Abstract
In this chapter we deal with the ways in which the two olivocochlear (OC) efferent systems, the medial (MOC) and lateral (LOC) systems, change the operation of the cochlea and how these changes may benefit hearing. To understand these changes, it is necessary to understand OC anatomy. OC anatomy is dealt with extensively in Brown (Chap. 2). Here we present the anatomy necessary for understanding OC physiology and function.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
3.1 Introduction
In this chapter we deal with the ways in which the two olivocochlear (OC) efferent systems, the medial (MOC) and lateral (LOC) systems, change the operation of the cochlea and how these changes may benefit hearing. To understand these changes, it is necessary to understand OC anatomy. OC anatomy is dealt with extensively in Brown (Chap. 2). Here we present the anatomy necessary for understanding OC physiology and function.
An important anatomical difference between MOC and LOC fibers is that MOC fibers are myelinated and can be recorded from and electrically stimulated, whereas LOC fibers are unmyelinated and have not been recorded from or directly stimulated. In addition, MOC fibers can be readily excited by acoustic stimulation, but it is not well established whether LOC fibers are acoustically excited, or not. The result is that we know a lot about the MOC system and very little about the LOC system.
Our focus is on OC function at the systems level of cochlear operation, covering important topics studied primarily since 1996. For a more elaborated review of older work, see the Guinan (1996) SHAR chapter. Sewell (Chap. 4) and Katz et al. (Chap. 5) present more detailed aspects of OC pharmacology, neurochemistry and the mechanisms of cholinergic inhibition. We will present enough of these topics for the reader to understand the material of this chapter.
The most important features of OC anatomy are shown in Fig. 3.1 (Smith 1961; Kimura and Wersäll 1962; Warr and Guinan 1979; Liberman 1980). Medial olivocochlear (MOC) fibers synapse on outer hair cells (OHCs), whereas lateral olivocochlear (LOC) fibers synapse on the dendrites of the type I afferent fibers, the fibers that make up the bulk of the auditory nerve (AN). Type II afferents send unmyelinated fibers to the brain and have much thicker processes that innervate OHCs with reciprocal synapses (i.e., the synapses go in both directions) (Thiers et al. 2002, 2008). These type II fibers spiral along the cochlea and receive innervation from MOC fibers at several places along their route (only one is shown in Fig. 3.1). The type II afferents may provide an additional way for MOC fibers to affect OHCs.
Afferent and efferent innervation of the cochlea. A schematic of the organ of Corti showing (1) radial, type I, afferent auditory-nerve (AN) fibers that innervate inner hair cells (IHCs), (2) lateral olivocochlear (LOC) efferent fibers that synapse on these radial afferent fibers, (3) medial olivocochlear (MOC) efferent innervation of outer hair cells (OHCs), and (4) spiral (spiraling not shown), type II afferent AN fibers that form reciprocal synapses on OHCs and receive synapses from MOC fibers. Omitted are synapses between LOC and MOC efferents in the tunnel of Corti, and MOC synapses onto supporting cells
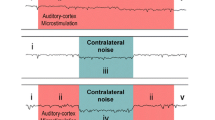
The main brain stem pathways of the MOC acoustic reflexes are shown in Fig. 3.2 (Guinan et al. 1983; Thompson and Thompson 1991; de Venecia et al. 2005). Inner hair cells (IHCs) sense cochlear mechanical movements and excite AN fibers. The AN fibers innervate neurons in the cochlear nucleus (CN). Reflex interneurons in the posteroventral cochlear nucleus send axons across the midline to innervate MOC neurons. These MOC neurons then send crossed or uncrossed axons that innervate OHCs. Note that the reflex signal crosses in the axons of the CN interneurons so that the contralateral MOC reflex (the MOC elicitor sound is contralateral to the side of the measurement) is mediated by the uncrossed MOC axons. Similarly, the ipsilateral reflex is mediated by the crossed MOC axons (it is a double-crossed reflex).
MOC acoustic-reflex pathways to the right (ipsilateral) cochlea. Schematic transverse brain stem section and cochlear cross sections showing the three-neuron contralateral and ipsilateral MOC acoustic reflexes: (1) Auditory nerve (VIIIth nerve) fibers (solid lines) to the cochlear nucleus (CN), (2) CN relay neurons with their crossed projections (dashed lines) to MOC cell bodies, and (3) MOC neuron axons to the cochlea (solid lines). Also shown are descending inputs to MOC cells (dotted lines)
In most laboratory animals there are twice as many crossed as uncrossed MOC fibers (review: Warr 1992) which is consistent with the ipsilateral reflex being twice as strong as the contralateral reflex (see later). In the squirrel monkey, there are 1.5 times as many crossed as uncrossed MOC fibers. In humans the ratio is unknown. Anatomical data indicate that the LOC projection to the cochlea is tonotopic but anatomical data are inconclusive for the MOC reflex (Guinan et al. 1984; Robertson et al. 1987). However, single-fiber labeling, which combines anatomical and physiological data, shows that the MOC reflex projection to the cochlea is also tonotopic.
3.2 MOC Effects in the Cochlea: Overview
MOC activation produces effects in the cochlea on two time scales, fast (∼100 ms) and slow (10s of seconds) (Sridhar et al. 1995; Cooper and Guinan 2003). The most common MOC effects are fast effects. Classic fast effects are due to turning down the gain of the cochlear amplifier, and this has different consequences depending on whether there is a background noise, or not. These classic fast effects are reviewed in Sects. 3.3 and 3.4. There are several MOC effects that are not due simply to turning down the gain of a cochlear amplified traveling wave. These effects appear to involve other vibrational motions of the organ of Corti in addition to motion that strictly follows basilar membrane (BM) motion. These are termed nonclassic MOC effects and are reviewed in Sect. 3.5. The MOC slow effect is reviewed in Sect. 3.6. The focus then changes, and Sects. 3.7 and 3.8 review MOC fiber responses to sound and MOC acoustic reflexes, followed in Sect. 3.9 by a discussion of MOC function in hearing. LOC physiology is reviewed in Sect. 3.10. This is short because we know little about LOC physiology. Finally, Sect. 3.11 summarizes the highlights of OC efferent physiology and makes suggestions for future research directions. But first, the next part of the present section provides some background and reviews two minor fast MOC electrical effects, an increase in cochlear microphonic (CM) and a decrease in endocochlear potential (EP).
3.2.1 MOC Activation Increases CM
The electrical effects produced by MOC efferents originate in the MOC synapses on OHCs. The neurochemistry and cellular physiology of these synapses are reviewed in detail in Sewell (Chap. 4) and Katz et al. (Chap. 5). Briefly stated, acetylcholine (ACh) released by the MOC presynaptic terminal acts on an unusual kind of ACh receptor that has a high calcium conductance (Elgoyhen et al. 1994, 2001). Activation of the ACh-receptors allows calcium (Ca2+) ions to flow into the OHC and these Ca2+ ions turn on nearby Ca2+-activated potassium (K+) channels (Housley and Ashmore 1991; Fuchs 1996). The resulting outflow of K+ ions overwhelms the smaller inflow of Ca2+ so that the net effect is a hyperpolarization of the OHC.
MOC stimulation increases CM (Fex 1959). This effect is produced primarily by the increase in OHC basolateral conductance brought about by the opening of both the K+ and the ACh channels. This increase in OHC synaptic conductance increases the receptor current through OHC stereocilia by increasing the conductance of the current return path. The receptor current is also increased slightly by the OHC hyperpolarization which slightly increases the voltage that drives current through the OHC stereocilia (this voltage is the difference between the ∼+100 mV EP and the ∼−60 mV OHC intracellular potential). The OHC receptor current flowing through the resistance of the surrounding tissue produces the CM voltage, and the MOC-induced increase in the receptor current increases this CM. Although MOC activation reduces basilar-membrane (BM) motion and the resulting receptor currents near the best-frequency place of a tone, this region contributes little to the externally measured CM because the phase of BM motion, and the resulting current flow, changes by more than 360° across this region and mostly cancels out when measured at a distant electrode. A distant electrode records primarily CM from the in-phase current sources basal to the peak of the traveling wave. Since there is no cochlear amplification in this region to be decreased, the MOC effect in this region is to increase the receptor current resulting in an increased CM seen from a distant electrode.
3.2.2 MOC Activation Decreases EP and Has Other Related Effects
The MOC-induced increase in OHC receptor current produces a small (a few millivolts) decrease in EP (Fex 1959; Gifford and Guinan 1987). The EP source in stria vascularis, like any battery, has an associated resistance, and the reduction in EP is due to a voltage drop in this resistance produced by the receptor current. The MOC-induced increase in receptor current increases the voltage drop resulting in a decreased EP.
The decrease in EP produces several other effects. A large part of the driving voltage at both IHC and OHC stereocilia comes from EP. In the OHCs, the MOC reduction in EP contributes to the reduction in cochlear amplification. In the IHC, the MOC reduction in EP produces a reduction in IHC receptor potentials and reduces AN responses. This may account for part of the MOC-induced reduction of AN responses at high sound levels where there is little cochlear amplification (Guinan and Stankovic 1996). The MOC reduction in EP also lowers the resting IHC potential and thereby reduces AN spontaneous activity (Guinan and Gifford 1988b). More detail and a circuit diagram illustrating these concepts are given in the Guinan (1996) SHAR chapter.
3.3 Classic MOC Fast Effects in a Silent Background
Before we consider MOC effects on cochlear amplification we must understand the basics of cochlear amplification. “Cochlear amplification” is the name given to the process by which OHCs increase the amplitude of BM responses to sound. Cochlear amplification comes about by a process in which BM motion bends OHC stereocilia thereby causing OHC motion that feeds energy back into BM motion. The gain of cochlear amplification comes from the interplay of all parts of this cycle, but two aspects are particularly important and reasonably well understood. The gain of forward transduction (stereocilia motion to receptor current) is set by the slope of the OHC-receptor-current vs. stereocilia-angle curve and is reduced during two-tone suppression (e.g., Geisler et al. 1990; Geisler 1992). The gain of backward transduction (OHC voltage to OHC motion) is set by the characteristics of the protein prestin, which produces OHC somatic motility by voltage-controlled changes in molecular conformation that cause OHC elongation and contraction (Santos-Sacchi 1991; Dallos et al. 2008). The least well understood part of cochlear amplification is the micromechanical motions involved, both the coupling of BM motion to the bending of OHC stereocilia (including tilting of the reticular lamina – Nowotny and Gummer (2006), and possible traveling waves in the tectorial membrane – Ghaffari et al. (2007), and the coupling of OHC elongation back into BM motion (see Cooper and Kemp 2009). Another poorly understood area is how fluctuations in OHC receptor current produce an adequate change in OHC voltage at frequencies far above the OHC membrane low-pass frequency of ∼1 kHz (see Lu et al. 2006 for one answer). Also poorly understood is the role of OHC stereocilia motility in mammalian cochlear amplification; its lack of a clear role is surprising considering that it is the main source of cochlear amplification in non-mammalian vertebrates (Hudspeth 2008).
Cochlear amplifier gain is changed during the MOC fast effect by two mechanisms: shunting and hyperpolarization. First, the MOC-induced increase in OHC synaptic conductance shunts the OHC receptor current thereby producing smaller changes in OHC voltage. The effect of OHC shunting should be less at frequencies above the OHC membrane low-pass frequency where the OHC capacitance dominates the OHC impedance (Guinan 1997). Second, the MOC-induced hyperpolarization of OHCs moves the operating point of reverse transduction away from its optimum point which reduces the OHC motion produced by a given OHC voltage change (Santos-Sacchi 1991). This effect should be independent of sound frequency. Finally, MOC fibers also synapse on type II afferent fibers and may indirectly exert effects on OHCs and cochlear amplification through the type II reciprocal synapses on OHCs (see Fig. 3.1).
3.3.1 Classic MOC Fast Effects on Basilar-Membrane Motion
MOC activation turns down the gain of cochlear amplification, and since amplification is greatest at low sound levels and at the best frequency, MOC activation has the largest effect at low sound levels and at the best frequency. In BM level functions, MOC activation shifts the response curve to higher levels and this shift is largest at low sound levels (Fig. 3.3a) (Murugasu and Russell 1996; Dolan et al. 1997; Cooper and Guinan 2003, 2006 a; but see Russell and Murugasu 1997 with Ren and Nuttall 2001). Since MOC activity turns down the gain, to get the same BM response the sound level must be turned up. The shift to higher sound levels (called the “level shift”) is a measure of how much cochlear amplification has been reduced. The level shift is greatest at the tip of the BM tuning curve (TC) and is near zero at much lower frequencies (Fig. 3.3b). This is consistent with the pattern of cochlear amplification deduced from measurements of BM motion in sensitive vs. damaged preparations (Robles and Ruggero 2001). Since MOC stimulation produces larger shifts near the best frequency (BF) than at the edges of the TC tip, the TC width is increased by MOC stimulation (Fig. 3.3b) (Murugasu and Russell 1996; Cooper and Guinan 2006a). MOC stimulation also produce phase leads in BM motion in the low-level tip portion of the response (Guinan and Cooper 2003).
Fast MOC effects on basilar membrane (BM) motion and auditory nerve (AN) firing. Left: BM motion (a) and AN firing rate (c, e) vs. sound level. Right: Tuning curves for BM motion (b) and AN firing (d, f) vs. tone frequency. Thin and thick lines are with and without MOC activity elicited by brain stem shocks. ((a, b) Adapted with permission from Cooper and Guinan 2006a, (c, e) Adapted with permission from Guinan and Stankovic 1996, (d) Adapted with permission from Guinan and Gifford 1988c, and (f) from unpublished data of Guinan and Gifford)
MOC effects on BM responses to clicks follow what would be expected from the MOC effects on BM responses to tones (Guinan and Cooper 2008). MOC inhibition, as a percentage of the response, is strongest at low click levels and becomes very small at high click levels (Fig. 3.4a). There is no MOC inhibition on the first half cycle of the BM response at any level, consistent with this initial response being passive. After the first half cycle, inhibition builds up gradually, and ultimately increases the decay rate of the BM click response. BM click responses in sensitive preparations show prominent waxing and waning, presumably from the interaction of two resonances (Recio et al. 1998; Guinan and Cooper 2008). MOC stimulation had little effect on the waxing and waning of the responses or response instantaneous frequency. MOC stimulation also produced small phase leads in the response waveforms.
