Abstract
Neurochemical imaging is frequently applied to measure markers of pathological change so as to understand mechanisms that create symptoms of major depressive disorder. For example, indices of greater monoamine oxidase A(MAO-A) level, particularly in the prefrontal and anterior cingulate cortex, are associated with depressed mood states, and high-risk states for onset of major depressive episodes. MAO-A metabolises monoamines, and greater metabolism of monoamines occurs when MAO-A is elevated in brain. Lower extracellular serotonin is associated with greater pessimism in humans and chronic serotonin deficiency is associated with upregulation of 5-HT2A (serotonin2A) receptors in cortex. During major depressive episodes when pessimism is more severe, greater 5-HT2A BPND, an index of density occurs in prefrontal and anterior cingulate cortex. These results argue for a mechanism of lowering extracellular serotonin in the prefrontal and anterior cingulate cortex, consequent to elevated MAO-A level. The relationship between elevated 5-HTT BPND and greater pessimism during major depressive episodes suggests that greater 5-HTT density in the context of elevated MAO-A level further contributes to serotonin deficiency in these brain regions. A similar mechanism may explain the association between neuroimaging indices of greater dorsal striatal D2 density, DAT density and symptoms of motor retardation: Greater MAO-A level and relatively greater DAT density lower extracellular dopamine in the dorsal striatum, leading to motor retardation. Indices of greater 5-HT1A density, particularly in the cingulate cortex, have been associated with major depressive disorder, and well as anxiety disorders, suggesting that this abnormality is mechanistically related to presence of anxiety symptoms. To date, abnormalities of Glx a measure reflecting glutamate and glutamine levels have been most strongly associated with presence of major depressive episodes, with greater levels in occipital cortex, and reduced levels in prefrontal cortex. Ultimately, the future for neurochemical imaging is to better understand the mechanisms that predispose toward onset of MDE so as to create biologically informed, novel, methods of prevention, and superior, more symptom-targeted treatments.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Monoamine oxidase A
- Serotonin2A receptors
- Serotonin transporter
- Serotonin1A receptors
- Dopamine2 receptors
- Dopamine transporter
- Glutamate
- Major depressive disorder
1 Introduction
Major depressive disorder affects 2–5 % of the population at any time and is the third leading cause of death and disability, according to the world health organisation (World Health Organization 2008). Given its high prevalence an important issue is to better understand the aetiology of symptoms so as to develop new strategies for prevention and more targeted approach for treatment.
Neurochemical imaging techniques offer an opportunity to measure markers of neurophysiological change to better understand mechanisms that contribute toward onset of symptoms during major depressive disorder. By identifying neurochemical abnormalities of MAO-A level, 5-HT2A (serotonin2A), 5-HTT (serotonin transporter), 5-HT1A (serotonin1A), D2, DAT as well as glutamate levels in functionally important regions, then relating these findings to symptoms, and illness state, neuroimaging investigations are making important contributions toward understanding the pathophysiology of major depressive disorder.
2 Monoamine Oxidase A and Depressed Mood
2.1 Monoamine Oxidase A, Monoamines and Mood
Monoamine oxidase-A (MAO-A) is an enzyme with diverse functions in the brain, some of which have important roles for influencing mood; for most brain tissues, MAO-A activity is the main route for serotonin metabolism, and a significant route of metabolism for other monoamines, including norepinephrine and dopamine (Youdim et al. 2006). MAO-A is detectable in cells that release these monoamines, with the highest levels in norepinephrine releasing neurons (Konradi et al. 1988, 1989; Luque et al. 1995; Moll et al. 1990; Saura et al. 1996); however, MAO-A is also present in cells that do not release monoamines, such as astrocytes and glia (Youdim et al. 2006). Within cells, MAO-A is mainly located on outer mitochondria membranes. MAO-A has a high density in brain regions that influence mood (Saura et al. 1992): While MAO-A density is highest in brainstem (within the locus coeruleus), it is moderately high in the cortex, hippocampus, striatum, much lower in cerebellar cortex and minimal in white matter tissue. In addition, MAO-A has pro-oxidant effect, via the production of hydrogen peroxide, and is functionally linked to apoptosis. The latter is based on the observations that MAO-A inhibitors reduce apoptosis and MAO-A expression is increased in cell lines that are in a pro-apoptotic state (Ou et al. 2006). Among these roles, it is the effect of MAO-A upon monoamine metabolism that is highly implicated in influencing mood state.
There is a considerable amount of data linking loss of extracellular serotonin, norepinephrine and dopamine in humans with onset of depressed mood and/or major depressive episodes. Overall, two temporal mechanisms have been observed, one acute and one chronic. For example, acute monoamine depletion, through either tryptophan depletion to lower brain serotonin or alpha-methyl-para-tyrosine to lower brain dopamine and norepinephrine, is associated with depressed mood (Freis 1954; Hasler et al. 2008; Neumeister et al. 2004b; Verhoeff et al. 2001; Young et al. 1985). The second type of mechanism observed is that long periods of monoamine depletion are associated with onset of MDE in humans as demonstrated by chronic reserpine administration (Freis 1954).
In brain tissue, the density of MAO-A correlates highly with the level of its metabolic activity. Thus, given the functional link between monoamine loss and low mood and/or MDE, and the role of MAO-A in metabolising multiple monoamines, states that raise of MAO-A level in affect modulating brain regions would be expected to influence overall mood toward a depression. Additional evidence for a relationship between MAO-A and mood has been that one longstanding class of antidepressants, MAO inhibitors, has been effective treatments; this provides another reason to investigate this target in depressed mood states (Youdim et al. 2006). The relationship between MAO-A density and depressed mood may now be tested: recent advances in positron emission tomography (PET) neuroimaging such as [11C]harmine PET enable optimal quantitation of MAO-A VT, a measurement proportional to MAO-A density.
