. 2000). The response of AREB/ARF regulon to dehydration and high salinity has been found to be conserved in rice also (Kagaya et al. 2002; Kobayashi et al. 2005). Further, the overexpression of NAC genes conferred tolerance to drought stress in transgenic Arabidopsis plants and up-regulated several stress-inducible genes (Fujita et al. 2004; Tran et al. 2004). It has also been demonstrated that NAC TFs act along with ZF-HD proteins to activate the expression of downstream target gene, EARLY RESPONSE TO DEHYDRATION 1 (ERD1) (Tran et al. 2006). In rice, one of the NAC TFs has been found to be responsive to ABA, abiotic stresses and biotic stresses, and its overexpression imparted enhanced stress tolerance in transgenic plants (Nakashima et al. 2007).
Taken together, TFs play important roles in abiotic stress responses and are powerful targets for engineering stress tolerance in transgenic plants because the overexpression of a single TF may lead to induction of diverse stress-responsive genes. Further, the role of many other stress-responsive TFs and their regulons need to be identified for understanding the molecular mechanisms of abiotic stress responses.
4 Homeobox Genes
Homeobox genes represent a class of TFs containing a conserved 180 bp long DNA sequence, which encodes for a 60 amino acid long DNA-binding domain termed as homeodomain (HD). The HD consists of three alpha helices forming a helix- turn-helix, which binds to specific DNA sequence and regulates the expression of target genes. The first homeobox genes were identified in Drosophila melanogaster and thereafter in all the eukaryotes. Homeobox genes are known to be the key regulators of various aspects of development, including cell fate determination and body plan specification.
Homeobox genes are represented by a large multigene family in plants, which have been classified into several distinct classes based on the amino acid sequence of HD and presence of other conserved domains. Initially, homeobox genes were classified into seven classes, including KNOX, BEL, ZM-HOX, HAT1, HAT2, ATHB8, and GL2 (Bharathan et al. 1997). Later on, Chan et al. (1998) classified homeobox genes into five groups (HD-ZIP, GLABRA, KNOTTED, PHD, and BEL). However, based on genome-wide analysis, 107 homeobox genes were classified into ten distinct subfamilies, including HD-ZIP I, HD-ZIP II, HD-ZIP III, HD-ZIP IV, ZF-HD, PHD, BLH, KNOXI, KNOXII, and WOX, in rice (Jain et al. 2008). Among these, HD-ZIP represented the largest family comprising of at least 48 members. The expansion of homeobox gene family has been attributed due to the chromosomal segmental duplications in rice, which might be responsible for the diversification of their function (Jain et al. 2008). Most recently, a comprehensive classification of plant homeobox genes based on the characterization of new motifs has been accomplished from the analysis of ten complete genomes of flowering plants, mosses, Selaginella, unicellular green algae, and red algae (Mukherjee et al. 2009). A total of 14 classes were identified across various plant species, namely HD-ZIP I-IV, PLINC, WOX, KNOX, BEL, PHD, DDT, NDX, LD, SAWADEE, and PINTOX. The conservation of homeobox genes across lineages emphasized their functional significance. In a recent study, it has also been shown that uncharacterized conserved motifs outside the HD-ZIP domain of HD-ZIP I subfamily confers functional diversity to members of this group of homeobox genes (Arce et al. 2011). Further, a greater number of homeobox genes in flowering plants have been related to their higher developmental and organizational complexity (Mukherjee et al. 2009).