MOC effects on BM motion (a) and AN firing (b) in response to clicks. (b) Compound histograms with the recovered-probability post-stimulus-time histogram from rarefaction plotted upwards and from condensation plotted downward. Black solid line = clicks alone; red dashed line = clicks plus MOC shocks (adapted with permission from Guinan et al. 2006)
3.3.2 Classic MOC Fast Effects on Otoacoustic Emissions
Otoacoustic emissions (OAEs) are sounds that originate from within the cochlea and travel backward through the middle ear into the ear canal. They are useful because they can be measured noninvasively and provide a window into the mechanical response of the cochlea. They have been most valuable when used in humans, and here they are paired with noninvasive eliciting of MOC activity by sound. Most of the new things learned using OAEs will be considered in Sect. 3.8.2 (the MOC acoustic reflexes). The present section considers how well MOC effects on OAEs fit with what has been learned from more direct measurement techniques, and how well the various OAE types serve as metrics for MOC effects.
For a detailed presentation of the three types of evoked OAEs: transient evoked, stimulus frequency, and distortion product (TEOAEs, SFOAEs, and DPOAEs), see Shera and Guinan (2007). At the sound levels usually used, TEOAEs and SFOAEs are due to coherent reflection, whereas DPOAEs are a combination of components from a distortion source and a coherent reflection sourceFootnote 1 (Shera and Guinan 1999, 2007). The two DPOAE sources have different phase properties and they interfere in the ear canal, that is, they can be in-phase and add, or out-of-phase and cancel.
The principal on which OAE measurements of MOC effects are based is that OAEs get most of their energy from cochlear amplification and MOC activity turns down the gain of the cochlear amplifier thereby lowering OAE amplitudes. MOC effects are obtained by first measuring the OAE without MOC activation and then with MOC activation; the difference is the MOC effect. MOC effects on OAEs are usually expressed in a normalized form as the decibel change from the original OAE amplitude. This dB change is more closely related to the decibel change in cochlear amplification than the absolute value of the change which varies with the OAE amplitude as well as with changes in cochlear amplification. An even better measure is the MOC-induced level shift. The relationship between MOC-induced changes in OAEs and in cochlear output (i.e., in AN responses), is unknown. In a study using DPOAEs, the relationship varied widely from nearly equal changes to much smaller OAE changes than N1 changes (Puria et al. 1996).
Since TEOAEs and SFOAEs originate from a single mechanism, linear coherent reflection, they show a simple pattern of MOC effects. The MOC effect on these emissions is almost always a reduction of their amplitude (e.g., Guinan 1990; Collet et al. 1990; Veuillet et al. 1991, 1996; Ryan and Kemp 1996; Guinan et al. 2003; Backus and Guinan 2006). In contrast, since the two DPOAE sources interfere, the MOC effect on DPOAEs is very complex and can even be an increased DPOAE (Siegel and Kim 1982; Moulin et al. 1993; Muller et al. 2005; Wagner et al. 2007). If the two DPOAE components normally cancel, and MOC stimulation inhibits one component more than the other, this inhibition reduces the cancelation and increases the DPOAE. Thus, the relative phases of the two DPOAE components greatly influences the DPOAE change measured. Since this phase relationship is unrelated to the MOC effect but strongly influences the result, the MOC change in DPOAE amplitude is not an accurate way of measuring MOC effects. Measuring MOC effects at response dips makes the value obtained larger, but, in humans, the result depends on the phase relationship more than on MOC strength.
A different kind of OAE paradigm is “DPOAE adaptation” which uses the DPOAE time course to measure the MOC effect (Liberman et al. 1996). The DPOAE primary tones are turned on abruptly and evoke MOC activity that builds up with a time constant of ∼100 ms. The difference between the DPOAE just after the onset (which is not yet affected by MOC activity) and the DPOAE after a few hundred ms (which is affected by the MOC activity elicited by the primary tones) provides a metric of the MOC effect. This method measures effects of the ipsilateral MOC reflex, which is an advantage. A disadvantage is that the primary tones also produce a slower DPOAE change (time constant ∼1 s) that remains after all efferents are cut and therefore is due to effects intrinsic to the cochlea (these intrinsic effects might be due to a build up of potassium around the OHC or an effect of the afferent type II neural network). The DPOAE adaptation technique has been applied in humans (e.g., Kim et al. 2001; Bassim et al. 2003; Muller et al. 2005) but, in humans, the results cannot be reliably interpreted because efferents cannot be cut and MOC effects cannot be separated from cochlear intrinsic effects.
In summary, MOC effects on TEOAEs and SFOAEs can be interpreted in a straightforward manner, but DPOAE measurements are complex because they originate from two separate cochlear mechanisms and places. DPOAE measurements of MOC effects can be made more accurate by separating the DPOAE into its two source components (e.g., Thompson et al. 2009; Abdala et al. 2009). Overall, OAEs provide imperfect measures of MOC effects on cochlear mechanical changes, but have the great advantage of being noninvasive.
3.3.3 Classic MOC Fast Effects on IHC and AN Responses
MOC effects were first observed on AN compound action potential (CAP) responses (N1) to clicks. In N1 responses evoked by clicks or tone-pips in a silent background, there are MOC level shifts as large as 20–30 dB at low sound levels (Galambos 1956; Desmedt 1962; Wiederhold and Peake 1966; Gifford and Guinan 1987). These N1 level shifts decrease to near zero at high sound levels, consistent with the MOC effect being to turn down the gain of the cochlear amplifier.
The dependence of MOC inhibition on MOC firing rate has been determined from MOC effects on AN N1 responses. The greatest inhibition of N1 is produced by shock rates of 200–400/s with little change in inhibition across this range. Inhibition diminishes rapidly as shock rates are lowered below 200/s, for example, at 60/s shocks the inhibition is about ¼ of the maximum value (Gifford and Guinan 1987). Existing data suggest that all MOC effects have a similar dependence on shock rate (Desmedt 1962; Konishi and Slepian 1971; Gifford and Guinan 1987; Rajan 1988). MOC fibers follow MOC shocks one-for-one for shock rates up to rates of 200/s, but not at 400/s (McCue and Guinan, unpublished). Thus, the effects shown by shocks at rates of 200/s, or less, indicate the effects that would be produced by sound-evoked MOC activity at the same rate. The effects produced by trains with different numbers shocks indicate that the MOC synapse shows strong, time-dependent facilitation, and that this facilitation is the origin of the dependence of MOC effects on shock rate (Cooper and Guinan 2006b).
For low-level sounds, MOC stimulation shifts sound level functions of IHC receptor potential and AN-fiber firing rate toward higher sound levels (Fig. 3.3c) and this shift is greatest for tones at the characteristic frequency (CF) (Fig. 3.3d), similar to the MOC effect on BM motion (Wiederhold 1970; Teas et al. 1972; Guinan and Gifford 1988a; Guinan and Stankovic 1996). The pattern of MOC-induced AN rate shifts across fiber CFs closely matches the pattern of MOC innervation of OHCs along the cochlea (Guinan and Gifford 1988c; Liberman et al. 1990; Maison et al. 2003). The characteristics of these cochlear responses to low level sound are readily explained by the classic MOC effect of turning down the gain of the cochlear amplifier.
MOC inhibition makes TCs wider for fibers with CFs >3 kHz and for most fibers with lower CFs. Again, this is what is expected from the pattern of cochlear amplification relative to CF and efferents turning down the gain of the cochlear amplifier by a fixed ratio at each frequency. However, in some low CF fibers, MOC stimulation narrows the TC (e.g., Fig. 3.3f); this is not a classic MOC effect and will be dealt with in Sect. 3.5.
3.4 Classic MOC Fast Effects in a Noisy Background
In the presence of a low-level background noise, MOC stimulation can increase the neural response to a brief sound (Winslow and Sachs 1987; Kawase et al. 1993). The mechanisms for this are illustrated in Fig. 3.5. The top panels show the MOC inhibition of AN fiber responses to short tone bursts at CF without a background noise, which is a shift of the rate-vs.-level function to higher levels. The bottom panels show responses to the same stimuli with an added background noise. The AN response with a background noise but no MOC activation (dashed line in Fig. 3.5c) shows an increased firing rate at very low tone-burst levels from excitation by the background noise. Because the noise is continuous, the increased AN firing is continuous. This continuous firing causes AN fiber adaptation, principally by using up vesicles at the IHC-AN synapse. The resulting vesicle depletion has the effect of lowering the AN rate to high-level tone bursts because there are fewer vesicles to release. The result of both effects is to reduce the dynamic range of the AN output, that is, the noise partially masks the response to the tone. MOC stimulation reduces cochlear amplifier gain, which reduces the response to the low-level background noise (Fig. 3.5d, bottom left). The noise then causes less adaptation so high-level tones can evoke higher firing rates. The net effect is to partially restore the output dynamic range of the auditory fiber (called MOC unmasking). Although there is little change in the threshold produced by the MOC activity, the increase in output dynamic range means that small changes in the tone are more robustly signaled to the central nervous system (CNS), which increases the discriminability of the tone in the noise. This increase in the in the discriminability of the tone, or of other brief signals such as the transitions in speech, is probably the most important function of MOC efferents in everyday hearing (see Sect. 3.9).
Schematic of MOC unmasking of AN fiber responses. Each panel shows rate-vs.-level functions for a single AN fiber with different conditions coded by line styles (key at the top of each panel). Schematized from the results of Wiederhold (1970); Winslow and Sachs (1987); Guinan and Gifford (1988a); and Kawase et al. (1993) (adapted with permission from Guinan 1996)
Another circumstance in which MOC inhibition of the cochlear amplifier has been suggested to affect psychophysical performance is the signal-in-noise “temporal effect,” also called “overshoot” (e.g., Zwicker 1965; Strickland and Krishnan 2005; Strickland 2008). The temporal effect is the phenomenon that a higher level of noise is needed to mask a brief tone when the tone is presented long after (>100 ms) the noise onset compared to just after the noise onset. One hypothesis is that the noise elicits efferent activity that turns down the gain of the cochlear amplifier thereby decreasing the response to the noise more than the response to the tone (Fig. 3.6). Since it takes 100 ms, or more, for MOC activity to reduce the gain, the S/N of the cochlear response is increased only for tones presented >100 ms after the onset of the noise. With SFOAEs as a monitor, the hypothesized increase in MOC activity was not seen even though the parameters used produced a substantial temporal effect (Keefe et al. 2009). However, the temporal effect shows a complicated dependence on the parameters of tone frequency and the noise spectrum relative to the tone. It may be that the temporal effect is due both to MOC effects and to other, perhaps central, effects, depending on the parameters used. Overall, it seems likely that both Figs. 3.5 and 3.6 illustrate ways in which MOC inhibition can increase the ability to hear brief sounds in noise.
The MOC-induced change in BM input–output function in the tone-detection-in-noise “temporal effect” (also called “overshoot”). The BM input–output function shows a large shift at low levels due to the noise-elicited MOC activity turning down the gain of cochlear amplification. The lowered gain means that a long-delayed tone in noise requires more noise to be just masked than a tone just after the noise onset (adapted with permission from Strickland and Krishnan 2005)
3.5 Nonclassic MOC Fast Effects in a Silent Background
Nonclassic MOC effects are effects that cannot be explained by the classic view that: (1) organ of Corti motion is a single vibration pattern that directly follows the BM motion of the traveling wave, (2) this motion is amplified by the cochlear amplifier, and (3) MOC effects are produced by turning down the gain of this amplifier. The part of this view that has the greatest need for revision is that organ of Corti motion is a single vibration pattern. Measurements in excised preparations show complex vibrational patterns of the organ of Corti consistent with the motion being the sum of motions from multiple vibrational modes (Mountain 1998). Further, some of these vibrational modes could lead to bending of IHC or OHC stereocilia without there being a direct coupling to BM motion (Nowotny and Gummer 2006; Karavitaki and Mountain 2007a, b; Ghaffari et al. 2007). It has not been possible to make micromechanical measurements in intact mammalian cochleas with demonstrated normal sensitivity. However, AN recording from intact cochleas with demonstrated normal sensitivity provide ample evidence that the motion that drives IHC stereocilia and leads to AN responses is due to multiple vibrational modes (e.g., Gifford and Guinan 1983; Liberman and Kiang 1984; Lin and Guinan 2000, 2004; Guinan et al. 2005).
MOC effects on BM motion have only been measured in the basal half of the cochlea (review: Cooper and Guinan 2006a). In fact, reliable measurements of mechanical responses of any kind in live preparations with thresholds shown to be normal (by tone-pip CAPs) are only from the basal half of the cochlea (Robles and Ruggero 2001). Measurements of cochlear motions have been made in the apex, but these have been without a good monitor of the preparation’s sensitivity in the frequency region tested. Furthermore, many apical measurements are also contaminated by artifactual motion from the cochlear fast wave (Cooper and Rhode 1996). The result is that there is little direct knowledge of cochlear motions in the apical half of the cochlea. Motion in the apical half of the cochlea is often thought to be similar to motion in the base because of apex-to-base similarities in cochlear anatomy and in many aspects of AN responses. However, mechanical measurements in excised preparations show qualitative differences in the apex compared to the base (Nowotny and Gummer 2006) and there are many apex-to-base differences in AN response patterns. Because of this, nonclassic MOC effects in the base and apex are considered separately.
3.5.1 Nonclassic MOC Fast Effects in the Basal Half of the Cochlea
In the basal turn of guinea pigs, MOC stimulation produces an increase in BM motion in response to high-level tones at frequencies well above the local best frequency (Dolan et al. 1997; Guinan and Cooper 2003). In plots of BM motion vs. sound level, there is typically a dip in BM motion at these frequencies. Below the dip MOC stimulation decreases the BM response, above the dip MOC stimulation increases the response, and at the dip there is a phase change close to a reversal (Guinan and Cooper 2003). A hypothesis that fits the data is that BM motion is due to a cochlear-amplified component that is large at low sound levels and saturates at high levels, plus a passive component that grows linearly and is out-of-phase with the amplified component. When the two components are equal in amplitude they cancel. MOC stimulation inhibits the amplified component which reduces the cancelation so that the resulting BM motion increases (Guinan and Cooper 2003). The origin of the unamplified component and its vibration pattern in the organ of Corti is not known, but one possibility is that this motion is a direct mechanical response to the fast pressure wave (Rhode 2007). It is unknown whether there is a similar effect in AN responses in the cochlear base because there are no suitable measurements of MOC effects for tones at high levels and frequencies above CF. However, this effect has many similarities to the MOC effects in AN fibers with CFs near 1 kHz (Gifford and Guinan 1983) (see Sect. 3.5.2).
