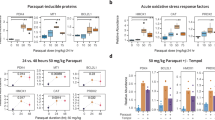
The “oxygen paradox” is that molecular oxygen is both essential for aerobic life but also can be toxic to cells largely because of the effects of oxygen-derived species collectively called “reactive oxygen species” (ROS) such as the hydroxyl radical. Cells have evolved elaborate defences against ROS but if these defences are decreased (as in ageing) or if the ROS challenge becomes too great (as in toxicity), a state of oxidative stress (OS) ensues. Proteins are the principal targets of ROS and redox proteomics uses proteomics tools to study redox-based effects on the cell’s protein complement. We have long used bivalve molluscs as sentinel organisms for study of pollution effects in estuaries, in particular looking at effects on stress-response proteins such as antioxidative enzymes, detoxification enzymes and heat shock proteins. Stress-response proteins are often affected by more than one stressor so these targets are likely to be of interest in other stress contexts. We are now applying redox proteomics approaches to study stress effects in bivalves. We detect carbonylation, glutathionylation, ubiquitination, effects on disulphide bridge patterns and changes in protein expression signatures in a range of electrophoresis formats. The effects are tissue- and treatment-specific. We find that many proteins targeted by OS are associated with either actin or protein disulphide isomerase. Many of the tools we use are species-independent and are appropriate for other stress scenarios.
Access provided by Autonomous University of Puebla. Download to read the full chapter text
Chapter PDF
Similar content being viewed by others
Keywords
- Mytilus Edulis
- Disulphide Bridge
- Aquatic Toxicology
- Protein Disulphide Isomerase
- Marine Environmental Research
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
References
Baty, JW, Hampton, MB, Winterburn, CC 2002. Detection of oxidant sensitive thiol proteins by fluorescence labelling and two dimensional electrophoresis. Proteomics 2: 1261–1266.
Bigelow, DJ, Squier, TC 2005. Redox modulation of cellular signalling and metabolism through reversible oxidation of methionine sensors in calcium regulatory proteins. Biochimica et Biophysica Acta 1703: 121–134.
Bota, DA, Van Remmen, H, Davies, KJA 2002. Modulation of Lon protease activity and aconitase turnover during ageing and oxidative stress. FEBS Letters 532: 103–106.
Bradley, BP, Shrader, EA, Kimmel, DG, Meiller, JC 2002. Protein expression signatures: an application of proteomics. Marine Environmental Research 54: 373–377.
Brooker, RJ, Slayman, CW 1983. [14C] N-ethylmaleimide labelling of the plasma membrane [H+]-ATP-ase of Neurospora crassa. Journal of Biological Chemistry 258: 222–226.
Dalle-Donne, I, Rossi, R, Milzani, A, Di Simplicio, P, Colombo, R 2001. The actin cytoskeleton response to oxidants: from small heat shock protein phosphorylation to changes in the redox state of actin itself. Free Radical Biology and Medicine 31: 1624–1632.
Davies, KJA 2001. Degradation of oxidized proteins by the 20S proteasome. Biochimie 83: 301–310.
Davies, MJ 2005. The oxidative environment and protein damage. Biochimica et Biophysica Acta 1703: 93–109.
Dowling, V, Sheehan, D 2006. Proteomics as a route to identification of toxicity targets in ecotoxicology Proteomics 6: 5597–5604.
Dowling, V, Hoarau, P, Romeo, M, O’Halloran, J, van Pelt, FNAM, O’Brien, NM, Sheehan, D 2006. Protein carbonylation and heat shock response in Ruditapes decussatus following p, p'-dichlorodiphenyldichloroethylene (DDE) exposure: a proteomic approach reveals DDE causes oxidative stress. Aquatic Toxicology 77: 11–18.
Eaton, P 2006. Protein thiol oxidation in health and disease: techniques for measuring disulfides and related modifications in complex protein mixtures. Free Radical Biology and Medicine 40: 1889–1899.
Eaton, P, Byers, HL, Leeds, N, Ward, MA, Shattock, MJ 2002. Detection, quantitation, purification, and identification of cardiac proteins S-thiolated during ischemia and reperfusion. Journal of Biological Chemistry 277: 9806–9811.
England, K, Cotter, T 2004. Identification of carbonylated proteins by MALDI-TOF mass spectroscopy reveals susceptibility of ER. Biochemical and Biophysical Research Communications 320: 123–130.
Friguet B 2006. Oxidized protein degradation and repair in ageing and oxidative stress. FEBS Letters 580: 2910–2916.
Görg, A 1991.Two-dimensional electrophoresis. Nature 349: 545–546.
Guo, ZY, Chang, CCY, Lu, XH, Chen, J, Chang, TY 2005. The disulfide linkage and the free sulfhydryl accessibility of acyl-coenzyme A: cholesterol acyltransferase 1 as studied by using mPEG5000-maleimide. Biochemistry 44: 6537–6546.
