Abstract
Hair cell receptors for the hearing and balance organs, and the lateral line, are unique among the senses by receiving an efferent innervation of the periphery. Olivocochlear (OC) neurons supply this efferent innervation, and they are the most peripheral of the many descending neural systems of the central auditory pathway (see Schofield, Chap. 9). OC neurons are named by their origins in the superior olivary complex and terminations in the cochlea (Fig. 2.1). In the cochlea, they innervate the hair cells and auditory-nerve fibers. This chapter mainly covers the new ground on OC anatomy in mammals since Warr’s (1992) comprehensive chapter on this topic about 15 years ago. Since that time, there is even stronger evidence for the separate innervation of the periphery by the two major groups of OC neurons. It is also now clear that both of these groups consist of distinct subgroups. There is additional information on the reflex pathways leading up to OC neurons that enables their response to sound. Overall, this anatomy may help to define the functions that OC neurons perform in the sense of hearing.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
2.1 Introduction
Hair cell receptors for the hearing and balance organs, and the lateral line, are unique among the senses by receiving an efferent innervation of the periphery. Olivocochlear (OC) neurons supply this efferent innervation, and they are the most peripheral of the many descending neural systems of the central auditory pathway (see Schofield, Chap. 9 ). OC neurons are named by their origins in the superior olivary complex and terminations in the cochlea (Fig. 2.1). In the cochlea, they innervate the hair cells and auditory-nerve fibers. This chapter mainly covers the new ground on OC anatomy in mammals since Warr’s (1992) comprehensive chapter on this topic about 15 years ago. Since that time, there is even stronger evidence for the separate innervation of the periphery by the two major groups of OC neurons. It is also now clear that both of these groups consist of distinct subgroups. There is additional information on the reflex pathways leading up to OC neurons that enables their response to sound. Overall, this anatomy may help to define the functions that OC neurons perform in the sense of hearing.
2.2 OC Neurons in the Brain Stem
2.2.1 Distributions of Lateral vs. Medial Olivocochlear Neurons
OC neurons were discovered by Rasmussen (1946, 1953). Later studies confirmed the interpretation of these early fiber degeneration studies with the use of retrograde labeling that filled the OC neurons with tracer (Warr 1975). The separation of OC neurons into the two major groups originated with Warr and Guinan (1979; Fig. 2.1). Generally, lateral olivocochlear (LOC) neurons originate in and/or near the lateral superior olive (LSO), whereas medial olivocochlear (MOC) neurons generally originate in the medial periolivary regions. The exact location of OC neurons depends on species. For example, many LOC neurons in rodents are found within the LSO (Campbell and Henson 1988; Vetter and Mugnaini 1992; Sánchez-González et al. 2003), whereas in cats they are found mainly in the dorsal hilus of the LSO (Warr 1975) and in squirrel monkey they appear to reside between the LSO and the medial superior olive (Thompson and Thompson 1986). Some LOC neurons are located in the anterolateral and dorsolateral periolivary areas (Warr et al. 2002).
Schematic showing OC neurons and the course of their axons to one cochlea, the cochlea on the right side of the figure designated “ipsilateral.” Lateral olivocochlear (LOC) neurons have cell bodies located around and/or in the lateral superior olive (LSO), whereas medial olivocochlear (MOC) neurons have cell bodies located in the more medial parts of the superior olivary complex. Axons from LOC neurons (gray lines representing unmyelinated axons) and MOC neurons (black lines representing myelinated axons) coalesce into the OC bundle. As the bundle projects laterally, branches are given off to the vestibular and cochlear nuclei. Inset below: Schematic of peripheral terminations of LOC fibers on auditory-nerve dendrites below inner hair cells (IHCs) and terminations of MOC fibers on outer hair cells (OHCs). Arrows indicate direction of spike propagation (adapted from Warren and Liberman 1989)
Two subgroups of LOC neurons were discovered by Vetter and Mugnaini (1992). LOC “intrinsic” neurons are small and contained within the body of the LSO, whereas LOC “shell” neurons are larger and found on the margins of the LSO (Fig. 2.2). In cats, where all LOC neurons are outside the LSO, the subgroup distinction is made by proximity and dendritic association with the LSO (Warr et al. 2002). LOC intrinsic neurons in rodents have dendrites running across the LSO (Fig. 2.3), presumably within an isofrequency plane. LOC shell neurons are about twice the size of intrinsic neurons. Some of their dendrites enter the LSO and others run into the reticular formation.
Photomicrograph of presumed OC neurons in the brain stem of a mouse. In this transverse section through the left side, the stain for acetylcholinesterase appears as a black color. Darkly stained MOC neurons in the ventral nucleus of the trapezoid body (VNTB) have large cell bodies and long dendrites that extend medially from these somata toward the trapezoid body. A few large stained neurons are seen in the dorsal periolivary nucleus (DPO); these are probably MOC neurons. MOC axons project dorsally to eventually form the OC bundle. LOC intrinsic neurons with small somata are seen within the LSO and LOC shell neurons are seen on its margins. LOC dendrites and axons are not well stained by acetylcholinesterase (from Brown and Levine 2008)
Drawings of OC somata in the brain and their peripheral terminations in the organ of Corti. There are distinct types of LOC neurons. LOC intrinsic neurons have somata within the LSO and project peripherally to form “unidirectional” fibers that run in one direction in the inner and tunnel spiral bundles beneath IHCs. Their peripheral terminations have limited spans along the cochlea. LOC shell neurons on the margins of the LSO project peripherally to form “bidirectional” fibers that run both directions in the spiral bundles and terminate over extensive spans. MOC neurons project peripherally to innervate OHCs (dots) in a “patchy” pattern. The endings are distributed over substantial spans (modified from Brown 1987a, b; Warr et al. 1997)
MOC neurons in most species are found predominantly in the ventral nucleus of the trapezoid body (VNTB). Other nuclei containing MOC neurons include the dorsomedial preolivary nucleus, the medial nucleus of the trapezoid body, and the dorsal periolivary nucleus (Fig. 2.2; Warr 1975; Brown and Levine 2008). The distribution of MOC neurons extends more rostrally than LOC neurons, reaching the caudal end of the ventral nucleus of the lateral lemniscus. MOC neurons have large somata (Fig. 2.2). They give rise to dendrites that radiate in different directions, and the longest are directed medially (Brown and Levine 2008). The nonplanar arrangement of these dendrites seems incongruous with MOC responses, which are sharply tuned to sound frequency (Robertson and Gummer 1985; Liberman and Brown 1986).
