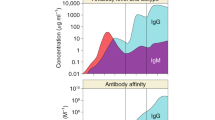
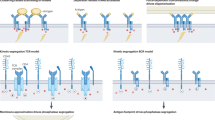
Abstract
The subunit structure of the B-cell antigen receptor (BCR) and its associated compart-mentalization of function confer enormous flexibility for generating signals and directing these toward specific and divergent cell fate decisions. Like all the multichain immune recognition receptors discussed in this volume, assembly of these multi-unit complexes sets these receptors apart from almost all other cell surface signal transduction proteins and affords them the ability to participate in almost all of the diverse aspects of, in this case, B-cell biology. We discuss here the structural aspects of the BCR and its associated coreceptors and relate these mechanistically to how BCR signaling can be directed towards specific fate decisions. By doing so, the BCR plays a pivotal role in ensuring the effective and appropriate B-cell response to antigen.
Access provided by Autonomous University of Puebla. Download to read the full chapter text
Chapter PDF
Similar content being viewed by others
Keywords
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
References
Schamel WW, Reth M. Monomerie and oligomeric complexes of the B-cell antigen receptor. Immunity 2000; 13(1):5–14.
Campbell KS, Hager EJ, Cambier JC. Alpha-chains of Igm and 1gd antigen receptor complexes are differentially n-glycosylated mb-1-related molecules. J Immunol 1991; 147(5):1575–1580.
Reth M. Antigen receptors on lymphocytes. Annu Rev Immunol 1992; 10:97–121.
Hombach J, Lottspeich F, Reth M. Identification of the genes encoding the Igm-alpha and Ig-beta components of the Igm antigen receptor complex by amino-terminal sequencing. Eur J Immunol 1990; 20(12):2795–2799.
Reth M, Wienands J, Tsubata T et al. Identification of components of the B-cell antigen receptor complex. Adv Exp Med Biol 1991; 292:207–214.
Mitchell RN, Shaw AC, Weaver YK et al. Cytoplasmic tail deletion converts membrane Immunoglobulin to a phosphatidylinositol-linked form lacking signaling and efficient antigen internalization functions. J Biol Chem 1991; 266(l4):8856–8860.
Grupp SA, Campbell K, Mitchell RN et al. Signaling-defective mutants of the lymphocyte antigen receptor fail to associate with Ig-alpha and Ig-beta/gamma. J Biol Chem 1993; 268(34):25776–25779.
Reth M. Antigen receptor tail clue. Nature 1989; 338(62l4):383–384.
Sigalov AB. Multichain immune recognition receptor signaling: Different players, same game? Trends Immunol 2004; 25(ll):583–589.
Kurosaki T. Genetic analysis of B-cell antigen receptor signaling. Annu Rev Immunol 1999; 17:555–592.
Tolar P, Sohn HW, Pierce SK. The initiation of antigen-induced B-cell antigen receptor signaling viewed in living cells by fluorescence resonance energy transfer. Nat Immunol 2005; 6(11): 1168–1176.
Fuentes-Panana EM, Monroe JG. Ligand-dependent and-independent processes in-cell-receptor-mediated signaling. Springer Semin Immunopathol 2001; 23(4):333–350.
Chung JB, Silverman M, Monroe JG. Transitional-cells: Step by step towards immune competence. Trends Immunol 2003; 24(6):343–349.
Koyama M, Ishihara K, Karasuyama H et al. CD79 alpha/CD79 beta heterodimers are expressed on pro-B-cell surfaces without associated mu heavy chain. Int Immunol 1997; 9(11):1767–1772.
Rolink A, Haasner D, Melchers F et al. The surrogate light chain in mouse B-cell development. Int Rev Immunol 1996; 13(4):341–356.
Grupp SA, Mitchell RN, Schreiber KL et al. Molecular mechanisms that control expression of the lymphocyte antigen receptor complex. J Exp Med 1995; 181(1):161–168.
Shaw AC, Mitchell RN, Weaver YK et al. Mutations of Immunoglobulin transmembrane and cytoplasmic domains: Effects on intracellular signaling and antigen presentation. Cell 1990; 63(2):381–392.
Wu Y, Pun C, Hozumi N. Roles of calnexin and Ig-alpha beta interactions with membrane igs in the surface expression of the B-cell antigen receptor of the igm and lgd classes. J Immunol 1997; 158(6):2762–2770.
King LB, Monroe JG. Immunobiology of the immature B-cell: Plasticity in the B-cell antigen receptor-induced response fine tunes negative selection. Immunol Rev 2000; 176:86–104.