A second nonclassic MOC effect is that AN fibers with low spontaneous rates (SRs) have rate vs. level functions that show greater level shifts at moderate-to-high sound levels than at low sound levels (Fig. 3.3e) (Guinan and Stankovic 1996). At low sound levels, the level shift appears to be due to MOC activity turning down the gain of cochlear amplification. However, at moderate to high sound levels the level shifts are too large to be fully accounted for by a MOC reduction of cochlear amplifier gain. Some additional reduction in AN firing rate must come from the MOC-induced reduction of EP (Sect. 3.2.2), but it is not clear that this is enough to account for the large level shifts observed. Another possibility is that AN drive at high levels is a combination of two out-of-phase components (as suggested in the previous paragraph), and that the more linear component approaches the cochlear-amplified component but never becomes larger than it (in the motion of IHC stereocilia). With this scenario, at high levels these two components would partially cancel and MOC inhibition, by reducing the larger component, would increase the cancelation. This would make the resulting level shift greater than the reduction of cochlear amplification. A similar mechanism may explain the large two-tone suppression observed in low-SR AN fibers (Cai and Geisler 1996).
Another nonclassic MOC effect is that in cat AN fibers with high CFs (>10 kHz), MOC stimulation inhibits the response at frequencies much lower than the TC tip (called “tail” frequencies; Fig. 3.3d) (Stankovic and Guinan 1999). BM measurements indicate that there is no cochlear amplification at tail frequencies and there is no comparable MOC inhibition of BM motion at similar tail frequencies (Fig. 3.3b) (Murugasu and Russell 1996; Dolan et al. 1997; Guinan and Cooper 2003; Cooper and Guinan 2006a). MOC inhibition of ∼1 dB at tail frequencies in AN fibers is attributable to the MOC-induced reduction of EP which reduces IHC receptor currents and the resulting AN response (Guinan and Gifford 1988b). However, the AN inhibition is particularly large (as much as 10 dB) near 2–3 kHz (Stankovic and Guinan 1999). It is noteworthy that AN response latencies (derived from phase-gradient group delays) at this 2–3 kHz region are slightly less than AN latencies at lower frequencies, despite the fact that the lowest frequency energy in the traditional traveling wave arrives first at any given cochlear location (Shera 2001; Rhode 2007). A hypothesis that fits these data is that: (1) there is a cochlear motion produced by sounds near 3 kHz that is inhibited by MOC efferents (which implies that the motion is derived from, or influenced by, OHCs), (2) this motion occurs slightly before the motion produced by low-frequency energy in the traveling wave, and (3) this motion produces bending of IHC stereocilia that excites AN fibers but with little or no associated BM motion (Guinan et al. 2005). The exact motion is unknown but one possibility is that the motion is a combination of fluid flow in the tunnel of Corti and an associated tilting of the reticular lamina (Karavitaki and Mountain 2007a; Nowotny and Gummer 2006).
3.5.2 Nonclassic MOC Fast Effects in the Apical Half of the Cochlea
Cat AN fibers with low CFs have rate and phase sound-level functions that show a sharp dip in rate at a high level (80–100 dB SPL) that is accompanied by an abrupt reversal of phase (Liberman and Kiang 1984). MOC stimulation reduces the firing rate at levels below the dip but not above the dip (Gifford and Guinan 1983). The reduction in rate below the dip follows the pattern of a classic MOC fast inhibition, but the whole phenomena of a rate dip and phase reversal does not. A hypothesis similar to the one for the MOC-induced enhancement of BM motion also fits these data (except that above the dip the AN response is not significantly enhanced), namely that the dip is due to the cancelation of two out-of-phase drives with the drive that is dominant at low levels being cochlear amplified and therefore inhibited by MOC activity. Presumably the AN fiber rate above the dip is not increased, whereas BM motion is, because the AN rate is limited by a separate saturation, for example, in the amount of transmitter that can be released. In both the high-CF BM enhancement and the low-CF AN dip phenomena, the low level component is presumably the cochlear amplified traveling wave drive. It is not known if the component that becomes dominant at high levels is the same for these two phenomena.
As noted earlier, for some low-CF AN fibers, MOC stimulation makes the TCs narrower instead of wider as expected. Guinan and Gifford (1988c) obtained some data that suggest an explanation. Low-CF AN fiber TCs, in addition to the basic V-shaped tuning, have side lobes. In at least some fibers, MOC produces a particularly large inhibition in the side-lobe region with level shifts far exceeding those seen at CF (Fig. 3.3f). A MOC-induced reduction of these side lobes might be the explanation for the MOC narrowing of TCs.
A particularly striking nonclassic MOC effect in the apical half of the cochlea is the MOC inhibition of the AN initial peak (ANIP) response to clicks (Guinan et al. 2005). In AN fibers with CFs <4 kHz, the synchronization of AN spike times is adequate to show individual peaks in post-stimulus-time histograms of responses to clicks. These peaks show the oscillations of the mechanical drive at frequencies near the local CF. AN fiber recordings with and without MOC stimulation show the MOC effects expected from the MOC effect seen in BM motion due to turning down the gain of the cochlear amplifier. That is, MOC stimulation completely inhibits AN click responses near threshold, and above threshold reduces the decaying part of the response consistent with the reduction in BM ringing due to widening cochlear tuning. However, in addition, MOC stimulation inhibits the initial peak of the AN response (the ANIP response). In BM motion (in the cochlear base) the first peak of the response is passive, grows linearly and is little changed by death (Recio et al. 1998; Guinan and Cooper 2008). The AN response was expected to have the same properties, so the inhibition of the first peak was unexpected. The ANIP inhibition cannot be explained by MOC stimulation turning down the gain of the classic cochlear amplifier because the BM first peak is not amplified. The ANIP inhibition implies that the first peak is produced by a motion that originates in (or is strongly modified by) OHCs and that the motion is distinct from the motion of the classic traveling wave which is not inhibited in the first peak. One possibility is that the presumed ANIP motion is due to tilting of the reticular lamina produced by OHC contractions (Nowotny and Gummer 2006; Karavitaki and Mountain 2007a) but another possibility is that it is a motion of the tectorial membrane (Ghaffari et al. 2007). The ANIP response might be the most behaviorally important response that shows nonclassic MOC effects because it is present at moderate to high sound levels and could have a large effect on the cue for the localization of transient sounds.
All of the nonclassic MOC effects imply the presence of two (or more) vibration modes of the organ of Corti. Most of these effects appear to be due to a classic MOC gain reduction in the amplification of the traveling wave vibrational mode which interacts with another, perhaps linear, response mode. The inhibition of the ANIP response, however, is different. It implies there is a nontraveling-wave mode that is MOC inhibited and therefore not linear and passive.
3.6 MOC Slow Effects
When MOC stimulation that produces a fast inhibition is continued for 10s of seconds, it produces an additional long-lasting inhibition called the MOC slow effect (Fig. 3.7). This effect was first seen in guinea-pig AN N1 responses to clicks and high-frequency tone pips (Sridhar et al. 1995, 1997). Since the slow AN N1 inhibition was accompanied by a slow CM increase, the slow inhibition was attributed to a slow increase in the MOC-OHC synaptic conductance. However, in BM motion, while fast and slow MOC effects both produce inhibition, there is a phase advance during the fast effect and a phase delay during the slow effect (Fig. 3.7b) (Cooper and Guinan 2003). These phase changes in opposite directions mean that different underlying mechanisms in the OHC must be involved in producing MOC fast and slow effects. A related discovery is that application of the MOC neurotransmitter, ACh, to isolated OHCs produces a slow decrease in OHC stiffness (Dallos et al. 1997). A hypothesis that fits these data is that the MOC slow effect is due to the decrease in OHC stiffness, whereas the MOC fast effect is due to the OHC hyperpolarization and conductance change (Cooper and Guinan 2003). The increase in CM during the slow effect suggests there is also an OHC conductance change (Sridhar et al. 1995) but the BM phase change indicates the slow effect is dominated by the other change (e.g., stiffness). The ACh-induced decrease in OHC stiffness takes place in prestin molecules and the associated OHC cytoskeleton which are distant from the MOC synapse (He et al. 2003). One possibility is that there is a calcium action potential (a “calcium spark”) that travels from the subsynaptic cistern at the OHC-MOC synapse to the OHC cytoskeleton and produces the slow change in stiffness and the MOC slow effect (see Sridhar et al. 1997). One reason for interest in MOC slow effects is that they may be involved in protecting the ear from acoustic trauma (Reiter and Liberman 1995). MOC slow effects are largest at high frequencies (>10 kHz) in guinea pigs, and it is not known if they are present in humans.
MOC fast and slow effects both produce reductions of BM motion but with different phase changes. Variations in BM response amplitude (top) and phase (bottom) vs. time. The gray bar indicates the period with MOC stimulation (adapted from Cooper and Guinan 2003)
3.7 MOC-Fiber Responses to Sound
There are two ways that we can learn about MOC responses to sound: (1) by recording from MOC fibers and (2) by measuring sound-evoked MOC effects. In this section we review recordings from MOC fibers, and in the next section we review sound-evoked MOC effects. Most recordings from MOC fibers have been done in anesthetized animals. A variety of evidence indicates that anesthesia reduces sound-evoked MOC activity (Robertson and Gummer 1985; Liberman and Brown 1986; Brown 1989; Boyev et al. 2002) so results from anesthetized animals must be viewed with caution, especially MOC firing rates. Sound-evoked MOC effects can measured in awake humans but the sound levels have be kept low to avoid eliciting middle-ear-muscle reflexes. Overall, for awake intact animals, we do not have a good quantitative assessment of MOC firing rates or how big a neural change can be produced by sound-evoked MOC activity.
Recordings from single medial efferents come from two species: cats and guinea pigs (Fex 1962, 1965; Cody and Johnstone 1982; Robertson 1984; Robertson and Gummer 1985; Liberman and Brown 1986; Liberman 1988a, b; Gummer et al. 1988; Brown 1989, 2001; Brown et al. 1998a, b). MOC fibers have been contacted at two places: (1) in the vestibular–cochlear anastomosis – also called the “bundle of Oort” – where the MOC fibers pass from the vestibular nerve to the cochlear nerve and (2) in the intraganglionic spiral bundle within the cochlea. MOC fibers were distinguished from other nearby fibers by three criteria: (1) They respond to sound. If some MOC fibers do not respond to sound, they would not have been identified. (2) They have “regular” firing patterns (i.e., their spike-interval distributions are approximately Gaussian) in both spontaneous activity (which is low or zero in most fibers) and responses to sound (Robertson and Gummer 1985; Liberman and Brown 1986), and (3) their latency to sound is 5 ms, or more.
The latency of the initial MOC response to sound is highly dependent on sound level. The first spike latency is tens of ms for sounds near threshold, but for high-level sounds it can be as short as 5 ms (Robertson and Gummer 1985; Liberman and Brown 1986; Brown 1989; Brown et al. 2003). This latency-vs.-level behavior suggests that MOC neurons integrate responses from their synaptic inputs and fire when threshold is reached. MOC response latency is more uniform, averaging 8.2 ± 1 ms, when measured as a modulation transfer function at moderate sound levels (Gummer et al. 1988). These data, particularly the 5 ms minimal latency, are consistent with the MOC reflex being the three-neuron arc shown in Fig. 3.2. However, additional contributions from longer pathways may also be present, for example, from the marginal shell of the anteroventral cochlear nucleus (Ye et al. 2000).
TCs from MOC fibers are similar to, or slightly wider than TCs from AN fibers, particularly at their tips (Fig. 3.8) (Cody and Johnstone 1982; Robertson 1984; Liberman and Brown 1986). Single MOC fibers labeled by dye injections show that each fiber innervates a cochlear frequency region close to the MOC fiber’s best frequency (i.e., MOC projections are tonotopic) and most individual fibers innervate OHCs over a 0- to 1-octave range of cochlear length (Robertson 1984; Liberman and Brown 1986; Brown 1989, 2002) although some rat MOC fibers innervate OHCs over more than 40% of the length of the cochlea (Warr and Boche 2003). The TCs and cochlear innervation patterns of MOC fibers have lead to the idea that MOC fibers provide frequency-specific feedback to the cochlea (Winslow and Sachs 1987). The extent to which the MOC acoustic reflex actually produces narrow, tonotopic effects on cochlear responses will be considered later.
MOC fibers have been divided into three types based upon the ear that activates them using monaural sound; this ear is called the “main ear.” The types are: Ipsi (these respond only to ipsilateral sound – ipsilateral re the ear innervated by the MOC fiber), Contra (these respond only to contralateral sound), and Either-Ear (these respond to sound in either ear). In cats and guinea pigs, most (∼2/3) MOC efferents are Ipsi, some (∼1/3) are Contra, and a few (4–11%) are Either-Ear. These percentages correspond well with the percentages of crossed and uncrossed MOC fibers (Warr 1975; Robertson 1985; Liberman 1988a; Brown 1989; Robertson and Gummer 1988) and the observation from a few labeled MOC fibers that crossed fibers respond to ipsilateral sound while uncrossed MOC fibers respond to contralateral sound (see Fig. 3.2). Although for monaural sound most MOC fibers only respond to one ear, almost all MOC fibers are binaural in that once they are activated by the main ear, sound in the opposite ear (re the main ear) can modulate their firing rate. Typically, sound in the opposite ear produces additional activation, although sometimes it inhibits the response (Robertson 1985; Liberman 1988 a; Brown 1989; Brown et al. 1998a). Opposite-ear sound usually widens MOC TCs (Fig. 3.8).
The reported firing rates of MOC fibers in response to monaural sound are relatively low. In anesthetized animals, using monaural tones or noise at nontraumatic levels (<∼90 dB SPL), the highest reported firing rate of a MOC fiber is ∼60/s and for most MOC fibers the highest rate is one half to one third of that (Fex 1962; Robertson and Gummer 1985; Liberman 1988 a; Brown 1989; Brown et al. 1998 a). When using binaural sounds, or after priming by a previously traumatic sound, rates up to 134/s (cats) and 140/s (guinea pigs) have been found (Liberman 1988 a; Brown et al. 1998a), however, on average, the highest MOC firing rate is much less.