2.2.2 Numbers of Neurons
Depending on the species, total numbers of OC neurons innervating one cochlea range between 341 (hamster) and 474 (mouse) on the lower end and 1,366 (cat) and 2,346 (guinea pig) on the higher end (reviews: Warr 1992; Sánchez-González et al. 2003). In humans, there are about 1,400 axons in the OC bundle (Arnesen 1984). In general, the smaller species have fewer neurons; thus, some of the variability in the number of OC neurons can be reduced by dividing by basilar membrane length, which is shorter in the smaller species (Bishop and Henson 1987). These numbers of OC neurons are dwarfed by the afferent neurons of the cochlea, which range from about 20,000 to 50,000 per cochlea in the various mammalian species (Nadol 1988).
The ratio of LOC to MOC neurons is also variable among species. In the extreme, a bat species similar to the horseshoe bat, Rhinolophus rouxi, lacks MOC neurons; in contrast, guinea pigs have approximately equal numbers of LOC and MOC neurons (Aschoff and Ostwald 1987). In cats and mice, the percentages are about 65% LOC and 35% MOC (Arnesen and Osen 1984; Campbell and Henson 1988; Warr et al. 2002). In humans, the percentages are about 70% LOC vs. 30% MOC (Arnesen 1984, counted as thin vs. thick axons in the OC bundle; see later). Shell neurons number about 15% of all LOC neurons in rats (Vetter and Mugnaini 1992).
OC neurons projecting to a single cochlea are distributed bilaterally in the brain stem (Fig. 2.1). LOC neurons are located predominantly on the same side of the brain as the cochlea that they innervate. For cats, the same side:opposite side ratio is about 3:1 (Warr et al. 2002), whereas for mice it is about 100:1 (Campbell and Henson 1988). MOC neurons are usually distributed unequally but their distribution is skewed toward more neurons on the side of the brain opposite to the innervated cochlea. For cats and most other species, there are about twice as many MOC neurons on the opposite side of the brain; for chinchillas the distribution is about even on the two sides (Azeredo et al. 1999). Opposite-side OC neurons have axons that cross the midline on their way to the innervated cochlea, and even uncrossed MOC axons can approach the midline (Fig. 2.1). About 5% of MOC neurons, but no LOC neurons, project bilaterally (Robertson et al. 1987a).
2.2.3 Axonal Characteristics
An important distinction between the groups of OC neurons is that LOC neurons have thin, unmyelinated axons and MOC neurons have thicker, myelinated axons (evidence reviewed by Guinan et al. 1983; Warr 1992). This distinction has important physiological implications because thin unmyelinated axons have much higher thresholds to electric stimulation. Thus, electric stimulation of the OC bundle is likely to activate the MOC axons exclusively (Guinan et al. 1983). Axons from both LOC shell neurons and intrinsic neurons are thin (Brown 1987a; Warr et al. 1997).
2.3 Peripheral Projections
2.3.1 Separate Terminations of LOC and MOC Neurons
The separate peripheral termination of LOC and MOC neurons was first demonstrated by Warr and Guinan (1979). Since then, the preponderance of the evidence suggests that LOC neurons project to inner hair cell (IHC)-associated targets, whereas MOC neurons project to outer hair cells (OHCs; Fig. 2.1, inset). Are there any OC neurons that innervate both IHCs and OHCs? Tracings of labeled fibers indicate separate innervation of the two types of hair cells, at least in the basal half of the cochlea (Fig. 2.3; Robertson and Gummer 1985; Liberman and Brown 1986; Brown 1987a). Also in support of separate innervation of the two types of hair cell, cuts of crossing MOC fibers reduce endings on OHCs, but have no effect on endings on IHCs in the mouse, where LOC neurons are almost entirely uncrossed (Maison et al. 2003). Separate innervations of the hair cells by the OC groups would parallel the separate innervation by afferent fibers, in which type I and type II auditory nerve fibers separately innervate IHC and OHC, respectively (Spoendlin 1971; Kiang et al. 1982).
2.3.2 Terminations of LOC Fibers
Both groups of OC neurons have fibers that branch extensively in the cochlea (Fig. 2.3). The end result of the branching is that a relatively small number of OC neurons gives rise to numerous synapses in the cochlea. LOC fibers synapse mainly on dendrites of auditory nerve fibers beneath IHCs. In the cat, those dendrites contacting the IHC on its modiolar side, which correspond to high-threshold, low-spontaneous rate fibers, receive an average of 15–25 synapses per fiber (Liberman et al. 1990). Those dendrites contacting the IHC on its pillar-cell side, which correspond to the low-threshold, high-spontaneous rate fibers, receive fewer synapses: an average of 5–10 synapses per fiber. Some LOC synapses are formed directly on the IHCs (Liberman 1980). In the inner spiral bundle, LOC synapses are formed by small en passant swellings (Fig. 2.4) and a few terminal branches. In some species, LOC swellings in the tunnel spiral bundle may contact MOC branches on their way to the OHCs (Iurato et al. 1978; Liberman 1980).
Cochlear immunostains showing cholinergic MOC terminals on OHCs and mixed population of terminals in the inner spiral bundle (ISB) just beneath IHCs. This cochlear whole-mount from a mouse was stained for vesicular acetylcholine transporter (VAT, red) and for tyrosine hydroxylase (TH, green). Large, VAT-positive endings of MOC neurons are present on the OHC. Small, VAT-positive endings of LOC neurons are present in the ISB. Intermingled with these cholinergic endings are a few TH-positive LOC endings. There are also numerous TH-positive swellings in the osseous spiral lamina (OSL) (figure generously provided by M.C. Liberman)
LOC intrinsic and shell neurons have very distinct terminal arbors (Fig. 2.3). Warr et al. (1997) studied this issue by injections of tracers either inside the LSO (to label intrinsic LOC neurons) or on its margins (to label shell neurons). Intrinsic neurons form peripheral fibers that turn one direction as they enter the inner spiral bundle. They terminate in arborizations of limited span along the organ of Corti. Shell neurons form peripheral fibers that bifurcate in the inner spiral bundle to run both apically and basally. They terminate in extensive arborizations over wide spans along the spiral bundles. Branching of axons giving rise to bidirectional fibers, but not unidirectional fibers, suggests that several bidirectional arborizations can arise from a single neuron (Brown 1987a).