5 Role of Homeobox Genes in Plant Development
The role of homeobox genes in plant developmental patterns has been extensively explored. The homeobox genes belonging to different subfamilies exhibit distinct expression patterns indicating their specific regulatory roles in tissue/organ differentiation and development (Chan et al. 1998). The molecular genetic analyses of several mutants have revealed that the KNOX family homeobox genes (SHOOTMERISTEMLESS, BREVIPEDICELLUS, KNAT2 and KNAT6 in Arabidopsis) are the key determinants in the maintenance of shoot apical meristem (Hake et al. 2004). The role of KNOX genes as versatile regulators of plant development and diversity have been comprehensively reviewed recently (Hay and Tsiantis 2010). KNOX proteins interact with HD proteins of BELL family to regulate the target genes that control hormone homeostasis (Hake et al. 2004; Hay et al. 2004; Hay and Tsiantis 2010; Smith et al. 2002). BELL family HD proteins are involved in pattern formation, stem-cell fate determination and tuber formation either independently or in coordination with KNOX proteins (Byrne et al. 2003; Chen et al. 2003; Reiser et al. 1995). WUSCHEL and other WOX family homeobox genes are involved in embryonic patterning, stem-cell maintenance, and organ formation (van der Graaff et al. 2009). WUSHEL has also been reported to be involved in regulation of cell differentiation during anther development (Deyhle et al. 2007). Very recently, it has been shown that the overexpression of WOX1 leads to meristem developmental defects in Arabidopsis (Zhang et al. 2011). Further, ZF-HD family proteins play a critical role in floral development in Arabidopsis (Tan and Irish 2006). The members of HD-ZIP family have been implicated in several developmental processes (Ariel et al. 2007). The ectopic expression of members of HD-ZIP I class suggested their role in the regulation of cotyledon development, leaf cell fate determination and blue-light perception signaling (Aoyama et al. 1995; Henriksson et al. 2005; Wang et al. 2003). HD-ZIP II class proteins have a role in plant development associated with shade avoidance responses (Sessa et al. 2005). The class III HD-ZIP proteins have been well characterized as regulators of apical meristem formation, vascular development, and maintenance of adaxial or abaxial polarity of leaves and embryo (Prigge et al. 2005; Talbert et al. 1995). HD-ZIP IV proteins are supposed to play specific roles in the outer cell layer of the plant organs (Nakamura et al. 2006). The presence of a large number of HD-ZIP proteins in plants may be responsible for the fine regulation of the developmental program as a result of the evolutionary pressure (Ariel et al. 2007).
HD-ZIP III class HD proteins are known to be the targets of miRNAs in Arabidopsis. It has been demonstrated that their miRNA-mediated post-transcriptional regulation controls the establishment of adaxial–abaxial polarity (McConnell et al. 2001; Rhoades et al. 2002; Tang et al. 2003). Recently, small RNA signatures were found to be associated with a significantly large fraction of homeobox genes in rice (Jain and Khurana 2008). Further, the results suggested an unusually high degree of small RNA regulation of rice homeobox genes during panicle development (Jain and Khurana 2008). This study provided evidence for a highly complex small RNAs-mediated regulation of homeobox genes involved in various cellular processes.
6 Role of Homeobox Genes in Abiotic Stress Responses
6.1 Differential Expression Under Abiotic Stress Conditions
The differential expression of individual homeobox genes in response to abiotic stresses has been reported in many plant species (Deng et al. 2002; Frank et al. 1998; Gago et al. 2002; Lee and Chun 1998; Soderman et al. 1996, 1999; Tran et al. 2006). In rice and Arabidopsis, the expression levels of quite a large number of homeobox genes have been found to be altered in response to various abiotic stress conditions. ATHB7 and ATHB12 transcripts were present in all organs at a basal level but there was phenomenal increase in the transcript level once the plant was exposed to drought conditions (Hjellstrom et al. 2003; Olsson et al. 2004; Soderman et al. 1996). Microarray analysis revealed that among a total of 107 homeobox genes, 37 were differentially expressed under desiccation, salt and/or cold stress conditions in rice seedlings (Jain et al. 2008). The expression of some of these genes was regulated by a specific stress and others by multiple stresses. A few HD-ZIP genes showed differential expression in the flowering stage of drought-sensitive and drought-tolerant rice cultivars also (Agalou et al. 2008; Bhattacharjee and Jain, unpublished), which further confirmed their role in abiotic stress responses. The activity of HD TFs was found to be regulated by cellular redox status also (Tron et al. 2002). Many of the homeobox genes, whose expression is regulated by abiotic stresses, were found to be preferentially expressed during specific developmental stage(s) (Jain et al. 2008). This suggested the role of homeobox TFs as mediators of plant growth response to different abiotic stress conditions during various stages of development. A novel homeobox gene, GhHB1 (HD-ZIP I class member) from cotton has also been identified, which was specifically expressed in roots (Ni et al. 2008). The expression of this gene was found to be up-regulated in the presence of exogenous salt and ABA.
