Abstract
This study aimed to compare flexible fiberoptic laryngoscopy (FFL) and polysomnography (PSG) findings in patients with mucopolysaccharidosis (MPS) type VI and to describe upper airway anatomical findings and abnormal PSG results in these patients. In this cross-sectional study, all MPS VI patients followed up at the genetic division of a hospital in southern Brazil were included. Overnight PSG was performed, and the results were classified as normal or mildly, moderately, or severely abnormal. FFL was performed between 7 days before and 7 days after PSG. FFL findings were classified as (1) no obstruction, (2) mild obstruction, (3) moderate obstruction, or (4) severe obstruction of the airways, using the highest score obtained in all the regions.
Eleven patients with MPS VI were included. FFL detected severe airway obstruction in eight (72.7%) patients, moderate obstruction in two (18.2%), and mild obstruction in one (9.1%). PSG revealed obstructive sleep apnea syndrome (OSAS) in nine (81.8%) patients. Among these, mild OSAS was observed in five (45.5%) patients, moderate OSAS in three (27.2%), and severe OSAS in one (9.1%). Moderate to severe hypertrophy of the nasal turbinates was found in 81.8% of the patients, and 64% had severe infiltration in the supraglottic region. There was no association between FFL and PSG findings (p = 0.454; κ = −0.09; 95%CI = −0.34 to 0.17), indicating no agreement between the two methods. In the present study, all patients with MPS showed some degree of airway obstruction. We suggest performing PSG in MPS patients to determine disease severity.
Competing interests: None declared
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
Introduction
Mucopolysaccharidoses (MPSs) consist of a rare group of inherited lysosomal storage diseases, specifically related to glycosaminoglycan (GAG) metabolism defects. Each type of MPS is caused by deficiency of a specific enzyme involved in the degradation of GAGs, causing accumulation of partially degraded products. MPS is estimated to affect one in every 20,000 live births (McKunsick and Neufeld 1983; Kakkis and Wynn 2015).
Head and neck are often affected by MPS, usually at an early stage (Wold et al. 2010). Obstructive sleep apnea, otitis media with effusion, sinusitis, frequent respiratory infections, adenotonsillar hyperplasia, and speech disorders are very common (Gönüldaş et al. 2014). Airway symptoms are the main cause of morbidity and mortality in these patients, airway obstruction is a common finding, and pharyngeal hypotonia may cause obstructed breathing during sleep (Leighton et al. 2001; Santamaria et al. 2007).
Mucopolysaccharidosis type VI (MPS VI), also known as Maroteaux-Lamy syndrome, is caused by N-acetylgalactosamine-4-sulfatase deficiency. As there is no central nervous system involvement in this case, cognitive capacity is not impaired. However, airway infiltration by GAGs is a major concern in these patients, as it leads to the obstructive sleep apnea syndrome (OSAS) and, consequently, to cor pulmonale. Death occurs early on, often in the second or third decades of life (Wraith 1995; Kakkis and Wynn 2015).
Polysomnography (PSG) is the gold standard for the diagnosis of OSAS. While flexible fiberoptic laryngoscopy (FFL) is a widely used diagnostic tool in upper airway obstructions in the general population, there is not any information about the agreement of its results with PSG findings in MPS VI patients or about the importance of identifying the site of obstruction for therapeutic planning (Leighton et al. 2001). Actually, no studies to date have compared the findings of FFL and PSG for the assessment of MPS VI patients with airway obstruction symptoms.
The aim of the present study is to compare FFL and PSG findings in MPS VI patients.
Materials and Methods
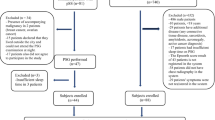
All MPS VI patients followed up at the Hospital de Clínicas de Porto Alegre, southern Brazil, and whose parents or legal representatives signed a consent form after being fully informed about the study were allowed to participate.
Both clinical and epidemiological data were collected from the patients after interviews with their parents or legal representatives and after analysis of medical charts.