The density of LOC terminals along the length of the cochlea is relatively even, with somewhat more innervation apically (Liberman et al. 1990). LOC neurons project to the cochlea in a way consistent with the known tonotopic mapping of the LSO (Guinan et al. 1984). For example, tracer injections into the lateral part of the rodent LSO (known to be a region of low characteristic frequency) result in labeling in the cochlear apex, whereas injections into the medial part of the LSO result in labeling the base (Stopp 1983; Robertson et al. 1987b). These results probably apply to intrinsic neurons; whether the mapping of shell neurons is tonotopic remains to be investigated.
2.3.3 Terminations of MOC Fibers
MOC endings on OHCs are larger than the LOC endings in the inner spiral bundle (Fig. 2.4). The density of these MOC terminals has a broad peak around the midpoint of the cochlea in mice (Fig. 2.5) or somewhat more basally in cats (Guinan et al. 1984). Compared to the more numerous terminals from crossed axons, terminals from uncrossed axons have a more even density and are distributed somewhat more apically (Guinan et al. 1984). In most species, as the terminations taper off apically, they do so initially for the third row of OHCs, next for the second row, and last for the first row (Liberman et al. 1990). However, in mouse, there is a relatively equal innervation of the three rows (Maison et al. 2003). The longitudinal distribution of endings is similar to the effects of stimulation in response to different frequencies (Fig. 2.5). The MOC terminals hyperpolarize OHCs, decrease the gain of the cochlear amplifier and the sensitivity of the IHCs, and finally reduce the responses of the auditory nerve fibers, which is the metric plotted in Fig. 2.5 (see Guinan, Chap. 3). OHCs in the cochlear base receive an average of nine MOC terminals (Liberman et al. 1990). Some MOC en passant swellings just below OHCs contact type II auditory nerve fibers (Thiers et al. 2002).
Longitudinal distribution of cochlear MOC terminals in the mouse shows a broad peak in the middle of the cochlea. The x-axis is distance along the cochlear spiral plotted as cochlear frequency correlate. Equivalent counts of immunostaining for glutamic acid decarboxylase (GAD, open circles), calcitonin gene-related peptide, (CGRP, shaded circles), or vesicular acetylcholine transporter (VAT, solid circles) results from extensive co-localization. Presumably, the peak in distribution of terminals is responsible for the peak of MOC effect, which is the average suppression of the compound action potential of the cochlea expressed as effective attenuation in decibels as a result of electrical stimulation of the OC bundle. MOC terminal data from Maison et al. (2003); MOC effects adapted from Vetter et al. (1999)
Reconstructions of single MOC fibers in the cochlea indicate substantial spans of the endings along the organ of Corti. For example, the guinea pig fiber in Fig. 2.3 contacts OHCs over a span of about 1.5 mm. Cat MOC fibers can span up to 3.2 mm, corresponding to a cochlear distance of about an octave (Liberman and Brown 1986). The innervation pattern from a single fiber is “patchy,” with clusters of OHCs innervated separated by long uninnervated regions; other fibers provide the innervation for those hair cells between the patches. MOC neurons project onto the cochlea in a mapping generally similar to the cochlear frequency mapping for auditory nerve fibers (Robertson and Gummer 1985; Liberman and Brown 1986; Brown 1989, 2002). For example, an MOC neuron with a characteristic frequency of 10 kHz projects to a region of the cochlea that has auditory-nerve fibers tuned to a similar characteristic frequency. MOC neurons thus affect the processing of information in the cochlea in a frequency-specific manner. Given the broad span of innervation it is expected that a single fiber would affect a band of frequencies rather than a single frequency (see Guinan, Chap. 3, for human data relevant to this issue).
MOC terminal anatomy is influenced by postsynaptic target. This has been demonstrated in mice lacking the α9 cholinergic receptor, a key component of the receptor for MOC action on OHCs (see Sewell, Chap. 4 and Katz et al., Chap. 5). In these mice, there is a single, large MOC terminal on each OHC rather than the multiple small terminals found in wild-type mice (Vetter et al. 1999). In these same mice, however, the central branches and somata of OC neurons have normal morphology (Brown and Vetter 2008), apparently because central receptors are different (see later) and are preserved in the knock-out mice.
2.4 Central Branches to the Cochlear and Vestibular Nuclei
MOC axons form branches to the cochlear nucleus en route to the periphery (Fig. 2.1). About two thirds of MOC axons in rodents form branches; crossing fibers as well as uncrossed fibers are branched (Brown 1993). There may be species differences in the number of branches formed. Mice and cats have many such branches (Osen et al. 1984), guinea pigs have fewer branches (Winter et al. 1989), and humans appear to lack them (Moore and Osen 1979). Some work (Brown et al. 1988) suggests that only the thick MOC axons form cochlear nucleus branches. Other studies (Ryan et al. 1990; Horvath et al. 2000), however, suggest branches from LOC neurons. Perhaps these differences arise from differences in the species used.
MOC branches terminate mainly in the edge regions and to a lesser extent the core of the cochlear nucleus (Osen et al. 1984; Ryan et al. 1990; Brown 1993; Brown and Vetter 2008). There are many branches at the medial edge (Fig. 2.6) and at the dorsal edges of the VCN where it abuts the granule cell lamina. Some branches are found in the core of the cochlear nucleus and on its superficial edge (Fig. 2.6), part of the shell of small and granule cells. Caudally and rostrally, the branches taper off. LOC processes are not well stained by acetylcholinesterase (Fig. 2.2), but LOC branches labeled by retrograde transport of amino acids terminate mainly in the core of the cochlear nucleus (Ryan et al. 1990).
OC branches to the cochlear nucleus. A mouse transverse section stained for acetylcholinesterase that shows the darkly stained olivocochlear bundle (OCB) giving off branches (br). In this section, most of the branches form endings at the medial edge (arrow), a few in the core of the anteroventral cochlear nucleus (AVCN), and one or two at its superficial edge (sup) (unpublished data from Brown and Levine)
The branches formed by the MOC give rise to numerous swellings, which are the site of synapses (Benson and Brown 1990). The synapses have round vesicles and are thus likely to be excitatory (Uchizono 1965). The most common targets of the synapses are large-diameter dendrites. Single dendrites receive multiple synapses from one branch, suggesting a powerful effect on the target. The target dendrites are likely to be from cochlear nucleus stellate/multipolar cells. In slice preparations, the type of neuron known as “T” stellate/multipolar cells is affected by cholinergic agonists (Fujino and Oertel 2001), indicating that it has the appropriate receptor for the acetylcholine released by OC branches. In vivo, electrical stimulation of the OC bundle causes excitation of neurons with an “onset chopper” response to sound (Mulders et al. 2007; see Robertson and Mulders, Chap. 10). Neurons with this response may correspond to a different subtype of stellate/multipolar cell and this difference remains to be resolved. The functional role of the OC branches to the cochlear nucleus also remains to be worked out. They may provide a form of “efferent copy” in which certain elements in the cochlear nucleus receive information about the type and amount of efferent feedback sent to the periphery (Benson and Brown 1990). For example, MOC neuron action on the periphery is inhibitory, in turn decreasing responses of cochlear nucleus neurons including those providing inputs to MOC neurons. This would cause MOC response to decrease, which might compromise important functions such as providing protection from acoustic overstimulation. A scheme to maintain MOC response at a constant level would be to have MOC branches excite those neurons providing inputs to MOC neurons.