Comparison of MAO-A DVs between depressed and healthy subjects. On average MAO-A DVs was elevated by 34 percent, or two standard deviations, in depressed individuals. Differences between groups were highly significant in each region: * p=0.001, ** p<0.0001, *** p<0.00001. Reprinted from Meyer et al. (2006a)
2.2 Prefrontal and Anterior Cingulate Cortex Monoamine Oxidase A Binding, Depressed Mood and Major Depressive Disorder
A key advantage of neuroimaging for investigating major depressive episodes (MDE) is that the in vivo measurement of MAO-A VT may be conducted in medication free subjects and in the specific grouping of early onset MDD. Most MDD is related to early onset, and it vital to differentiate early from late onset for investigations of MDD, because late onset MDD is associated with neurodegenerative disease such as Parkinson’s disease or Alzheimer’s disease, which can be viewed as different pathologies. The issue of selecting early onset medication free subjects was not readily addressable in the first three postmortem studies of MAO-A levels and/or activity, hence prior to 2006 it was unknown whether MAO-A density or activity was increased in early onset major depressive disorder. In 2006, MAO-A VS, an index of MAO-A density, was measured using [11C] harmine PET in medication free MDE secondary to early onset MDD (Meyer et al. 2006a). Subjects with MDE were drug free for at least 5 months and most were antidepressant naive. All MDE subjects and controls were otherwise healthy. The MAO-A VS was highly significantly elevated (p < 0.001 each region, average magnitude 34 % (or two standard deviations)) during MDE (see Fig. 1). This was the first definitive study of MAO-A binding in MDE because the clinical sample was carefully defined to focus upon MDD, the effect size was large and the PET radiotracer [11C] harmine has outstanding qualities for measuring MAO-A binding.
Later studies have been highly consistent in support of this finding: Barton et al. (2008) reported elevated brain serotonin turnover in unmedicated depressed patients, a phenomenon which could be explained by greater brain MAO-A level (Barton et al. 2008). In 2009, the finding of greater MAO-A binding in MDE was replicated with [11C] harmine PET in the same laboratory, and in 2011 the finding was replicated in antidepressant free MDE subjects in postmortem study of orbitofrontal cortex in a different laboratory applying Western blot (Johnson et al. 2011; Meyer et al. 2009).
Monoamine lowering processes may lead to lowered mood, hence, in recovery from MDD, it might be expected that MAO-A levels would normalise with euthymic mood. However, the recovered state of MDD is also a state of high risk for another MDE. The risk for a recurrent MDE over 2 years is 20–50 % depending upon treatment conditions. Elevated MAO-A binding may be considered an index of a monoamine lowering process and in the 1950s during treatment with reserpine-based antihypertensives, it was discovered that chronic monoamine lowering is associated with subsequent onset of MDEs which typically occurred 2 weeks to 4 months later (Meyer et al. 2009). The in vivo nature of neuroimaging enabled measurement of MAO-A VT, an index of MAO-A density, in a study of recovered MDD. In this study, MAO-A binding was significantly elevated in prefrontal cortex, anterior cingulate cortex, striatum, hippocampus, thalamus and midbrain in a sample of 18 medication free recovered MDD subjects compared to 28 healthy controls (Meyer et al. 2009). Recovered MDD subjects who had recurrence of their MDE in the subsequent 6 months had the highest levels of MAO-A binding in the prefrontal and anterior cingulate cortex at the time of scanning (Meyer et al. 2009). The prefrontal cortex and anterior cingulate cortex were prioritised because these regions (and/or subregions of these structures) are often activated in mood induction studies (reflecting processes that generate sad mood) (Liotti et al. 2002) and these regions participate in cognitive functions like pessimism which create sad mood (Sharot et al. 2007; Tom et al. 2007). In this MDD sample (Meyer et al. 2009), other factors related to recurrence were accounted for: Subjects were medication free for at least 1 year, had no cognitive behavioural therapy within 3 years, were currently asymptomatic, and had no comorbid medical, psychiatric or substance abuse illnesses. Given the link between elevated MAO-A binding in prefrontal and anterior cingulate cortex and subsequent MDE, a stronger case can be made that new therapeutics are needed to decrease MAO-A levels in these brain regions, ideally with persistence beyond the duration of administration of the therapeutic to prevent recurrence.
2.3 Monoamine Oxidase A and Depressed Mood During Postpartum
Depressed mood is common during early postpartum and can be categorised into three different syndromes. One common syndrome, occurring up to 75 % of the time, and considered within the healthy range of experience, is the “baby blues” or maternity blues. This consists of sad mood typically peaking on day 5 and ending with the first week postpartum accompanied by some irritability, desire to be alone, insomnia and trouble concentrating (O’Hara and Swain 1996). The second syndrome, which is also common with a prevalence rate of 13 %, is a full, clinical level MDE occurring within the first 1–3 months after delivery (O’Hara and Swain 1996; American Psychiatric Association 1994). The third syndrome, postpartum psychosis, is relatively rare with a prevalence rate of 0.1–0.2 %, and includes a combination of a MDE with hallucinations or delusions (Brockington et al. 1981; Kendell et al. 1987).
The first investigation of MAO-A in the postpartum period occurred in 2010: A [11C] harmine PET study measured MAO-A VT during postpartum blues (with scanning done between days 4 and 6 postpartum). A highly significant elevation of MAO-A binding was found, which, on average, was 43 % greater across the brain regions assayed (prefrontal cortex, anterior cingulate cortex, striatum, thalamus, hippocampus and midbrain) as compared to women not recently pregnant (Sacher et al. 2010). A voxel-based analysis demonstrated that the elevation in MAO-A binding was present throughout the grey matter of the brain (see Fig. 2) (Sacher et al. 2010).