The patients were submitted to overnight PSG, including electroencephalogram, electrooculogram, and electromyography for the classification of sleep stages. Leg electromyography, electrocardiogram, transcutaneous hemoglobin saturation (oximetry), snoring intensity detected with a microphone attached to the neck, and body position were recorded continually. An oronasal thermal sensor was used to detect the absence of airflow. Apnea was defined as blocked airflow for >10 s. Obstructive sleep apnea was defined as absence of airflow for >10 s associated with the presence of continued or increased chest and/or abdominal movements throughout the period of airflow obstruction. Hypopnea was considered whenever nasal airflow decreased by at least 30%, compared to the previous two ventilations, and when arterial oxygenation dropped by at least 4% in the subsequent 30 s. The apnea/hypopnea index (AHI), i.e., the number of apneas and hypopneas per hour of sleep, was calculated. Arousals were regarded as a sudden increase in EEG frequency for 3 or more seconds. The arousal index, defined as the number of arousals divided by the number of hours of sleep, and sleep efficiency, defined as sleep time divided by the number of hours in bed, were also measured (Santamaria et al. 2007; Iber et al. 2007; Epstein et al. 2009).
PSG was classified as normal or mildly (AHI ≥5 and <15/h), moderately (AHI ≥15 and ≤30/h), or severely (AHI >30/h) abnormal, according to the criteria established for adults (Iber et al. 2007).
All patients were also examined in the outpatient clinic, using FFL without sedation 7 days before to 7 days after PSG. The examination was performed with the patient sitting and after administration of a local topical anesthetic inside the nose (xylocaine 2% gel). A flexible fiberoptic 2.2-mm laryngoscope (Olympus®, model ENF-XP, Olympus America, Melville, NY, USA), employed exclusively to this end and connected to a Storz® microcamera (Karl Storz, Tuttlingen, Germany) and to a Storz® light source, was used. The examination was recorded on DVD and later reviewed by a researcher blinded to the PSG findings and widely experienced in pediatric airways (D.M.), who classified airway obstructions according to the following parameters:
Nasal cavities (lower nasal turbinates): classified, according to the grade of hypertrophy, into normal turbinates or mildly, moderately, or severely hypertrophic turbinates or fully obstructed nasal cavities
Cavum (pharyngeal or adenoid tonsils): classified, according to Brodsky and Koch (1992), based on the percentage of cavum filled by the pharyngeal tonsils, absence of hyperplasia, mild hyperplasia (pharyngeal tonsils filling up to 25% of the cavum), moderate hyperplasia (26–50%), severe hyperplasia (51–75%), or total obstruction (76–100%) (Brodsky 1989; Brodsky and Koch 1992)
Retropalatal region (palate and posterior and lateral walls): classified, according to the level of infiltration, into normal, mild, moderate, or severe infiltration or total obstruction
Oropharynx (palatine tonsils): classified, according to Brodsky (1989), into absence of tonsils, grade 1 hypertrophy (up to 25% of the oropharynx filled by the tonsils), grade 2 hypertrophy (26–50%), grade 3 hypertrophy (51–75%), and grade 4 hypertrophy (76–100%) (Brodsky 1989; Brodsky and Koch 1992)
Oropharynx (base of the tongue and epiglottis): classified, according to the level of infiltration, into normal, mild, moderate, or severe infiltration or total obstruction
Supraglottis (arytenoids/aryepiglottic folds): classified, according to the level of infiltration, into normal, mild, moderate, or severe infiltration or total obstruction
Glottis (vocal folds, ventricles of Morgani): classified, according to the level of infiltration, into normal, mild, moderate, or severe infiltration or total obstruction
Hypopharynx (pyriform sinuses): classified, according to the level of infiltration, into normal, mild, moderate, or severe infiltration or total obstruction/effacement of pyriform sinuses
After the assessment, taking into account the high grade of obstruction observed in different regions, the patients were classified as having: (1) no airway obstruction, (2) mild obstruction, (3) moderate obstruction, and (4) severe obstruction.
The data were analyzed by the SPSS® software program, version 21.0. The quantitative variables were described as mean and standard deviation or median and interquartile range. The categorical variables were described by absolute and relative frequencies. Pearson’s chi-square test was used to assess the association between the diagnostic methods. The kappa statistic was used to assess the amount of agreement. The significance level was set at 5%.
Results
Eleven MPS VI patients aged 17.8 ± 3.2 years, seven (63.6%) of whom were male were included in the study. Snoring was the most frequent clinical manifestation (72.7%), followed by nasal obstruction (54.5%). Secretion, rebound upper airway infection, swallowing difficulty, and dysphagia were also common (45.5%). Doctor-diagnosed sleep apnea was reported by 36.4% of the patients. No patient had a previous history of stridor or cyanosis. Most patients (72.7%) had a previous history of intubation and the use of CPAP was reported by 27.3% of them. Prior airway surgeries had been necessary in 45.5% of the individuals and consisted of adenoidectomy, tonsillectomy, and nasal turbinate cautery (Table 1).
