Abstract
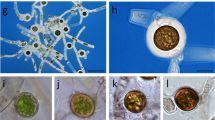
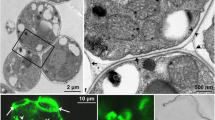
The attachment of spores to a substratum is essential for their germination and, therefore, to the completion of the life cycle of the red algae. In most red algae, spores are liberated without a cell wall, within a sheath of mucilage which is responsible for their primary attachment. Utilizing fluorescent-labeled lectins, we identified carbohydrate residues and their locations in the mucilage and cell walls of spores of Gelidium floridanum. Cell wall formation and mucilage composition were studied with calcofluor, toluidine blue — O (AT-O), alcian blue (AB) and periodic acid-Schiff (PAS). In the mucilage we identified α-D mannose, α-D glucose, β-D-galactose, N-acetyl-glucosamine and N-acetyl-galactosamine. The first two sugar residues were not found in the cell wall of the germ tube but they were present on the rhizoid’s cell wall indicating their importance to substrate adhesion. A cell wall is produced soon after the spore’s attachment, beginning with a polar deposition of cellulose and its gradual spread around the spore as indicated by calcofluor. The cell wall matrix was positive to AB and metachromatic to AT-O, indicating acidic polysaccharides, while cellulose microfibrills were positive to PAS. A polar disorganization of the cell wall triggers the process of germination. As spores are the natural form of propagation of Gelidium, the understanding of the mechanisms of spore attachment may contribute to the cultivation of this valuable seaweed.
Access provided by Autonomous University of Puebla. Download to read the full chapter text
Chapter PDF
Similar content being viewed by others
References
Apple ME, Harlin MM, Norris JH (1996) Characterization of Champia parvula (Rhodophyta) tetraspore mucilage end rhizoids with histochemical stains and FITC-labeled lectins. Phycologia 35:245–252
Avanzini A (1989) La ultraestructura de las esporas de Rhodophyta. Insula 19:7–10
Callow JA(1985) Sexual recognition and fertilization in brown algae. J Cell Sci 2:219–232
Chamberlain AHL, Evans LV (1973) Aspects of spore production in the red alga Ceramium. Protoplasma 76:139–159
Chamberlain AHL, Evans LV (1981) Chemical and histochemical studies on the spore adhesive of Ceramium. In: Fogg, GE, Jones, WE (eds) Proceedings of the 8th International Seaweed Symposium, Menai Bridge, pp 539–542
Cole KM, Park CM, Reid PE, Sheath RG (1985) Comparative studies in the cell walls of sexual and asexual Bangia atropurpurea (Rhodophyta): I. Histochemistry of polysaccharides. J Phycol 21:585–592
Costas E, López-Rodas V (1994) Identification of marine dinoflagellates using fluorescent lectins. J Phycol 30:987–990
Costas E, Gonzalez-Chavarri E, Aguilera E, Gonzalez A, Gil S, Lopez Rodas V (1993) Use of lectins to recognize and differentiate unicellular algae. Bot Marina 36:1–4
Gahan PB (1984) Plant histochemistry and cytochemistry: an introduction. Academic Press, London
Gordon-Mills EM, McCandless EC (1978) Carrageenans in the cell walls of Chondrus crispus Stack. (Rhodophyceae, Gigartinales): III. Metachromasia and the topootical reaction. Phycologia 17:95–104
Hommersand MH, Fredericq S (1988) An investigation of cystocarp development in Gelidium pteridifolium with a revised description of the Gelidiales (Rhodophyta). Phycologia 27:254–272
Hori K, Ogata T, Kamiya H, Mimuro M (1996) Lectin-like compounds and lectin receptors in marine microalgae: hemagglutination and reactivity with purified lectins. J Phycol 32:783–790
Kim GH, Fritz L (1993) Ultrastucture and cytochemistry of early spermatangial development in Antithamnion nipponicum (Ceramiaceae, Rhodophyta). J Phycol 29:797–805
Northcote DH, Pickett-Heaps JD (1965) A function of the Golgi apparatus in polysaccharide synthesis and transport in the root cap cells of wheat. Biochem J 98:159–167
Pueschel CM (1979) Ultrastructure of tetrasporogenesis in Palmaria palmata (Rhodophyta). J Phycol 15:409–424
Ramus J (1974) In vitro molybdate inhibition of sulfate transfer to Porphyridium capsular polysaccharide. Plant Physiol 54:945–949
Rascio N, Mariani P, Vecchia FD, Trevisan R (1991) The vegetative thallus of Pterocladia capillacea (Gelidiales, Rhodophyta): II. Pit connections. Bot Marina 34:187–194
Ravetto C (1964) Alcian blue-alcian yellow: a new method for the identification of different acidic groups. J Histochem Cytochem 12:44–45
Villemez CL, MacNab JC, Albersheim P (1968) Formation of plant cell wall polysaccharides. Nature 218:878–880
von Sengbusch P, Müller U (1983) Distribution of glycoconjugates at algal cell surfaces as monitored by FITC-conjugates lectins. Studies on selected species from Cyanophyta, Pyrrhophyta, Raphidophyta, Euglenophyta, Chromophyta and Chlorophyta. Protoplasma 114:103–113
von Sengbusch P, Mix M, Wachholz I, Manshard E (1982) FITC labeled lectins and calcofluor white ST as probes for the investigation of the molecular architecture of cell surfaces. Studies on Conjugatophycean species. Protoplasma 111:38–52
Walko RM, Furtula V, Nothnagel EA (1987) Analysis of labeling of plant protoplast surface by fluorophore-conjugated lectins. Protoplasma 141:33–46
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2006 Springer
About this paper
Cite this paper
Bouzon, Z.L., Ouriques, L.C., Oliveira, E.C. (2006). Spore adhesion and cell wall formation in Gelidium floridanum (Rhodophyta, Gelidiales). In: Anderson, R., Brodie, J., Onsøyen, E., Critchley, A.T. (eds) Eighteenth International Seaweed Symposium. Developments in Applied Phycology, vol 1. Springer, Dordrecht. https://doi.org/10.1007/978-1-4020-5670-3_8
Download citation
DOI: https://doi.org/10.1007/978-1-4020-5670-3_8
Publisher Name: Springer, Dordrecht
Print ISBN: 978-1-4020-5669-7
Online ISBN: 978-1-4020-5670-3
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)