These sound-evoked MOC firing rates are distinctly less than the 200/s shock rates typically used to excite MOC fibers. The sound/shock rate discrepancy has raised the question of whether sound-evoked MOC activity produces significant effects (Pfalz 1969). Sound-evoked MOC effects on OAEs in awake humans are typically only a few dB (see later), again raising the same question. There are several factors which may account for this apparent discrepancy. First, all early MOC recordings and most experiments using OAEs used monaural sound; binaural sounds evoke much higher rates. Second, anesthesia is likely to have reduced the measured firing rates, perhaps by as much as a factor of two considering that MOC effects decreased approximately by a factor of 2 when awake animals were anesthetized with a barbiturate (Boyev et al. 2002). Third, in many MOC fibers, especially fibers with BFs >2 kHz, the firing rate continues to increase as long as the sound level increased, that is, no rate saturation was found. This suggests that much higher firing rates would be produced by sounds at traumatic levels. Finally, only a fraction of MOC fibers are activated by shocks (as shown by recordings of MOC fibers at the bundle of Oort while stimulating at the floor of the fourth ventricle; McCue and Guinan, unpublished). In contrast, it is thought that all, or almost all, MOC fibers are activated by sound. Thus, to produce the same effect, shock activation of MOC fibers might require higher rates than sound activation.
MOC response properties vary with fiber BF. MOC fibers with low BFs have shorter latencies, lower thresholds and higher maximum rates than high-BF fibers (Liberman and Brown 1986). During the presentation of a BF ipsilateral tone, binaural facilitation by contralateral tones is largest for low BF MOC fibers, whereas binaural facilitation by contralateral noise is largest for high-BF MOC fibers (Liberman 1988 a).
3.8 MOC Acoustic Reflexes
MOC fibers respond to sound and form ipsilateral, contralateral and bilateral MOC acoustic reflexes, terms that refer to the ear stimulated relative to the measurement ear. The MOC effects on cochlear responses presented in Sects 3.2–3.7 were mostly from shock-activation of MOC fibers. The same MOC effects are also produced by the MOC acoustic reflexes, but many aspects of the patterns of these effects may be altered by the brain stem control of MOC activation. For instance, shock activation of MOC fibers is not frequency specific, but sound activation may be frequency specific. In Sect. 3.8, first we consider the effects of sound-elicited MOC activation on AN responses. AN responses provide a direct measure of MOC effects on the output of the cochlea. Next, we consider MOC effects as seen by the changes induced in OAEs. OAEs are an indirect measure but have the advantage of being noninvasive so they can be measured in humans. Finally, we consider the influence of descending projections to MOC neurons.
3.8.1 Sound-Elicited MOC Effects on AN Fibers
Most MOC fast effects on AN responses produced by shocks are also produced by contralateral sound, but they are smaller when evoked by contralateral sound. Inhibition of AN N1 has been reported by many papers (e.g., Buño 1978; Folsom and Owsley 1987; Liberman 1989; Warren and Liberman 1989a, b; Aran et al. 2000). In single AN fibers, contralateral sound shifts rate and synchrony sound-level functions to higher levels but has little effect on phase functions (Warren and Liberman 1989a) which is similar to findings with shocks (Gifford and Guinan 1983). In both of these studies, MOC effects were greatest with the probe sound at the CF of the AN fiber. In fibers that showed dips in rate-vs.-level functions, contralateral sound lowered the rate below the dip and produced little change above the dip, again, the same pattern as found with shock activation of MOC fibers (Gifford and Guinan 1983; Warren and Liberman 1989a). Both contralateral sound and shocks produced small decreases in AN spontaneous activity, presumably due to the small MOC-induced decrease in EP.
For AN fibers excited by an ipsilateral CF tone, the addition of a contralateral tone inhibited the AN response (Warren and Liberman 1989b). The inhibition had a complicated pattern across fiber CFs, but was always largest for contralateral tones at frequencies near the AN-fiber CF. Fibers with CFs of 2–5 kHz showed the largest inhibitions for contralateral tones 0.5–1 octave below the CF (Fig. 3.9a). In fibers with much lower CFs, the largest inhibitions were for contralateral tones at frequencies above CF, and in fibers with CFs higher than 5 kHz, the largest inhibitions were for tones near CF.
Tuning of the MOC reflex is skewed when measured at midfrequencies by (a) AN responses, or (b) the change in stimulus frequency emissions (SFOAEs). (a) Responses to a characteristic frequency (CF) tone (2.14 kHz) from a cat AN fiber. Solid line: Inhibition from MOC activity elicited by a contralateral tone swept in frequency. X’s: a sweep with the contra tone off. (b) The normalized SFOAE change from a 1-kHz probe tone vs. elicitor center frequency for 60 dB SPL half-octave noise-band elicitors. Compare the solid curve in (a) with the contralateral curve in (b) ((a) adapted with permission from Warren and Liberman 1989b, (b) adapted from Lilaonitkul and Guinan 2009a)
Several lines of evidence indicate that all of the sound-evoked efferent effects described above are due to MOC and not LOC activity. First, sound evokes firing in MOC fibers that could produce these effects (see Sect. 3.7). Second, the fast time course and the patterns of the neural inhibition evoked by sound are qualitatively compatible with the inhibition produced by shocks to the olivocochlear bundle (OCB) (Warren and Liberman 1989a, b), and all of the effects elicited by OCB shocks are attributable to MOC fibers (review: Guinan 1996). Third, many studies show sound-evoked effects on OAEs that are similar to the effects on neural responses (see Sect. 3.8.2), and OAE changes are mechanical changes that cannot be produced by LOC synapses. Finally, the LOC effects that have been measured to date show very slow changes (Groff and Liberman 2003), and these are much slower than any of the sound-evoked fast effects reviewed above. Overall, it seems highly likely that the fast sound-evoked efferent effects described so far are all due to MOC efferents.
In addition to the fast effects, slower effects due to contralateral sound have been reported. Lima da Costa et al. (1997) found that a contralateral broad-band noise reduced ipsilateral round-window noise (RWN) with fast and slow time courses. RWN near 1 kHz is dominated by small contributions from far-field potentials from the random firing of AN fibers (Dolan et al. 1990). With no ipsilateral sound, RWN is due to spontaneous AN activity (or AN activity from animal noise) and a reduction of AN spontaneous activity can be produced by MOC activation (Guinan and Gifford 1988b). The fast reduction of RWN found by Lima da Costa et al. was blocked by a gentamicin injection, which blocks MOC synapses, and seems likely to be a MOC effect. The slower reduction of RWN by contralateral sound was attributed to the MOC slow effect by Lima da Costa et al., but was not blocked by the same gentamicin dose that blocked the fast effect. Blocking at the same ACh concentration is expected because fast and slow effects are both produced by the same ACh synapses on OHCs (Sridhar et al. 1995). An alternative explanation is that the slow effect of Lima da Costa et al. is due to LOC efferents (Yoshida et al. 1999). A somewhat similar slow increase in RWN along with a slow increase in AN N1 and a slow decrease in DPOAEs, was found by Larsen and Liberman (2009). This constellation of changes indicates that MOC efferents are involved. Larsen and Liberman attributed the slow change to a centrally mediated slow increase in the MOC fast effect, rather than to a MOC slow effect. More work is needed to sort out the contributions of the various possible candidates for these slow sound-evoked efferent effects.
3.8.2 Sound-Elicited MOC Effects on Otoacoustic Emissions
The measurement of sound-elicited effects using OAEs has been one of the most productive ways of measuring MOC effects because it is noninvasive and can be done in humans. However, OAEs provide an indirect measure of cochlear mechanical responses and before considering results from them, we first consider the issues in their use.
Most measurements of MOC effects on OAEs used contralateral sound to elicit MOC activity because this is the easiest method. However, measurements can also be made with ipsilateral or bilateral elicitors if the two main problems with these are avoided. First, the high-level acoustic waveform of the ipsilateral elicitor can be canceled out by reversing sign of the elicitor on alternate presentations and averaging an even number of responses. The second problem, “two tone suppression”Footnote 2 produced by an ipsilateral elicitor, is more difficult. Two tone suppression is produced by energy in the elicitor that is near in frequency to the probe frequency and turns down the gain of the probe-frequency cochlear amplifier by bending OHC stereocilia into their nonlinear range (Geisler et al. 1990; Geisler 1992). There are two ways to get around this: (1) select ipsilateral elicitors that do not have energy near the probe frequency (e.g., notched noise, Backus and Guinan 2006; Lilaonitkul and Guinan 2009b), or (2) separate the effects in time using the difference in their decay rates, that is, two-tone suppression decays in a few milliseconds but MOC inhibition decays with a time constant of ∼100 ms (Guinan 1990). This second technique can be achieved by measuring in a “post elicitor window” that is after the two-tone suppression has died out but before the MOC inhibition has decayed away (Guinan et al. 2003). With the second technique, the MOC effect is measured while it is decaying and is not as large as it was during the elicitor. To make comparable measurements of ipsilateral and contralateral MOC effects, the same window has to be used for both.
There are several other issues that must be considered when using OAEs to measure MOC effects. First, the signal-to-noise ratio (S/N) must be adequate. Although this seems obvious, the S/N criterion has almost never been applied correctly in the literature. Most commonly, the S/N criterion has been applied to the OAE measurement. However, when measuring MOC effects, the “signal” of interest is the change in the OAE, not the OAE. To have an accurate measurement, the S/N of the change in the OAE must be adequate (e.g., 6 dB). A second consideration is to have an adequate number of alterations of elicitor-on vs. elicitor-off to remove any systematic drift. Third, there must be no middle-ear-muscle (MEM) contractions. MEM contractions interfere by changing sound transmission through the middle ear and by changing the impedance of the ear as seen by the acoustic source. Weak MEM contractions are not always shown by clinical MEM instruments (Feeney and Keefe 2001; Feeney et al. 2004). A more sensitive test for MEM contractions is the suppressed-OAE test (Lilaonitkul and Guinan 2009a, b). Finally, the sound used to evoke the OAE may also elicit unintentional MOC activity (Guinan et al. 2003). Such unintended MOC activity is certainly undesirable, but it is not known how much this changes measurements using contralateral elicitors.
MOC effects on OAEs in humans have been measured over a frequency range from ∼0.5 to 5 kHz with the largest effects often at 1–2 kHz (e.g., Collet et al. 1990; Moulin et al. 1993; Lilaonitkul and Guinan 2009b). In animals, MOC effects on OAEs have been measured up to 30 kHz in cats and over 60 kHz in bats (Guinan 1986; Henson et al. 1995). Considering that MOC innervation peaks in the basal half of the cochlea in animals (the pattern in humans is assumed to be similar), it might seem surprising that the largest MOC effects are often at 1–2 kHz. However, contralateral sound evokes the highest MOC firing rates at these frequencies (Liberman 1988 a). In addition, the change in OAEs may vary across frequency due to the processes by which OAEs are generated. Considering this, MOC effects vs. OAE probe frequency must be interpreted cautiously.
The threshold for MOC effects on OAEs is slightly (e.g., 10–15 dB) above the hearing threshold, and above that, MOC effects increase as the elicitor level increases (e.g., Collet et al. 1990; Ryan et al. 1991; Backus and Guinan 2006). Note, however, that the elicitor level range is limited by the need to avoid MEM activation, so it is not known if MOC effects saturate at higher elicitor levels or continue to increase. Presumably, the growth of MOC effects on OAEs with increases in sound level is due to, and provide a window on, the growth in MOC fiber activity.
The time course of MOC effects in humans is shown most clearly by ΔSFOAEs (Backus and Guinan 2006). For probes near 1 kHz, there is a delay of ∼25 ms from sound onset, or offset, to the beginning of the change in the ΔSFOAE; ∼20 ms of this is the delay to the beginning of the change in cochlear amplification, and ∼5 ms is for this change to be carried backward in the cochlea to the ear canal. After the initial delay, there is a monotonic rise in the ΔSFOAE with an overall time constant of a few hundred ms, and after the offset delay there is a decrease in the ΔSFOAE that is typically faster than the rise. In some subjects the ΔSFOAE shows a small, short overshoot lasting a few tens of ms. The time course of MOC effects on TEOAEs has not been adequately demonstrated because of the noncontinuous nature of TEOAEs. However, this time course should be the same as for SFOAEs. In contrast to the above, MOC effects on DPOAEs can show very complicated time patterns, presumably because of the complicated mixing of the two different sources of DPOAEs (Muller et al. 2005). Overall, the OAE data indicate that the MOC fast effect is not fast enough to significantly change the hearing of speech on a syllable-by-syllable basis.
During continuous or intermittent contralateral noise, MOC effects are maintained for many minutes with little evidence of adaptation but with some evidence for a slow increase in the effect (Giraud et al. 1997a; Lima da Costa et al. 1997; Larsen and Liberman 2009; Zyl et al. 2009). After a long stimulation evoking MOC effects, there is sometimes a rebound enhancement lasting seconds (Zyl et al. 2009). This enhancement may be a rebound from a MOC slow effect, or may arise from an entirely different cause (see Maison et al. 2007).
3.8.2.1 MOC Reflex Tuning
OAE measurements in awake humans show there is tuning in the MOC reflex, but the largest MOC effects are not always centered on the elicitor frequency. Measurements of the MOC effects produced by 1/3 octave noise-band elicitors using DPOAEs or tone-pip TEOAEs show tuning in the MOC reflex for probes near 1 and 2 kHz but not 3 and 4 kHz (Veuillet et al. 1991; Chéry-Croze et al. 1993). Both studies concluded that the largest MOC effects were when the elicitor noise band was centered on the probe frequency, but both studies also show cases where elicitor bands below the probe frequency had larger effects than those at the probe frequency. Lilaonitkul and Guinan measured the MOC effects produced by 60 dB SPL tones or half-octave noise-bands using SFOAEs from probes at 0.5, 1, and 4 kHz. The change in SFOAEs (ΔSFOAE) for probes near 1 kHz show broad, skewed tuning with the most effective elicitor frequencies 0.5–1 octave below the probe frequency (Fig. 3.9b) (Lilaonitkul and Guinan 2009a). For 0.5-kHz probes, there was also broad tuning but with a skew in the opposite direction, whereas for 4-kHz probes, the tuning had a narrower peak and a broad low-frequency activation region (Lilaonitkul and Guinan, unpublished). These ΔSFOAE tuning patterns are similar to the effects of contralateral sound on AN fibers found by Warren and Liberman (1989b) (see Fig. 3.9 and Sect. 3.7) except that the human pattern is shifted down by about an octave from the cat pattern (presumably because the human hearing range is about an octave lower than cats). In contrast, a different SFOAE metric, the change in the magnitude of the SFOAE (SFOAEmoc) showed MOC effects that were much narrower and centered on the probe frequency (Lilaonitkul and Guinan, unpublished). The correspondence between ΔSFOAE and the neural data gives support to the ΔSFOAE metric for MOC effects in humans. However, the more centered results found with TEOAEs, DPOAEs and SFOAEmoc indicates that we still do not fully understand the ways that MOC activity affects OAEs and the tuning of the MOC reflexes. Also, the above experiments were done with passive listening; perhaps in the context of a task such as identifying a 1-kHz signal in noise, the MOC effect might be more focused on 1 kHz.
