Abstract
The clinical significance of the presence of Clostridium difficile in children’s faeces remains uncertain using current diagnostic procedures. Clostridium difficile is a relatively common finding in infants with no symptoms of gastrointestinal disease, suggesting it may be an incidental finding and form part of the normal gut micro-flora in this age group. On the other hand, particularly in older children or those with significant co-morbidity, there are examples where C. difficile causes disease and exerts considerable morbidity and even mortality (C. difficile infection, CDI). Between these extremes lie a substantial group of children who have both diarrhoea and C. difficile in their stools but where the nature of the association is not clear: Clostridium difficile associated disease (CDAD). We review the significance of C. difficile in children presenting recently uncovered paediatric data from a large UK epidemiological study that informs some key unanswered questions.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Clostridium Difficile
- Asymptomatic Child
- Toxigenic Culture
- Nosocomial Diarrhoea
- National Health Service Hospital Trust
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
4.1 Introduction
The clinical significance of Clostridium difficile colonisation in children is crucial in determining testing and treatment policies . C. is the leading cause of nosocomial diarrhoea in adults and one of the Healthcare Acquired Infections (HAI) specifically targeted by the National Health Service (NHS) in England [1]. Mandatory monitoring in the UK currently requires reporting of all diarrhoeal samples positive for C. difficile in those over the age of 2 years [2, 3]. C. difficile infection (CDI) in adults has been extensively studied and there are clear guidelines for its diagnosis and management [3–8]. The situation is much less clear in children because [9, 10] of the following issues:
-
a.
Clostridium difficile carriage rates are high in young children, especially infants: Asymptomatic carriage of C. difficile in children is much more frequent than in adults. Carriage can be found in up to 64 % of neonates (under 4 weeks old), who appear to acquire C. difficile environmentally in the first few weeks of life [10]. Asymptomatic carriage is so high in infants under 1 year of age that testing for C. difficile is of questionable benefit [11]. Carriage declines with age, [12, 13] whilst the proportion with symptomatic disease increases to approach levels in adults (5–8 %, [10]).
-
b.
Clostridium difficile may be an incidental finding: Acute diarrhoeal illness is common in young children, and due to a wide range of potential pathogens [12, 14–17], although in a substantial proportion no pathogen can be identified even after extensive testing for a range of bacteria, viruses and parasites . Given that so many children suffer diarrhoea without an identifiable cause, it is possible that even in those cases where C. difficile is identified, it is in fact not the causative agent but an incidental finding. It is even possible that colonisation with C. difficile may be protective [18, 19] against CDI .
-
c.
The relationship between prior antibiotic usage and C. difficile is considerably weaker in younger children: Not only do many children with C. difficile in their stool sample not have a recent history of antibiotic exposure [10, 20, 21], but a significant proportion of them have concurrent infection with other pathogenic organisms [14, 20, 21]. Thus, two of the original diagnostic criteria for CDI in adults [6] would exclude a large proportion of symptomatic children with C. difficile positive stools. Although there are recent guidelines for diagnosing CDI in adults [7, 8], no equivalent guidelines exist for children, where the evidence base is much more limited.
Despite this, there is a consensus that C. difficile can cause serious disease in children [10, 22]. It is helpful to maintain a distinction between examples of CDI, where C. difficile is the accepted cause of children’s symptoms, and “C. difficile associated disease” (CDAD) where the exact nature of the association between C. difficile and gastrointestinal symptoms remains unclear. Although variably defined, for example “gastrointestinal symptoms in a child with C. difficile toxin positive stools” [23], or “children with a clinical diagnosis of C. difficile infection on discharge who had C. difficile testing and antibiotic therapy for CDAD” [24], the incidence of CDAD and/or CDI in children appears to be on the increase, particularly in cases arising from the community [25]. Recent reviews have highlighted the importance of considering CDAD/CDI in children and outside the hospital setting [26–28]. Difficult questions are raised for clinicians faced with children suffering from diarrhoea [9, 10]:
-
1.
When should C. difficile be considered as a cause for the child’s symptoms and what is the appropriate testing strategy?
-
2.
Should the presence of C. difficile in diarrhoea in children be treated, and if so how?
With current technologies, testing for C. difficile in children is controversial [11]. This is especially true for children under the age of 1, who are the most likely to suffer from diarrhoea, have the highest prevalence of asymptomatic carriage of C. difficile, and also form a group for whom the incidence of CDAD is increasing [23]. Devising the most appropriate policy has significant implications for all acute NHS Hospital Trusts in England, who face financial penalties if their rates of C. difficile infection fail to meet national targets under a system that has been criticised as unfair due to arbitrary thresholds [29]. This is a potential disincentive to test in populations expected to have higher prevalence of colonisation with C. difficile, or to improve the accuracy of testing strategies in ways that increase reportable rates of CDI [30] . Such conflicts can only be resolved by a better understanding of the significance of C. difficile colonisation and infection in children that can inform new guidelines.
4.2 The Complexity of Testing for C. difficile and Diagnosing C. difficile Infection
Conventional detection of C. difficile relies on selective culture of the organism from faecal samples or detection of toxin by cell culture cytotoxicity assay (CCA) or enzyme immunoassay (EIA) [4]. Clostridium difficile’s pathogenicity is associated primarily with expression of toxin B (cytotoxin), with the role of toxin A (enterotoxin) being less certain [31, 32]. Not all strains are capable of producing toxin A [33, 34] so tests directed against this alone may miss CDI. Molecular assays based on amplification of C. difficile toxin gene sequences (PCRCDT) provide rapid and sensitive results but may not be as specific for CDI as phenotypic testing methods (e.g. CCA) which detect toxin production [35]. Clostridium difficile exhibits considerable genomic variation [36], allowing development of genetic typing schemes using techniques such as PCR ribotyping [37]. Genetic variation within the toxin genes may affect the performance of both phenotypic and genetic detection methods [38, 39]. Detection of C. difficile will therefore depend on the strain present and the test used (Table 4.1) [4, 7, 40]. A two-stage testing strategy has recently been implemented in the UK, [3] based on an observational diagnostic study in four UK laboratories [41] that confirmed that C. difficile toxin EIAs are not suitable as stand alone tests for the diagnosis of CDI or detection of C. difficile. The new UK recommendation contains a two test screening protocol comprising a GDH EIA (or toxin gene PCR) followed by a sensitive toxin EIA. If the first test (GDH or toxin gene test) is negative, the second test (sensitive toxin EIA) does not need to be performed.