Regional distribution of p-values reflecting elevated MAO-A binding in immediate postpartum period. Parametric maps of elevated monoamine oxidase A binding in the postpartum group vs the control group. Maps are superimposed on a T1-weighted magnetic resonance image that is normalized to the T1-weighted template (SPM2; Department of Cognitive Neurology, Wellcome Trust Centre for Neuroimaging, London, UK). a Transverse. b Sagittal. c Coronal. Individual voxel threshold was set at P<0.05; 86 412 voxels comprised a single cluster, which had a cluster-corrected significance of P=0.03. Mean regional difference was 43%
This discovery led to a new neurobiological explanation for postpartum blues, involving a rapid decline in estrogen, followed by a rapid rise in MAO-A levels in affect-modulating brain regions, and finally subsequent sad mood with postpartum blues. During pregnancy, estradiol and estriol levels rise more than 100-fold and during the first week postpartum, with loss of the placenta, there is an enormous reduction in 17β-estradiol and estriol levels (O’Hara et al. 1991). Most of the decline in estrogens occur in the first 4 days followed by a more modest decline thereafter (O’Hara et al. 1991). Although MAO-A had never been previously investigated in the postpartum, it is known that estrogen decline is associated with a rise in either MAO-A density, activity or mRNA (see review (Sacher et al. 2010)). The 43 % elevation in MAO-A VT in early postpartum confirms that this inverse relationship is applicable to the early postpartum period, after the early estrogen decline. MAO-A VT can be viewed as an index of MAO-A levels and MAO-A levels correlate highly with MAO-A activity in brain tissue (Saura et al. 1992). Hence, the acute rise in MAO-A VT in the early postpartum period represents a monoamine-lowering process and acute monoamine lowering processes are associated with sad mood (Ruhe et al. 2007).
2.4 Cigarette Withdrawal, Depressed Mood and MAO-A
Sad mood is an important problem for people who smoke cigarettes. First, during early withdrawal, sad mood frequently occurs, and when sad mood is prominent during withdrawal, it is associated with greater likelihood of relapse during quit attempts (Carey et al. 1993; Kenford et al. 2002). Second, cigarette smoking predisposes to MDD and vice versa; consequently, there is a very high comorbidity between cigarette smoking and major depressive disorder with 50 % of people with MDD also smoking cigarettes (Anda et al. 1990; Breslau et al. 1998).
The initial impression of the neuroimaging field was that MAO-A binding is reduced in those who smoke cigarettes in the active smoking state (Fowler et al. 1996). Given that, the plasma half-life of the key MAO-A binding substances found in cigarette smoke (harman and norharman) is only an hour (Rommelspacher et al. 2002), there was reason to specifically assess MAO-A VT during both active smoking and withdrawal conditions. When MAO-A VT was assessed in both conditions, it was discovered that prefrontal and anterior cingulate cortex MAO-A VT rose during withdrawal in those who smoke heavily, that is, more than one pack of cigarettes per day, but not in those who smoke more moderately at less than one pack per day (see Fig. 3 from Bacher et al. (2011)). Interestingly, it is the heavy cigarette smoking group who are known to be at much greater risk for MDD (Pratt and Brody 2010), and it was this group that had 25 % elevated MAO-A VT in the prefrontal and anterior cingulate cortex during withdrawal as compared to healthy controls, arguing for a process of elevated MAO-A level during withdrawal as a mechanism to create risk for MDD. Hence repeated exposure of elevated MAO-A level, in the prefrontal and anterior cingulate cortex, a mechanism associated MDD, occurring after a short period of 8 h withdrawal could explain the predisposition to MDD in people who smoke cigarettes heavily.
Monoamine Oxidase-A binding in cigarette smoking subjects during active smoking and withdrawal. Repeated measures MANOVA found a highly significant effect between smoking severity and change in MAO-A VT in the prefrontal cortex and anterior cingulate cortex regions (F1, 22 = 25.58, p<0.001). Repeated measures MANOVA also found a significant effect between smoking severity and change in MAO-A VT for all the regions assayed (F1, 22 = 28.24, p<0.001). The effect was confirmed in each region with a repeated measures ANOVA (interaction between smoking severity and change in MAO-A VT, repeated measures ANOVA, F1, 22 = 11.16 to 28.87, p = 0.003 to p<0.001). a p<0.001 b p = 0.003. Greater MAO-A binding during acute cigarette withdrawal in heavy smoking group compared with healthy (MAO-A VT within groups were compared with an independent t-test. a p ≤ 0.001 b p ≤ 0.005 c p ≤ 0.01 d p ≤ 0.05 e p ≤ 0.1 Arch Gen Psych (in press))
As mentioned earlier, a second problem with mood in people who smoke cigarettes is the depressed mood of acute withdrawal (Carey et al. 1993; Kenford et al. 2002). In the imaging study of MAO-A during cigarette withdrawal, those who smoked heavily, the magnitude of rise in MAO-A VT in prefrontal and anterior cingulate cortex during withdrawal was significantly correlated with the shift in visual analogue scales toward depressed mood (Bacher et al. 2011). This rise in MAO-A VT also correlated with the decline in the MAO-A binding substance harman in those who smoke heavily. These results have significant implications for quitting heavy smoking. They suggest that rapid removal of harman from occupying MAO-A sites leaves a high level of available MAO-A for metabolising monoamines in prefrontal and anterior cingulate cortex resulting in depressed mood. This argues for testing of MAO-A inhibitor treatments in people who experience sad mood during early cigarette withdrawal as a strategy to assist in quitting, an important issue since 50 % of people tend to relapse in the first few days of trying to quit cigarette smoking (Garvey et al. 1992; Law and Tang 1995).
In conclusion, greater MAO-A VT occurs, particularly in the prefrontal and anterior cingulate cortex, in a number of low mood states that are also associated with predisposition to major depressive disorder (Bacher et al. 2011; Meyer et al. 2009; Sacher et al. 2010). This data has major implications for preventing major depressive disorder as it suggests that better understanding and regulation of MAO-A may be helpful in avoiding the onset of major depressive episodes.
3 Pessimistic Perspective, 5-HT2A Receptor and 5-HTT Receptor Imaging
3.1 5-HT2A Receptor Imaging in Prefrontal and Anterior Cingulate Cortex
Negativistic thinking often occurs during major depressive episodes and it is important because high levels of hopelessness, a key component of pessimism, are associated with greater risk of suicide. Pessimism during major depressive episodes has also been captured by the concept of ‘dysfunctional attitudes’. While a modest level of dysfunctional attitude can viewed as adaptive, dysfunctional attitudes increase significantly during major depressive episodes (Simons et al. 1986; Weissman 1979). Greater pessimism during major depressive episodes contributes to negative thoughts, and subsequent sad mood and this underlying pessimism is targeted by cognitive therapy (Simons et al. 1986; Weissman 1979). Dysfunctional attitudes may be measured with the dysfunctional attitudes scale (DAS), a measure that is sensitive for detecting negativistic thinking during major depressive episodes (Simons et al. 1986; Weissman 1979), and also demonstrates very good internal consistency (Cronbach alpha = 0.85–0.87) (Cane et al. 1986; Oliver and Baumgart 1985) and has high test–retest reliability (Oliver and Baumgart 1985; Weissman 1979).