3.8.2.2 MOC Reflex Amplitude as a Function of Elicitor Bandwidth
MOC effects on OAEs from elicitors of various bandwidths show little evidence of a frequency selective reflex. MOC effects measured with elicitors at a fixed level, but with increasing bandwidths, show that MOC activation increases as bandwidth increases up to 4–6.7 octaves (Fig. 3.10) (Maison et al. 2000; Lilaonitkul and Guinan 2009b). Because the elicitor SPL was held constant as bandwidth increased, the spectral level near the probe frequency decreased. However, despite the resulting decrease in MOC activation in frequency regions near the probe, the overall activation does not decrease, even at the widest bandwidths. This indicates that the increased activation from frequency regions remote from the probe frequency must have compensated for the activation lost near the probe frequency. These experiments indicate that the MOC reflex must integrate activation from practically the whole cochlea.
MOC inhibition grows as elicitor noise bandwidth is increased despite the overall noise level being held constant at 60 dB SPL. MOC effect is the change in the SFOAE normalized by the magnitude of the SFOAE. BBN broad-band noise (0.1–10 kHz, or 6.67 octaves). Error bars are standard errors of the mean. Horizontal lines: Solid = noise-floor mean, dotted = 1 std. above the noise mean (adapted with permission from Lilaonitkul and Guinan 2009b)
The wide frequency integration profile of the MOC reflex shown by the bandwidth experiments helps to explain why a wide variety of experiments found that the most potent elicitor of MOC effects is broad band noise (Ryan et al. 1991; Veuillet et al. 1991; Chéry-Croze et al. 1993; Norman and Thornton 1993; Maison et al. 2000; Guinan et al. 2003). Potency in eliciting MOC activity appears to increase in the following order: single tones, two tones (DPOAE primaries), repetitive tone pips, repetitive clicks and broad-band noise (Guinan et al. 2003). Note that the first four of these sounds are used to evoke OAEs, but they also elicit MOC activity.
White noise that is amplitude modulated (AM) at 100 Hz has been reported to produce larger MOC activations than unmodulated noise (Maison et al. 1999). Our experiments with AM noise show an increase for some subjects but not others, and it is unclear if these are two separate groups or a continuum (Backus and Guinan 2004). The extent to which AM might be used to increase MOC effects in humans is not yet clear.
3.8.2.3 MOC Reflex Laterality
The ratio of ipsilateral/contralateral MOC fibers is approximately 2:1 in small mammals but is unknown in humans. This ratio was derived largely from labeling the MOC neurons that project to one cochlea. Such data show that twice as many MOC fibers originate from the contralateral side as from the ipsilateral side (reviewed by Warr 1992) (keep in mind the innervation pattern of Fig. 3.2). Comparable experiments cannot be done on humans, but relevant information might be obtained by comparing the effects of ipsilateral vs. contralateral sound on OAEs. As shown in Fig. 3.10, binaural sound produces the largest MOC effects, often twice as large as the ipsilateral or contralateral effects alone. Monaural broad-band noise elicits ipsilateral and contralateral effects of very similar amplitudes (Fig. 3.10, the points labeled BBN). In contrast, for narrow-band noise, ipsilateral elicitors produce changes that are approximately twice as large as contralateral elicitors. The ratio of ipsilateral/ contralateral MOC fibers cannot change with elicitor bandwidth. These results show that the laterality of MOC effects is strongly influenced by central processes that change the activity in crossed vs. uncrossed MOC fibers according to the bandwidth of the stimulus.
One theory to explain the change in MOC laterality with elicitor bandwidth is that having the reflexes be equal when they produce large effects prevents the MOC effects from producing interaural time differences that disrupt binaural hearing. 60 dB SPL contralateral noise produces low-frequency cochlear phase advances in the 0.5 ms range (Francis and Guinan 2010), and this phase change would have a profound effect on the interaural time cue for binaural localization if it were not binaurally balanced. In cats, a species that hears at low enough frequencies to use interaural time differences, the crossed/uncrossed MOC fiber ratio is a function of the cochlear frequency region, that is, the overall ipsi/contra reflex ratio is 2/1, but at low frequencies the ratio is near 1:1 (Guinan et al. 1984). In contrast the ratio stays near 2:1 across frequency in the mouse, a species that does not have significant low-frequency hearing (Maison et al. 2003). These patterns are consistent with the hypothesis that MOC reflex equality at low frequencies evolved to enable good binaural localization at low frequencies.
3.8.2.4 MOC Reflex Strength
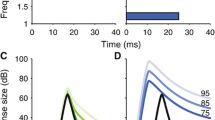
Most studies of MOC effects in humans used averages across groups of subjects. Such studies show that MOC efferents produced a change across the group as a whole, but not whether the change was present in each individual. To date, only one study applied an adequate S/N criterion to insure that the MOC effect on each subject was significantly different from the measurement noise (Backus and Guinan 2007). This study measured normalized ΔSFOAEs elicited by 60 dB SPL contralateral sound and found more variation between measurements at nearby frequencies than could be accounted for by the expected variation across frequency of the MOC effect. One explanation is that the MOC effect changes the weighting of the cochlear irregularities involved in the coherent reflection that produces SFOAEs (Backus and Guinan 2007). To get consistent measures of MOC strength, MOC effects had to be averaged across measurements at several nearby frequencies. The resulting MOC strength distribution is shown in Fig. 3.11. The distribution is approximately Gaussian with a mean of 36% (∼3 dB) and a range of a factor of 5 between subjects with weak effects and subjects with strong effects. Thus, whatever benefits MOC reflexes provide, these benefits must vary considerably across individuals.
A histogram of the MOC strengths at 1 kHz for 25 subjects (mean = 36.6%, SD = 11.7%). MOC activation from 60 dB SPL contralateral wide-band noise. MOC effect was measured by the SFOAE change. The curve is a Gaussian fit to the data (adapted with permission from Backus and Guinan 2007)
3.8.3 Descending Influences on MOC Acoustic Reflex Properties in Humans
Reviews of the anatomy of the descending auditory system and its influences within the CNS are given in Schofield (Chap. 9), Robertson and Mulders (Chap. 10), and Suga et al. (Chap. 11). Here we concentrate on descending influences on MOC neurons, particularly in humans. There are three areas that appear to show descending influences: (1) MOC reflex modulation by attention, (2) right-vs.-left ear differences, and (3) auditory learning.
There is an extensive literature on the effects of attention on cochlear processes. In one class of experiments, attention to a visual task is alternated with attention to an auditory task (reviews: Guinan 1996; Delano et al. 2007). Numerous experiments of this kind show changes in cochlear potentials and/or OAEs that indicate there is increased MOC activation during the visual task. However, such changes were not seen in all cases so perhaps the MOC activation is present in some subjects but not others. In other experiments, comparisons were made across auditory tasks, or MOC effects were elicited by contralateral noise (e.g., Giard et al. 1994; Michie et al. 1996; Maison et al. 2001; de Boer and Thornton 2007). A theme that may explain the diverse results from these experiments is that MOC efferents are activated for tasks when the MOC activity produces a benefit (e.g., reducing a distracting sound during a visual task, or aiding in difficult signal-in-noise tasks) but not when there is no benefit (e.g., doing an easy auditory task while ignoring a visual stimulus, or counting tone pips embedded in a click train).
An interesting attentional experiment was conducted by Scharf et al. (1997) using the probe-signal method, which focuses on subject detection of an “odd-ball” stimulus presented in a small fraction of the trials. Using a signal just above threshold in a background noise, Scharf et al. found that on-frequency signals were heard, but odd-ball signals that differed in frequency by 5% were not heard. Scharf et al. concluded that the off-frequency tone was MOC inhibited because the effect was not present in subjects with efferents cut for medical reasons. More recently, Tan et al. (2008) performed a set of similar experiments and drew an opposite conclusion. Tan et al. concluded that MOC efferents are activated by the tone cue used in the probe-signal method and produce a MOC benefit at the cued on-frequency, whereas targets at nearby off-frequencies get little or no MOC benefit and so their perception is worse than tones at the cued frequency.
There are right–left asymmetries in various aspects of the peripheral auditory system. There are small handedness and gender differences in OAE amplitudes, and in MOC effects on OAEs (e.g., Aidan et al. 1997; Khalfa et al. 1998; Morlet et al. 1999; Sininger and Cone-Wesson 2004). Benzodiazepines reduce contralaterally elicited MOC inhibition in the right ear, but not the left, according to Morand et al. (2001); Morand-Villeneuve et al. (2005), who suggested that this is because there are more benzodiazepine receptors in the left cortex than the right. At the cortical level, right–left asymmetries are well established. An attractive hypothesis is that these cortical right–left differences, through descending projections, produce MOC asymmetries and these produce the OAE asymmetries (see Khalfa et al. 2001).
A variety of evidence suggests that auditory training can have an effect on MOC reflex strength. Musicians have stronger MOC reflexes than people who never had musical training (Perrot et al. 1999). In a study in children with reading disabilities, certain children showed an absence of the asymmetry favoring the right ear that is found in average-reading children (Veuillet et al. 2007). After auditory training that improved their score in a speech task, these children’s MOC function changed and their asymmetry became closer to normal. In a study using adults, subjects were given a 5-day training regimen on a speech-discrimination-in-noise task, and their MOC activation was measured on each day (de Boer and Thornton 2008). Subjects who originally had weaker MOC activation showed greater improvement in the speech-in-noise task and also showed increases in MOC activation so that after the training their MOC activation was similar to the subjects who originally had larger MOC activation (Fig. 3.12). More work is needed to show whether the improvement in perception is brought about by the increased MOC activity, or whether the increased MOC activity is simply a byproduct of other central changes. In either case, the work suggests that a test for MOC strength might predict subjects who would benefit from auditory training.
MOC reflex activity and signal-in-noise detection change over 5 days of auditory training in some subjects, but not others. Subjects were divided into learners and nonlearners based their learning over 5 days. The discrimination thresholds for these two groups are shown in (a). On day 1, nonlearners had significantly lower MOC shifts than learners, but training erased this difference (b). MOC shift was induced by 40 dB SL contralateral noise. MOC shifts were measured by click-evoked TEOAEs (adapted with permission from de Boer and Thornton 2008)
3.9 MOC Function in Hearing
There are two areas for which there is good evidence for a MOC function in hearing: aiding discrimination of signals in noise, and preventing or reducing acoustic trauma. In a third area, attention and learning, the evidence that the measured effects are primarily due to efferents is not clear, but there may be an important MOC function in this area as well.
3.9.1 MOC Activity Changes the Dynamic Range of Hearing and Thereby Increases the Discriminability of Transients in Background Noise
The hypothesis that MOC efferents aid in the discrimination of signals in noise originated from animal results showing that when AN output dynamic range is reduced by background noise, the dynamic range can be partially restored by MOC activation (see Fig. 3.5 and Sect. 3.4). There have been two main ways in which this “MOC unmasking” hypothesis has been tested: (1) by cutting MOC fibers and (2) by looking for correlations between psychophysical performance and MOC strength.
In many animal studies, MOC fibers were cut and performance deficits were investigated (review: Guinan 1996). In a noisy background, lesions of MOC efferents in cats reduce their performance in speech discrimination, high-frequency tone discrimination, and sound localization (May et al. 1995; Hienz et al. 1998; May et al. 2004). In contrast, functional MOC lesions in mice produced by altering the ACh receptor did not reveal any deficit in detecting signals in noise (May et al. 2002). This surprising result may be because the conditions tested were not right for mice. In humans, surgical cuts of the vestibular nerve (MOC fibers exit the brain in this nerve) revealed a MOC benefit in detecting speech in noise in some patients, but not others (Zeng and Shannon 1994; Zeng et al. 2000; Giraud et al. 1997b). Perhaps the inconsistent results were because these lesions interrupt MOC fibers to varying degrees (Zeng and Shannon 1994; Giraud et al. 1995; Chays et al. 2003). Scharf et al. (1997) performed signal-in-noise tests on humans with vestibular nerve cuts without finding a deficit, but the tests used conditions for which no MOC benefit is expected (tones and noise that were both continuous or both equal-duration bursts; see Sect. 3.4).
Measurements across subjects of psychophysical performance and MOC activation, with MOC activation sometimes increased by contralateral noise, have found correlations between performance and MOC activation that indicate both MOC benefits and handicaps (e.g., Micheyl and Collet 1996; Micheyl et al. 1997; Giraud et al. 1997b; Kumar and Vanaja 2004). Using the correct stimulus conditions appears to be the key to finding a MOC benefit. Subjects with strong MOC reflexes are better than those with weak reflexes in detecting signals in noise when the S/N is moderate (Fig. 3.13) but not near threshold or with high-level noise (Kumar and Vanaja 2004). In studies that found no correlation between MOC activation and subject ability to detect a signal in noise (e.g., Wagner et al. 2008; Mukari and Mamat 2008), the negative results may be due to the methods used (e.g., MOC-induced changes in human DPOAEs are poor MOC metrics; see earlier) and/or to using the wrong stimulus conditions (e.g., signals too close to threshold). Overall, the data support the hypothesis that one function of MOC efferents is to aid in the detection of signals in noise, although the conditions that produce this benefit are not well documented.
A positive correlation across subjects between the improvement of speech perception in noise and MOC inhibition of transient evoked otoacoustic emissions (TEOAEs), which indicates that MOC activity provides a benefit in the detection of signals in noise. Correlation coefficient, r = 0.48, p = 0.001. MOC inhibition elicited by 30 dB SL contralateral noise. Speech at 50 dB HL and 10 dB S/N re ipsilateral noise (adapted with permission from Kumar and Vanaja 2004)
3.9.2 MOC Activity Helps to Protect Against Acoustic Trauma
Data from many animal experiments leave little doubt that MOC activity helps to prevent both temporary and permanent threshold shifts (TTS and PTS) due to traumatic sounds (review: Rajan 2000). We highlight one study, a prospective PTS study in which animals were exposed to traumatizing sounds after being classified into those with weak, average, and strong MOC reflexes using OAE measurements (Maison and Liberman 2000). The animals with strong MOC reflexes had the least PTS and the animals with weak MOC reflexes had the most PTS (Fig. 3.14). This result indicates that MOC activity reduces PTS, and also that an OAE-based test may show which subjects are susceptible to acoustic trauma.
