Abstract
A young old man, with frequent episodes of VT, referred for ablation suspected for sarcoidosis and ARVC in CMR, after ICD implantation presented with VT storm, without any endocardial scar and with extensive epicardial scar in voltage mapping, so epicardial VT ablation was done.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
Patient was a 36 y/o man, referred from elsewhere with diagnosis of RVOT–VT and hemodynamic compromise and termination of the arrhythmia with D/C shock. Echocardiography revealed moderate RV dysfunction and mild LV dysfunction, so cardiac MRI was recommended for patient.
Cardiac MRI revealed patchy areas of Late gadolinium enhancement (LGE) suggesting fibrosis in the RV inflow and outflow tract and another site of LGE in the mid posterolateral wall of LV with LVEF about 45%, and moderate RV dysfunction.
Patient was infertile.
The first diagnosis in cardiac MRI was Sarcoidosis and ARVC was recommended as the second diagnosis (Fig. 27.1a, b).
ICD was implanted for patients.
He refused ablation so was referred for Rheumatology consult.
Three weeks later he presented with VT storm.
Ablation
We tried to do endocardial biopsy using guide of 3D voltage mapping, but interestingly there was not any low voltage area in bipolar voltage mapping, but unipolar mapping revealed large scar burden, so ARVC with RV epicardial involvement was considered for him.
We did not do an endocardial biopsy because of the absence of any site of the endocardial scar.
So we tried to do an epicardial approach for VT ablation.
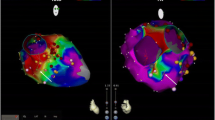
Subxiphoid puncture was done and 3D electroanatomical mapping of the epicardium revealed multiple sites of patchy scars in the RVOT and RV inflow areas.
Because of hemodynamic deterioration with VTs (Figs. 27.2 and 27.3), substrate modification was done for patients (Fig. 27.4a, b).
Before ablation coronary angiography was done determination of the position of the coronary arteries (Fig. 27.5).
We ablated all of the entrance conducting channels and late potentials in the RVOT and RV inflow (Figs. 27.6 and 27.7).
No arrhythmia was inducible after ablation with three ventricular extrastimuluses and Isuprel infusion (Fig. 27.8), so ablation was terminated and no arrhythmia was present in the 1 and 3 months follow-ups later.
Discussion
Scar distribution patterns in cardiac MRI may be helpful by indicating disease-specific patterns of fibrosis on LGE images. Cardiac Sarcoidosis is a diagnostic challenge and work up for it requires biopsy [1,2,3].
A scar pattern of basal septal RV involvement may be infavor of Sarcoidosis, but does not prove it.
In ARVC, the disease process starts in the epicardial RV where fibrofatty tissue replaces myocardial tissue.
In about 50% of the patients with ARVC, for cardiac MRI and voltage mapping was done, endocardial voltage mapping failed to detect areas of scar, especially in the inferobasal part of RV [4,5,6].
Involvement of LV is present in up to ¾ of the ARVC patients [7,8,9].
An epicardial approach is often necessary to eliminate VT in ARCV patients. The presence of epicardial scar could be assessed by unipolar mapping.
Low amplitude electrograms in bipolar mapping are defined in the endocardium by less than 1.5 mv voltage and in the epicardium by less than 1 mv voltage.
Endocardial unipolar signals suggesting midmyocardial or epicardial substrates are defined as less than 5.5 mv in RV and 8.3 mv in LV [10, 11].
In this patient we did scar dechanneling with ablation of the conducting channels entrance sites within scar, characterized by earliest late potentials following global ventricular activation and end point of the ablation was the elimination of all conducting channels into scar. Following targeted ablation at entrance sites and noninducibility of VT with three ventricular extrastimuluses with and without Isuprel infusion.
IN this patient ablation was successfully terminated, and intrapericardial triamcinolone was administered before long sheet withdrawn. Procedure was done without any complication [12, 13].