Halliwell, B 1996. Oxidative stress, nutrition and health: experimental strategies for optimization of nutritional antioxidant intake in humans. Free Radical Research 25: 57–74.
Halliwell, B, Gutteridge, JMC 2007. Free Radicals in Biology and Medicine Fourth Edition Oxford University Press, Oxford.
He, QY, Chiu, JF 2003. Proteomics in biomarker discovery and drug development. Journal of cellular biochemistry 89: 868–886.
Holland, HD 1994. Early proterozoic atmospheric change. In: Early Life on Earth, Nobel Symposium No. 84: 237–244.
Jaffrey, SR, Erdjument-Bromage, H, Ferris, CD, Tempst, P, Snyder, SH 2001. Protein S-nitrosylation: a physiological signal for neuronal nitric oxide. Nature Cell Biology 3: 193–197.
Levine, RL, Stadtman, ER 2001. Oxidative modification of proteins during aging. Experimental Gerontology 36: 1495–1502.
Levine, RL, Garland, D, Oliver, CN, Amici, A, Climent, I, Lenz, A, Ahn, BW, Shaltiel, S, Stadtman, ER 1990. Determination of carbonyl content in oxidatively modified proteins. Methods in Enzymology 186: 464–478.
Marques, C, Pereira, P, Taylor, A, Liang, JN, Reddy, VN, Szweda, LI, Shang, F 2004. Ubiquitin-dependent lysosomal degradation of the HNE-modified proteins in lens epithelial cells. FASEB Journal 18: 1424–1426.
Masella, R, Di Benedetto, R, Vari, R, Filesi, C, Giovannini, C 2005. Novel mechanisms of natural antioxidant compounds in biological systems: involvement of glutathione and glutathione-related enzymes. Journal of Nutritional Biochemistry 16: 577–586.
Mates, JM, Sanchez-Jimenez, FM 2000. Role of reactive oxygen species in apoptosis: implications for cancer therapy. International Journal of Biochemistry and Cell Biology 32: 157–170.
McDonagh, B, Sheehan, D 2006. Redox proteomics in the blue mussel Mytilus edulis: carbonylation is not a pre-requisite for ubiquitination in acute free radical-mediated oxidative stress. Aquatic Toxicology 79: 325–333.
McDonagh, B, Tyther, R, Sheehan, D 2006. Redox proteomics in the mussel Mytilus edulis. Marine Environmental Research 62: S101–104.
McDonagh, B, Tyther, R, Sheehan, D 2005. Carbonylation and glutathionylation of proteins in the blue mussel Mytilus edulis detected by proteomic analysis and Western blotting: actin as a target for oxidative stress. Aquatic Toxicology 73: 315–326.
O’Farrell, PH 1975. High-resolution two-dimensional electrophoresis of proteins. Journal of Biological Chemistry 250: 4007–4021.
Patton, WF 2002. Detection technologies in proteome analysis. Journal of Chromatography. B–Analytical Technologies in the Biomedical and Life Sciences 771: 3–31.
Petroski, MD, Deshaies, RJ 2005. Mechanism of lysine-48-linked ubiquitin chain synthesis by the cullin-RING ubiquitin-ligase complex SCF-Cdc34. Cell 123: 1107–1120.
Romero-Ruiz, A, Carrascal, M, Alhama, J, Gomez-Ariza, JL, Albian, J, Lopez-Barea, J 2006. Utility of proteomics to assess pollutant response of clams from the Donana bank of Guadalquivir estuary (SW Spain). Proteomics 6: S245–S255.
Shepard, JL, Olsson, M, Tedengren, M, Bradley, BP 2000. Protein expression signatures identified in Mytilus edulis exposed to PCBs, copper and salinity stress. Marine Environmental Research 50: 337–340.
Valavanidis, A, Vlahogianni, T, Dassenakis, M, Scoullos, M 2006. Molecular biomarkers of oxidative stress in aquatic organisms in relation to toxic environmental pollutants. Ecotoxicology and Environmental Safety 64: 178–189.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2007 Springer
About this paper
Cite this paper
Sheehan, D., Tyther, R., Dowling, V., McDonagh, B. (2007). Redox Proteomics – A Route to the Identifi cation of Damaged Proteins. In: Mothersill, C., Mosse, I., Seymour, C. (eds) Multiple Stressors: A Challenge for the Future. NATO Science for Peace and Security Series. Springer, Dordrecht. https://doi.org/10.1007/978-1-4020-6335-0_20
Download citation
DOI: https://doi.org/10.1007/978-1-4020-6335-0_20
Publisher Name: Springer, Dordrecht
Print ISBN: 978-1-4020-6333-6
Online ISBN: 978-1-4020-6335-0
eBook Packages: Earth and Environmental ScienceEarth and Environmental Science (R0)