6.2 ABA-Mediated Regulation of Homeobox Genes
The plant hormone ABA plays a key role in adaptive stress responses to environmental stimuli (Cutler et al. 2010; Fujita et al. 2011; Raghavendra et al. 2010). Much information is not available about the role of homeobox genes in ABA-dependent abiotic stress response pathways. Many of the Arabidopsis HD-ZIP class homeobox genes, including ATHB6, ATHB7 and ATHB12 have been shown to be induced in response to exogenous ABA (Henriksson et al. 2005; Lee and Chun 1998; Soderman et al. 1996). However, no induction of ATHB7 and ATHB12 was observed in the ABA-deficient mutants (Olsson et al. 2004; Soderman et al. 1996). Further, the mutants of these genes exhibited reduced sensitivity and transgenic Arabidopsis plants overexpressing them were hypersensitive to exogenous ABA as compared to wild type, indicating their role in ABA-dependent abiotic stress response pathways (Olsson et al. 2004). Another homeobox gene, ATHB5, has been characterized as a positive regulator of ABA response in developing seedlings (Johannesson et al. 2003). ATHB6 protein has been identified as the interacting protein of protein phosphatase ABI1 and acts as negative regulator of ABA signaling pathway downstream of ABI1 (Himmelbach et al. 2002). The N-terminal domain of ATHB6 and protein phosphatase domain of ABI1 were found to be crucial for their interaction. It is known that the phosphorylation of transcription factors play a major role in DNA binding and in this case, the PP2C activity of ABI1 has been shown to be responsible for the interaction between ATHB6 and ABI1. A single point mutation is sufficient to prevent the interaction to take place, further suggesting the importance of these phosphorylation reactions for interacting proteins. The binding site of ATHB6 protein (CAATTATTA) has also been identified, which along with minimal promoter was sufficient to mediate ABA-dependent activation of gene expression (Himmelbach et al. 2002). We have also found the expression of some rice homeobox genes to be regulated by exogenous ABA [Bhattacharjee and Jain, unpublished]. Altogether, these studies suggest that HD TFs may be implicated mainly in ABA-dependent abiotic stress response pathways. However, their role in ABA-independent pathways cannot be ruled out.
6.3 Implication of Homeobox Genes in Abiotic Stress Tolerance
Only a few mutant and transgenic studies have been performed in plants, which implicate the homeobox genes in abiotic stress responses. In Arabidopsis, a few mutants have been isolated and analyzed, which validate the role of homeobox genes in abiotic stress responses. A homeobox gene mutant, hos9 hypersensitive to freezing before and after cold acclimation as compared to wild-type plants, was identified by large scale screening of Arabidopsis mutants (Zhu et al. 2004). The transcript levels of RD29A and other stress-inducible genes increased in the hos9 mutant as compared to wild-type plants after cold treatment. However, there was no alteration in the expression level of CBF genes, which are otherwise believed to play an important role in cold acclimation. Further, none of the CBF family genes were differentially expressed in the hos9 mutant, suggesting the role of HOS9 homeobox TF in mediating cold tolerance via CBF-independent pathway (Zhu et al. 2004).
More recently, one mutant with improved drought tolerance, enhanced drought tolerance1 (edt1), was isolated from Arabidopsis in a gain-of-function genetic screen (Yu et al. 2008). The enhanced drought tolerance of the mutant was found due to the activated expression of a HD-ZIP IV class member, HDG11. The mutant exhibited more extensive root system, higher levels of superoxide dismutase activity and elevated levels of ABA and proline as compared to wild-type plants. Further, the overexpression of this homeobox gene conferred drought tolerance in transgenic tobacco plants, which was attributed to the improved root architecture and reduced leaf stomatal density. Unlike other TFs, such as DREB (Kasuga et al. 1999), the constitutive expression of HDG11 did not cause growth retardation (Yu et al. 2008), which is agronomically very important for crop improvement. Several stress-responsive genes involved in ABA signaling and calcium signaling were induced in the mutant plants. It has been suggested that HDG11 may regulate a complex network of genes to impart stress tolerance. Further, the possibility of binding of START domain of HDG11 to a lipid ligand to provide drought tolerance had also not been ruled out. Interestingly, HDG11 was found to be expressed specifically in flower buds, flowers and immature siliques, and did not respond to stress conditions (Yu et al. 2008). Although it is not clear that how exactly HDG11 mediates stress tolerance, it has been suggested that the expression pattern of this gene has allowed it to gain novel function in drought tolerance (Yu et al. 2008).
The transgenic plants overexpressing ATHB7 and ATHB12 showed altered phenotype mimicking stress conditions and suggested their involvement in the growth response to water-deficit in shoot and root (Olsson et al. 2004). In addition, ATHB12 functionally complemented the NaCl-sensitive phenotype of a calcineurin-deficient yeast mutant and increased salt tolerance by regulating sodium ion homeostasis (Shin et al. 2004). In another study, the cDNA encoding for ZFHD1 TF was identified in a yeast one-hybrid screen using 62 bp promoter region of ERD1 containing ZF-HD recognition sequence (Tran et al. 2006). The transgenic plants overexpressing ZFHD1 revealed a significant improvement in drought tolerance and upregulation of several stress-inducible genes. Further, yeast two-hybrid analysis identified NAC proteins as interacting partners of ZFHD1. Interestingly, the ERD1 transcripts accumulated at higher level in the plants overexpressing both ZFHD1 and NAC proteins simultaneously, but not in transgenics overexpressing ZFHD1 only (Tran et al. 2006). These results validated the earlier observation that induction of ERD1 gene requires coordinated activity of ZFHDRS and NACRS cis-regulatory elements (Simpson et al. 2003) and provides evidence for regulation of abiotic stress responses by homeobox TFs in conjunction with other transcription factors.
