A regulatory network comprised of transcription factors PU.1, Ikaros, E2A, EBF, and Pax5 control B cell fate specification and differentiation. Early B Cell Factor-1 (EBF-1) is essential for B cell fate specification while Pax5 is required for B cell development. Mice deficient in Pax5 or EBF-1 have a developmental arrest of B cell differentiation at the pro-B cell stage, which results in the absence of mature B cells. We analyzed the bone phenotype of Pax5 and EBF-1 wild-type (+/+) and homozygous mutant (–/–) mice to determine if the loss of these transcription factors regulated bone cell development.
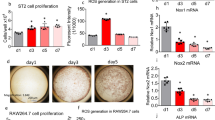
Bones from Pax5–/– mice were strikingly osteopenic 15 days after birth, with increased numbers of osteoclasts, and decreased trabecular number. The number of osteoblasts in Pax5–/– bones and their function in vitro were not different from controls. In addition, Pax5 was not expressed by wild-type osteoblasts. To investigate the origin of the in vivo increase in osteoclasts, Pax5–/– or +/+ spleen cells were cultured with M-CSF and RANKL and multinucleated, TRAP+ cells counted. Cells from Pax5–/– spleen produced 5-10 times more osteoclasts than did controls.
Tibia from EBF-1–/– mice had a striking increase in osteoblasts lining bone surfaces. Consistent with this was an increase in osteoid thickness and in the bone formation rate. This correlated with a 2-fold increase in serum osteocalcin. However, in vitro proliferation and ALP of mutant osteoblasts did not differ from control. In contrast, osteoclast number was similar in 4 week-old +/+ and –/– mice; however, at 12 weeks the number of osteoclasts was more than twice that of controls. These data correlated with an increase in bone volume at 12 weeks of age. The most striking aspect of the EBF-1–/– bones was the presence of adipocytes, which filled the marrow space. The adipocytes in the marrow were present at both 4 and 12 weeks of age. Increased fat was also seen in the liver of mutant mice. However, subcutaneous fat was almost absent in EBF-1–/– mice. Importantly, EBF-1 mRNA was expressed in wild-type osteoblasts and in adipocytes. Loss of EBF-1 and Pax5 causes distinct, non-overlapping bone phenotypes. It is important to understand why this network of transcription factors, which are so important for B cell development, have such striking effects on bone cell growth and development.
Access provided by Autonomous University of Puebla. Download to read the full chapter text
Chapter PDF
Similar content being viewed by others
Keywords
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
References
Adams, B., P. Dorfler, A. Aguzzi, Z. Kozmik, P. Urbanek, I. Maurer-Fogy, and M. Busslinger. 1992. Pax5 encodes the transcription factor BSAP and is expressed in B-lymphocytes, the developing CNS, and adult testis. Genes Dev 6: 1589–1607.
Akerblad, P., U. Lind, D. Liberg, K. Bamberg, and M. Sigvardsson. 2002. Early B-cell factor (O/E-1) is a promoter of adipogenesis and involved in control of genes important for terminal adipocyte differentiation. Mol Cell Biol 22: 8015–8025.
Baldwin, C.T., C.F. Hoth, J.A. Amos, E.O. da-Silva, and A. Milunsky. 1992. An exonic mutation in the HuP2 paired domain gene causes Waardenburg’s syndrome. Nature 355: 637–638.
Balling, R., U. Deutsch, and P. Gruss. 1988. Undulated, a mutation affecting the development of the mouse skeleton, has a point mutation in the paired box of Pax1. Cell 55: 531–535.
Baumgartner, M., D. Bopp, M. Burri, and L.M. Nol. 1987. Structure of two genes at the gooseberry locus related to the paired gene and their spatial expression during Drosophila embryogenesis. Genes Dev 1: 1247–1267.
Bopp, D., M. Burri, S. Baumgartner, G. Frigerio, and M. Noll. 1986. Conservation of a large protein domain in the segmentation gene paried and in functionally related genes of Drosphila. Cell 47: 1033–1040.
Burri, M., Y. Tromvoulis, D. Bopp, G. Frigerio, and L.M. Nol. 1989. Conservation of the paired domain in metazoans and its structure in three isolated human genes. EMBO J 8: 1183–1190.
Campos-Ortega, J.A. 1998. The genetics of the Drosophila achaete-scute gene complex: a historical appraisal. Int J Dev Biol 42: 291–297.
Crozatier, M., D. Valle, L. Dubois, S. Ibnsouda, and A. Vincent. 1996. Collier, a novel regulator of Drosophila head development, is expressed in a single mitotic domain. Curr. Biol 6: 707–718.
Dambly-Chaudiere, C., and M. Vervoort. 1998. The bHLH genes in neural development. Int J Dev Biol. 42: 269–273.
Dowell, P., and D.W. Cooke. 2002. Olf-1/early B cell factor is a regulator of gult4 gene expression in 3T3–L1 adipocytes. J Biol Chem 277: 1712–1718.
Epstein, D.J., M. Vekemans, and P. Gros. 1991. Splotch (Sp2H), a mutation affecting development of the mouse neural tube, shows a deletion within the paired homeodomain of Pax3. Cell 57: 767–774.