Unfortunately, nearly all of the studies comparing the performance of different testing strategies have been conducted in adults and the clinical significance of the results will be subject to different interpretations in paediatric and neonatal populations. One study that investigated both children and young adults is a recent prospective study from Texas that compared direct stool EIA toxin A/B and direct stool PCRCDT with toxigenic culture (TGC, testing for toxin with either EIA or PCRCDT after the organism has been cultured selectively from faeces) [42]. Stools from 96 patients (age 15 days–25 years, median 4 years) suspected of having CDAD were tested using EIA toxin A/B and PCRCDT, both before (direct) and after (TGC) isolation and selective culture. Although lacking an independent gold standard, the “reference standard” for calculating sensitivities and specificities was any stool specimen where at least four of the six tests used (stool EIA, postculture EIA, stool PCRCDT (TcdA), stool PCRCDT (TcdB), postculture PCRCDT (TcdA), postculture PCRCDT (TcdB)) were positive. Direct stool PCRCDT had the greatest sensitivity of all methods used (95 %, compared to only 35 % for EIA; specificity of both PCRCDT and EIA: 100 %). Interestingly, positivity rates for C. difficile have doubled (from ~ 8 to 16 %) since the introduction of PCRCDT as the standard testing strategy at this US institution (while the number of samples sent for testing has stayed the same).
4.3 Epidemiology of C. difficile in Children
Few published studies are well placed to establish when C. difficile should be considered the cause of a child’s diarrhoea, or when it should be treated. We therefore reanalysed paediatric data from the English community-based Infectious Intestinal Disease Study (IIDS) [12, 43] which have not previously been reported within the peer reviewed literature. The IIDS data is informative as the study attempted to: (a) test each sample for a range of viral, bacterial and parasitic pathogens to identify co-infection and the possibility that C. difficile is an “innocent bystander”; (b) use an appropriately sensitive testing strategy for C. difficile (including selective culture); (c) test both symptomatic and asymptomatic children to assess the association between C. difficile and symptoms; and (d) present data within clinically relevant age ranges. A second study (IID2) is currently in progress [44].
4.3.1 Clostridium difficile in Children in the Community
Several community-based studies have demonstrated that detection of C. difficile and/or C. difficile toxin is as common in asymptomatic children as in those suffering with diarrhoea (Table 4.2). Conducted between 1993 and 1996, the IIDS collected data and stools from over 6,000 cases of infectious intestinal diseases in children and adults, as well as from controls [12, 43]. The IIDS included two components: a community cohort recruited randomly from 70 General Practitioner (GP) practices across England and a GP-based case-control study which included subjects presenting spontaneously with symptoms of diarrhoea to one of 34 practices. All faecal samples were subject to extensive testing for a range of potential pathogens including C. difficile. Asymptomatic carriage of C. difficile was highest in infants under 1 year of age: 21 % in those recruited as part of the community cohort and 16.6 % in the age and sex-matched controls for those presenting to their GP with diarrhoea. Carriage rates in infants with diarrhoea were not much greater (28.6 % in the community cohort) or even less (7.2 % in infants presenting to their GP). 1 % of children aged 1–4 years of age had C. difficile in their faeces, and C. difficile was rarely found in children 5 years or older. There was no significant association of C. difficile with diarrhoeal symptoms for children under the age of 5 in either the GP study (derived odds ratio for diarrhoea if C. difficile positive 0.59; 95 % confidence interval (CI) 0.34–1.02) or the community cohort (odds ratio 1.21; 95 % CI 0.37–3.9).
Two other studies comparing C. difficile in symptomatic children with asymptomatic controls showed similar results. Boenning et al. [45] showed that the prevalence of C. difficile amongst paediatric outpatients was actually higher in those with non-diarrhoeal illnesses (14.8 %) than those with diarrhoea (7.0 %; Odds ratio 0.43, 95 % CI 0.20–0.92), and found no association with antibiotic exposure in the preceding month. More recently, Vernacchio et al. [17] carried out a prospective cohort study of healthy children in the community in which baseline normal stools were cultured and compared to those from the same children who subsequently developed diarrhoea during the study period. Of healthy baseline stools, 3.5 % contained C. difficile compared to just 1.9 % in diarrhoeal specimens (matched relative risk 0.54, 95 % CI 0.20–1.50). Contrasting results were obtained from an earlier study of diarrhoeal outbreaks in community day centres [46], where rates of C. difficile isolation were greater amongst children with diarrhoea (57 %) than in their healthy classmates (9 %; OR 13.3, 95 % CI 3.5–51).
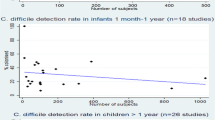
Other community-based studies have either tested only asymptomatic children [13] or children with diarrhoea [14–16] , and therefore cannot provide comparative data regarding the association of C. difficile with symptoms. They nevertheless demonstrate that C. difficile is a relatively common finding (5–9 %, Table 4.2) in children presenting from the community.
Although the prevalence in different age ranges have varied between studies, a high prevalence of C. difficile has largely been found in young children and infants, with an average age in these studies typically 18 months or less. For example, in the recent US study [24] the mean age was 15.2 (± 7.5) months, with most children under the age of 3 years. In studies that report prevalence for infants separately this is universally higher than for older children.
A small study in Japan was notable for the particularly high prevalence of C. difficile in asymptomatic children at two day nurseries and a kindergarten [13]. All the infants tested had C. difficile in their faeces but carriage rates declined steadily with increasing age to less than 25 % by 5 years of age. Nearly all (21/22) of the C. difficile strains (by PCR ribotype and PFGE type) isolated from environmental swabs at one nursery were identical to those found in the faeces of infants in that nursery, suggesting acquisition from the day-care environment.
Community onset diarrhoea is relatively common in young children and infants, with a range of potential pathogens other than C. difficile. Table 4.3 summarises data from five studies which carried out extensive testing for pathogens in children with community-acquired diarrhoea . A number of trends are revealed across the different settings, methodologies and even countries where the studies were based. Despite the investigators’ best efforts to test for a large range of pathogens, none could be identified in 30–75 % of the cases of diarrhoea. Viruses were the commonest identified pathogen in all studies, especially in infants and pre-school age children. Rotavirus was most frequently encountered ( ~ 14–20 %, except in an Austrian study, where there is routine vaccination against rotavirus [16]), followed by norovirus (where tested, ~ 9–11 %), adenovirus ( ~ 4–10 %) and astrovirus ( ~ 2–7 %). Clostridium difficile was the commonest identified bacterium in most studies. Other common isolates were C. perfringens, pathogenic E. coli, Aeromonas spp., Salmonella spp. and Campylobacter spp. (notably the latter two were more commonly isolated in older children) .