Two findings initially suggested a relationship between manipulations of extracellular serotonin and dysfunctional attitudes. The first is that dysfunctional attitudes normalise during the response to selective serotonin reuptake inhibitor (SSRI) treatment (Fava et al. 1994; Simons et al. 1986). The second is that raising extracellular serotonin after administration of intravenous d-fenfluramine rapidly shifts dysfunctional attitudes toward optimism in healthy individuals (Meyer et al. 2003). These results argue that one of the roles of serotonin is to modulate dysfunctional attitudes in humans. More recently, the rostral anterior cingulate cortex and subregions of prefrontal cortex (dorsolateral, and medial prefrontal cortex) have been demonstrated to participate in functions related to optimism and pessimism (Elliott et al. 2002; Sharot et al. 2007).
Direct evidence that serotonin is low in brain is difficult to obtain: Brain serotonin cannot be directly measured in vivo and it is likely, based upon animal simulations of postmortem delay, that serotonin levels are very unstable, even within 24 h of death (Kontur et al. 1994). Moreover, postmortem investigations reviewed by Mann et al. (1996), Stockmeier (2003) have difficultly sampling medication free subjects.
While one cannot measure extracellular serotonin directly during major depressive episodes with neuroimaging either, one may measure an index of regional 5-HT2A receptor density such as 5-HT2A BP or 5-HT2A BPND (an index of specific binding relative to free and non-specific binding). 5-HT2A density has an inverse relationship to extracellular serotonin such that binding increases when extracellular serotonin is chronically lowered (O’Regan et al. 1987; Roth et al. 1987; Stockmeier and Kellar 1986; Todd et al. 1995). Therefore, if extracellular serotonin loss occurred in the prefrontal and anterior cingulate cortex during MDE, increased 5-HT2A BPND would occur in these regions. A review of the initial set of 5-HT2A imaging studies of MDE find a reduction in those with recent antidepressant use, and no change in those with no recent antidepressant use (see Table 1) (Meyer 2008). One could interpret the reductions in 5-HT2A binding in recently treated subjects as being consequent to recent antidepressant use. The other set of findings, reflecting the medication free state of MDE, which found no change in 5-HT2A BPND would suggest either abandoning the notion of reduced extracellular serotonin in the prefrontal and anterior cingulate cortex during MDE or creating an alternative hypothesis.
One alternative perspective is that monoamine loss during MDD is heterogenous and that the loss is greatest in those with the most severe symptoms. The first investigations of this revision began with prefrontal cortex 5-HT2A BPND measurement and its relationship to dysfunctional attitudes. A strong correlation was observed between severity of dysfunctional attitudes (pessimism) and elevation in prefrontal and anterior cortex 5-HT2A BPND. Furthermore, cortex 5-HT2A BPND was significantly elevated in subjects with MDE and severe pessimism (Meyer et al. 2003). For example, in the prefrontal cortex region of interest centred on Brodman’s area 9, 5-HT2A BPND was elevated 29 % in depressed subjects with dysfunctional attitude scores greater (more pessimistic) than the median for the group. There was also a strong, significant correlation between severity of pessimism and prefrontal cortex 5-HT2A BPND (see Fig. 4). A study by Bhagwagar et al. replicated this relationship between dysfunctional attitudes severity and prefrontal cortex 5-HT2A BPND in recovered depressed subjects(Bhagwagar et al. 2006). In another study of a large sample of healthy subjects, two personality factors related to pessimism, vulnerability and anxiety, also positively correlated with prefrontal cortex, temporal cortex and left insula 5-HT2A BPND (Frokjaer et al. 2008).
5-HT2A Binding Potential is Greater During Major Depressive Episodes (MDE) With Highly Abnormal Dysfunctional Attitudes. (5-HT2A receptor binding potential in averaged bilateral middle frontal gyrus (Brodmann's area 9) is plotted against age to show the relationship between depressed and healthy subjects. The 22 depressed patients were divided into high and low dysfunctional attitudes scale (DAS) groups depending upon whether their DAS scores were above or below the median DAS score for the MDE group. This median score was 166. MDE subjects with high DAS scores had significantly higher 5-HT2A receptor binding potential as compared to healthy subjects (ANCOVA (age covariate), diagnosis, F1,19=11, p=0.003). (Age was an expected covariate in the model which was designed to identify a disease effect influencing 5-HT2A receptor availability.))
These results provide an explanation to interpret the investigations of suicide victims which had been a key focus of the mood disorders field between the mid 1984 and 2000. At that time, the most consistent postmortem biological abnormality in suicide victims was increased serotonin2A receptor density in the prefrontal cortex, most commonly in Brodmann’s area 9 (Arango et al. 1990, 1992; Arora and Meltzer 1989; Hrdina and Vu 1993; Mann et al. 1986; Pandey et al. 2002; Stanley and Mann 1983; Stockmeier 2003; Stockmeier et al. 1997; Turecki et al. 1999; Yates et al. 1990). At the time the studies reported the abnormality as alterations in serotonin2 receptor binding, but it is generally accepted that these studies investigated serotonin2A receptors given that ligand binding to 5-HT2C receptors in cortex is extremely low (Hoyer et al. 1986; Marazziti et al. 1999) and mRNA of 5-HT2B receptors is extremely low in cortex (Schmuck et al. 1994). Although these findings occurred in studies in which the diagnosis of the suicide victim was unrestricted, these findings were more consistent in the subsample of studies of suicide victims who had major depressive disorder and were medication free (Hrdina and Vu 1993; Yates et al. 1990).