Garel, S., F. Marin, M.G. Mattei, C. Vesque, A. Vincent, and P. Charnay. 1997. Family of Ebf/Olf-1-related genes potentially involved in neuronal differentiation and regional specification in the central nervous system. Dev Dyn 210: 191–205.
Georgopoulos, K., M. Bigby, J-H. Want, A. Molnar, P. Wu, S. Winandy, and A. Sharpe. 1994. The Ikaros gene is required for the development of all lymphoid lineages. Cell 79: 143–156.
Hagman, J., C. Belanger, A. Travis, C.W. Turck, and R. Grosschedl. 1993. Cloning and functional characterization of early B-cell factor, a regulator of lymphocyte-specific gene expression. Genes Dev 7: 760–773.
Hardy, R.R., C.E. Carmack, S.A. Shinton, J.D. Kemp, and K. Hayakawa. 1991. Resolution, and characterization of pro-B and Pre-pro-B cell stages in normal mouse bone marrow. J Exp Med 173: 1213–1225.
Hata, A., J. Seoane, G. Lagna, E. Montalvo, A. Hemmati-Brivanlou, and J. Massague. 2000. OAZ uses distinct DNA- and protein-binding zinc fingers in separate BMP-Smad and Olf signaling pathways. Cell 100: 229–240.
Hill, E.R., J. Favor, B.L.M. Hogan, C.C. Ton, G.F Saunders, I.M. Hanson, J. Prosser, T. Jordan, N.D. Hastie, and V. van Heyningen. 1991. Mouse Small eye results from mutations in a paired-like homeobox-containing gene. Nature 354: 522–525.
Hofbauer, L.C., and A.E. Heufelder. 2000. The role of receptor activator of nuclear factor-HB ligand and osteoprotegerin in the pathogenesis and treatment of metabolic bone diseases. J Clin Endocrinol Metab 85(7): 2355–2363.
Horowitz, M.C., Y. Xi, D.L. Pflugh, D.G.T. Hesslein, D.G. Schatz, J.A. Lorenzo, and A.L.M. Bothwell. 2004. Pax5 deficient mice exhibit early onset osteopenia with increased osteoclast progenitors. J Immunol 173: 6583–6591.
Horowitz, M.C., A.L.M. Bothwell, D.G.T. Hesslein, D.L. Pflugh, and D.G. Schatz. 2005. B cells and osteoblast and osteoclast development. Immunol Rev 208: 141–153.
Lin, H., and R. Grosschedl. 1995. Failure of B-cell differentiation in mice lacking the transcription factor EBF. Nature 376: 263–267.
O’Riordan, M., and R. Grosschedl. 1999. Coordinate regulation of B cell differentiation by the transcription factors EBF and E2A. Immunity 11: 21–31.
Ton, C.C., H. Hirvonen, H. Miwa, M.M. Weil, P. Monaghan, T. Jordan, V. van Heyningen, N.D. Hastie, H. Meijers-Heijboer, M. Drechsler, B. Royer-Pokora, F. Collins, A. Swaroop, L.C. Strong, and G.F. Saunders. 1991. Positional cloning and characterization of a paired-box-and homeobox-containing gene from the anirida region. Cell 57: 1059–1074.
Tondravi, M.M., S.R. McKercher, K. Anderson, J.M. Erdmann, M. Quiroz, R. Maki, and S.L. Teitelbaum. 1997. Osteopetrosis in mice lacking haematopoietic transcription factor PU.1. Nature 386: 81–84.
Urbánek, P., Z-Q. Wang, I. Fetka, E.F. Wagner, and M. Busslinger. 1994. Complete block of early B cell differentiation and altered patterning of the posterior midbrain in mice lacking Pax5/BSAP. Cell 79: 901–912.
Walther, C., J.I. Guenet, D. Simon, U. Deutsch, B. Jostes, M.D. Goulding, D. Plachov, R. Balling, P. Gruss. 1991. Pax: A murine multigene family of paired box-containing genes. Genomics 11: 424–434.
Wang, M.M., and R.R. Reed. 1993. Molecular cloning of the olfactory neuronal transcription factor Olf-1 by genetic selection in yeast. Nature 364: 121–126.
Wang, S.S., R.Y.L. Tsai, and R.R. Reed. 1997. The characterization of the Olf-1/EBF-like HLH transcription factor family: implications in olfactory gene regulation and neuronal development. J Neurosci 17: 4149–4158.
Wang, S.S., A.G. Betz, and R.R. Reed. 2002. Cloning of a novel Olf-1/EBF-like gene, O/E-4, by degenerate oligo-based direct selection. Mol Cell Neurosci 20: 404–414.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2007 Springer Science+Business Media, LLC
About this paper
Cite this paper
Horowitz, M.C., Lorenzo, J.A. (2007). Immunologic Regulation of Bone Development. In: Choi, Y. (eds) Osteoimmunology. Advances in Experimental Medicine and Biology, vol 602. Springer, Boston, MA. https://doi.org/10.1007/978-0-387-72009-8_6
Download citation
DOI: https://doi.org/10.1007/978-0-387-72009-8_6
Publisher Name: Springer, Boston, MA
Print ISBN: 978-0-387-72008-1
Online ISBN: 978-0-387-72009-8
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)