A proportion of C. difficile positive children were co-infected with other known pathogens. In Klein et al.’s [14] study, 25 (6.7 %) of the 372 specimens that underwent extensive testing were positive for C. difficile toxin B, but nine of these had concomitant viral or bacterial pathogens. Only 4.3 % of children with diarrhoea had C. difficile as the only identifiable pathogen [14] and some common causes (e.g. norovirus) were not tested for, so it is possible that there may have been underestimation of co-infection with C. difficile. Denno et al. tested for the same range of organisms, and found one case of co-infection out of 75 children undergoing ‘complete’ analysis (one of the eight stools positive for C. difficile was also positive for adenovirus) [15]. Co-infection is also common in hospitalised children, with other pathogens reported in 36 % [21], 44 % [20] 27 % [47] and 23–38 % of C. difficile culture positive stools [48]. In three recently reported cases of severe CDAD in children, it was noted that two had co-infection with viruses (rotavirus and calicivirus), although these two also had underlying chronic medical conditions (Hirschsprung’s disease and Down’s syndrome) [49].
4.3.1.1 Explanations for High Levels of Asymptomatic Colonisation
The high rates of carriage of C. difficile may be explained by a lack of ‘colonisation resistance’ in infants as the intestinal micro-flora is in the process of becoming established [50], but this cannot explain the absence of symptomatic disease (CDI). Asymptomatic carriage does not simply occur because the strains of C. difficile present are non-toxigenic, for most of the studies discussed previously demonstrated the presence of toxin in faeces from asymptomatic children. One hypothesis is that the strains typically found in children have reduced virulence compared to those in adults. Factors other than toxin expression, such as those influencing adherence and intestinal colonisation, are also recognised contributors to virulence and may play a part in childhood disease [51] .
The hypervirulent strain of C. difficile associated with outbreaks of severe CDI in adults (PCR ribotype 027) has greatly increased in vitro levels of expression of tcdA (16-fold) and tcdB (23-fold) [51]. This strain was responsible for a high proportion (19.4 %) of C. difficile toxin positive stools in symptomatic children in a recent study in the USA [52]. A study of hospitalised but asymptomatic children in Thailand demonstrated relatively high carriage rates for C. difficile (11.0 % of infants, and 21.1 % of 1–11 year olds) but low rates of toxin A gene detection (0.9 % of infants and none of the older children; toxin B not tested) [53]. 87.2 % of the strains isolated in asymptomatic children in the Japanese day-care study were also non-toxigenic (tcdA - /tcdB - ) [13]. Unfortunately neither study examined rates of carriage or toxin expression in symptomatic children for comparison. A small study in Brazil attempted to compare strains of C. difficile in hospitalised children with acute diarrhoea with asymptomatic children recruited from day-care centres, but none of the controls were culture positive for C. difficile. Nine out of the ten strains isolated from symptomatic patients were toxigenic: six were tcdA + /tcdB + and three were tcdA - /tcdB + [54].
Only the IID study provides direct data comparing C. difficile colonisation and toxin B detection for cases of diarrhoea and asymptomatic controls [12] . Below 2 years of age, children presenting to their GP with diarrhoea had slightly higher rates of C. difficile colonisation (24 % of 374 cf. 19.5 % of 385 cultured) but lower rates of toxin B detection than asymptomatic age- and sex-matched controls (4.3 % of 391 cf. 9.0 % of 423 tested). For children 2 years or older, controls had lower rates of both toxin B detection (0.2 % of 1,616 cf. 1.1 % of 1,866 tested) and C. difficile colonisation (0.4 % of 1,613 cf. 1.0 % of 1,845 cultured). These data suggest that above the age of 2 years, rates of colonisation drop dramatically, and toxin production is more likely to be associated with disease .
4.3.2 Clostridium difficile in Hospitalised Children
The prevalence of C. difficile in hospitalised children is higher than in the community but appears similar in children with and without diarrhoea (Table 4.4). Karsch et al. [55] found high rates of carriage, particularly in infants (30 %), with most isolates producing toxin (82 % toxin A and 43 % toxin B), but no significant difference between symptomatic children and controls, and no clear association with previous antibiotic therapy. A prospective study in Denmark also showed significantly higher isolation of C. difficile in infants and no relationship to antibiotic exposure (p < 0.001), but it was the only identified pathogen in 12 % of children of all ages with acute gastroenteritis , and this was significantly (p < 0.01) higher than for asymptomatic controls [21]. As 78 % of the positive cultures were obtained within 2 days of admission this suggests acquisition in the community rather than nosocomial infection. A retrospective cohort study in the US reported that 26 % of cases of CDAD occured in infants [24], but it remains unclear whether C. difficile was truly the underlying cause. Of the 56 % of specimens from paediatric inpatients in Canada with nosocomial diarrhoea where a pathogen was identified, most were viruses (38 % of episodes, viral diagnosis typically in younger children with mean age 0.8 years) [56]. Clostridium difficile was identified in 18 % of all episodes (mainly in older children, mean age 3.9 years). A retrospective case control study in Canada was unable to demonstrate any difference in clinical characteristics between infants with C. difficile toxin in their stool and those without toxin present, nor could it identify a significant treatment effect of metronidazole [11]. Colonisation with C. difficile was relatively high at two US institutions (49 % and 26 %) , but toxigenic colonisation was less common (14 % and 15 %), and many isolates were unique (92 % within the general hospital), indicating that they did not arise from a common source [48]. Underlying medical conditions and exposure to two or more antibiotics were associated with increased toxigenic strain colonisation . In Turkey, although C. difficile toxin was found more frequently in hospitalised children with nosocomial diarrhoea (22 %) than asymptomatic controls (10 %), this was not significant (odds ratio 2.54, 95 % CI 0.90–7.17) and co-infection was found in six cases (27 %) of CDAD (rotavirus was the only viral pathogen tested) [47]. Children with CDAD were older (mean 5.4 years) than those asymptomatic controls with C. difficile toxin in their stools (all under 2 years).