The investigations correlating severity of dysfunctional attitudes with greater 5-HT2A BPND (Meyer et al. 2003; Bhagwagar et al. 2006) explains at a diagnostic and symptom specific level what clinical phenomenon was studied in these postmortem studies: Fifty percent of suicide victims have major depressive disorder (Barraclough et al. 1974; Robins et al. 1959). The dysfunctional attitudes scale is highly correlated with hopelessness measured with the Beck Hopelessness Scale (Bouvard et al. 1992; Cannon et al. 1999; DeRubeis et al. 1990; Norman et al. 1988). Given that hopelessness is a risk factor for suicide (Beck et al. 1985; Beck et al. 1989), it is likely that investigations of suicide victims reporting increased 5-HT2A BPND sampled depressed subjects with greater severity of pessimism. See Fig. 5 which represents the relationship of sampling studies for major depressive disorder, dysfunctional attitudes and suicide. Interestingly, while these findings are consistent with postmortem study in suicide victims, they are also consistent with a model of heterogeneous extracellular serotonin loss in prefrontal cortex in MDD such that extracellular serotonin is lowest in major depressive episodes with more severe pessimism.
3.2 5-HTT Imaging and Pessimism
3.2.1 5-HTT Radioligands
In 2000, the first high quality radiotracer for measuring indices of serotonin transporter binding occurred with the development of [11C] DASB. The two previous radiotracers applied for in vivo imaging had significant limitations: 2-beta-carbomethoxy-3-beta-(4-iodophenyl)-tropane (β-CIT) single photon emission tomography (SPECT), the first technique developed had a specific binding signal that could be detected in the midbrain (Brucke et al. 1993; Innis et al. 1993; Kuikka et al. 1993), but it has almost equal affinity for the dopamine transporter as compared to the serotonin transporter (Carroll et al. 1995; Laruelle et al. 1994). Since there is high dopamine transporter density in the substantia nigra (Ciliax et al. 1999), the relative contributions of specific binding to dopamine and serotonin transporters cannot be differentiated in midbrain and it was the midbrain for which this radiotracer technique was applied as an index of serotonin transporter binding. The second applied radiotracer, [11C](+)McN5652, had better selectivity, but also had a low ratio of specific binding relative to free and non-specific binding. This disadvantage, in combination with modest reversibility makes valid, reliable quantitation difficult in regions other than the thalamus, and impossible in human cortex (Buck et al. 2000; Ikoma et al. 2002; Kent et al. 2002; Parsey et al. 2000). Thus, the radiotracer [11C] (DASB, 3-amino-4-(2-dimethylaminomethyl phenylsulfanyl)-benzonitrile) represented a major advance due to its high selectivity, reversibility, greater specific binding relative to free and non-specific binding and reliability (Ginovart et al. 2001; Houle et al. 2000; Ichise et al. 2003; Meyer et al. 2001b, 2004a, b; Praschak-Rieder et al. 2005; Wilson et al. 2000, 2002).
3.2.2 Interpretations of 5-HTT Binding Measurement
Measurement of serotonin transporter binding can be a useful index for several different models of disease that may affect extracellular serotonin levels. There are at least four such models may be considered in relationship to how serotonin transporter binding may be abnormal in a disease that lowers extracellular brain serotonin (Meyer 2007). These are referred to subsequently as models one through four. Abnormalities in serotonin transporter binding during major depressive episodes may be discussed in the context of these models.
Model one is a lesion model that reduces monoamine releasing neurons. In a lesion model, lowered 5-HTT binding measures occur. Model two is a secondary change in serotonin transporter binding as a sequelae to serotonin lowering via a different process. Model two is unlikely to be relevant for serotonin transporter binding. Acute reductions in serotonin have repeatedly shown reductions in 5-HTT mRNA (Linnet et al. 1995; Xiao et al. 1999; Yu et al. 1995). However, long-term reductions or elevations in serotonin typically show no effect upon regional 5-HTT density (Benmansour et al. 1999; Dewar et al. 1992; Graham et al. 1987). Model three is increased clearance of extracellular monoamine via greater monoamine transporter density. In model three, greater available serotonin transporter binding leads to greater clearance of monoamines from extracellular locations.
Model four is endogenous displacement and endogenous displacement is the property of a few radioligands to express different binding after short-term manipulations of their endogenous neurotransmitter, such that greater binding occurs during depletion of endogenous neurotransmitter and reduced binding occurs during elevations of endogenous neurotransmitter. For [11C] DASB, endogenous displacement may occur with large magnitude changes in extracellular 5-HT, but this is unlikely to occur with extracellular 5-HT changes that are physiologically tolerable for humans (Meyer 2007). For other PET radiotracers such as [11C]HOMADAM, [11C] MADAM, [11C] (+) McN5652, or SPECT radiotracers [123I]-B-CIT SPECT or [123I] ADAM SPECT, it is unknown whether endogenous levels of serotonin influence binding levels. This fourth model is unlikely to apply to PET imaging studies with [11C] DASB in humans, but it is unclear as to whether this model applies to other serotonin transporter radiotracers since the question has not been tested.
3.2.3 Dysfunctional Attitudes During Major Depressive Episodes and 5-HTT Binding
Dysfunctional attitudes, an index of pessimism, are elevated during major depressive disorder, and, as noted above, represent important symptoms because they generates sad mood and are strongly related to suicide. Hopelessness (Beck et al. 1985, 1989) and difficulty seeing positive reasons for living (Malone et al. 2000) are also significant risk factors for suicide and it has been demonstrated in four separate samples of subjects with major depressive episodes that greater hopelessness is highly positively correlated with greater severity of dysfunctional attitudes as measured with the dysfunctional attitudes scale (Bouvard et al. 1992; Cannon et al. 1999; DeRubeis et al. 1990; Norman et al. 1988).
In the two postmortem investigations of 5-HTT density in subjects with recent symptoms of depressive episodes no changes in 5-HTT density in the dorsal raphe or the locus coeruleus were found (Bligh-Glover et al. 2000; Klimek et al. 2003). Other postmortem investigations of 5-HTT density sampled subjects with a history of a depressive episode and these investigations usually studied the prefrontal cortex and/or dorsal raphe nucleus. Findings ranged from decreased 5-HTT density (Arango et al. 2001, Austin et al. 2002; Crow et al. 1984; Mann et al. 2000; Perry et al. 1983) to no difference in 5-HTT density (Hendricksen et al. 2004; Hrdina et al. 1990; Lawrence et al. 1990; Leake et al. 1991; Little et al. 1997). In several of these studies, subjects were medication free (Hrdina et al. 1990; Lawrence et al. 1990; Mann et al. 2000) and for many of these investigations, average postmortem delay was less than 1 day (Austin et al. 2002; Bligh-Glover et al. 2000; Klimek et al. 2003; Little et al. 1997; Mann et al. 2000; Perry et al. 1983). Further detail may be found in the review of Stockmeier (2003). Other sampling issues that influence postmortem investigations are inclusion of subjects with bipolar disorder and lack of differentiation between early versus late onset MDD. None of the postmortem studies investigated the relationship between 5-HTT binding and hopelessness or pessimism.