MOC reflex strength predicts acoustic trauma. Twelve guinea pigs were grouped by the preexposure strength of their MOC reflexes. The noise-induced permanent threshold shifts (PTSs) measured from AN compound action potentials were least in the group with the strongest reflexes. The results imply that MOC reflexes reduce PTS. Error bars indicate SEM (adapted with permission from Maison and Liberman 2000)
3.9.3 Possible Roles of MOC Activity in Attention and Learning
Changes in MOC activation during attention and learning were shown in Sect. 3.8.3. At least for some subjects and conditions, MOC activation brought about by attention helps to reduce distracting sounds or aids in discriminating the attended sound when it is in a background noise. More data are needed to understand the extent to which attention modifies the function of the MOC acoustic reflex in everyday hearing.
Attention is important in learning and may bring about changes in MOC activity that are important for learning a task. The de Boer and Thornton (2008) results (see Sect. 3.8.3) suggest that some normal subjects had to learn to turn on their MOC efferents so that they could detect a signal in noise as well as was done by other subjects who required no training (Fig. 3.12). Learning to control MOC efferents seems to have been the key factor in this learning experiment. On the other hand, the change in MOC activation and gaining of normal right–left differences in the reading impaired children of Veuillet et al. (1996) seems more likely to be a byproduct of some other central change. There may be an important MOC role in learning, but more work is needed before it is understood.
3.10 LOC Physiology and Function
Before we review LOC physiology, we briefly review LOC anatomy. There are two types of LOC neurons based on their position around the lateral superior olivary nucleus: intrinsic and shell neurons. In the cochlea, these appear to correspond to unidirectional fibers and bidirectional fibers, respectively (Brown 1987; Warr et al. 1997). In the mouse, intrinsic-unidirectional neurons are cholinergic and shell-bidirectional neurons are dopaminergic (Darrow et al. 2006b); whether this holds for other species is unknown. A wide variety of other neurotransmitters and neuroactive substances have been found in LOC neurons. For further details on LOC anatomy and neurochemistry, see Brown (Chap. 2) and Sewell (Chap. 4).
3.10.1 LOC Effects in the Cochlea
The fibers of both LOC groups are unmyelinated, and neither has been recorded from or stimulated. However, indirect activation of LOC neurons has been achieved by electrical stimulation in the inferior colliculus (IC). Depending on where the IC is stimulated, AN responses can be enhanced or reduced by effects that are attributable to LOC efferents (Groff and Liberman 2003). Considering that the two LOC groups have different neurotransmitter contents, an attractive hypothesis is that excitation is produced when one LOC group is activated and inhibition when the other group is activated. When LOC efferents are lesioned, AN activity is depressed (Liberman 1990; Le Prell et al. 2003) or enhanced (Darrow et al. 2006a). The difference in results might be explained by the lesions in different studies affecting the two LOC groups to different extents.
All of the effects attributable to LOC efferents are very slow (Groff and Liberman 2003). This is not surprising because these fibers are unmyelinated and have slow conduction velocities. In Groff and Liberman experiments, the LOC effects decayed with time constants (τ’s) of minutes (the onset time courses of LOC effects were obscured by shock artifacts and MOC effects). Thus, efferent effects occur on three time scales: MOC fast effects with τ’s of ∼100 ms, MOC slow effects with τ’s of 10’s of seconds, and LOC effects with τ’s of minutes.
3.10.2 LOC Response to Sound
LOC neurons receive innervation from the ipsilateral ventral cochlear nucleus and this innervation could form the basis of an ipsilateral LOC acoustic reflex (Thompson and Thompson 1991). There are no known inputs to LOC neurons from the contralateral side. However, most neurons in the vicinity of LOC neurons are excited by sound in the ipsilateral ear and inhibited by sound in the contralateral ear (Guinan et al. 1972), and MOC neurons might be similar. There are no recording from LOC neurons in intact preparations, so there are no definitive data on LOC responses to sound. LOC acoustic reflexes, if they exist, can be expected to act on a slow (minutes) time scale.
3.10.3 LOC Function in Hearing
LOC fibers synapse directly on AN fibers and can change their firing patterns. The wealth of neurotransmitters in the two types of LOC fibers suggests that LOC fibers have multiple functions; however, we know relatively little about these functions.
LOC fibers appear to reduce the acoustic-trauma-induced excitotoxic effect of the IHC neurotransmitter, that is, they reduce the swelling and bursting of AN fibers produced by over stimulating IHCs (e.g., Ruel et al. 2001). See Groff and Liberman (2003) for a review of the evidence that LOC fibers reduce acoustic trauma in this and other ways.
Guinan (1996) suggested that LOC neurons act to achieve balance in the outputs from the two ears to enable binaural localization based on interaural level differences. Support for this hypothesis comes from LSO lesions that produced an imbalance in ABR responses from the right ear vs. the left ear (Darrow et al. 2006a). However, more evidence is needed before this hypothesis can be considered as established.
3.11 Summary and Future Directions
Most MOC effects are classical effects owing to turning down the gain of cochlear amplified traveling waves. At high sound levels, the traveling wave interacts with a more linear mechanical response and produces most of the nonclassic MOC effects. Click responses in the apical half of the cochlea show there is an additional motion that is MOC inhibited and that produces the ANIP response. Work is needed to determine the exact nature of the ANIP motion. Another unknown is what tone response corresponds to the ANIP click response. One possibility is TC side lobes (Fig. 3.3f). Overall, progress in understanding MOC effects requires a better understanding of the multiple motions involved in cochlear mechanics.
In addition to MOC fast effects, there is a MOC slow effect that has been seen only at high frequencies in guinea pigs. Its presence and frequency range in other species remains to be determined. It is also unknown whether the MOC slow effect originates from an OHC stiffness change and whether it has any relationship to protection from TTS.
Sound in either ear evokes MOC activity and produces MOC effects. More work is needed to show the single-fiber MOC responses evoked by ipsilateral, contralateral and bilateral noise of various bandwidths. Measurements in awake animals are necessary for a full understanding of MOC responses to sound.
Aiding the detection and discrimination of signals in noise is probably the most important MOC function. Physiological mechanisms capable of producing this benefit have been demonstrated, but we do not yet understand how powerful these mechanisms are or the conditions under which MOC activity provides a benefit and when it does not. Although it seems likely that MOC activity plays an important role in the signal-detection-in-noise temporal effect (i.e., “overshoot”), the MOC role needs to be more clearly shown. For instance, the fact that narrow-band noise centered on the signal frequency produces little temporal effect may be because narrow-band noise elicits little MOC effect (Fig. 3.10).
A wide variety of data indicate that both MOC and LOC activity reduce acoustic trauma, but the mechanisms for this are largely unknown. Small reductions in sound level can greatly reduce both TTS and PTS, so one possibility is that a small MOC-induced reduction in cochlear mechanical motion may be involved, but such reductions have not yet been demonstrated at traumatic sound levels. Although dopamine released by LOC activity may reduce TTS by blocking excitotoxity from too much transmitter released by IHCs (Ruel et al. 2001), the role of the many other neuroactive substances released by LOC synapses needs to be elucidated, for TTS, PTS, and on neural signaling. Prospective tests are needed to determine whether a MOC strength test can determine whether a person is particularly susceptible to noise damage. MOC reflex testing needs to be done with an adequate S/N whenever the results are to apply to a single individual.
Finally, at least under some circumstances, MOC activation changes during learning. Whether the learning causes the change in MOC activation or is caused by MOC activation needs to be determined. Also, work is needed to show whether MOC activation tests can predict which subjects will be aided by auditory training.
Notes
- 1.
- 2.
In the literature the term “contralateral suppression” is often used to mean the effect of MOC activity elicited by contralateral sound. We avoid this term because it does not distinguish between MOC inhibition elicited by contralateral sound and two-tone suppression produced by acoustic crosstalk from the contralateral to the ipsilateral ear. Instead, we use the term “contralateral inhibition” or “contralateral MOC inhibition”.
References
Abdala C, Mishra SK, Williams TL (2009) Considering distortion product otoacoustic emission fine structure in measurements of the medial olivocochlear reflex. J Acoust Soc Am 125:1584–1594
Aidan D, Lestang P, Avan P, Bonfils P (1997) Characteristics of transient-evoked otoacoustic emissions (TEOES) in neonates. Acta Otolaryngol 117:25–30
Aran JM, Pajor AM, de Sauvage RC, Erre JP (2000) Role of the efferent medial olivocochlear system in contralateral masking and binaural interactions: an electrophysiological study in guinea pigs. Audiology 39:311–321
Backus BC, Guinan JJ Jr (2004) The efficacy of AM noise for activating the human MOC reflex measured using SFOAEs. Assoc Res Otolaryngol Abstr 27:535
Backus BC, Guinan JJ Jr (2006) Time course of the human medial olivocochlear reflex. J Acoust Soc Am 119:2889–2904
Backus BC, Guinan JJ Jr (2007) Measurement of the distribution of medial olivocochlear acoustic reflex strengths across normal-hearing individuals via otoacoustic emissions. J Assoc Res Otolaryngol 8:484–496
Bassim MK, Miller RL, Buss E, Smith DW (2003) Rapid adaptation of the 2f1–f2 DPOAE in humans: binaural and contralateral stimulation effects. Hear Res 182:140–152
Boyev KP, Liberman MC, Brown MC (2002) Effects of anesthesia on efferent-mediated adaptation of the DPOAE. J Assoc Res Otolaryngol 3:362–373
Brown MC (1987) Morphology of labeled efferent fibers in the guinea pig cochlea. J Comp Neurol 260:605–618
Brown MC (1989) Morphology and response properties of single olivocochlear fibers in the guinea pig. Hear Res 40:93–110
Brown MC (2001) Response adaptation of medial olivocochlear neurons is minimal. J Neurophysiol 86:2381–2392
Brown MC (2002) Cochear projections of single medial olivocochlear (MOC) axons in the guinea pig. Asso Res Otolaryngol Abstr 25:310
Brown MC, Kujawa SG, Duca ML (1998a) Single olivocochlear neurons in the guinea pig. I. Binaural facilitation of responses to high-level noise. J Neurophysiol 79:3077–3087
Brown MC, Kujawa SG, Liberman MC (1998b) Single olivocochlear neurons in the guinea pig. II. Response plasticity due to noise conditioning. J Neurophysiol 79:3088–3097
Brown MC, de Venecia RK, Guinan JJ (2003) Responses of medial olivocochlear neurons: specifying the central pathways of the medial olivocochlear reflex. Exp Brain Res 153:491–498
Buño W Jr (1978) Auditory nerve fiber activity influenced by contralateral ear sound stimulation. Exp Neurol 59:62–74
Cai Y, Geisler CD (1996) Suppression in auditory-nerve fibers of cats using low-side suppressors. II. Effect of spontaneous rates. Hear Res 96:113–125
Chays A, Maison S, Robaglia-Schlupp A, Cau P, Broder L, Magnan J (2003) Are we sectioning the cochlear efferent system during vestibular neurotomy? Rev Laryngol Otol Rhinol (Bord) 124:53–58
Chéry-Croze A, Moulin A, Collet L (1993) Effect of contralateral sound stimulation on the distortion product 2f1–f2 in humans: evidence of a frequency specificity. Hear Res 68:53–58
Cody AR, Johnstone BM (1982) Acoustically evoked activity of single efferent neurons in the guinea pig cochlea. J Acoust Soc Am 72:280–282
Collet L, Kemp DT, Veuillet E, Duclaux R, Moulin A, Morgon A (1990) Effect of contralateral auditory stimuli on active cochlear micro-mechanical properties in human subjects. Hear Res 43:251–262
Cooper NP, Guinan JJ Jr (2003) Separate mechanical processes underlie fast and slow effects of medial olivocochlear efferent activity. J Physiol 548:307–312
Cooper NP, Guinan JJ Jr (2006a) Efferent-mediated control of basilar membrane motion. J Physiol 576:49–54
Cooper NP, Guinan JJ Jr (2006b) The dynamics of medial olivocochlear efferent fast effects on basilar membrane motion. Assoc Res Otolaryngol Abstr 30:273
Cooper NP, Kemp DT (2009) Concepts and challenges in the biophysics of hearing. World Scientific, Singapore
Cooper NP, Rhode WS (1996) Fast travelling waves, slow travelling waves and their interactions in experimental studies of apical cochlear mechanics. Audit Neurosci 2:289–299
Dallos P, He DZ, Lin X, Sziklai I, Mehta S, Evans BN (1997) Acetylcholine, outer hair cell electromotility, and the cochlear amplifier. J Neurosci 17:2212–2226
Dallos P, Wu X, Cheatham MA, Gao J, Zheng J, Anderson CT, Jia S, Wang X, Cheng WH, Sengupta S, He DZ, Zuo J (2008) Prestin-based outer hair cell motility is necessary for mammalian cochlear amplification. Neuron 58:333–339
Darrow KN, Maison SF, Liberman MC (2006a) Cochlear efferent feedback balances interaural sensitivity. Nat Neurosci 9:1474–1476