The cochlear nucleus contains neither the α9 nor α10 nicotinic receptor subunits that are present at MOC synapses in the cochlea. The central receptor for the MOC branches is most likely composed of the α7 nicotinic cholinergic receptor. Immunostaining (Yao and Godfrey 1999) and the presence of mRNA for this receptor (Happe and Morely 1998) overlap with the regional distributions of branches. The pharmacological profile of the effect of acetylcholine on cochlear nucleus stellate cells is also consistent with the involvement of an α7 receptor as well as the presence of other cholinergic receptors (Fujino and Oertel 2001).
MOC and LOC neurons also give off branches to some of the vestibular nuclei (Brown et al. 1988; Ryan et al. 1990; Brown 1993). These branches form swellings that, like those in the cochlear nucleus, produce synapses (Benson and Brown 1996). The postsynaptic targets in the inferior vestibular nucleus are thick dendrites and sometimes cell bodies. The function of these vestibular-nucleus branches is obscure.
2.5 Neurochemistry
Both groups of OC neurons are predominantly cholinergic, but the LOC group probably has additional neurons that are not cholinergic (see Sewell, Chap. 4 and Katz et al., Chap. 5). In immunohistochemical studies of the rat, about half of the LOC neurons are reported to stain for glutamic acid decarboxylase (GAD, an indicator of GABAergic neurotransmission), and the other half are reported to stain for calcitonin gene-related peptide (CGRP) (Vetter et al. 1991). In mouse, though, there is extensive co-localization of immunolabeling for these transmitter-related substances along with vesicular acetylcholine transporter (an indicator of cholinergic neurotransmission). This co-labeling suggests that LOC neurons are predominantly cholinergic. LOC shell neurons may be in large part dopaminergic because they immunostain for tyrosine hydroxylase (Darrow et al. 2006). These LOC neurons apparently account for the tyrosine hydroxylase-positive endings in the IHC area (Fig. 2.4). Some LOC shell neurons, however, may stain for acetylcholinesterase (Brown and Levine 2008). Clarification of the neurotransmitters of LOC neurons is an active area of research.
MOC neurons are cholinergic. At least in some species, there is co-localization of other transmitters in the terminals (Maison et al. 2003). For example, there are similar numbers of mouse OHC terminals stained for vesicular acetylcholine transporter (an indicator of cholinergic neurotransmission), GAD, and CGRP (Fig. 2.5). These numbers are similar to the numbers of OHC terminals stained for SNAP25, a marker of all vesiculated terminals, so there are apparently no other types of terminals. Consistent with their cholinergic neurotransmission, MOC neurons, their axons, and their processes all stain darkly for acetylcholinesterase (Fig. 2.2; Schuknecht and Nomura 1965; Osen et al. 1984). LOC neurons also stain, but their axons and dendrites are not as darkly stained (Fig. 2.2; Brown and Levine 2008). OC neurons also stain for the cholinergic marker, choline acetyltransferase (Thompson and Thompson 1986).
2.6 Ultrastructure of Synaptic Inputs to OC Neurons
MOC neurons receive several types of synaptic terminals (Fig. 2.7; White 1984, 1986; Spangler et al. 1986; Helfert et al. 1988; Benson and Brown 2006). One type contains large, round vesicles (Fig. 2.7a). These terminals are large in size and form up to seven synapses per terminal. Some of them are associated with spines of MOC neurons, which were first described by Mulders and Robertson (2000b). Both simple and “mushroom” spines are formed (Benson and Brown 2006). The type of terminal containing large, round vesicles is apparently lacking on MOC neurons from the cat (White 1984, 1986; Spangler et al. 1986). The second type (Fig. 2.7b) contains smaller oval-to-round vesicles as well as a few dense core vesicles. The presence of dense core vesicles is an important distinguishing feature, since morphometric measurement of vesicle area suggest a continuum in average vesicle size between the types containing “large, round” and “small, round” types (Fig. 2.7d). The small, round terminals are of moderate size and form up to four synapses per terminal. This type of terminal may originate from neurons in the cochlear nucleus and may excite the MOC neurons to respond to sound as part of the MOC reflex (Benson and Brown 2006). Interestingly, LOC neurons receive many such terminals (Helfert et al. 1988). A third type (Fig. 2.7c) contains flattened or pleomorphic vesicles and is thus likely to be inhibitory (Uchizono 1965). This type of terminal is small and forms a single synapse per terminal. In recordings of single MOC neurons, inhibition of activity has been reported for frequencies outside the excitatory area and sometimes for sounds in the nondominant ear (Liberman and Brown 1986; Brown 1989).