FFL revealed moderate to severe hypertrophy of nasal turbinates in 81.8% of the patients, moderate to severe hypertrophy of pharyngeal tonsils in 27.2%, and grades 3 and 4 hypertrophy of palatine tonsils in 27.2%. Approximately 64% of the patients had moderate to severe obstruction in the supraglottic region, 36.4% revealed moderate to severe obstruction in the retropalatal region, and 54.5% had moderate to severe obstruction at the base of the tongue.
Taking into account the high incidence of obstruction in the regions, FFL detected severe obstruction in eight patients (72.7%), moderate obstruction in two patients (18.2%), and mild obstruction in one patient (9.1%).
PSG revealed OSAS in nine patients (81.8%). Among these, mild OSAS was observed in five (45.5%), moderate OSAS in three (27.2%), and severe OSAS in one (9.1%).
Sleep efficiency averaged 76.4% (±12.8%), and a median of 7.9 (0.4–16.9) apneas per hour and of 3.4 (0.3–11.8) hypopneas per hour was observed. Mean oxygen saturation was as high as 95.6% (±3.7) while minimum saturation was 80.2% (±8.6). A median of 12 (2–40) microarousals per hour was obtained.
As shown in Fig. 1 and in Tables 1 and 2, there was no association between FFL and PSG findings (p = 0.454), which shows lack of agreement between the diagnostic methods (κ = −0.09; 95%CI = −0.34 to 0.17).
Discussion
The present study was the first one to describe these findings exclusively in MPS VI patients, thereby providing a more homogeneous sample of individuals.
Airway obstruction is quite common in patients with this disease, which is progressive and may be fatal. Although nearly 50% of the individuals included in the study (45.5%) had already undergone surgical procedures to treat airway obstruction, 81.8% of them, according to the PSG findings, still had OSAS. This finding was highly frequent and corroborates the literature data, according to which the prevalence of this symptom ranges from 40 to 90% in patients with different types of MPS (Leighton et al. 2001; Santamaria et al. 2007).
Similarly to our study, Yeung et al. (2009) used FFL and PSG to assess some patients with different types of MPS. They found that 19 of their 27 patients presented with upper airway obstruction. Of these 19 patients, seven were submitted to PSG and had an AHI between 10 and 17. Only five of the patients were submitted to direct laryngoscopy, which revealed the presence of macroglossia and of redundant tissues in the oropharynx and in the supraglottis.
In our study, in which all individuals were submitted to FFL, major obstruction was observed in the nasal fossae, at the base of the tongue, and in the supraglottic region. Presumably, this is due to the accumulation of GAGs in these regions, causing airway narrowing.
Santamaria et al. (2007) assessed 11 patients (five children and six adults) with MPS. They also performed PSG and FFL in all of them, in addition to computerized tomography (CT) of the upper airways. They found OSAS in 100% of the children and in 17% of the adults. In the CT scan, they observed a smaller retropalatal and retroglossal space whereas nasal endoscopy revealed hypertrophic adenoids in all of the patients. However, they assessed only the size of pharyngeal tonsils and did not perform a thorough examination of the larynx and pharynx as we did in the present study.
The FFL with sedation, with consequent loss of muscle tone, could be ideal for the assessment of upper airways during sleep. Nevertheless, the high anesthetic risk should only be taken in cases where the procedure has actual potential to change the clinical outcomes of these patients (Simmons et al. 2005). In our population, all the individuals tolerated the procedure without sedation, and the information could be obtained as expected.
Finally, by comparing FFL and PSG findings, we did not observe good agreement between them. Thus, not all those patients in which FFL showed airway obstruction had an abnormal PSG. These data suggest it is necessary to perform PSG in MPS patients to identify those at risk for OSAS.
Conclusions
MPS VI is a heterogeneous and progressive disease with major systemic involvement. All the assessed patients have some airway obstruction. We suggest performing PSG in all individuals in order to verify the severity of each case. The role of FFL in the identification of obstructive lesions in this population still needs clarification.