Darrow KN, Simons EJ, Dodds L, Liberman MC (2006b) Dopaminergic innervation of the mouse inner ear: evidence for a separate cytochemical group of cochlear efferent fibers. J Comp Neurol 498:403–414
de Boer J, Thornton AR (2007) Effect of subject task on contralateral suppression of click evoked otoacoustic emissions. Hear Res 233:117–123
de Boer J, Thornton AR (2008) Neural correlates of perceptual learning in the auditory brainstem: efferent activity predicts and reflects improvement at a speech-in-noise discrimination task. J Neurosci 28:4929–4937
de Venecia RK, Liberman MC, Guinan JJ Jr, Brown MC (2005) Medial olivocochlear reflex interneurons are located in the posteroventral cochlear nucleus: a kainic acid lesion study in guinea pigs. J Comp Neurol 487:345–360
Delano PH, Elgueda D, Hamame CM, Robles L (2007) Selective attention to visual stimuli reduces cochlear sensitivity in chinchillas. J Neurosci 27:4146–4153
Desmedt JE (1962) Auditory-evoked potentials from cochlea to cortex as influenced by activation of the efferent olivocochlear bundle. J Acoust Soc Am 34:1478–1496
Dolan DF, Nuttall AL, Avinash G (1990) Asynchronous neural activity recorded from the round window. J Acoust Soc Am 87:2621–2627
Dolan DF, Guo MH, Nuttall AL (1997) Frequency-dependent enhancement of basilar membrane velocity during olivocochlear bundle stimulation. J Acoust Soc Am 102:3587–3596
Elgoyhen AB, Johnson DS, Boulter J, Vetter DE, Heinemann S (1994) Alpha 9: an acetylcholine receptor with novel pharmacological properties expressed in rat cochlear hair cells. Cell 79:705–715
Elgoyhen AB, Vetter DE, Katz E, Rothlin CV, Heinemann SF, Boulter J (2001) Alpha10: a determinant of nicotinic cholinergic receptor function in mammalian vestibular and cochlear mechanosensory hair cells. Proc Natl Acad Sci USA 98:3501–3506
Fahey PF, Stagner BB, Martin GK (2008) Source of level dependent minima in rabbit distortion product otoacoustic emissions. J Acoust Soc Am 124:3694–3707
Feeney MP, Keefe DH (2001) Estimating the acoustic reflex threshold from wideband measures of reflectance, admittance, and power. Ear Hear 22:316–332
Feeney MP, Keefe DH, Sanford CA (2004) Wideband reflectance measures of the ipsilateral acoustic stapedius reflex threshold. Ear Hear 25:421–430
Fex J (1959) Augmentation of cochlear microphonic by stimulation of efferent fibers to the cochlea. Acta Otolaryngol 50:540–541
Fex J (1962) Auditory activity in centrifugal and centripetal cochlear fibers in cat. Acta Physiol Scand 55:2–68
Fex J (1965) Auditory activity in the uncrossed centrifugal cochlear fibers in cat. A study of a feedback system, II. Acta Physiol Scand 64:43–57
Folsom RC, Owsley RM (1987) N1 action potentials in humans. Influence of simultaneous contralateral stimulation. Acta Otolaryngol (Stockh) 103:262–265
Francis NA, Guinan JJ Jr (2010) Acoustic stimulation of human medial olivocochlear efferents reduces stimulus-frequency and click-evoked otoacoustic emission delays: Implications for cochlear filter bandwidths. Hear Res 267:36–45
Fuchs PA (1996) Synaptic transmission at vertebrate hair cells. Curr Opin Neurobiol 6:514–519
Galambos R (1956) Suppression of auditory activity by stimulation of efferent fibers to the cochlea. J Neurophysiol 19:424–437
Geisler CD (1992) Two-tone suppression by a saturating feedback model of the cochlear partition. Hear Res 63:203–210
Geisler CD, Yates GK, Patuzzi RB, Johnston BM (1990) Saturation of outer hair cell receptor currents causes two-tone suppression. Hear Res 44:241–256
Ghaffari R, Aranyosi AJ, Freeman DM (2007) Longitudinally propagating traveling waves of the mammalian tectorial membrane. Proc Natl Acad Sci USA 104:16510–16515
Giard M-H, Collet L, Bouchet P, Pernier J (1994) Auditory selective attention in the human cochlea. Brain Res 633:353–356
Gifford ML, Guinan JJ Jr (1983) Effects of crossed-olivocochlear-bundle stimulation on cat auditory nerve fiber responses to tones. J Acoust Soc Am 74:115–123
Gifford ML, Guinan JJ Jr (1987) Effects of electrical stimulation of medial olivocochlear neurons on ipsilateral and contralateral cochlear responses. Hear Res 29:179–194
Giraud AL, Collet L, Chery-Croze S, Magnan J, Chays A (1995) Evidence of a medial olivocochlear involvement in contralateral suppression of otoacoustic emissions in humans. Brain Res 705:15–23
Giraud AL, Collet L, Chery-Croze S (1997a) Suppression of otoacoustic emission is unchanged after several minutes of contralateral acoustic stimulation. Hear Res 109:78–82
Giraud AL, Garnier S, Micheyl C, Lina G, Chays A, Chery Croze S (1997b) Auditory efferents involved in speech-in-noise intelligibility. Neuroreport 8:1779–1783
Groff JA, Liberman MC (2003) Modulation of cochlear afferent response by the lateral olivocochlear system: activation via electrical stimulation of the inferior colliculus. J Neurophysiol 90:3178–3200
Guinan JJ Jr (1986) Effect of efferent neural activity on cochlear mechanics. Scand Audiol Suppl 25:53–62
Guinan JJ Jr (1990) Changes in stimulus frequency otoacoustic emissions produced by two-tone suppression and efferent stimulation in cats. In: Dallos P, Geisler CD, Matthews JW, Steele CR (eds) Mechanics and biophysics of hearing. Springer, New York, pp 170–177
Guinan JJ Jr (1996) The physiology of olivocochlear efferents. In: Dallos PJ, Popper AN, Fay RR (eds) The cochlea. Springer, New York, pp 435–502
Guinan JJ Jr (1997) Efferent inhibition as a function of efferent stimulation parameters and sound frequency: testing the OHC-shunt hypothesis. In: Lewis ER, Long GR, Lyon RF, Narins PM, Steele CR, Hecht-Poinar E (eds) Diversity in auditory mechanics. World Scientific, Singapore, pp 368–375
Guinan JJ Jr, Cooper NP (2003) Fast effects of efferent stimulation on basilar membrane motion. In: Gummer AW, Dalhoff E, Nowotny M, Scherer MP (eds) The biophysics of the cochlea: molecules to models. World Scientific, Singapore, pp 245–251
Guinan JJ Jr, Cooper NP (2008) Medial olivocochlear efferent inhibition of basilar-membrane responses to clicks: evidence for two modes of cochlear mechanical excitation. J Acoust Soc Am 124:1080–1092
Guinan JJ Jr, Gifford ML (1988a) Effects of electrical stimulation of efferent olivocochlear neurons on cat auditory-nerve fibers. I. Rate-level functions. Hear Res 33:97–114
Guinan JJ Jr, Gifford ML (1988b) Effects of electrical stimulation of efferent olivocochlear neurons on cat auditory-nerve fibers. II. Spontaneous rate. Hear Res 33:115–128
Guinan JJ Jr, Gifford ML (1988c) Effects of electrical stimulation of efferent olivocochlear neurons on cat auditory-nerve fibers. III. Tuning curves and thresholds at CF. Hear Res 37:29–46
Guinan JJ Jr, Stankovic KM (1996) Medial efferent inhibition produces the largest equivalent attenuations at moderate to high sound levels in cat auditory-nerve fibers. J Acoust Soc Am 100:1680–1690
Guinan JJ Jr, Norris BE, Guinan SS (1972) Single auditory units in the superior olivary complex II: locations of unit categories and tonotopic organization. Int J Neurosci 4:147–166
Guinan JJ Jr, Warr WB, Norris BE (1983) Differential olivocochlear projections from lateral vs. medial zones of the superior olivary complex. J Comp Neurol 221:358–370
Guinan JJ Jr, Warr WB, Norris BE (1984) Topographic organization of the olivocochlear projections from the lateral and medial zones of the superior olivary complex. J Comp Neurol 226:21–27
Guinan JJ, Backus BC, Lilaonitkul W, Aharonson V (2003) Medial olivocochlear efferent reflex in humans: otoacoustic emission (OAE) measurement issues and the advantages of stimulus frequency OAEs. J Assoc Res Otolaryngol 4:521–540
Guinan JJ Jr, Lin T, Cheng H (2005) Medial-olivocochlear-efferent inhibition of the first peak of auditory-nerve responses: evidence for a new motion within the cochlea. J Acoust Soc Am 118:2421–2433
Guinan JJ Jr, Lin T, Cheng H, Cooper N (2006) Medial-Olivocochlear-Efferent Effects on Basilar-Membrane and Auditory-Nerve Responses to Clicks: Evidence for a New Motion within the Cochlea. In: Nuttall AL, Ren T, Gillespie PG, Grosh K, de Boer E, eds. Auditory Mechanisms: Processes and Models. World Scientific, Singapore, pp:1–9
Gummer M, Yates GK, Johnstone BM (1988) Modulation transfer function of efferent neurons in the guinea pig cochlea. Hear Res 36:41–52
He DZ, Jia S, Dallos P (2003) Prestin and the dynamic stiffness of cochlear outer hair cells. J Neurosci 23:9089–9096
Henson OW, Xie DH, Keating AW, Henson MM (1995) The effect of contralateral stimulation on cochlear resonance and damping in the mustached bat: the role of the medial efferent system. Hear Res 86:111–124
Hienz RD, Stiles P, May BJ (1998) Effects of bilateral olivocochlear lesions on vowel formant discrimination in cats. Hear Res 116:10–20
Housley GD, Ashmore JF (1991) Direct measurement of the action of acetylcholine on isolated outer hair cells of the guinea pig cochlea. Proc R Soc Lond B 244:161–167
Hudspeth AJ (2008) Making an effort to listen: mechanical amplification in the ear. Neuron 59:530–545
Karavitaki KD, Mountain DC (2007a) Evidence for outer hair cell driven oscillatory fluid flow in the tunnel of corti. Biophys J 92:3284–3293
Karavitaki KD, Mountain DC (2007b) Imaging electrically evoked micromechanical motion within the organ of Corti of the excised gerbil cochlea. Biophys J 92:3294–3316
Kawase T, Delgutte B, Liberman MC (1993) Anti-masking effects of the olivocochlear reflex, II: Enhancement of auditory-nerve response to masked tones. J Neurophysiol 70:2533–2549
Keefe DH, Schairer KS, Ellison JC, Fitzpatrick DF, Jesteadt W (2009) Use of stimulus-frequency otoacoustic emissions to investigate efferent and cochlear contributions to temporal overshoot. J Acoust Soc Am 125:1595–1604
Khalfa S, Veuillet E, Collet L (1998) Influence of handedness on peripheral auditory asymmetry. Eur J Neurosci 10:2731–2737
Khalfa S, Bougeard R, Morand N, Veuillet E, Isnard J, Guenot M, Ryvlin P, Fischer C, Collet L (2001) Evidence of peripheral auditory activity modulation by the auditory cortex in humans. Neuroscience 104:347–358
Kim DO, Dorn PA, Neely ST, Gorga MP (2001) Adaptation of distortion product otoacoustic emission in humans. J Assoc Res Otolaryngol 2:31–40
Kimura R, Wersäll J (1962) Termination of the olivocochlear bundle in relation to the outer hair cells of the organ of Corti in guinea pig. Acta Otolaryng (Stockh) 55:11–32
Konishi T, Slepian JZ (1971) Effects of the electrical stimulation of the crossed olivocochlear bundle on cochlear potentials recorded with intracochlear electrodes in guinea pigs. J Acoust Soc Am 49:1762–1769
Kumar UA, Vanaja CS (2004) Functioning of olivocochlear bundle and speech perception in noise. Ear Hear 25:142–146
Larsen E, Liberman MC (2009) Slow build-up of cochlear suppression during sustained contralateral noise: central modulation of olivocochlear efferents? Hear Res 256:1–10
Le Prell CG, Shore SE, Hughes LF, Bledsoe SC Jr (2003) Disruption of lateral efferent pathways: functional changes in auditory evoked responses. J Assoc Res Otolaryngol 4:276–290
Liberman MC (1980) Efferent synapses in the inner hair cell area of the cat cochlea: An electron microscopic study of serial sections. Hear Res 3:189–204
Liberman MC (1988a) Response properties of cochlear efferent neurons: monaural vs. binaural stimulation and the effects of noise. J Neurophysiol 60:1779–1798
Liberman MC (1988b) Physiology of cochlear efferent and afferent neurons: direct comparisons in the same animal. Hear Res 34:179–192
Liberman MC (1989) Rapid assessment of sound-evoked olivocochlear feedback: suppression of compound action potentials by contralateral sound. Hear Res 38:47–56
Liberman MC (1990) Effects of chronic cochlear de-efferentation on auditory-nerve response. Hear Res 49:209–224
Liberman MC, Brown MC (1986) Physiology and anatomy of single olivocochlear neurons in the cat. Hear Res 24:17–36
Liberman MC, Kiang NYS (1984) Single-neuron labeling and chronic cochlear pathology. IV. Stereocilia damage and alterations in rate- and phase-level functions. Hear Res 16:75–90
Liberman MC, Dodds LW, Pierce S (1990) Afferent and efferent innervation of the cat cochlea: quantitative analysis with light and electron microscopy. J Comp Neurol 301:443–460
Liberman MC, Puria S, Guinan JJ Jr (1996) The ipsilaterally evoked olivocochlear reflex causes rapid adaptation of the 2f1–f2 distortion product otoacoustic emission. J Acoust Soc Am 99:3572–3584
Lilaonitkul W, Guinan JJ Jr (2009a) Reflex control of the human inner ear: a half-octave offset in medial efferent feedback that is consistent with an efferent role in the control of masking. J Neurophysiol 101:1394–1406
Lilaonitkul W, Guinan JJ Jr (2009b) Human medial olivocochlear reflex: effects as functions of contralateral, ipsilateral, and bilateral elicitor bandwidths. J Assoc Res Otolaryngol 10:459–470
Lima da Costa DL, Chibois A, Erre JP, Blanchet C, de Sauvage RC, Aran JM (1997) Fast, slow, and steady-state effects of contralateral acoustic activation of the medial olivocochlear efferent system in awake guinea pigs: action of gentamicin. J Neurophysiol 78:1826–1836
Lin T, Guinan JJ Jr (2000) Auditory-nerve-fiber responses to high-level clicks: interference patterns indicate that excitation is due to the combination of multiple drives. J Acoust Soc Am 107:2615–2630
Lin T, Guinan JJ Jr (2004) Time-frequency analysis of auditory-nerve-fiber and basilar-membrane click responses reveal glide irregularities and non-characteristic-frequency skirts. J Acoust Soc Am 116:405–416
Lu TK, Zhak S, Dallos P, Sarpeshkar R (2006) Fast cochlear amplification with slow outer hair cells. Hear Res 214:45–67
Maison SF, Liberman MC (2000) Predicting vulnerability to acoustic injury with a noninvasive assay of olivocochlear reflex strength. J Neurosci 20:4701–4707