Ultrastructure of synaptic input to MOC neurons, showing the three types of synaptic terminals (a–c) found on guinea pig MOC neurons. The MOC neuron is at the top in each panel, and the synaptic specialization is denoted by arrowheads. (a) Terminal with relatively large, round (spherical) vesicles. (b) Terminal with relatively smaller, round-to-oval vesicles and dense core vesicles (dcv, one indicated with arrow). (c) Terminal with pleomorphic or variously shaped vesicles. Scale bar = 1 μm. (d) Vesicle morphometry of synaptic terminals on MOC neurons. The terminals were first classified visually using size and shape of vesicles and presence of dense core vesicles (over all the serial sections containing the terminal). To make the measurements, all clear vesicles within 1 μm of a synaptic specialization in one or two sections from each terminal were measured using ImageJ, which defines circularity as 4π(area/perimeter2). The number of vesicles measured per terminal ranged from 20 to 92; the means of these circularity/area measurements are plotted as a single point for each terminal. Dense core vesicles were not included. Data are from two guinea pigs ((a)–(c) from Benson and Brown 2006; (d) is unpublished data from Benson and Brown)
2.7 Neural Pathway of the Medial Olivocochlear Reflex
2.7.1 Direct Reflex Pathway
Medial OC neurons respond to sound as part of the MOC reflex (Fig. 2.8). The three neurons of the reflex pathway are auditory nerve fibers, cochlear nucleus neurons, and MOC neurons. Direct projections from the ventral cochlear nucleus to OC neurons have been demonstrated using neural tracers (Robertson and Winter 1988; Thompson and Thompson 1991; Ye et al. 2000). These direct projections are consistent with the short latency of the MOC neuron response to sound (Robertson and Gummer 1985; Liberman and Brown 1986; Brown et al. 2003). MOC neurons consist of two major subgroups defined on the basis of which ear excites their response (see Guinan, Chap. 3). Ipsi units respond to monaural sound in the ipsilateral ear whereas Contra units respond to monaural sound in the contralateral ear (Robertson and Gummer 1985; Liberman and Brown 1986). An additional minor subgroup responds to sound in either ear. Because of the distinct distribution of MOC neurons by the two major response classes, separate pathways can be drawn for the MOC reflex in response to ipsilateral vs. contralateral sound (black vs. gray pathways in Fig. 2.8).
Pathways of the sound-evoked MOC reflexes to one cochlea, the ipsilateral cochlea on the right of the figure. Indicated are the positions of MOC neurons that respond to sound in the ipsilateral ear (Ipsi Neurons) and those that respond to sound in the contralateral ear (Contra Neurons). The reflex pathway in response to ipsilateral sound begins in the ipsilateral cochlea with the responses of the hair cells and the auditory nerve. Nerve fibers project centrally into the cochlear nucleus. Here, MOC reflex interneurons from the cochlear nucleus send axons across the midline (black pathway) to innervate Ipsi neurons. These Ipsi neurons send axons back across the midline to innervate the ipsilateral cochlea. The reflex pathway in response to contralateral sound begins with hair cells and nerve fibers in the contralateral cochlea. From the contralateral cochlear nucleus, MOC reflex interneurons send axons that cross the midline (gray pathway) to innervate Contra neurons. Contra neurons in turn project without crossing to the ipsilateral cochlea. In addition to these dominant inputs, both types of MOC neurons receive inputs that facilitate the response to the dominant ear (small arrows, “Facilitatory Inputs”) (modified from Liberman and Guinan 1998)
The cochlear nucleus projections to OC neurons originate in two very different locations: the posteroventral subdivision (PVCN) (Thompson and Thompson 1991) and the shell of the anteroventral subdivision (AVCN) (Ye et al. 2000). The functional importance of the various projections has been explored by making small lesions using kainic acid. Lesions of the PVCN (Fig. 2.9), but not the AVCN, interrupt the ipsilateral MOC reflex (de Venecia et al. 2005), demonstrating that this cochlear nucleus subdivision contains the interneurons of the MOC reflex. Some data suggest a similar PVCN site for the neurons mediating the contralateral reflex. The PVCN contains projection neurons of three types: octopus cells, stellate/multipolar cells, and globular bushy cells (Osen 1969; Hackney et al. 1990). Octopus cells have onset responses to tone bursts at the characteristic frequency (Rhode et al. 1983; Rouiller and Ryugo 1984), but MOC neurons have very sustained responses (Brown 2001). Thus, octopus cells are unlikely to be the interneurons (Brown et al. 2003), leaving stellate/multipolar and bushy cells as possibilities.
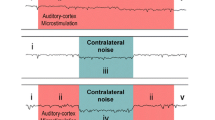
Determination of the site of the MOC reflex interneurons in the cochlear nucleus. Graph below: Effect of a kainic-acid lesion on the adaptation of the DPOAE. Before the lesion, there was a large, positive-going adaptation of the DPOAE. Presumably, this takes place because the two primary tones (f1, f2 at bottom) activate Ipsi MOC neurons to respond to sound, and their influence on OHC alters the DPOAE. For particular frequency/level combinations of tones in which the DPOAE is composed of several components that are close to equal and opposite, a relatively small alteration of one can have a large effect on the sum. The DPOAE adaptation magnitude (arrows at right) was used as a metric for the MOC reflex because cuts of the OC bundle greatly reduce the adaptation (at least in the guinea pigs used here: Kujawa and Liberman 2001). After the kainic acid lesion, the adaptation was greatly reduced (dashed line), presumably because the reflex interneurons were lesioned. Post-experiment histology from the same experiment (drawings above), showing a region of complete cell loss (gray shading) centered in the dorsal and caudal portions of the posteroventral subdivision of the cochlear nucleus (PVCN), an area common to other reflex-interrupting lesions. In this particular lesion, there was also some involvement of the lamina (lam), the octopus cell area (oca), the superficial granular layer (sup), and the dorsal cochlear nucleus (DCN) (graph adapted from Brown et al. 2003; drawing from de Venecia et al. 2005)
Stellate/multipolar neurons have projections that are consistent with a role as intermediaries of the MOC reflex. At least some stellate/multipolar neurons project to the VNTB (Smith et al. 1993; Doucet and Ryugo 2003), the nucleus that contains most of the MOC neurons. The projection is likely formed by “planar” stellate/multipolar neurons. These neurons have dendrites confined to isofrequency laminae in the cochlear nucleus (Doucet and Ryugo 2003) and may, like MOC neurons, be sharply tuned to sound frequency. Another type of stellate/multipolar neuron, the radiate neuron, is inhibitory and thus cannot provide the excitation needed to drive the MOC response to sound. Which synaptic terminals on MOC neurons might correspond to those of the MOC reflex interneurons? Stellate/multipolar terminal ultrastructure in the inferior colliculus (Oliver 1987) and locally in the cochlear nucleus (Smith et al. 1993) is consistent with the type that has small, round vesicles and a few dense core vesicles (Fig. 2.7b), but this remains to be directly demonstrated. Another type of cochlear nucleus neuron, the globular bushy cell, is less likely to play a direct role in the MOC reflex. Some bushy cells have projections to the VNTB (Spirou et al. 1990; Smith et al. 1991). However, the most effective reflex-interrupting lesions in the MOC reflex were in caudal PVCN locations (de Venecia et al. 2005), and the distribution of bushy cells is in restricted to more rostral PVCN and AVCN in the guinea pig (Hackney et al. 1990). Also, the waveform peaks of the auditory brain stem response, which are generated by bushy cells, could persist even though the MOC reflex was interrupted (de Venecia et al. 2005), suggesting independence of bushy cells and the reflex.