The first investigation of [11C]DASB PET imaging of major depressive disorder examined the relationship of 5-HTT BPND to severity of dysfunctional attitudes and presence of a major depressive episode. Meyer et al., sampled 20 subjects with major depressive episodes (from early onset major depressive disorder) and 20 healthy controls (Meyer et al. 2004a). Subjects were medication free for at least 3 months, had no other comorbid axis I illnesses, were nonsmoking, and had early onset major depressive disorder. There was no difference in 5-HTT BPND in either cortical or subcortical regions (including medial prefrontal cortex, dorsolateral prefrontal cortex and anterior cingulate cortex) between the group with major depressive episodes and healthy controls. However, subjects with severely pessimistic dysfunctional attitudes who were in the midst of major depressive episodes had significantly higher 5-HTT BPND, compared to healthy in brain regions sampling serotonin nerve terminals (dorsolateral and medial prefrontal cortex, anterior cingulated cortex, thalamus, bilateral caudate and bilateral putamen). On average, 5-HTT BPND was 21 % greater in these regions in subjects who were in the midst of major depressive episodes with severely pessimistic dysfunctional attitudes. Moreover, within the major depressive episode group, greater 5-HTT BPND was highly correlated with more negativistic dysfunctional attitudes in the same brain regions (see Fig. 6). The interpretation was that given serotonin transporters have an important role in influencing extracellular serotonin, greater regional 5-HTT levels provide greater vulnerability to low extracellular 5-HT and symptoms of extremely negativistic dysfunctional attitudes. This interpretation, in subjects with high levels of pessimism during MDE, corresponds to the third model discussed earlier under “Interpretations of 5-HTT Binding Measurement”.
Correlations between dysfunctional attitudes (DAS) and serotonin transporter binding potential (5-HTT BP) in some of the larger regions in depressed subjects. Highly significant correlations were found: prefrontal cortex (P = 0.0004), anterior cingulate (P = 0.002), bilateral putamen (P = 0.0002), bilateral thalamus (P = 0.001). Reprinted from the Archives of General Psychiatry
Neuroimaging investigations, sampling subjects with early onset MDD who are medication free for greater than 2 months, are non-smoking and do not have comorbid axis I disorders, and also apply better quality radiotracer technology, tend to find either no change in regional 5-HTT binding or an increase in regional 5-HTT binding (Cannon et al. 2007; Herold et al. 2006; Ichimiya et al. 2002; Meyer et al. 2004a). Investigations which include sampling of subjects with late onset MDD, comorbid axis I psychiatric disorders, recent antidepressant use, current cigarette smoking and do not apply a selective radiotracer are more likely to report a reduction in regional 5-HTT binding (Joensuu et al. 2007; Malison et al. 1998; Newberg et al. 2005; Parsey et al. 2006a; Selvaraj et al. 2009). Only the first study of [11C] DASB PET concurrently investigated a measure of pessimism or hopelessness.
3.2.4 Serotonin Transporter Binding and Seasonal Behaviour
Seasonal affective disorder is an important problem for countries with regions located at greater extremes of latitude. Rates of SAD typically range from 1 to 6 % in regions of 40′ latitude or greater (Magnusson 2000). Furthermore, at these latitudes, 25 % of healthy individuals experience lower mood, less energy, greater appetite and increased sleep in the winter (Kasper et al. 1989; Rosen et al. 1990).
There is significant evidence for seasonal variations in markers of serotonin physiology. In postmortem study of serotonin concentrations, Carlsson et al. reported a seasonal variation of serotonin levels in human hypothalamus with lower levels in late winter and higher levels in late summer (Carlsson et al. 1980). More recently, Lambert et al. found seasonal fluctuations in whole brain serotonin turnover in humans (Lambert et al. 2002). In rodents, reduced light exposure is associated with greater 5-HTT density (Rovescalli et al. 1989), lower 5-HT release (Blier et al. 1989) and greater 5-HT clearance (Rovescalli et al. 1989) in the hypothalamus and suprachiasmatic nucleus. It is possible that these findings were region specific or that the effect of light is more detectable in these regions of high 5-HT concentration and density.
While seasonal variation in pessimism has not been studied, the relationship of 5-HTT BPND to season has been subject to a number of interesting investigations. In a study of 5-HTT BPND with [11C] DASB PET in 88 healthy, non-smoking humans in Toronto, Canada, greater 5-HTT binding occurred in the fall/winter as compared to spring/summer in a number of affect modulating brain regions (Praschak-Rieder et al. 2008) (see Fig. 7). Another centre in Copenhagen, located at a latitude more north than Toronto replicated the effect of season in a sample of 54 subjects using [11C] DASB PET (Kalbitzer et al. 2009, seasonal changes in brain serotonin transporter, personal communication). In a combined sample of 49 healthy subjects and 49 depressed subjects, Ruhe et al. reported a similar relationship between midbrain binding and season (Ruhe et al. 2009) (although [123I]B-CIT SPECT is not very selective for 5-HTT). Buchert et al. reported the same seasonal finding in the midbrain in a sample of 39 subjects using [11C] McN5652 PET but not in the thalamus (Buchert et al. 2006). Although the Buchert study reported a positive result in only one of the two regions assayed, the sensitivity of this study to detect seasonal change was more modest: the sample is smaller than the other studies, and [11C] McN5652 is a less sensitive technique than [11C] DASB (Meyer 2007, 2008; Kent et al. 2002; Meyer et al. 2001b, 2004b). There are two studies in the literature that do not report greater 5-HTT binding in winter and both have a small sample size (n = 12 or less) (Koskela et al. 2008; Neumeister et al. 2000). To date, most studies of large sample size and reasonably northern latitude report greater 5-HTT BPND in most brain regions in the fall/winter as compared to spring/summer.