References
Cesario DA, Vaseghi M, Boyle NG, et al. Value of high-density endocardial and epicardial mapping for catheter ablation of hemodynamically unstable ventricular tachycardia. Heart Rhythm. 2006;3:1–10.
Garcia FC, Bazan V, Zado ES, Ren JF, Marchlinski FE. Epicardial substrate and outcome with epicardial ablation of ventricular tachycardia in arrhythmogenic right ventricular cardiomyopathy/dysplasia. Circulation. 2009;120:366–75.
Bai R, Di Biase L, Shivkumar K, et al. Ablation of ventricular arrhythmias in arrhythmogenic right ventricular dysplasia/cardiomyopathy: arrhythmia – free survival after endo-epicardial substrate based mapping and ablation. Circ Arrhythm Electrophysiol. 2011;4:478–85.
Santangeli P, Di Biase L, Lakkireddy D, et al. Radiofrequency catheter ablation of ventricular arrhythmias in patients with hypertrophic cardiomyopathy: safety and feasibility. Heart Rhythm. 2010;7:1036–42.
Dukkipati SR, d’Avila A, Soejima K, et al. Long-term outcomes of combined epicardial and endocardial ablation of monomorphic ventricular tachycardia related to hypertrophic cardiomyopathy. Circ Arrhythm Electrophysiol. 2011;4:185–94.
Sosa E, Scanavacca M, D’Avila A, Bellotti G, Pilleggi F. Radiofrequency catheter ablation of ventricular tachycardia guided by nonsurgical epicardial mapping in chronic Chagasic heart disease. Pacing Clin Electrophysiol. 1999;22:128–30.
Sosa E, Scanavacca M, d’Avila A. Transthoracic epicardial catheter ablation to treat recurrent ventricular tachycardia. Curr Cardiol Rep. 2001;3:451–8.
Reimer KA, Lowe JE, Rasmussen MM, Jennings RB. The wavefront phenomenon of ischemic cell death. 1. Myocardial infarct size vs duration of coronary occlusion in dogs. Circulation. 1977;56:786–94.
Jincun G, Faguang Z, Weibin H, Yan W, Kang D, Tung R. Outside-in subepicardial dissection during percutaneous epicardial ventricular tachycardia ablation. Circ Arrhythm Electrophysiol. 2016;9.
Valles E, Bazan V, Marchlinski FE. ECG criteria to identify epicardial ventricular tachycardia in nonischemic cardiomyopathy. Circ Arrhythm Electrophysiol. 2010;3:63–71.
Berruezo A, Fernández-Armenta J, Andreu D, et al. Scar dechanneling: new method for scar-related left ventricular tachycardia substrate ablation. Circ Arrhythm Electrophysiol. 2015;8:326–36.
Kuck KH, Schaumann A, Eckardt L, Willems S, Ventura R, Delacrétaz E, Pitschner HF, Kautzner J, Schumacher B, Hansen PS, VTACH Study Group. Catheter ablation of stable ventricular tachycardia before defibrillator implantation in patients with coronary heart disease (VTACH): a multicentre randomised controlled trial. Lancet. 2010;375:31–40.
Shani MM, Haghjoo M, Vasheghani A, Madadi S, Hashemi SN, Shabani R, Taraghi N. Evaluation of diagnostic characteristics and predictors of appropriate ICD therapy in patients with arrhythmogenic right ventricular cardiomyopathy. IHJ. 2016.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 Springer-Verlag London Ltd., part of Springer Nature
About this chapter
Cite this chapter
Madadi, S. (2021). Epicardial Approach for VT Ablation in an ARVC Case. In: Maleki, M., Alizadehasl, A. (eds) Case-Based Clinical Cardiology. Springer, London. https://doi.org/10.1007/978-1-4471-7496-7_27
Download citation
DOI: https://doi.org/10.1007/978-1-4471-7496-7_27
Published:
Publisher Name: Springer, London
Print ISBN: 978-1-4471-7495-0
Online ISBN: 978-1-4471-7496-7
eBook Packages: MedicineMedicine (R0)