Maison S, Micheyl C, Collet L (1999) Sinusoidal amplitude modulation alters contralateral noise suppression of evoked otoacoustic emissions in humans. Neuroscience 91:133–138
Maison S, Micheyl C, Andeol G, Gallego S, Collet L (2000) Activation of medial olivocochlear efferent system in humans: influence of stimulus bandwidth. Hear Res 140:111–125
Maison S, Micheyl C, Collet L (2001) Influence of focused auditory attention on cochlear activity in humans. Psychophysiology 38:35–40
Maison SF, Adams JC, Liberman MC (2003) Olivocochlear innervation in the mouse: immunocytochemical maps, crossed versus uncrossed contributions, and transmitter colocalization. J Comp Neurol 455:406–416
Maison SF, Vetter DE, Liberman MC (2007) A novel effect of cochlear efferents: in vivo response enhancement does not require alpha9 cholinergic receptors. J Neurophysiol 97:3269–3278
May BJ, McQuone SJ, Lavoie A (1995) Effects of olivocochlear lesions on intensity discrimination in cats. Assoc Res Otolaryngol Abstr 18:146
May BJ, Prosen CA, Weiss D, Vetter D (2002) Behavioral investigation of some possible effects of the central olivocochlear pathways in transgenic mice. Hear Res 171:142–157
May BJ, Budelis J, Niparko JK (2004) Behavioral studies of the olivocochlear efferent system: learning to listen in noise. Arch Otolaryngol Head Neck Surg 130:660–664
Micheyl C, Collet L (1996) Involvement of the olivocochlear bundle in the detection of tones in noise. J Acoust Soc Am 99:1064–1610
Micheyl C, Perrot X, Collet L (1997) Relationship between auditory intensity discrimination in noise and olivocochlear efferent system activity in humans. Behav Neurosci 111:801–807
Michie PT, LePage EL, Solowlij N, Haller M, Terry L (1996) Evoked otoacoustic emissions and auditory selective attention. Hear Res 98:54–67
Morand N, Bouvard S, Ryvlin P, Mauguiere F, Fischer C, Collet L, Veuillet E (2001) Asymmetrical localization of benzodiazepine receptors in the human auditory cortex. Acta Otolaryngol 121:293–296
Morand-Villeneuve N, Veuillet E, Perrot X, Lemoine P, Gagnieu MC, Sebert P, Durrant JD, Collet L (2005) Lateralization of the effects of the benzodiazepine drug oxazepam on medial olivocochlear system activity in humans. Hear Res 208:101–106
Morlet T, Goforth L, Hood LJ, Ferber C, Duclaux R, Berlin CI (1999) Development of human cochlear active mechanism asymmetry: involvement of the medial olivocochlear system? Hear Res 134:153–162
Moulin A, Collet L, Duclaux R (1993) Contralateral auditory stimulation alters acoustic distortion products in humans. Hear Res 65:193–210
Mountain DC (1998) Modal analysis: a new paradigm for cochlear mechanics. Assoc Res Otolaryngol Abstr 21:61
Mukari SZ, Mamat WH (2008) Medial olivocochlear functioning and speech perception in noise in older adults. Audiol Neurootol 13:328–334
Muller J, Janssen T, Heppelmann G, Wagner W (2005) Evidence for a bipolar change in distortion product otoacoustic emissions during contralateral acoustic stimulation in humans. J Acoust Soc Am 118:3747–3756
Murugasu E, Russell IJ (1996) The effect of efferent stimulation on basilar membrane displacement in the basal turn of the guinea pig cochlea. J Neurosci 16:325–332
Norman M, Thornton ARD (1993) Frequency analysis of the contralateral suppression of evoked otoacoustic emissions by narrow-band noise. Br J Audiol 27:281–289
Nowotny M, Gummer AW (2006) Nanomechanics of the subtectorial space caused by electromechanics of cochlear outer hair cells. Proc Natl Acad Sci USA 103:2120–2125
Perrot X, Micheyl C, Khalfa S, Collet L (1999) Stronger bilateral efferent influences on cochlear biomechanical activity in musicians than in non-musicians. Neurosci Lett 262:167–170
Pfalz RKJ (1969) Absence of a function for the crossed olivocochlear bundle under physiological conditions. Arch Klin Exp Ohren Nasen Kehlkopfheilkd 193:89–100
Puria S, Guinan JJ Jr, Liberman MC (1996) Olivocochlear reflex assays: effects of contralateral sound on compound action potentials vs. ear-canal distortion products. J Acoust Soc Am 99:500–507
Rajan R (1988) Effect of electrical stimulation of the crossed olivocochlear bundle on temporary threshold shifts in auditory sensitivity. I. Dependence on electrical stimulation parameters. J Neurophysiol 60:549–568
Rajan R (2000) Centrifugal pathways protect hearing sensitivity at the cochlea in noisy environments that exacerbate the damage induced by loud sound. J Neurosci 20:6684–6693
Recio A, Rich NC, Narayan SS, Ruggero MA (1998) Basilar-membrane responses to clicks at the base of the chinchilla cochlea. J Acoust Soc Am 103:1972–1989
Reiter ER, Liberman MC (1995) Efferent-mediated protection from acoustic overexposure: relation to slow effects of olivocochlear stimulation. J Neurophysiol 73:506–514
Ren T, Nuttall AL (2001) Recording depth of the heterodyne laser interferometer for cochlear vibration measurement. J Acoust Soc Am 109:826–829
Rhode WS (2007) Basilar membrane mechanics in the 6–9 kHz region of sensitive chinchilla cochleae. J Acoust Soc Am 121:2792–2804
Robertson D (1984) Horseradish peroxidase injection of physiologically characterized afferent and efferent neurones in the guinea pig spiral ganglion. Hear Res 15:113–121
Robertson D (1985) Brainstem location of efferent neurons projecting to the guinea pig cochlea. Hear Res 20:79–84
Robertson D, Gummer M (1985) Physiological and morphological characterization of efferent neurons in the guinea pig cochlea. Hear Res 20:63–77
Robertson D, Gummer M (1988) Physiology of cochlear efferents in the mammal. In: Syka J, Masterton RB (eds) Auditory pathways: structure and function. Plenum, New York, pp 269–278
Robertson D, Anderson C-J, Cole KS (1987) Segregation of efferent projections to different turns of the guinea pig cochlea. Hear Res 25:69–76
Robles L, Ruggero MA (2001) Mechanics of the mammalian cochlea. Physiol Rev 81:1305–1352
Ruel J, Nouvian R, Gervais d’Aldin C, Pujol R, Eybalin M, Puel JL (2001) Dopamine inhibition of auditory nerve activity in the adult mammalian cochlea. Eur J Neurosci 14:977–986
Russell IJ, Murugasu E (1997) Medial efferent inhibition suppresses basilar membrane responses to near characteristic frequency tones of moderate to high intensities. J Acoust Soc Am 102:1734–1738
Ryan S, Kemp DT (1996) The influence of evoking stimulus level on the neural suppression of transient evoked otoacoustic emissions. Hear Res 94:140–147
Ryan S, Kemp DT, Hinchcliffe R (1991) The influence of contralateral acoustic stimulation on click-evoked otoacoustic emission in humans. Br J Audiol 25:391–397
Santos-Sacchi J (1991) Reversible inhibition of voltage-dependent outer hair cell motility and capacitance. J Neurosci 11:3096–3110
Scharf B, Magnan J, Chays A (1997) On the role of the olivocochlear bundle in hearing: 16 case studies. Hear Res 103:101–122
Shera CA (2001) Frequency glides in click responses of the basilar membrane and auditory nerve: their scaling behavior and origin in traveling-wave dispersion. J Acoust Soc Am 109:2023–2034
Shera CA, Guinan JJ Jr (1999) Evoked otoacoustic emissions arise by two fundamentally different mechanisms: a taxonomy for mammalian OAEs. J Acoust Soc Am 105:782–798
Shera CA, Guinan JJ Jr (2007) Cochlear traveling-wave amplification, suppression, and beamforming probed using noninvasive calibration of intracochlear distortion sources. J Acoust Soc Am 121:1003–1016
Siegel JH, Kim DO (1982) Efferent neural control of cochlear mechanics? Olivocochlear bundle stimulation affects cochlear biomechanical nonlinearity. Hear Res 6:171–182
Sininger YS, Cone-Wesson B (2004) Asymmetric cochlear processing mimics hemispheric specialization. Science 305:1581
Smith CA (1961) Innervation pattern of the cochlea. Ann Oto Rhinol Laryngol 70:504–527
Sridhar TS, Liberman MC, Brown MC, Sewell WF (1995) A novel cholinergic “slow effect” of olivocochlear stimulation on cochlear potentials in the guinea pig. J Neurosci 15:3667–3678
Sridhar TS, Brown MC, Sewell WF (1997) Unique post-synaptic signaling at the hair cell efferent synapse permits calcium to evoke changes on two different time scales. J Neurosci 17:428–437
Stankovic KM, Guinan JJ Jr (1999) Medial efferent effects on auditory-nerve responses to tail-frequency tones I: rate reduction. J Acoust Soc Am 106:857–869
Strickland EA (2008) The relationship between precursor level and the temporal effect. J Acoust Soc Am 123:946–954
Strickland EA, Krishnan LA (2005) The temporal effect in listeners with mild to moderate cochlear hearing impairment. J Acoust Soc Am 118:3211–3217
Tan MN, Robertson D, Hammond GR (2008) Separate contributions of enhanced and suppressed sensitivity to the auditory attentional filter. Hear Res 241:18–25
Teas DC, Konishi T, Nielsen DW (1972) Electrophysiological studies on the spatial distribution of the crossed olivocochlear bundle along the guinea pig cochlea. J Acoust Soc Am 51:1256–1264
Thiers FA, Burgess BJ, Nadol JB (2002) Reciprocal innervation of outer hair cells in a human infant. J Assoc Res Otolaryngol 3:269–278
Thiers FA, Nadol JB Jr, Liberman MC (2008) Reciprocal synapses between outer hair cells and their afferent terminals: evidence for a local neural network in the mammalian cochlea. J Assoc Res Otolaryngol 9:477–489
Thompson AM, Thompson GC (1991) Posteroventral cochlear nucleus projections to olivocochlear neurons. J Comp Neurol 303:267–285
Thompson S, Abdelrazeq S, Long GR, Henin S (2009) Differential effects of efferent stimulation by contralateral bandpass noise on the two major components of distortion product otoacoustic emissions. Assoc Res Otolaryngol Abstr 32:244
Veuillet E, Collet L, Duclaux R (1991) Effect of contralateral acoustic stimulation on active cochlear micromechanical properties in human subjects: dependence on stimulus variables. J Neurophysiol 65:724–735
Veuillet E, Duverdy-Bertholon F, Collet L (1996) Effect of contralateral acoustic stimulation on the growth of click-evoked otoacoustic emissions in humans. Hear Res 93:128–135
Veuillet E, Magnan A, Ecalle J, Thai-Van H, Collet L (2007) Auditory processing disorder in children with reading disabilities: effect of audiovisual training. Brain 130:2915–2928
Wagner W, Heppelmann G, Muller J, Janssen T, Zenner HP (2007) Olivocochlear reflex effect on human distortion product otoacoustic emissions is largest at frequencies with distinct fine structure dips. Hear Res 223:83–92
Wagner W, Frey K, Heppelmann G, Plontke SK, Zenner HP (2008) Speech-in-noise intelligibility does not correlate with efferent olivocochlear reflex in humans with normal hearing. Acta Otolaryngol 128:53–60
Warr WB (1975) Olivocochlear and vestibular efferent neurons of the feline brain stem: their location, morphology and number determined by retrograde axonal transport and acetylcholinesterase histochemistry. J Comp Neurol 161:159–182
Warr WB (1992) Organization of olivocochlear efferent systems in mammals. In: Webster DB, Popper AN, Fay RR (eds) Mammalian auditory pathway: neuroanatomy. Springer, New York, pp 410–448
Warr WB, Boche JE (2003) Diversity of axonal ramifications belonging to single lateral and medial olivocochlear neurons. Exp Brain Res 153:499–513
Warr WB, Guinan JJ Jr (1979) Efferent innervation of the organ of Corti: two separate systems. Brain Res 173:152–155
Warr WB, Beck Boche JE, Neely ST (1997) Efferent innervation of the inner hair cell region: origins and terminations of two lateral olivocochlear systems. Hear Res 108:89–111
Warren EH III, Liberman MC (1989a) Effects of contralateral sound on auditory-nerve responses. I. Contributions of cochlear efferents. Hear Res 37:89–104
Warren EH III, Liberman MC (1989b) Effects of contralateral sound on auditory-nerve responses. II. Dependence on stimulus variables. Hear Res 37:105–122
Wiederhold ML (1970) Variations in the effects of electric stimulation of the crossed olivocochlear bundle on cat single auditory-nerve-fiber responses to tone bursts. J Acoust Soc Am 48:966–977
Wiederhold ML, Peake WT (1966) Efferent inhibition of auditory nerve responses: dependence on acoustic stimulus parameters. J Acoust Soc Am 40:1427–1430
Winslow RL, Sachs MB (1987) Effect of electrical stimulation of the crossed olivocochlear bundle on auditory nerve response to tones in noise. J Neurophysiol 57:1002–1021
Ye Y, Machado DG, Kim DO (2000) Projection of the marginal shell of the anteroventral cochlear nucleus to olivocochlear neurons in the cat. J Comp Neurol 420:127–138
Yoshida N, Liberman MC, Brown MC, Sewell WF (1999) Gentamicin blocks both fast and slow effects of olivocochlear activation in anesthetized guinea pigs. J Neurophysiol 82:3168–3174
Zeng F-G, Shannon RV (1994) Loudness-coding mechanisms inferred from electric stimulation of the human auditory system. Science 264:564–566
Zeng F, Martino KM, Linthicum FH, Soli SD (2000) Auditory perception in vestibular neurectomy subjects. Hear Res 142:102–112
Zurek PM (1985) Acoustic emissions from the ear: a summary of results from humans and animals. J Acoust Soc Am 78:340–344
Zwicker E (1965) Temporal effects in simultaneous masking by white-noise bursts. J Acoust Soc Am 37:653–663
Zyl AV, Swanepoel DW, Hall JW III (2009) Effect of prolonged contralateral acoustic stimulation on transient evoked otoacoustic emissions. Hear Res 254:77–81
Acknowledgments
This work was supported by NIH NIDCD RO1 000235 and RO1 005977.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2011 Springer Science+Business Media, LLC
About this chapter
Cite this chapter
Guinan, J.J. (2011). Physiology of the Medial and Lateral Olivocochlear Systems. In: Ryugo, D., Fay, R. (eds) Auditory and Vestibular Efferents. Springer Handbook of Auditory Research, vol 38. Springer, New York, NY. https://doi.org/10.1007/978-1-4419-7070-1_3
Download citation
DOI: https://doi.org/10.1007/978-1-4419-7070-1_3
Published:
Publisher Name: Springer, New York, NY
Print ISBN: 978-1-4419-7069-5
Online ISBN: 978-1-4419-7070-1
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)