In crop plants, the role of only one homeobox gene from rice, OsBIHD1, encoding a BELL-type TF, in stress responses has been analyzed in transgenics so far (Luo et al. 2005). The overexpression of OsBIHD1 in transgenic tobacco plants elevated the levels of defense-related PR-1A gene expression. Various analyses revealed that these transgenic lines had developed enhanced disease resistance against tomato mosaic virus, tobacco mosaic virus, and Phytopthora parasitica. However, in contrast, these transgenics exhibited enhanced sensitivity to salt and oxidative stresses. It has been suggested that OsBIHD1 may be acting as a negative regulator of stress tolerance by suppressing the abiotic stress signaling cascade in overexpression transgenic tobacco plants. Further, these results suggested that OsB1HD1 might be involved in different pathways to regulate abiotic and biotic stress responses (Luo et al. 2005).
The functional characterization of homeobox genes in abiotic stress responses has been carried out in other plants also. For example, Hahb-4, a sunflower HD-ZIP gene acts as a developmental regulator and has been shown to confer drought tolerance in Arabidopsis plants (Dezar et al. 2005). Additionally, in sunflower, the Hahb-4 gene has been potentially shown to be involved in ABA-dependent responses to water stress (Gago et al. 2002). Moreover, in resurrection plant Craterostigma plantagineum, identification of five novel dehydration-responsive HD-ZIP proteins has been done (Deng et al. 2002). In Brassica napus, a HD-ZIP gene, BnHB6, has been reported to be involved in both biotic and abiotic stress responses (Yu et al. 2005). These evidences suggest crucial involvement of the homeobox genes in stress-responsive signaling in various plant species.
Very little is known about the identity of downstream target genes of HD TFs. Although a few studies have reported the identification of putative targets of homeobox TFs, convincing evidences are not available as of now. For example, a late embryogenesis abundant/dehydrin gene, CdeT6-19, has been identified as the potential target of CpHB-7 gene in Craterostigma plantageneum (Deng et al. 2006). In addition, based on the macroarray analysis and mining of HD-ZIP binding site containing genes, several known ABA-responsive genes were proposed to be the targets of CpHB-7. Further, in another study, microarray analysis identified the genes involved in ethylene signaling and synthesis as targets in transgenic Arabidopsis plants overexpressing Hahb-4 gene (Manavella et al. 2006). The identification of downstream targets of homeobox genes will be very important as it would help in unraveling the exact role of these homeobox TFs in the gene regulatory network of abiotic stress responses.
7 Conclusion and Perspectives
Plants face tremendous challenge when they are exposed to various abiotic stresses. Hence, they develop several modes of adaptability to protect themselves against these stresses. The recognition of key components which can reduce the deleterious effects of abiotic stresses to plants is very important for understanding the molecular mechanisms responsible for stress response and tolerance. Several genes including TFs involved in stress responses have been identified, but their exact role has not been studied yet in crop plants. So, there is a need to identify the master regulators and their regulatory pathways involved in stress adaptation. Further, it is imperative to select suitable candidates for conferring stress tolerance in plants via genetic engineering. The homeobox genes represent a family of transcription factors, which regulate the expression of a plethora of target genes. It is already well established that these genes play central role in regulating various developmental processes. The recent studies suggest that homeobox genes are promising candidate targets for manipulating abiotic stress tolerance in plants and can be used for crop improvement. These genes have largely overlapping roles in development and stress responses and thus, may offer multiple advantages, when attempts are made to raise overexpression transgenic plants. As the evidences related to the role of homeobox genes in providing stress tolerance available as of now are preliminary, it will be important to carry out their detailed functional analysis. Probably, the overexpression of homeobox genes may be able to impart stress tolerance in transgenics without compromising the yield, growth, and development of plants. Further, the identification of other regulatory components and target genes of stress-responsive homeobox TFs may lead to identification of novel pathways and better understanding of underlying molecular mechanisms. In addition, it would be very interesting to study the role of small RNAs in HD TFs-mediated regulation of abiotic stress responses.