Srivastava B, Lindsley RC, Nikbakht N et al. Models for peripheral B-cell development and homeostasis. Semin Immunol 2005; 17(3):175–182.
Kraus M, Alimzhanov MB, Rajewsky N et al. Survival of resting mature lymphocytes depends on BCR signaling via the Igalpha/beta heterodimer. Cell 2004; 117(6):787–800.
Lam KP, Kuhn R, Rajewsky K. In vivo ablation of surface Immunoglobulin on mature-cells by induc-ible gene targeting results in rapid cell death. Cell 1997; 90(6):1073–1083.
Campbell MA, Sefton BM. Association between B-lymphocyte membrane Immunoglobulin and multiple members of the sre family of protein tyrosine kinases. Mol Cell Biol 1992; 12(5):2315–2321.
Yamanashi Y, Kakiuchi T, Mizuguchi J et al. Association of B-cell antigen receptor with protein tyrosine kinase lyn. Science 1991; 251(4990): 192–194.
Schmitz R, Baumann G, Gram H. Catalytic specificity of phosphotyrosine kinases blk, lyn, c-src and syk as assessed by phage display. J Mol Biol 1996; 260(5):664–677.
Rolli V, Gallwitz M, Wossning T et al. Amplification of B-cell antigen receptor signaling by a syk/itam positive feedback loop. Mol Cell 2002; 10(5):1057–1069.
Kurosaki T, Johnson SA, Pao L et al. Role of the syk autophosphorylation site and SH2 domains in B-cell antigen receptor signaling. J Exp Med 1995; 182(6):1815–1823.
Takata M, Sabe H, Hata A et al. Tyrosine kinases lyn and syk regulate B-cell receptor-coupled Ca2+ mobilization through distinct pathways. EMBO J 1994; 13(6):1341–1349.
Engels N, Wollscheid B, Wienands J. Association of slp-65/BLNK with the B-cell antigen receptor through a non-itam tyrosine of Ig-alpha. Eur J Immunol 2001; 31(7):2126–2134.
Kabak S, Skaggs BJ, Gold MR et al. The direct recruitment of BLNK to Immunoglobulin alpha couples the B-cell antigen receptor to distal signaling pathways. Mol Cell Biol 2002; 22(8):2524–2535.
Patterson HC, Kraus M, Kim YM et al. The B-cell receptor promotes B-cell activation and proliferation through a non-itam tyrosine in the Igalpha cytoplasmic domain. Immunity 2006; 25(1):55–65.
Janssen E, Zhu M, Zhang W et al. Lab: A new membrane-associated adaptor molecule in B-cell activation. Nat Immunol 2003; 4(2):117–123.
DeFranco AL, Raveche ES, Paul WE. Separate control of lymphocyte early activation and proliferation in response to anti-Igm antibodies. J Immunol 1985; 135(1):87–94.
Kremyanskaya M, Monroe JG. Ig-independent ig beta expression on the surface of lymphocytes after B-cell receptor aggregation. J Immunol 2005; 174(3):1501–1506.
Kim JH, Cramer L, Mueller H et al. Independent trafficking of Ig-alpha/Ig-beta and mu-heavy chain is facilitated by dissociation of the B-cell antigen receptor complex. J Immunol 2005; 175(1):147–154.
Vilen BJ, Nakamura T, Cambier JC. Antigen-stimulated dissociation of BCR mlg from Ig-alpha/Ig-beta: Implications for receptor desensitization. Immunity 1999; 10(2):239–248.
Lang P, Stolpa JC, Freiberg BA et al. TCR-induced transmembrane signaling by peptide/MHC class II via associated Ig-alpha/beta dimers. Science 2001; 291(5508): 1537–1540.
Hou P, Araujo E, Zhao T et al. B-cell antigen receptor signaling and internalization are mutually exclusive events. PLoS Biol 2006;4(7):e.200
Monroe JG. ITAM-mediated tonic signalling through pre-BCR and BCR complexes. Nat Rev Immunol 2006; 6(4):283–294.
Pierce SK. Lipid rafts and B-cell activation. Nat Rev Immunol 2002; 2(2):96–105.
Laude AJ, Prior IA. Plasma membrane microdomains: Organization, function and trafficking. Mol Membr Biol 2004; 21(3):193–205.
Edidin M. Shrinking patches and slippery rafts: Scales of domains in the plasma membrane. Trends Cell Biol 2001; 11(12):492–496.
Cheng PC, Dykstra ML, Mitchell RN et al. A role for lipid rafts in B-cell antigen receptor signaling and antigen targeting. J Exp Med 1999; 190(11): 1549–1560.