References
Brodsky L (1989) Modern assessment of the tonsils and adenoids. Pediatr Clin North Am 36:1551–1569
Brodsky L, Koch J (1992) Anatomic correlates of normal and diseased adenoids in children. Laryngoscope 102:1268–1274
Epstein L, Kristo D, Strollo P et al (2009) Clinical guideline for the evaluation, management and long-term care of obstructive sleep apnea in adults. J Clin Sleep Med 5:263–276
Gönüldaş B, Yilmaz T, Sivri HS, Gucer KS, Kilinç K, Genç GA et al (2014) Mucopolysaccharidosis: otolaryngologic findings, obstructive sleep apnea and accumulation of glucosaminoglycans in lymphatic tissue of the upper airway. Int J Ped Otorhinolaryngol 78:944–949
Iber C, Ancoli-Israel S, Chesson A Jr, Quan S (2007) The AASM manual for the scoring of sleep and associated events: rules, terminology and technical specifications. American Academy of Sleep Medicine, Westchester
Kakkis E, Wynn R (2015) Mucopolysaccharidoses: clinical features and diagnosis. UpToDate. Wolters Kluwer Health. http://www.uptodate.com/contents/mucopolysaccharidoses-clinical-features-and-diagnosis. Cited 9 Mar 2015
Leighton S, Papsin B, Vellodi A, Dinwiddie R, Lane R (2001) Disordered breathing during sleep in patients with mucopolysaccharidoses. Int J Ped Otorhinolaryngol 58:127–138
McKunsick VA, Neufeld EF (1983) The mucopolysaccharidoses storage diseases. In: Stanbury JB, Wyngaarden JB, Fredrickson DS (eds) The metabolic basis of inherited diseases, 5th edn. McGraw-Hill, New York, pp 342–365
Santamaria F, Andreucci MV, Parenti G et al (2007) Upper airway obstructive disease in mucopolysaccharidoses: polysomnography, computed tomography and nasal endoscopy findings. J Inherit Metab Dis 30:743–749
Simmons MA, Bruce IA, Penney S, Wraith E, Rothera MP (2005) Otorhinolaryngological manifestations of the mucopolysaccharidoses. Int J Ped Otorhinolaryngol 69:589–595
Wold SM, Derkay CS, Darrow DH, Proud V (2010) Role of the pediatric otolaryngologist in diagnosis and management of children with mucopolysaccharidoses. Int J Ped Otorhinolaryngol 74:27–31
Wraith JE (1995) The mucopolysaccharidoses: a clinical review and guide to management. Arch Dis Child 72:263–267
Yeung AH, Cowan MJ, Horn B, Rosbe KW (2009) Airway management in children with mucopolysaccharidoses. Arch Otolaryngol Head Neck Surg 135:73–79
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Additional information
Communicated by: Verena Peters
Appendices
Take Home Message (Synopsis)
Obstructive sleep apnea syndrome is frequently found in patients with mucopolysaccharidosis type VI and should be investigated with polysomnography.
Compliance with Ethics Guidelines
Details of the Contributions of Individual Authors
D.R.R.P., C.S., and D.M. were responsible for the design of the study and for data collection. D.R.R.P., C.S., and S.F. carried out the tests. G.K. and P.J.C.M. participated in the design of the study and performed the statistical analysis. C.F.S. and R. G. participated in its design and helped to draft the manuscript. D.R.R.P. was responsible for data analysis and interpretation and helped to draft the manuscript. All authors read and approved the final manuscript.
Name of the Author Who Serves as Guarantor
Denise Rotta Ruttkay Pereira
Competing Interest Statement
Denise Rotta Ruttkay Pereira, Claudia Schweiger, Carolina Fischinger de Souza, Simone Fagondes, Denise Manica, Roberto Giugliani, Gabriel Kuhl, and Paulo José Cauduro Marostica declare that they have no conflict of interest.
Details of Funding
The authors have no financial relationships relevant to this article to disclose.
Details of Ethics Approval
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2000.
Patient Consent Statement
Patients’ parents or legal representatives signed a consent form after being fully informed about the study.
Rights and permissions
Copyright information
© 2015 SSIEM and Springer-Verlag Berlin Heidelberg
About this chapter
Cite this chapter
Pereira, D.R.R. et al. (2015). Correlation Between Flexible Fiberoptic Laryngoscopic and Polysomnographic Findings in Patients with Mucopolysaccharidosis Type VI. In: Morava, E., Baumgartner, M., Patterson, M., Rahman, S., Zschocke, J., Peters, V. (eds) JIMD Reports, Volume 29. JIMD Reports, vol 29. Springer, Berlin, Heidelberg. https://doi.org/10.1007/8904_2015_513
Download citation
DOI: https://doi.org/10.1007/8904_2015_513
Received:
Revised:
Accepted:
Published:
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-662-53277-5
Online ISBN: 978-3-662-53278-2
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)