References
Agalou A, Purwantomo S, Overnas E et al (2008) A genome-wide survey of HD-Zip genes in rice and analysis of drought-responsive family members. Plant Mol Biol 66:87–103
Aoyama T, Dong CH, Yan WU et al (1995) Ectopic expression of the Arabidopsis transcriptional activator ATHB-1 alters leaf cell fate in tobacco. Plant Cell 7:1773–1785
Arce AL, Raineri J, Capella M, Cabello JV, Chan RL (2011) Uncharacterized conserved motifs outside the HD-Zip domain in HD-Zip subfamily I transcription factors; a potential source of functional diversity. BMC Plant Biol 11:42
Ariel FD, Manavella PA, Dezar CA, Chan RL (2007) The true story of the HD-Zip family. Trends Plant Sci 12:419–426
Bartels D, Sunkar R (2005) Drought and salt tolerance in plants. Crit Rev Plant Sci 24:23–58
Bharathan G, Janssen BJ, Kellogg EA, Sinha N (1997) Did homeodomain proteins duplicate before the origin of angiosperms, fungi, and metazoa? Proc Natl Acad Sci USA 94:13749–13753
Byrne ME, Groover AT, Fontana JR, Martienssen RA (2003) Phyllotactic pattern and stem cell fate are determined by the Arabidopsis homeobox gene BELLRINGER. Development 130:3941–3950
Chan RL, Gago GM, Palena CM, Gonzalez DH (1998) Homeoboxes in plant development. Biochim Biophys Acta 1442:1–19
Chen H, Rosin FM, Prat S, Hannapel DJ (2003) Interacting transcription factors from the three-amino acid loop extension superclass regulate tuber formation. Plant Physiol 132:1391–1404
Chinnusamy V, Schumaker K, Zhu JK (2004) Molecular genetic perspectives on cross-talk and specificity in abiotic stress signaling in plants. J Exp Bot 55:225–236
Choi H, Hong J, Ha J, Kang J, Kim SY (2000) ABFs, a family of ABA-responsive element binding factors. J Biol Chem 275:1723–1730
Cutler SR, Rodriguez PL, Finkelstein RR, Abrams SR (2010) Abscisic acid: Emergence of a core signaling network. Annu Rev Plant Biol 61:651–679
Deng X, Phillips J, Meijer AH, Salamini F, Bartels D (2002) Characterization of five novel dehydration responsive homeodomain leucine zipper genes from the resurrection plant Craterostigma plantagineum. Plant Mol Biol 49:601–610
Deng X, Phillips J, Bräutigam A et al (2006) A homeodomain leucine zipper gene from Craterostigma plantagineum regulates abscisic acid responsive gene expression and physiological responses. Plant Mol Biol 61:469–489
Deyhle F, Sarkar AK, Tucker EJ, Laux T (2007) WUSCHEL regulates cell differentiation during anther development. Dev Biol 302:154–159
Dezar CA, Gago GM, Gonzalez DH, Chan RL (2005) Hahb-4, a sunflower homeobox-leucine zipper gene, is a developmental regulator and confers drought tolerance to Arabidopsis thaliana plants. Transgenic Res 14:429–440
Dubouzet JG, Sakuma Y, Ito Y et al (2003) OsDREB genes in rice, Oryza sativa L, encode transcription activators that function in drought, high-salt and cold-responsive gene expression. Plant J 33:751–763
Frank W, Phillips J, Salamini F, Bartels D (1998) Two dehydration-inducible transcripts from the resurrection plant Craterostigma plantagineum encode interacting homeodomain leucine zipper proteins. Plant J 15:413–421
Fujii H, Verslues PE, Zhu JK (2007) Identification of two protein kinases required for abscisic acid regulation of seed germination, root growth and gene expression in Arabidopsis. Plant Cell 19:485–494
Fujita M, Fujita Y, Maruyama K et al (2004) A dehydration induced NAC protein, RD26, is involved in a novel ABA-dependent stress-signaling pathway. Plant J 39:863–876
Fujita Y, Fujita M, Satoh R et al (2005) AREB1 is a transcription activator of novel ABRE-dependent ABA-signaling that enhances drought stress tolerance in Arabidopsis. Plant Cell 17:3470–3488
Fujita M, Fujita Y, Noutoshi Y et al (2006) Crosstalk between abiotic and biotic stress responses: a current view from the points of convergence in the stress signaling networks. Curr Opin Plant Biol 9:436–442