4.3.3 Clostridium difficile in Neonates
Neonates acquire C. difficile from the environment, resulting in high rates of carriage within the first few weeks of birth [57–60]. In one study, no neonatal faeces cultured C. difficile on day 1 after birth but 17 % did by day 4, and most strains were toxigenic (58–65 %) [60]. None of the maternal rectal swabs and only one vaginal swab cultured C. difficile, whereas 13 % of environmental samples did and these were all of the same strain (matching 11 of the 31 neonatal strains typed). On one neonatal intensive care unit (NICU), C. difficile acquisition reached 33 % after 2 weeks with all cultures toxigenic [61]. In common with previous studies [58, 59], there was no association between C. difficile acquisition and gastrointestinal symptoms. In another NICU, 90 % of samples cultured C. difficile after only 6 days, and although toxin was detected directly in only 36 % of these, 94 % of the isolates were found to be toxigenic in vitro [57]. One small study suggested that neonates with toxin A positive stools are likely to experience increased numbers of days with frequent and abnormal stools [62], but most demonstrate asymptomatic carriage of C. difficile in neonates, even when a high proportion of stools test positive for toxin.
4.4 Burden of CDAD in Children
The high asymptomatic prevalence of C. difficile in young children initially led to the conclusion that CDI was not a problem in this age group [45, 63]. However, several studies have suggested that CDAD (and by implication CDI) is an increasing problem in children. A retrospective cohort study in the USA showed that from 2001 to 2006 the incidence rate of CDAD (defined as “clinical symptoms, such as diarrhoea or bloody stools, in a patient whose stool specimen tested positive for C. difficile toxin”) amongst outpatients increased by 11 % (from 1.24 to 1.38 cases per 1,000 visits) [23]. The incidence of CDAD in patients attending the emergency department increased 2.5 fold, largely due to an increase in community-associated CDAD (from 0.84 to 2.04 cases per 1,000 visits), while inpatient incidence of CDAD decreased over the same period [23]. In another study the annual incidence of CDAD (identified by the combination of discharge diagnosis of CDI, positive test assay for CDI and treated with antibiotics against CDI) increased from 2.6 to 4.0 cases/1,000 admissions over the same time period [24]. Hospitalisation rates for children with CDAD almost doubled between 1997 and 2006 [64]. Complications of CDAD can be severe, including pseudomembranous colitis [22, 49, 65, 66], rectal prolapse [67], osteomyelitis [68] and reactive arthritis [69, 70].
4.5 When to Consider CDAD and How to Test for it
The new UK Department of Health testing algorithm [41] contains a two test screening protocol comprising a GDH EIA (or toxin gene PCR) followed by a sensitive toxin EIA. If the first test (GDH or toxin gene test) is negative, the second test (sensitive toxin EIA) does not need to be performed.
4.5.1 Children Without Co-morbidities
Because the prevalence of C. difficile in children is so dependent on age it might be more appropriate to tailor the testing and management strategy accordingly (Table 4.5). Given the very limited data on the performance of different testing strategies in children (Table 4.1), adult guidelines may eventually be shown to perform poorly in children, where PCRCDT might be considered a more appropriate formal second line test after GDH EIA due to its rapid turnaround time and good correlation with toxigenic culture (Table 4.1).
Infants have high asymptomatic prevalence, so testing stools for C. difficile is not recommended unless illness is severe and there is a high level of clinical suspicion [71]. Serious cases of CDI (pseudomembranous colitis ) have been reported in infants, but have generally been associated with other conditions such as prematurity, Hirschprung’s disease, obstruction or necrotising enterocolitis [65, 72, 73], and the role of C. difficile in the pathogenesis has not been confirmed . These reports suggest symptomatic infants should only be tested for C. difficile in carefully selected circumstances. Other organisms may also be responsible for disease and viral testing in particular should be undertaken alongside testing for C. difficile.
It would be helpful to identify reliable predictors for development of CDI in children. Despite the conflicting evidence for an association between antibiotic usage and CDI in children (against association: [11, 20, 21, 45, 55, 74, 75]; for association: [14, 47, 48, 63, 76–79]), severe diarrhoea in the context of recent antibiotic therapy is likely to remain one such predictor that clinicians will use.
4.5.2 Children with Co-morbidities
Certain co-morbitities are associated with higher rates of C. difficile colonisation , and some of these children may be at particular risk of CDI: inflammatory bowel disease (IBD), Hirschprung’s disease, cystic fibrosis, cancer patients and organ transplant recipients.
The prevalence of C. difficile is significantly greater in children with IBD compared to controls (indicating an increased risk of colonisation) and also greater in children with IBD experiencing active disease compared to those with inactive disease (indicating a potential role of the organism in the symptom exacerbation) [80]. It is important to use an appropriate testing strategy to prevent missing CDI in the context of inflammatory bowel disease, as the diagnosis may be missed in up to 41 % of IBD patients if a single-toxin assay is used [81]. Such errors could lead to misattribution of symptoms to an exacerbation of underlying IBD and result in inappropriate treatment, potentially even colectomy. Steroids given to treat IBD without appropriate antibiotics to treat CDI are likely to exacerbate symptoms rather than resolve them [82].
Case reports of severe CDI associated with Hirschsprung’s disease suggest that this may form another group of children at increased risk [66, 83]. Increased C. difficile was found in Hirschsprung’s patients with enterocolitis compared with asymptomatic Hirschsprung’s patients or healthy controls [84].
Carriage rates of C. difficile in patients with cystic fibrosis have been reported to be 22–46 %, double that of control patients receiving antibiotics [85–87] . These high carriage rates were described in older patients (median age 18.5, youngest 15 years) in the absence of symptoms of diarrhoea or abdominal pain, despite the presence of toxigenic strains [87]. Nevertheless, severe cases of CDI do occur in children with cystic fibrosis, emphasizing the importance of considering the diagnosis [88]. Patients may present atypically, without watery diarrhoea but rather abdominal distension and reduced bowel motions, risking confusion with faecal impaction or meconium ileus equivalent [89, 90]. There also appears to be a greater risk of CDI following lung transplantation for cystic fibrosis, with patients experiencing a fulminant course resulting in high mortality [91, 92].
Cancer patients are thought to have increased carriage of C. difficile due to their chemotherapy treatment and increased exposure to antimicrobials [74, 77] . Most reported cases have been associated with haematological malignancies, but a recent Italian study demonstrated that 6 % of children with solid tumours had C. difficile toxin A in their stools and gastrointestinal symptoms, with three out of nine of these being under 1 year old [93]. An earlier prospective study of oncology patients demonstrated higher rates of toxin detection in asymptomatic (19 %) than in symptomatic (8.7 %) children, questioning the significance of C. difficile as a pathogen in this patient group [74].