Regional 5-HTT BPND versus month (n=88 healthy subjects). Reciprocal peaks and troughs of brain serotonin transporter binding and duration of sunshine in 88 healthy study participants. Serotonin transporter binding potential values were measured in six brain regions. Circles represent bimonthly moving average means. Error bars represent 95 per cent confidence intervals of the mean. The shaded areas represent the average duration of sunshine in Toronto, Ontario, Canada. Regional 5-HTT BPND was significantly greater in spring/summer than fall/winter by 10 to 16%, p<0.02 for each region. Differences in peak to trough ranged from 22 to 42%. Reprinted from Meyer et al., Archives of General Psychiatry 65(9):1072–1078
3.3 5-HT1A Receptor Binding and Anxiety
Most [11C] WAY-100635 PET studies report lower 5-HT1A BPND in subregions of prefrontal cortex (dorsolateral, ventrolateral, orbitofrontal), anterior cingulate cortex, temporal cortex and raphe during major depressive episodes and continuance of this reduction during remission (Bhagwagar et al. 2004; Drevets et al. 1999; Sargent et al. 2000). There is one study with a different result and it may be that the selection of white matter as a reference region, and/or sampling characteristics may account for the difference (Parsey et al. 2006b). A potential problem with applying white matter as a reference tissue is that in contrast to using grey matter in cerebellar cortex, the properties of white matter are more likely to be different from grey matter, and the modelling method requires the assumption that the free and non-specific binding in the reference tissue is similar to grey matter tissue. Hence, a grey matter region with low specific binding is preferable as a reference tissue such as the cerebellar cortex excluding the vermis.
There is reason to consider that finding in MDD may actually be more strongly associated with anxiety, which often occurs during major depressive episodes or presence of comorbid anxiety disorders, which also frequently occur with MDD since reduced 5-HT1A binding also occurs in affect modulating regions also occurs in anxiety disorders. In a [18F]trans-4-fluoro-N-2-[4-(2-methoxyphenyl)piperazin-1-yl]ethyl]-N-(2-pyridyl) cyclohexanecarboxamide ([18F]-FCWAY) study of panic disorder (with comorbid MDD in almost half the subject sample), significant reductions in 5-HT1A binding in anterior cingulate, posterior cingulate and raphe regions were reported (Neumeister et al. 2004a). (Quantitation with this radiotracer is valid for subcortical regions but not cortical regions due to bone uptake of radiotracer metabolite.) A second study applying [11C] WAY100635 reported similar findings with the greatest magnitude being in orbitofrontal cortex, temporal cortex and midbrain (Nash et al. 2008). In social anxiety disorder, reduced 5-HT1A binding occurs in most brain regions (insula, anterior cingulate cortex, medial orbitofrontal cortex, amygdala and midbrain) (Lanzenberger et al. 2007). Since the neuroimaging studies of 5-HT1A binding in MDD did not exclude comorbid anxiety disorders, one interpretation of the reduced 5-HT1A binding in the brain regions assayed in MDD, particularly in orbitofrontal cortex, anterior cingulate cortex and midbrain, is that it reflects comorbid anxiety disorders. MDD with anxiety is often treatment resistant, and this interpretation is appealing because there could be an opportunity to target pathologies related to this abnormality in this treatment resistant subgroup (Trivedi et al. 2006).
A limitation of this interpretation is that a postmortem study with subjects who mainly did not have comorbid anxiety disorders reported decreased 5-HT1A antagonist binding in orbitofrontal cortex (Stockmeier et al. 2009) and another explanation could be that abnormally low 5-HT1A density in prefrontal and cingulate cortex is associated with both MDD and anxiety disorders. Future work should investigate the cellular mechanism, and identify the clinical subgroup of MDD with this finding so as to optimise therapeutics. One explanation is that lower 5-HT1A receptor density reflects decreased 5-HT1A receptors on GABA interneurons or the segments of pyramidal cell axons proximal to extensions from GABA containing chandelier interneurons (Stockmeier et al. 2009).
4 D2, DAT and Motor Slowing
4.1 Dorsal Striatal [11C]Raclopride Binding and Motor Retardation in Major Depressive Disorder
Among the symptoms of MDEs, motor retardation and anhedonia both reflect functions modulated by dopamine release in the dorsal putamen and nucleus accumbens respectively. The second symptom, anhedonia, is more difficult to investigate with neuroimaging markers of dopamine: The nucleus accumbens is a smaller structure. Anhedonia is difficult to measure during major depressive episodes since it can be biased by negativistic perspective if the measure involves self report or motor speed if the measure involves movement. However, given recent advances in the development of neuroimaging markers for D3 receptors for the ventral striatum, and ongoing development of quantitation of hedonic measures in depression, it is likely that future investigations will focus upon this direction (Willeit et al. 2008). The first symptom, motor retardation, is straightforward to investigate with quantitative measures because dorsal putamen is large, motor retardation can be measured with well validated motor tests such as the finger tapping test, and [11C] raclopride has a strong specific binding signal in dorsal putamen. [11C] raclopride is a widely available PET radiotracer selective for D2 type receptors whose specific binding changes inversely relative to changes in neurotransmitter levels (Laruelle 2000).
In addition to the straightforward ability to quantify D2 BPND in dorsal striatum, there were other reasons to investigate this biomarker in relation to motor retardation. One is that greater D2 BPND and reduced dopamine levels occur in dorsal striatum in Parkinson’s disease and other similar diseases that involve reduced movement speed (Kim et al. 2002; Kish et al. 1988). The second is that D2 BPND found with [11C]raclopride PET, is inversely proportional to extracellular dopamine levels in animal and human paradigms that manipulate dopamine levels (Laruelle 2000). The third reason is that decreased cerebrospinal fluid levels of the dopamine metabolite homovanillic acid is often reported during MDE with motor retardation, suggesting that some regions of the brain have lower levels of dopamine when motor retardation is present during major depressive episodes (Korf and Praag 1971; Post et al. 1973; Praag et al. 1975).