Fujita Y, Fujita M, Shinozaki K, Yamaguchi-Shinozaki K (2011) ABA-mediated transcriptional regulation in response to osmotic stress in plants. J Plant Res 124:509–525. http://dx.doi.org/10.1007/s10265-011-0412-3
Furihata T, Maruyama K, Fujita Y et al (2006) ABA-dependent multisite phosphorylation regulates the activity of a transcription activator AREB1. Proc Natl Acad Sci USA 103:1988–1993
Gago GM, Almoguera C, Jordano J, Gonzalez DH, Chan RL (2002) Hahb-4, a homeobox-leucine zipper gene potentially involved in abscisic acid-dependent responses to water stress in sunflower. Plant Cell Environ 25:633–640
Grant MR, Jones JDG (2009) Hormone (dis)harmony molds plant health and disease. Science 324:750–752
Hadiarto T, Tran LS (2011) Progress studies of drought-responsive genes in rice. Plant Cell Rep 30:297–310
Hake S, Smith HM, Holtan H, Magnani E, Mele G, Ramirez J (2004) The role of knox genes in plant development. Annu Rev Cell Dev Biol 20:125–151
Hay A, Tsiantis M (2010) KNOX genes: Versatile regulators of plant development and diversity. Development 137:3153–3165
Hay A, Craft J, Tsiantis M (2004) Plant hormones and homeoboxes: bridging the gap? Bioessays 26:395–404
Henriksson E, Olsson A, Johannesson H, Hanson J, Engstrom P, Soderman E (2005) Homeodomain leucine zipper class I genes in Arabidopsis expression patterns and phylogenetic relationships. Plant Physiol 139:509–518
Himmelbach A, Hoffmann T, Leube M, Hohener B, Grill E (2002) Homeodomain protein ATHB6 is a target of the protein phosphatase ABI1 and regulates hormone responses in Arabidopsis. EMBO J 21:3029–3038
Hirayama T, Shinozaki K (2007) Perception and transduction of abscisic acid signals: keys to the function of the versatile plant hormone ABA. Trends Plant Sci 12:343–351
Hirayama T, Shinozaki K (2010) Research on plant abiotic stress responses in the post-genome era: past, present and future. Plant J 61:1041–1052
Hjellstrom M, Olsson A, Engstrom P, Soderman E (2003) Constitutive expression of the water deficit-inducible homeobox gene ATHB7 in transgenic Arabidopsis causes a suppression of stem elongation growth. Plant Cell Environ 26:1127–1136
Jain M, Khurana JP (2008) Small RNA regulation of rice homeobox genes. Plant Signal Behav 3:1024–1025
Jain M, Khurana JP (2009) Transcript profiling reveals diverse roles of auxin-responsive genes during reproductive development and abiotic stress in rice. FEBS J 276:3148–3162
Jain M, Tyagi AK, Khurana JP (2008) Genome-wide identification, classification, evolutionary expansion and expression analyses of homeobox genes in rice. FEBS J 275:2845–2861
Johannesson H, Wang Y, Hanson J, Engstrom P (2003) The Arabidopsis thaliana homeobox gene ATHB5 is a potential regulator of abscisic acid responsiveness in developing seedlings. Plant Mol Biol 51:719–729
Kagaya Y, Hobo T, Murata M, Ban A, Hattori T (2002) Abscisic acid induced transcription is mediated by phosphorylation of an abscisic acid response element binding factor, TRAB1. Plant Cell 14:3177–3189
Kasuga M, Liu Q, Miura S, Yamaguchi-Shinozaki K, Shinozaki K (1999) Improving plant drought, salt and freezing tolerance by gene transfer of a single stress-inducible transcription factor. Nat Biotechnol 17:287–291
Kobayashi Y, Murata M, Minami H et al (2005) Abscisic acid-activated SNRK2 protein kinases function in the gene-regulation pathway of ABA signal transduction by phosphorylating ABA response element-binding factors. Plant J 44:939–949
Lee YH, Chun JY (1998) A new homeodomain-leucine zipper gene from Arabidopsis thaliana induced by water stress and abscisic acid treatment. Plant Mol Biol 37:377–384
Luo H, Song F, Zheng Z (2005) Overexpression in transgenic tobacco reveals different roles for the rice homeodomain gene OsBIHD1 in biotic and abiotic stress responses. J Exp Bot 56:2673–2682
Mahajan S, Tuteja N (2005) Cold, salinity and drought stresses: An overview. Arch Biochem Biophys 444:139–158