4.6 Treatment Strategies
If C. difficile is the only pathogen identified and seems the most likely cause of disease in a child over the age of 1 then treatment should be considered [10]. In adults, where exposure to broad spectrum antibiotics is a recognised trigger for CDI, cessation of the antibiotic, if clinically possible, is an important step . As previously discussed, the association between antibiotic use and CDAD in children is much weaker, but it is sensible to discontinue broad spectrum antibiotics where possible . Oral metronidazole and, if this fails, vancomycin are the standard treatments for adults with severe CDI requiring treatment and are also used in children. Up to 25 % of patients experience a further episode of CDI within 2 months, and 50–65 % of these will suffer from further recurrences [94].
More recently, interest has moved to alternative treatment strategies for CDI, in particular to prevent recurrence of disease [10, 95]. Alternative strategies may be divided into those that attempt to re-establish colonisation resistance (use of probiotics, faecotherapy), direct chemical neutralisation of the toxins (toxin binding by ion exchange resins and polymers) and immunotherapy (passive immunoglobulin therapy) [94, 95]. Unfortunately, few randomised controlled trials have been conducted in adult populations , and none have been done in children. A recent Cochrane review [96] found little evidence that probiotics were useful in adults, identifying only one study that showed a beneficial effect when added to antibiotic therapy [97]. Extrapolation of such data to children is problematic given that in a child’s early years the natural intestinal flora is different from adults and evolves over time. None of the alternative strategies explored so far has demonstrated sufficient success to recommend incorporation into general clinical practice [95].
4.7 Conclusions
Clostridium difficile is a relatively common finding in the faeces of infants under 1 year of age, and is very unlikely to signify disease, even when toxins are produced. Children with diarrhoea may have C. difficile in their stools, but viruses are a more likely cause of symptoms. It is not possible to identify the aetiological agent in a large proportion of childhood diarrhoea, but the disease is typically self-limiting and requires only supportive care [71].
CDAD appears to be increasing in children and should be considered as adult-type CDI whenever symptoms are particularly severe, protracted or the child belongs to a known at risk group such as inflammatory bowel disease or cystic fibrosis following lung transplantation. The use of predictors for CDAD such as recent antibiotic usage or prior hospitalisation is generally unhelpful in children, where these associations are much weaker and colonisation appears to occur in the community .
Few diagnostic tests in routine clinical use for C. difficile infection have been evaluated in children, where the true sensitivities and specificities are likely to be different from adult populations. The evidence on which to base decisions of when and how to treat CDAD in children remains limited and many important, interrelated research questions are yet unanswered (Table 4.6).
4.8 Search Strategy and Selection Criteria
This review was prompted by re-appraisal of data contained in a large, UK government-funded investigation report (The Infectious Intestinal Diseases (IID) Study in England [12]) in the light of a growing appreciation that C. difficile in children differs substantially from adults [9, 10, 98]. The IID study produced substantial data on the incidence of C. difficile in children in the community not contained in the original journal article [43], which combined data for children and adults. Other data and references were obtained by searching PubMed using “infant”, “child(ren)”, “p(a)ediatric” and “C. difficile” as search terms. Retrieved titles and abstracts were screened and full text versions obtained of suitable articles in English. Where necessary, odds ratios and confidence intervals were derived from original data presented in the manuscript by a standard method [99].
References
(2008) National target to reduce Clostridium difficile infections: SHA envelopes. Department of Health
(2008) Changes to the mandatory healthcare associated infection surveillance system for Clostridium difficile infection (CDI) from 1st January 2008. Department of Health
Infection ACoARaHA. UK Department of Health Updated Guidance on the Diagnosis and Reporting of Clostridium difficile 2012. http://www.dh.gov.uk/prod_consum_dh/groups/dh_digitalassets/@dh/@en/documents/digitalasset/dh_133016_OnlinePDF.pdf
Brazier JS (1998) The diagnosis of Clostridium difficile-associated disease. J Antimicrob Chemother 41(Suppl C):29–40
Delmee M (2001) Laboratory diagnosis of Clostridium difficile disease. Clin Microbiol Infect 7(8):411–416
Fekety R (1997) Guidelines for the diagnosis and management of Clostridium difficile-associated diarrhea and colitis. American College of Gastroenterology, Practice Parameters Committee. Am J Gastroenterol 92(5):739–750
Crobach MJ, Dekkers OM, Wilcox MH, Kuijper EJ (2009) European society of clinical microbiology and infectious diseases (ESCMID): data review and recommendations for diagnosing Clostridium difficile-infection (CDI). Clin Microbiol Infect 15(12):1053–1066
Cohen SH, Gerding DN, Johnson S, Kelly CP, Loo VG, McDonald LC, Pepin J, Wilcox MH (2010) Clinical practice guidelines for Clostridium difficile infection in adults: 2010 update by the society for healthcare epidemiology of America (SHEA) and the infectious diseases society of America (IDSA). Infect Control Hosp Epidemiol 31(5):431–455
Wilson ME (2006) Clostridium difficile and childhood diarrhea: cause, consequence, or confounder. Clin Infect Dis 43(7):814–816
McFarland LV, Brandmarker SA, Guandalini S (2000) Pediatric Clostridium difficile: a phantom menace or clinical reality? J Pediatr Gastroenterol Nutr 31(3):220–231