2.7.2 Modulatory Pathways
Both groups of OC neurons receive inputs that presumably modulate the response to sound. Serotoninergic inputs (Thompson and Thompson 1995) project to OC neurons and these inputs may arise from the midline raphe nuclei. Such neurons are labeled after cochlear injections of transneuronal labeling agents that first label OC neurons and then pass across synapses to neurons providing input to OC neurons (Horvath et al. 2003). Noradrenaline is present in varicosities surrounding OC neurons in the brain stem (Mulders and Robertson 2005); its likely source is the locus coeruleus. Serotonin and noradrenaline have been demonstrated to affect VNTB and OC neurons in slice preparations (Wang and Robertson 1997a, b).
MOC neurons receive inputs from higher centers that also presumably modulate the reflex. Descending inputs to MOC neurons arise from the inferior colliculus (Faye-Lund 1986; Thompson and Thompson 1993; Vetter et al. 1993) and the auditory cortex (Mulders and Robertson 2000a). Perhaps these higher centers mediate the trainable effects of the MOC reflex that have been documented in humans (de Boer and Thornton 2008). Such effects might be mediated by the synaptic terminals containing large, round vesicles (Fig. 2.7a), because these terminals are associated with spines. Spine-associated neural systems in the hippocampus mediate such plastic changes (reviews: Matsuzaki 2007; Bourne and Harris 2008). Working out the anatomical substrate for plasticity in the OC reflexes is a future goal.
2.8 Summary
Our knowledge of the anatomy of OC neurons has advanced, but is still incomplete. The separate innervations of the two hair cell groups by LOC and MOC neurons is clear, and it is apparent that MOC neuron action on OHCs decreases cochlear sensitivity. What separate role is played by LOC neurons is much less clear. By their limited dendritic extents and cochleotopic projections, the LOC intrinsic subgroup seems poised to operate within narrow bands of frequency information, whereas their radiating dendrites and extensive cochlear spans implies that the LOC shell subgroup operates in a much broader fashion. However, even whether the two subgroups differ in excitation vs. inhibition of the periphery has yet to be completely worked out. Our knowledge of MOC anatomy has advanced so that the basic reflex pathway is established although modulatory inputs and their roles are less well defined. The locations of MOC neurons responsive to sound in either ear is not indicated in Fig. 2.8 because they have not been established; similarly, how response characteristics of MOC neurons vary with brain stem location is not known. LOC neurons probably also participate in a sound-evoked LOC reflex, but the participating elements in cochlear nucleus and even whether it is a three-neuron or four-neuron pathway remains to be resolved. Further work on both groups of OC neurons will better establish their anatomical characteristics and roles in hearing.
References
Arnesen AR (1984) Fibre population of the vestibulocochlear anastomosis in humans. Acta Otolaryngol 98:501–518
Arnesen AR, Osen KK (1984) Fibre spectrum of the vestibulo-cochlear anastomosis in the cat. Acta Otolaryngol 98:255–269
Aschoff A, Ostwald J (1987) Different origins of cochlear efferents in some bat species, rats, and guinea pigs. J Comp Neurol 264:56–72
Azeredo WJ, Kliment ML, Morley BJ, Relkin E, Slepecky NB, Sterns A, Warr WB, Weekly JM, Woods CI (1999) Olivocochlear neurons in the chinchilla: A retrograde fluorescent labelling study. Hear Res 134:57–70
Benson TE, Brown MC (1990) Synapses formed by olivocochlear axon branches in the mouse cochlear nucleus. J Comp Neurol 295:52–70
Benson TE, Brown MC (1996) Synapses from medial olivocochlear branches in the inferior vestibular nucleus. J Comp Neurol 372:176–188
Benson TE, Brown MC (2006) Ultrastructure of synaptic input to medial olivocochlear neurons. J Comp Neurol 499:244–257
Bishop AL, Henson OW Jr (1987) The efferent cochlear projections of the superior olivary complex in the mustached bat. Hear Res 31:175–182
Bourne JN, Harris KM (2008) Balancing structure and function at hippocampal dendritic spines. Annu Rev Neurosci 31:47–67
Brown MC (1987a) Morphology of labeled efferent fibers in the guinea pig cochlea. J Comp Neurol 260:605–618
Brown MC (1987b) Morphology of labeled afferent fibers in the guinea pig cochlea. J Comp Neurol 260:591–604
Brown MC (1989) Morphology and response properties of single olivocochlear fibers in the guinea pig. Hear Res 40:93–110
Brown MC (1993) Fiber pathways and branching patterns of biocytin-labeled olivocochlear neurons in the mouse brainstem. J Comp Neurol 337:600–613
Brown MC (2001) Response adaptation of medial olivocochlear neurons is minimal. J Neurophysiol 86:2381–2392
Brown MC (2002) Cochlear projections of single medial olivocochlear (MOC) axons in the guinea pig. ARO Absts 25:310
Brown MC, Levine JL (2008) Dendrites of medial olivocochlear (MOC) neurons in mouse. Neuroscience 154:147–159
Brown MC, Vetter DE (2008) Olivocochlear somata and central branches are normal in α9 knockout mice. J Assoc Res Otolaryngol 10:64–75
Brown MC, Liberman MC, Benson TE, Ryugo DK (1988) Brainstem branches from olivocochlear axons in cats and rodents. J Comp Neurol 278:591–603
Brown MC, de Venecia RK, Guinan JJ Jr (2003) Responses of medial olivocochlear (MOC) neurons: Specifying the central pathways of the MOC reflex. Exp Brain Res 153:491–498
Campbell JP, Henson MM (1988) Olivocochlear neurons in the brainstem of the mouse. Hear Res 35:271–274
Darrow KN, Simons EJ, Dodds L, Liberman MC (2006) Dopaminergic innervation of the mouse inner ear: Evidence for a separate cytochemical group of cochlear efferent fibers. J Comp Neurol 498:403–414
de Boer E, Thornton AR (2008) Neural correlates of perceptual learning in the auditory brainstem: efferent activity predicts and reflects improvement at a speech-in-noise discrimination task. J Neurosci 28:4929–4937