Manavella PA, Arce AL, Dezar CA (2006) Cross-talk between ethylene and drought signaling pathways is mediated by the sunflower Hahb-4 transcription factor. Plant J 48:125–137
McConnell JR, Emery J, Eshed Y, Bao N, Bowman J, Barton MK (2001) Role of PHABULOSA and PHAVOLUTA in determining radial patterning in shoots. Nature 411:709–713
Mukherjee K, Brocchieri L, Burglin TR (2009) A comprehensive classification and evolutionary analysis of plant homeobox genes. Mol Biol Evol 26:2775–2794
Nakamura M, Katsumata H, Abe M et al (2006) Characterization of the class IV homeodomain-leucine zipper gene family in Arabidopsis. Plant Physiol 141:1363–1375
Nakashima K, Tran LS, Van Nguyen D et al (2007) Functional analysis of a NAC-type transcription factor OsNAC6 involved in abiotic and biotic stress-responsive gene expression in rice. Plant J 51:617–630
Nakashima K, Ito Y, Yamaguchi-Shinozaki K (2009) Transcriptional regulatory networks in response to abiotic stresses in Arabidopsis and grasses. Plant Physiol 149:88–95
Ni Y, Wang X, Li D, Wu Y, Xu W, Li X (2008) Novel cotton homeobox gene and its expression profiling in root development and in response to stresses and phytohormones. Acta Biochim Biophy Sin 40:78–84
Olsson A, Engstrom P, Soderman E (2004) The homeobox genes ATHB12 and ATHB7 encode potential regulators of growth in response to water deficit in Arabidopsis. Plant Mol Biol 55:663–677
Pieterse CMJ, Leon-Reyes A, Van der Ent S, Van Wees SCM (2009) Networking by small-molecule hormones in plant immunity. Nat Chem Biol 5:308–316
Prigge MJ, Otsuga D, Alonso JM, Ecker JR, Drews GN, Clark SE (2005) Class III homeodomain-leucine zipper gene family members have overlapping, antagonistic and distinct roles in Arabidopsis development. Plant Cell 17:61–76
Qin F, Sakuma Y, Li J et al (2004) Cloning and functional analysis of a novel DREB1/CBF transcription factor involved in cold-responsive gene expression in Zea mays L. Plant Cell Physiol 45:1042–1052
Raghavendra AS, Gonugunta VK, Christmann A, Grill E (2010) ABA perception and signaling. Trends Plant Sci 15:395–401
Reiser L, Modrusan Z, Margossian L et al (1995) The BELL1 gene encodes a homeodomain protein involved in pattern formation in the Arabidopsis ovule primordium. Cell 83:735–742
Rhoades MW, Reinhart BJ, Lim LP, Burge CB, Bartel B, Bartel DP (2002) Prediction of plant microRNA targets. Cell 110:513–520
Sessa G, Carabelli M, Sassi M et al (2005) A dynamic balance between gene activation and repression regulates the shade avoidance response in Arabidopsis. Genes Dev 19:2811–2815
Shin D, Koo YD, Lee J et al (2004) Athb-12, a homeobox-leucine zipper domain protein from Arabidopsis thaliana, increases salt tolerance in yeast by regulating sodium exclusion. Biochem Biophys Res Commun 323:534–540
Shinozaki K, Yamaguchi-Shinozaki K, Seki M (2003) Regulatory network of gene expression in the drought and cold stress responses. Curr Opin Plant Biol 6:410–417
Simpson SD, Nakashima K, Narusaka Y, Seki M, Shinozaki K, Yamaguchi-Shinozaki K (2003) Two different novel cis-acting elements of erd1, a clpA homologous Arabidopsis gene, function in induction by dehydration stress and dark-induced senescence. Plant J 33:259–270
Smith HM, Boschke I, Hake S (2002) Selective interaction of plant homeodomain proteins mediates high DNA- binding affinity. Proc Natl Acad Sci USA 99:9579–9584
Soderman E, Mattsson J, Engstrom P (1996) The Arabidopsis homeobox gene ATHB-7 is induced by water deficit and by abscisic acid. Plant J 10:375–381
Soderman E, Hjellstrom M, Fahleson J, Engstrom P (1999) The HD-Zip gene ATHB6 in Arabidopsis is expressed in developing leaves, roots and carpels and up-regulated by water deficit conditions. Plant Mol Biol 40:1073–1083
Suzuki N, Sejima H, Tam R, Schlauch K, Mittler R (2011) Identification of the MBF1 heat-response regulon of Arabidopsis thaliana. Plant J 66:844–851
Talbert PB, Adler HT, Parks DW, Comai L (1995) The REVOLUTA gene is necessary for apical meristem development and for limiting cell divisions in the leaves and stems of Arabidopsis thaliana. Development 121:2723–2735