Tang P, Roscoe M, Richardson SE (2005) Limited clinical utility of Clostridium difficile toxin testing in infants in a pediatric hospital. Diagn Microbiol Infect Dis 52(2):91–94
(2000) A Report of the Study of Infectious Intestinal Disease in England. The Stationery Office, London
Matsuki S, Ozaki E, Shozu M, Inoue M, Shimizu S, Yamaguchi N, Karasawa T, Yamagishi T, Nakamura S (2005) Colonization by Clostridium difficile of neonates in a hospital, and infants and children in three day-care facilities of Kanazawa, Japan. Int Microbiol 8(1):43–48
Klein EJ, Boster DR, Stapp JR, Wells JG, Qin X, Clausen CR, Swerdlow DL, Braden CR, Tarr PI (2006) Diarrhea etiology in a Children’s Hospital Emergency Department: a prospective cohort study. Clin Infect Dis 43(7):807–813
Denno DM, Stapp JR, Boster DR, Qin X, Clausen CR, Del Beccaro KH, Swerdlow DL, Braden CR, Tarr PI (2005) Etiology of diarrhea in pediatric outpatient settings. Pediatr Infect Dis J 24(2):142–148
Huhulescu S, Kiss R, Brettlecker M, Cerny RJ, Hess C, Wewalka G, Allerberger F (2009) Etiology of acute gastroenteritis in three sentinel general practices, Austria 2007. Infection 37(2):103–108
Vernacchio L, Vezina RM, Mitchell AA, Lesko SM, Plaut AG, Acheson DW (2006) Diarrhea in American infants and young children in the community setting: incidence, clinical presentation and microbiology. Pediatr Infect Dis J 25(1):2–7
Kyne L, Warny M, Qamar A, Kelly CP (2000) Asymptomatic carriage of Clostridium difficile and serum levels of IgG antibody against toxin A. N Engl J Med 342(6):390–397
Jangi S, Lamont JT (2010) Asymptomatic colonization by Clostridium difficile in infants: implications for disease in later life. J Pediatr Gastroenterol Nutr 51(1):2–7
Niyogi SK, Dutta P, Dutta D, Mitra U, Sikdar S (1991) Clostridium difficile and its cytotoxin in hospitalized children with acute diarrhea. Indian Pediatr 28(10):1129–1132
Tvede M, Schiotz PO, Krasilnikoff PA (1990) Incidence of Clostridium difficile in hospitalized children. A prospective study. Acta Paediatr Scand 79(3):292–299
Brook I (2005) Pseudomembranous colitis in children. J Gastroenterol Hepatol 20(2):182–186
Benson L, Song X, Campos J, Singh N (2007) Changing epidemiology of Clostridium difficile- associated disease in children. Infect Control Hosp Epidemiol 28(11):1233–1235
Kim J, Smathers SA, Prasad P, Leckerman KH, Coffin S, Zaoutis T (2008) Epidemiological features of Clostridium difficile-associated disease among inpatients at children’s hospitals in the United States, 2001–2006. Pediatrics 122(6):1266–1270
Pituch H (2009) Clostridium difficile is no longer just a nosocomial infection or an infection of adults. Int J Antimicrob Agents 33(Suppl 1):S42–45
DuPont HL, Garey K, Caeiro JP, Jiang ZD (2008) New advances in Clostridium difficile infection: changing epidemiology, diagnosis, treatment and control. Curr Opin Infect Dis 21(5):500–507
Cohen MB (2009) Clostridium difficile infections: emerging epidemiology and new treatments. J Pediatr Gastroenterol Nutr 48(Suppl 2):S63–65
McFarland LV, Beneda HW, Clarridge JE, Raugi GJ (2007) Implications of the changing face of Clostridium difficile disease for health care practitioners. Am J Infect Control 35(4):237–253
Walker AS, Spiegelhalter D, Crook DW, Wyllie D, Morris J, Peto TE (2008) Fairness of financial penalties to improve control of Clostridium difficile. BMJ 337:a2097
Goldenberg SD, Price NM, Tucker D, Wade P, French GL (2011) Mandatory reporting and improvements in diagnosing Clostridium difficile infection: An incompatible dichotomy? J Infect 62(5):363–370
Lyras D, O’Connor JR, Howarth PM, Sambol SP, Carter GP, Phumoonna T, Poon R, Adams V, Vedantam G, Johnson S, Gerding DN, Rood JI (2009) Toxin B is essential for virulence of Clostridium difficile. Nature 458(7242):1176–1179
Kuehne SA, Cartman ST, Heap JT, Kelly ML, Cockayne A, Minton NP (2010) The role of toxin A and toxin B in Clostridium difficile infection. Nature 467(7316):711–713
Drudy D, Fanning S, Kyne L (2007) Toxin A-negative, toxin B-positive Clostridium difficile. Int J Infect Dis 11(1):5–10
Kyne L, Farrell RJ, Kelly CP (2001) Clostridium difficile. Gastroenterol Clin North Am 30(3):753–77
Wilcox MH, Planche T, Fang FC, Gilligan P (2010) What is the current role of algorithmic approaches for diagnosis of Clostridium difficile infection? J Clin Microbiol. 48(12):4347–4353
Sebaihia M, Wren BW, Mullany P, Fairweather NF, Minton N, Stabler R, Thomson NR, Roberts AP, Cerdeno-Tarraga AM, Wang H, Holden MT, Wright A, Churcher C, Quail MA, Baker S, Bason N, Brooks K, Chillingworth T, Cronin A, Davis P, Dowd L, Fraser A, Feltwell T, Hance Z, Holroyd S, Jagels K, Moule S, Mungall K, Price C, Rabbinowitsch E, Sharp S, Simmonds M, Stevens K, Unwin L, Whithead S, Dupuy B, Dougan G, Barrell B, Parkhill J (2006) The multidrug-resistant human pathogen Clostridium difficile has a highly mobile, mosaic genome. Nat Genet 38(7):779–786
Stubbs SL, Brazier JS, O’Neill GL, Duerden BI (1999) PCR targeted to the 16S-23S rRNA gene intergenic spacer region of Clostridium difficile and construction of a library consisting of 116 different PCR ribotypes. J Clin Microbiol 37(2):461–463
Rupnik M (2001) How to detect Clostridium difficile variant strains in a routine laboratory. Clin Microbiol Infect 7(8):417–420
Tenover FC, Novak-Weekley S, Woods CW, Peterson LR, Davis T, Schreckenberger P, Fang FC, Dascal A, Gerding DN, Nomura JH, Goering RV, Akerlund T, Weissfeld AS, Baron EJ, Wong E, Marlowe EM, Whitmore J, Persing DH (2010) Impact of strain type on detection of toxigenic Clostridium difficile: comparison of molecular diagnostic and enzyme immunoassay approaches. J Clin Microbiol 48(10):3719–3724
Carroll KC (2011) Tests for the diagnosis of Clostridium difficile infection: The next generation. Anaerobe 17(4):170–174
Wilcox MH, Planche T Defining a testing algorithm to improve the laboratory diagnosis of CDI. http://www.hpa.org.uk/webc/HPAwebFile/HPAweb_C/1317132979562