de Venecia RK, Liberman MC, Guinan JJ Jr, Brown MC (2005) Medial olivocochlear reflex interneurons are located in the posteroventral cochlear nucleus. J Comp Neurol 487: 345–360
Doucet JR, Ryugo DK (2003) Axonal pathways to the lateral superior olive labeled with biotinylated dextran amine injections in the dorsal cochlear nucleus in rats. J Comp Neurol 461:452–465
Faye-Lund H (1986) Projection from the inferior colliculus to the superior olivary complex in the albino rat. Anat Embryol (Berl) 175:35–52
Fujino K, Oertel D (2001) Cholinergic modulation of stellate cells in the mammalian ventral cochlear nucleus. J Neurosci 21:7372–7383
Guinan JJ Jr, Warr WB, Norris BE (1983) Differential olivocochlear projections from lateral vs. medial zones of the superior olivary complex. J Comp Neurol 221:358–370
Guinan JJ Jr, Warr WB, Norris BE (1984) Topographic organization of the olivocochlear projections from the lateral and medial zones of the superior olivary complex. J Comp Neurol 226:21–27
Hackney CM, Osen KK, Kolston J (1990) Anatomy of the cochlear nuclear complex of the guinea pig. Anat Embryol 182:123–149
Happe HK, Morely BJ (1998) Nicotinic acetylcholine receptors in rat cochlear nucleus: [125I]-a-bungarotoxin receptor autoradiography and in situ receptor autoradiography of a7 nAChR subunit mRNA. J Comp Neurol 397:163–180
Helfert RH, Schwartz IR, Ryan AF (1988) Ultrastructural characterization of gerbil olivocochlear neurons based on differential uptake of 3H-d-aspartic acid and a wheatgerm agglutinin-horseradish peroxidase conjugate from the cochlea. J Neurosci 8:3111–3123
Horvath M, Kraus KS, Illing R-B (2000) Olivocochlear neurons sending axon collaterals into the ventral cochlear nucleus of the rat. J Comp Neurol 422:95–105
Horvath M, Ribari O, Repassy G, Toth IE, Boldogkoi Z, Palkovits M (2003) Intracochlear injection of pseudorabies virus labels descending auditory and monoaminerg projections to olivocochlear cells in guinea pig. Eur J Neurosci 18:1439–1447
Iurato S, Smith CA, Eldredge DH, Henderson D, Carr C, Ueno Y, Cameron S, Richter R (1978) Distribution of the crossed olivocochlear bundle in the chinchilla’s cochlea. J Comp Neurol 182:57–76
Kiang NYS, Rho JM, Northrop CC, Liberman MC, Ryugo DK (1982) Hair-cell innervation by spiral ganglion cells in adult cats. Science 217:175–177
Kujawa S, Liberman MC (2001) Effects of olivocochlear feedback on distortion product otoacoustic emissions in guinea pig. J Assoc Res Otolaryngol 2:268–278
Liberman MC (1980) Efferent synapses in the inner hair cell area of the cat cochlea: An electron microscopic study of serial sections. Hear Res 3:189–204
Liberman MC, Brown MC (1986) Physiology and anatomy of single olivocochlear neurons in the cat. Hear Res 24:17–36
Liberman MC, Guinan JJ Jr (1998) Feedback control of the auditory periphery: Anti-masking effects of middle ear muscles vs. olivocochlear efferents. J Commun Disord 31:471–483
Liberman MC, Dodds LW, Pierce S (1990) Afferent and efferent innervation of the cat cochlea: Quantitative analysis with light and electron microscopy. J Comp Neurol 301:443–460
Maison SF, Adams JC, Liberman MC (2003) Olivocochlear innervation in the mouse: Immunocytochemical maps, crossed versus uncrossed contributions, and transmitter colocalization. J Comp Neurol 455:406–416
Matsuzaki M (2007) Factors critical for the plasticity of dendritic spines and memory storage. Neurosci Rev 57:1–9
Moore JK, Osen KK (1979) The cochlear nuclei in man. Am J Anat 154:393–418
Mulders WHAM, Robertson D (2000a) Evidence for direct cortical innervation of medial olivocochlear neurones in rats. Hear Res 144:65–72
Mulders WHAM, Robertson D (2000b) Morphological relationships of peptidergic and noradrenergic nerve terminals to olivocochlear neurones in the rat. Hear Res 144:53–64
Mulders WHAM, Robertson D (2005) Catecholaminergic innervation of guinea pig superior olivary complex. J Chem Neuroanat 30:230–242
Mulders WHAM, Harvey AR, Robertson D (2007) Electrically-evoked responses in onset chopper neurons in guinea pig cochlear nucleus. J Neurophysiol 97:3288–3297
Nadol JB Jr (1988) Comparative anatomy of the cochlea and auditory nerve in mammals. Hear Res 34:253–266
Oliver DL (1987) Projections to the inferior colliculus from the anteroventral cochlear nucleus in the cat: Possible substrates for binaural interaction. J Comp Neurol 264:24–46
Osen KK (1969) Cytoarchitecture of the cochlear nuclei in the cat. J Comp Neurol 136:453–484
Osen KK, Mugnaini E, Dahl A-L, Christiansen AH (1984) Histochemical localization of acetylcholinesterase in the cochlear and superior olivary nuclei. A reappraisal with emphasis on the cochlear granule cell system. Arch Ital Biol 122:169–212
Rasmussen GL (1946) The olivary peduncle and other fiber connections of the superior olivary complex. J Comp Neurol 84:141–219
Rasmussen GL (1953) Further observations of the efferent cochlear bundle. J Comp Neurol 99:61–94
Rhode WS, Oertel D, Smith PH (1983) Physiological response properties of cells labeled intracellularly with horseradish peroxidase in cat ventral cochlear nucleus. J Comp Neurol 213:448–463
Robertson D, Gummer M (1985) Physiological and morphological characterization of efferent neurons in the guinea pig cochlea. Hear Res 20:63–77
Robertson D, Winter IM (1988) Cochlear nucleus inputs to olivocochlear neurones revealed by combined anterograde and retrograde labelling in the guinea pig. Brain Res 462:47–55
Robertson D, Cole KS, Corbett K (1987a) Quantitative estimate of bilaterally projecting medial olivocochlear neurons in the guinea pig brainstem. Hear Res 27:177–181
Robertson D, Anderson C-J, Cole KS (1987b) Segregation of efferent projections to different turns of the guinea pig cochlea. Hear Res 25:69–76
Rouiller EM, Ryugo DK (1984) Intracellular marking of physiologically characterized cells in the ventral cochlear nucleus of the cat. J Comp Neurol 225:167–186