Tan QK, Irish VF (2006) The Arabidopsis zinc finger-homeodomain genes encode proteins with unique biochemical properties that are coordinately expressed during floral development. Plant Physiol 140:1095–1108
Tang G, Reinhart BJ, Bartel DP, Zamore PD (2003) A biochemical framework for RNA silencing in plants. Genes Dev 17:49–63
Tran LS, Nakashima K, Sakuma Y et al (2004) Isolation and functional analysis of Arabidopsis stress-inducible NAC transcription factors that bind to a drought-responsive cis-element in the early responsive to dehydration stress 1 promoter. Plant Cell 16:2481–2498
Tran LS, Nakashima K, Sakuma Y et al (2006) Co-expression of the stress-inducible zinc finger homeodomain ZFHD1 and NAC transcription factors enhances expression of the ERD1 gene in Arabidopsis. Plant J 49:46–63
Tron AE, Bertoncini CW, Chan RL, Gonzalez DH (2002) Redox regulation of plant homeodomain transcription factors. J Biol Chem 277:34800–34807
Uno Y, Furihata T, Abe H, Yoshida R, Shinozaki K, Yamaguchi-Shinozaki K (2000) Arabidopsis basic leucine zipper transcription factors involved in an abscisic acid-dependent signal transduction pathway under drought and high-salinity conditions. Proc Natl Acad Sci USA 97:11632–1163725
Urano K, Kurihara Y, Seki M, Shinozaki K (2010) ‘Omics’ analyses of regulatory networks in plant abiotic stress responses. Curr Opin Plant Biol 13:1–7
van der Graaff E, Laux T, Rensing SA (2009) The WUS homeobox-containing (WOX) protein family. Genome Biol 10:248.1–248.9
Vij S, Tyagi AK (2007) Emerging trends in the functional genomics of the abiotic stress response in crop plants. Plant Biotechnol J 5:361–380
Wang Y, Henriksson E, Soderman E, Henriksson KN, Sundberg E, Engstrom P (2003) The Arabidopsis homeobox gene, ATHB16, regulates leaf development and the sensitivity to photoperiod in Arabidopsis. Dev Biol 264:228–239
Yamaguchi-Shinozaki K, Shinozaki K (2006) Transcriptional regulatory networks in cellular responses and tolerance to dehydration and cold stresses. Annu Rev Plant Biol 57:781–803
Yu SW, Zhang LD, Zuo KJ, Tang DQ, Sun XF, Tang KX (2005) Brassica napus L. homeodomain leucine zipper gene BnHB6 responds to abiotic and biotic stresses. J Integr Plant Biol 47:1236–1248
Yu H, Chen X, Hong YY et al (2008) Activated expression of an Arabidopsis HD-START protein confers drought tolerance with improved root system and reduced stomatal density. Plant Cell 20:1134–1151
Zhang Y, Wu R, Qin G, Chen Z, Gu H, Qu LJ (2011) Over-expression of WOX1 leads to defects in meristem development and polyamine homeostasis in Arabidopsis. J Integr Plant Biol 53:493–506
Zhu JK (2002) Salt and drought stress signal transduction in plants. Annu Rev Plant Biol 53:247–273
Zhu J, Shi H, Lee BH et al (2004) An Arabidopsis homeodomain transcription factor gene, HOS9, mediates cold tolerance through a CBF-independent pathway. Proc Natl Acad Sci U S A 101:9873–9878
Acknowledgements
The work is supported financially by the Department of Science and Technology (grant file number SR/S0/PS/07/2011), Government of India, New Delhi and core grant from NIPGR. AB acknowledges the award of research fellowship from the Council of Scientific and Industrial Research, New Delhi.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2013 Springer Science+Business Media New York
About this chapter
Cite this chapter
Bhattacharjee, A., Jain, M. (2013). Homeobox Genes as Potential Candidates for Crop Improvement Under Abiotic Stress. In: Tuteja, N., Singh Gill, S. (eds) Plant Acclimation to Environmental Stress. Springer, New York, NY. https://doi.org/10.1007/978-1-4614-5001-6_7
Download citation
DOI: https://doi.org/10.1007/978-1-4614-5001-6_7
Published:
Publisher Name: Springer, New York, NY
Print ISBN: 978-1-4614-5000-9
Online ISBN: 978-1-4614-5001-6
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)