Luna RA, Boyanton BL, Jr, Mehta S, Courtney EM, Webb CR, Revell PA, Versalovic J (2011) Rapid Stool-Based Diagnosis of Clostridium difficile Infection by Real-Time PCR in a Children’s Hospital. J Clin Microbiol 49(3):851–857
Wheeler JG, Sethi D, Cowden JM, Wall PG, Rodrigues LC, Tompkins DS, Hudson MJ, Roderick PJ (1999) Study of infectious intestinal disease in England: rates in the community, presenting to general practice, and reported to national surveillance. The Infectious Intestinal Disease Study Executive. BMJ 318(7190):1046–1050
Sandora TJ, Fung M, Flaherty K, Helsing L, Scanlon P, Potter-Bynoe G, Gidengil CA, Lee GM (2011) Epidemiology and risk factors for Clostridium difficile infection in children. Pediatr Infect Dis J 30(7):580–584
Boenning DA, Fleisher GR, Campos JM, Hulkower CW, Quinlan RW (1982) Clostridium difficile in a pediatric outpatient population. Pediatr Infect Dis 1(5):336–338
Kim K, DuPont HL, Pickering LK (1983) Outbreaks of diarrhea associated with Clostridium difficile and its toxin in day-care centers: evidence of person-to-person spread. J Pediatr 102(3):376–382
Oguz F, Uysal G, Dasdemir S, Oskovi H, Vidinlisan S (2001) The role of Clostridium difficile in childhood nosocomial diarrhea. Scand J Infect Dis 33(10):731–733
Rexach CE, Tang-Feldman YJ, Cantrell MC, Cohen SH (2006) Epidemiologic surveillance of Clostridium difficile diarrhea in a freestanding pediatric hospital and a pediatric hospital at a university medical center. Diagn Microbiol Infect Dis 56(2):109–114
Pokorn M, Radsel A, Cizman M, Jereb M, Karner P, Kalan G, Grosek S, Andlovic A, Rupnik M (2008) Severe Clostridium difficile-associated disease in children. Pediatr Infect Dis J 27(10):944–946
Wilson KH (1993) The microecology of Clostridium difficile. Clin Infect Dis 16(Suppl 4):S214–218
Deneve C, Janoir C, Poilane I, Fantinato C, Collignon A (2009) New trends in Clostridium difficile virulence and pathogenesis. Int J Antimicrob Agents 33(Suppl 1):S24–28
Toltzis P, Kim J, Dul M, Zoltanski J, Smathers S, Zaoutis T (2009) Presence of the epidemic North American Pulsed Field type 1 Clostridium difficile strain in hospitalized children. J Pediatr 154(4):607–608
Wongwanich S, Pongpech P, Dhiraputra C, Huttayananont S, Sawanpanyalert P (2001) Characteristics of Clostridium difficile strains isolated from asymptomatic individuals and from diarrheal patients. Clin Microbiol Infect 7(8):438–441
Ferreira CE, Nakano V, Durigon EL, Avila-Campos MJ (2003) Prevalence of Clostridium spp. and Clostridium difficile in children with acute diarrhea in Sao Paulo city, Brazil. Mem Inst Oswaldo Cruz 98(4):451–454
Karsch W, Strelau E, Grahlow WD, Fischer E, Schulz R (1989) Occurrence and significance of Clostridium difficile in faecal specimens of hospitalized children. Zentralbl Bakteriol Mikrobiol Hyg A 270(3):441–448
Langley JM, LeBlanc JC, Hanakowski M, Goloubeva O (2002) The role of Clostridium difficile and viruses as causes of nosocomial diarrhea in children. Infect Control Hosp Epidemiol 23(11):660–664
Al-Jumaili IJ, Shibley M, Lishman AH, Record CO (1984) Incidence and origin of Clostridium difficile in neonates. J Clin Microbiol 19(1):77–78
Zedd AJ, Sell TL, Schaberg DR, Fekety FR, Cooperstock MS (1984) Nosocomial Clostridium difficile reservoir in a neonatal intensive care unit. Pediatr Infect Dis 3(5):429–432
Delmee M, Verellen G, Avesani V, Francois G (1988) Clostridium difficile in neonates: serogrouping and epidemiology. Eur J Pediatr 147(1):36–40
Martirosian G, Kuipers S, Verbrugh H, van Belkum A, Meisel-Mikolajczyk F (1995) PCR ribotyping and arbitrarily primed PCR for typing strains of Clostridium difficile from a Polish maternity hospital. J Clin Microbiol 33(8):2016-2021
el-Mohandes AE, Keiser JF, Refat M, Jackson BJ (1993) Prevalence and toxigenicity of Clostridium difficile isolates in fecal microflora of preterm infants in the intensive care nursery. Biol Neonate 63(4):225–229
Enad D, Meislich D, Brodsky NL, Hurt H (1997) Is Clostridium difficile a pathogen in the newborn intensive care unit? A prospective evaluation. J Perinatol 17(5):355–359
Vesikari T, Isolauri E, Maki M, Gronroos P (1984). Clostridium difficile in young children. Association with antibiotic usage. Acta Paediatr Scand 73(1):86–91
Zilberberg MD, Shorr AF, Kollef MH (2008) Increase in Clostridium difficile-related hospitalizations among infants in the United States, 2000-2005. Pediatr Infect Dis J 27(12):1111–1113
Zwiener RJ, Belknap WM, Quan R (1989) Severe pseudomembranous enterocolitis in a child: case report and literature review. Pediatr Infect Dis J 8(12):876–882
Qualman SJ, Petric M, Karmali MA, Smith CR, Hamilton SR (1990) Clostridium difficile invasion and toxin circulation in fatal pediatric pseudomembranous colitis. Am J Clin Pathol 94(4):410–416
Harris PR, Figueroa-Colon R (1995) Rectal prolapse in children associated with Clostridium difficile infection. Pediatr Infect Dis J 14(1):78–80
Gaglani MJ, Murray JC, Morad AB, Edwards MS (1996) Chronic osteomyelitis caused by Clostridium difficile in an adolescent with sickle cell disease. Pediatr Infect Dis J 15(11):1054–1056
Durand CL, Miller PF (2009) Severe Clostridium difficile colitis and reactive arthritis in a 10-year-old child. Pediatr Infect Dis J 28(8):750–751
Loffler HA, Pron B, Mouy R, Wulffraat NM, Prieur AM (2004) Clostridium difficile-associated reactive arthritis in two children. Joint Bone Spine 71(1):60–62
Clinical guideline 84 (CG84). (2009) Diarrhoea and vomiting in children under 5: full guideline. National Institute for Health and Clinical Excellence, London
Singer DB, Cashore WJ, Widness JA, Campognone P, Hillemeier C (1986) Pseudomembranous colitis in a preterm neonate. J Pediatr Gastroenterol Nutr 5(2):318–320
Adler SP, Chandrika T, Berman WF (1981) Clostridium difficile associated with pseudomembranous colitis. Occurrence in a 12-week-old infant without prior antibiotic therapy. Am J Dis Child 135(9):820–822
Burgner D, Siarakas S, Eagles G, McCarthy A, Bradbury R, Stevens M (1997) A prospective study of Clostridium difficile infection and colonization in pediatric oncology patients. Pediatr Infect Dis J 16(12):1131–1134