Chung JB, Sater RA, Fields ML et al. CD23 defines two distinct subsets of immature B-cells which differ in their responses to ? cell help signals. Int Immunol 2002; 14(2): 157–166.
Norvell A, Mandik L, Monroe JG. Engagement of the antigen-receptor on immature murine B lym-phocytes results in death by apoptosis. J Immunol 1995; 154(9):4404–4413.
King LB, Norvell A, Monroe JG. Antigen receptor-induced signal transduction imbalances associated with the negative selection of immature B-cells. J Immunol 1999; 162(5):2655–2662.
Karnell FG, Brezski RJ, King LB et al. Membrane cholesterol content accounts for developmen-tal differences in surface B-cell receptor compartmentalization and signaling. J Biol Chem 2005; 280(27):25621–25628.
Chung JB, Baumeister MA, Monroe JG. Cutting edge: Differential sequestration of plasma membrane-associated B-cell antigen receptor in mature and immature B-cells into glycosphingolipid-enriched domains. J Immunol 2001; 166(2):736–740.
Nitschke L. The role of CD22 and other inhibitory coreceptors in B-cell activation. Curr Opin Im-munol. 2005; 17(3):290–297.
Cornall RJ, Cyster JG, Hibbs ML et al. Polygenic autoimmune traits: Lyn, CD22 and SHP-1 are limiting elements of a biochemical pathway regulating bcr signaling and selection. Immunity 1998; 8(4):497–508.
Dempsey PW, Allison ME, Akkaraju S et al. C3d of complement as a molecular adjuvant: Bridging innate and acquired immunity. Science 1996; 271(5247):348–350.
Cherukuri A, Cheng PC, Sohn HW et al. The CD19/CD21 complex functions to prolong B-cell antigen receptor signaling from lipid rafts. Immunity 2001; 14(2): 169–179.
Cherukuri A, Shoham T, Sohn HW et al. The tetraspanin CD81 is necessary for partitioning of coli-gated CD 19/CD21-B-cell antigen receptor complexes into signaling-active lipid rafts. J Immunol 2004; 172(1):370–380.
Singh DK, Kumar D, Siddiqui Z et al. The strength of receptor signaling is centrally controlled through a cooperative loop between Ca2+ and an oxidant signal. Cell 2005; 121(2):281–293.
Reth M. Hydrogen peroxide as second messenger in lymphocyte activation. Nat Immunol 2002; 3(12):1129–1134.
Shao D, Segal AW, Dekker LV. Lipid rafts determine efficiency of NADPH oxidase activation in neu-trophils. FEBS Lett 2003; 550(1-3):101–106.
Wienands J, Larbolette O, Reth M. Evidence for a preformed transducer complex organized by the B-cell antigen receptor. Proc Natl Acad Sci USA 1996; 93(15):7865–7870.
Tze LE, Schräm BR, Lam KP et al. Basal Immunoglobulin signaling actively maintains developmental stage in immature B-cells. PLoS Biol 2005; 3(3):e.82.
Bannish G, Fuentes-Panana EM, Cambier JC et al. Ligand-independent signaling functions for the lymphocyte antigen receptor and their role in positive selection during lymphopoiesis. J Exp Med 2001; 194(11):1583–1596.
Fuentes-Panana EM, Bannish G, Shah N et al. Basal Igalpha/Igbeta signals trigger the coordinated initiation of pre-B-cell antigen receptor-dependent processes. J Immunol 2004; 173(2):1000–1011.
Fuentes-Panana EM, Bannish G, van der Voort D et al. Ig alpha/lg beta complexes generate signals for B-cell development independent of selective plasma membrane compartmentalization. J Immunol 2005; 174(3):1245–1252.
Shaffer AL, Schlissel, MS. A truncated heavy chain protein relieves the requirement for surrogate light chains in early B-cell development. J Immunol 1997; 159(3): 1265–75.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2008 Landes Bioscience and Springer Science+Business Media
About this chapter
Cite this chapter
Brezski, R.J., Monroe, J.G. (2008). B-Cell Receptor. In: Sigalov, A.B. (eds) Multichain Immune Recognition Receptor Signaling. Advances in Experimental Medicine and Biology, vol 640. Springer, New York, NY. https://doi.org/10.1007/978-0-387-09789-3_2
Download citation
DOI: https://doi.org/10.1007/978-0-387-09789-3_2
Publisher Name: Springer, New York, NY
Print ISBN: 978-0-387-09788-6
Online ISBN: 978-0-387-09789-3
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)