Ryan AF, Keithley EM, Wang Z-X, Schwartz IR (1990) Collaterals from lateral and medial olivocochlear efferent neurons innervate different regions of the cochlear nucleus and adjacent brainstem. J Comp Neurol 300:572–582
Sánchez-González MA, Warr WB, López DE (2003) Anatomy of olivocochlear neurons in the hamster studied with FluoroGold. Hear Res 185:65–76
Schuknecht HF, Nomura Y (1965) The efferent fibers in the cochlea. Ann Otol Rhinol Laryngol 74:289–303
Smith PH, Joris PX, Carney LH, Yin TCT (1991) Projections of physiologically characterized globular bushy cell axons from the cochlear nucleus of the cat. J Comp Neurol 304:387–407
Smith PH, Joris PX, Banks MI, Yin TCT (1993) Responses of cochlear nucleus cells and projections of their axons. In: Merchan MA, Juiz JM, Godfrey DA, Mugnaini E (eds) The mammalian cochlear nuclei: Organization and function. Plenum Press, New York, pp 349–360
Spangler KM, White JS, Warr WB (1986) Electron microscopic features of axon terminals on olivocochlear neurons in the cat. Assoc Res Otolaryngol Abstr 9:37–38
Spirou GA, Brownell WE, Zidanic M (1990) Recordings from cat trapezoid body and HRP labeling of globular bushy cell axons. J Neurophysiol 63:1169–1190
Spoendlin H (1971) Degeneration behaviour of the cochlear nerve. Arch Klin Exp Ohren Nasen Kehlkopfheilkd 200:275–291
Stopp PE (1983) The distribution of the olivocochlear bundle and its possible role in frequency/intensity coding. In: Klinke R, Hartmann R (eds) Hearing-physiological bases and physchophysics. Springer, Berlin, pp 176–179
Thiers FA, Burgess BJ, Nadol JB Jr (2002) Axodendritic and dendrodendritic synapses within outer spiral bundles in a human. Hear Res 164:97–104
Thompson GC, Thompson AM (1986) Olivocochlear neurons in the squirrel monkey brainstem. J Comp Neurol 254:246–258
Thompson AM, Thompson GC (1991) Posteroventral cochlear nucleus projections to olivocochlear neurons. J Comp Neurol 303:267–285
Thompson AM, Thompson GC (1993) Relationship of descending inferior colliculus projections to olivocochlear neurons. J Comp Neurol 335:402–412
Thompson AM, Thompson GC (1995) Light microscopic evidence of serotoninergic projections to olivocochlear neurons in the bush baby (Otolemur garnettii). Brain Res 695:263–266
Uchizono K (1965) Characteristics of excitatory and inhibitory synapses in the central nervous system of the cat. Nature (Lond) 207:642–643
Vetter DE, Mugnaini E (1992) Distribution and dendritic features of three groups of rat olivocochlear neurons. A study with two retrograde cholera toxin tracers. Anat Embryol (Berl) 185:1–16
Vetter DE, Adams JC, Mugnaini E (1991) Chemically distinct rat olivocochlear neurons. Synapse 7:21–43
Vetter DE, Saldana E, Mugnaini E (1993) Input from the inferior colliculus to medial olivocochlear neurons in the rat: A double label study with PHA-L and cholera toxin. Hear Res 70:173–186
Vetter DE, Liberman MC, Mann J, Barhanin J, Boulter J, Brown MC, Saffiote-Kolman J, Heinemann SF, Elgoyhen AB (1999) Role of a9 nicotinic ACh receptor subunits in the development and function of cochlear efferent innervation. Neuron 23:93–103
Wang X, Robertson D (1997a) Effects of bioamines and peptides on neurones in the ventral nucleus of the trapezoid body and rostral periolivary regions of the rat superior olivary complex: An in vitro investigation. Hear Res 106:20–28
Wang X, Robertson D (1997b) Two types of actions of noradrenaline on identified auditory efferent neurons in rat brainstem slices. J Neurophysiol 78:1800–1810
Warr WB (1975) Olivocochlear and vestibular efferent neurons of the feline brainstem: Their location, morphology, and number determined by retrograde axonal transport and acetylcholinesterase histochemistry. J Comp Neurol 161:159–182
Warr WB (1992) Organization of olivocochlear efferent systems in mammals. In: Webster DB, Popper AN, Fay RR (eds) The mammalian auditory pathway: Neuroanatomy. Springer, New York, pp 410–448
Warr WB, Guinan JJ Jr (1979) Efferent innervation of the organ of Corti: Two separate systems. Brain Res 173:152–155
Warr WB, Beck Boche JE, Neely ST (1997) Efferent innervation of the inner hair cell region: Origins and terminations of two lateral olivocochlear systems. Hear Res 108:89–111
Warr WB, Boche JEB, Ye Y, Kim DO (2002) Organization of olivocochlear neurons in the cat studied with the retrograde tracer cholera toxin-B. J Assoc Res Otolaryngol 3:457–478
Warren EH III, Liberman MC (1989) Effects of contralateral sound on auditory-nerve responses I. Contributions of cochlear efferents. Hear Res 37:89–104
White JS (1984) Fine structure of medial olivocochlear neurons in the rat. Soc Neurosci Abstr 10:393
White JS (1986) Differences in the ultrastructure of labyrinthine efferent neurons in the albino rat. ARO Abstr 9:34–35
Winter IM, Robertson D, Cole KS (1989) Descending projections from auditory brainstem nuclei to the cochlea and cochlear nucleus of the guinea pig. J Comp Neurol 280:143–157
Yao W, Godfrey DA (1999) Immunolocalization of alpha4 and alpha7 subunits of nicotinic receptor in rat cochlear nucleus. Hear Res 128:97–102
Ye Y, Machado DG, Kim DO (2000) Projection of the marginal shell of the anteroventral cochlear nucleus to olivocochlear neurons in the cat. J Comp Neurol 420:127–138
Acknowledgments
I thank Dr. M. Charles Liberman for comments on the manuscript and Ms. Marie Drottar for assistance with the figures. This work was Supported by NIH grant DCD 01089.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2011 Springer Science+Business Media, LLC
About this chapter
Cite this chapter
Brown, M.C. (2011). Anatomy of Olivocochlear Neurons. In: Ryugo, D., Fay, R. (eds) Auditory and Vestibular Efferents. Springer Handbook of Auditory Research, vol 38. Springer, New York, NY. https://doi.org/10.1007/978-1-4419-7070-1_2
Download citation
DOI: https://doi.org/10.1007/978-1-4419-7070-1_2
Published:
Publisher Name: Springer, New York, NY
Print ISBN: 978-1-4419-7069-5
Online ISBN: 978-1-4419-7070-1
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)