Tullus K, Aronsson B, Marcus S, Mollby R (1989) Intestinal colonization with Clostridium difficile in infants up to 18 months of age. Eur J Clin Microbiol Infect Dis 8(5):390–393
Brady MT, Pacini DL, Budde CT, Connell MJ (1989) Diagnostic studies of nosocomial diarrhea in children: assessing their use and value. Am J Infect Control 17(2):77–82
Schuller I, Saha V, Lin L, Kingston J, Eden T, Tabaqchali S (1995) Investigation and management of Clostridium difficile colonisation in a paediatric oncology unit. Arch Dis Child 72(3):219–222
Ferroni A, Merckx J, Ancelle T, Pron B, Abachin E, Barbut F, Larzul J, Rigault P, Berche P, Gaillard JL (1997) Nosocomial outbreak of Clostridium difficile diarrhea in a pediatric service. Eur J Clin Microbiol Infect Dis 16(12):928–933
Gogate A, De A, Nanivadekar R, Mathur M, Saraswathi K, Jog A, Kulkarni MV (2005) Diagnostic role of stool culture & toxin detection in antibiotic associated diarrhoea due to Clostridium difficile in children. Indian J Med Res 122(6):518–524
Pascarella F, Martinelli M, Miele E, Del Pezzo M, Roscetto E, Staiano A (2009) Impact of Clostridium difficile infection on pediatric inflammatory bowel disease. J Pediatr 154(6):854–858
Markowitz JE, Brown KA, Mamula P, Drott HR, Piccoli DA, Baldassano RN (2001) Failure of single-toxin assays to detect clostridium difficile infection in pediatric inflammatory bowel disease. Am J Gastroenterol 96(9):2688–2690
Issa M, Ananthakrishnan AN, Binion DG (2008) Clostridium difficile and inflammatory bowel disease. Inflamm Bowel Dis 14(10):1432–1442
Parsons SJ, Fenton E, Dargaville P (2005) Clostridium difficile associated severe enterocolitis: a feature of Hirschsprung’s disease in a neonate presenting late. J Paediatr Child Health 41(12):689–690
Thomas DF, Fernie DS, Bayston R, Spitz L, Nixon HH (1986) Enterocolitis in Hirschsprung’s disease: a controlled study of the etiologic role of Clostridium difficile. J Pediatr Surg 21(1):22–25
Welkon CJ, Long SS, Thompson CM Jr Gilligan PH (1985). Clostridium difficile in patients with cystic fibrosis. Am J Dis Child 139(8):805–808
Yahav J, Samra Z, Blau H, Dinari G, Chodick G, Shmuely H (2006) Helicobacter pylori and Clostridium difficile in cystic fibrosis patients. Dig Dis Sci 51(12):2274–2279
Peach SL, Borriello SP, Gaya H, Barclay FE, Welch AR (1986) Asymptomatic carriage of Clostridium difficile in patients with cystic fibrosis. J Clin Pathol 39(9):1013–1018
Rivlin J, Lerner A, Augarten A, Wilschanski M, Kerem E, Ephros MA (1998) Severe Clostridium difficile-associated colitis in young patients with cystic fibrosis. J Pediatr 132(1):177–179
Binkovitz LA, Allen E, Bloom D, Long F, Hammond S, Buonomo C, Donnelly LF 1999 Atypical presentation of Clostridium difficile colitis in patients with cystic fibrosis. AJR Am J Roentgenol 172(2):517–521
Barker HC, Haworth CS, Williams D, Roberts P, Bilton D (2008) Clostridium difficile pancolitis in adults with cystic fibrosis. J Cyst Fibros 7(5):444–447
Yates B, Murphy DM, Fisher AJ, Gould FK, Lordan JL, Dark JH, Corris PA (2007) Pseudomembranous colitis in four patients with cystic fibrosis following lung transplantation. Thorax 62(6):554–556
Theunissen C, Knoop C, Nonhoff C, Byl B, Claus M, Liesnard C, Estenne MJ, Struelens MJ, Jacobs F (2008) Clostridium difficile colitis in cystic fibrosis patients with and without lung transplantation. Transpl Infect Dis 10(4):240–244
Castagnola E, Battaglia T, Bandettini R, Caviglia I, Baldelli I, Nantron M, Moroni C, Garaventa A (2009) Clostridium difficile-associated disease in children with solid tumors. Support Care Cancer 17(3):321–324
McFarland LV (2005) Alternative treatments for Clostridium difficile disease: what really works? J Med Microbiol 54(Pt 2):101–111
Bauer MP, van Dissel JT (2009) Alternative strategies for Clostridium difficile infection. Int J Antimicrob Agents 33(Suppl 1):S51–56
Pillai A, Nelson R (2008) Probiotics for treatment of Clostridium difficile-associated colitis in adults. Cochrane Database Syst Rev 23(1):CD004611
McFarland LV, Surawicz CM, Greenberg RN, Fekety R, Elmer GW, Moyer KA, Melcher SA, Bowen KE, Cox JL, Noorani Z et al (1994) A randomized placebo-controlled trial of Saccharomyces boulardii in combination with standard antibiotics for Clostridium difficile disease. JAMA 271(24):1913–1918
Bryant K, McDonald LC (2009) Clostridium difficile infections in children. Pediatr Infect Dis J 28(2):145–146
Bland JM, Altman DG (2000) Statistics notes. The odds ratio. BMJ 320(7247):1468
Stamper PD, Alcabasa R, Aird D, Babiker W, Wehrlin J, Ikpeama I, Carroll KC (2009) Comparison of a commercial real-time PCR assay for tcdB detection to a cell culture cytotoxicity assay and toxigenic culture for direct detection of toxin-producing Clostridium difficile in clinical samples. J Clin Microbiol 47(2):373–378
Acknowledgements
This review was carried out as part of OM’s UK National Institute of Health Research (NIHR) Academic Foundation placement at the University of Southampton and was supported by the University of Southampton NIHR Wellcome Trust Clinical Research Facility (WTCRF). SNF and MT were funded by the UK NIHR via the NIHR WTCRF and and NIHR Clinical Lectureship respectively.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2013 Springer Science+Business Media New York
About this chapter
Cite this chapter
Morris, O. et al. (2013). Clostridium difficile in Children: A Review of Existing and Recently Uncovered Evidence. In: Curtis, N., Finn, A., Pollard, A. (eds) Hot Topics in Infection and Immunity in Children IX. Advances in Experimental Medicine and Biology, vol 764. Springer, New York, NY. https://doi.org/10.1007/978-1-4614-4726-9_4
Download citation
DOI: https://doi.org/10.1007/978-1-4614-4726-9_4
Published:
Publisher Name: Springer, New York, NY
Print ISBN: 978-1-4614-4725-2
Online ISBN: 978-1-4614-4726-9
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)