Unfortunately, early imaging studies of dopamine receptors did not sample depressed subjects who were medication free and non-smoking or addressing the confounding effect of age (Meyer 2008). Since D2 BPND declines with age and motor retardation increases with age, to assess this relationship in a manner that addressed the age bias, one must correct for the effect of age upon each variable prior to investigating the relationship between the two. There is only one study that meets all of these criteria, and there is no other study meeting two of these three criteria (Meyer 2008). The main findings of the one, unbiased study, of striatal D2 BPND and motor retardation were that the caudate and putamen D2 BPND were elevated in the depressed group as compared to the healthy group, and that greater putamen D2 BPND was significantly correlated with more severe motor retardation in the depressed group (Meyer et al. 2006b) (see Fig. 8). The findings support a specific, neuromodulatory role for striatal dopamine loss during MDE, particularly when motor retardation is present.
(a) Correlation between bilateral putamen D2 receptor binding potential and motor speed in depressed (n=19) subjects (The correlation was significant (r = -0.53, p = 0.02). In order to reduce variance related to age and gender, individual putamen D2 BP values were normalized to age 30 using the slope of the linear decline in D2 BP with age. Motor speed was measured with the finger tapping test. Individual finger tapping scores were normalized similarly to a 30 year old male (Meyer et al. 2006a, b). (b) Striatal D2 Receptor Binding Potential in Motor Retarded Depressed (n=10), Other Depressed (n=11) and Healthy Subjects (n=21) (Striatal D2 Receptor Binding Potential in Motor Retarded Depressed (n=10), Other Depressed (n=11) and Healthy Subjects (n=21) (D2 binding potential values were normalized to a 30-year-old subject using the slope of the age-related decline. Differences between the healthy group and the depressed subjects with motor retardation assessed by means of independent sample t tests. *p ≤ 0.05 **p ≤ 0.01 ***p ≤ 0.005)
4.2 Striatal DAT Binding and Motor Retardation During Major Depressive Episodes
With regard to dopamine transporter (DAT) imaging in MDD, studies sampling medication free subjects typically report lower striatal DAT BPND (Meyer et al. 2001a; Neumeister et al. 2001; Sarchiapone et al. 2006), whereas studies sampling subjects with recent antidepressant treatment sometimes report higher striatal DAT binding (Brunswick et al. 2003). Most studies address the age-related decline in DAT and some address the confound of cigarette smoking. Mechanisms to explain reduced striatal DAT binding include lesions to dopamine releasing neurons or a downregulation model (in response to another monoamine lowering process). In contrast to the serotonin transporter, dopamine transporters in the striatum downregulate when subacute to chronic dopamine depletion occurs (Gordon et al. 1996; Han et al. 1999; Ikawa et al. 1993; Kilbourn et al. 1992).
The downregulation model may explain reduced striatal DAT binding during MDD since greater MAO-A levels occur in striatum in major depressive episodes and MAO-A metabolises dopamine (Meyer et al. 2006a, 2009). In addition, greater striatal D2 BPND was found during major depressive episodes with [11C]raclopride PET and this index is increased when extracellular dopamine is reduced (Laruelle 2000).
Also, the correlation between lower dorsal putamen DAT BPND values and less impaired performance on the finger tapping test has a particular interpretation (Meyer et al. 2001a). The finger tapping test is a measure of motor slowing in MDD (Meyer et al. 2001a, 2006b). Slower performance on the finger tapping test is correlated with greater putamen D2 BPND during MDE and greater D2 BPND from [11C] raclopride occurs when extracellular dopamine is lower (Laruelle 2000). The data can be interpreted as follows: Major depressive episodes without motor retardation are associated with lower DAT BPND and demonstrate a compensatory protective mechanism (Meyer et al. 2001a). Downregulation of DAT occurs when dopamine is chronically low in striatum (Gordon et al. 1996; Han et al. 1999; Ikawa et al. 1993; Kilbourn et al. 1992), but reduced DAT levels decrease clearance of extracellular dopamine. Compared with the healthy state, the compensated state has only mildly reduced extracellular striatal dopamine concentrations with downregulated DAT. This is the process whereby dorsal striatal DAT BPND is decreased protects against motor slowing.
5 Behavioural Correlates of With Abnormalities in Glutatmate Regulation
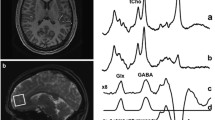
Magnetic resonance spectroscopy (MRS) can measure ‘Glx’ in brain tissue, an index mainly composed of glutamate (intracellular and extracellular), and glutamine. Exposure to elevated glutamate levels has been proposed as a mechanism that leads to sad mood because treatment resistant MDD subjects often experience a rapid, short term, mood elevation after single dose ketamine, a NMDA receptor antagonist and some subtypes of MDE have a response to lamotrigine, a medication that can reduce glutamate release (Barbee and Jamhour 2002; Barbee et al. 2011; Zarate et al. 2006). Also, elevations in extracellular glutamate have been proposed as being relevant to mood symptoms because reductions in glia are reported in orbitofrontal, dorsolateral prefrontal and anterior cingulate cortex in MDD and glia clear glutamate via excitatory amino acid transporters (Rajkowska and Miguel-Hidalgo 2007).
There are several studies applying proton MRS to measure Glx, in reasonably large samples of MDD with region specific results. For example, greater Glx levels were reported in the occipital cortex, a reduction was reported in the medial prefrontal cortex and no change was reported in the pregenual cingulate cortex (Hasler et al. 2007; Sanacora et al. 2004; Walter et al. 2009). A reasonable explanation for the anatomical variation is that glutamate levels are generally elevated in the brain during MDD, and that regions with reduced glial cell density (Rajkowska and Miguel-Hidalgo 2007) have reduced Glx because the intracellular contribution of Glx signal are lower. Some level of homogenous abnormality in brain glutamate regulation during MDD is suggested by a recent [11C]ABP688 study reporting reduced prefrontal cortex, cingulate cortex, insula, thalamus and hippocampus mGlu5 receptor binding in conjunction with a postmortem study of reduced mGlu5 density in prefrontal cortex (Deschwanden et al. 2011). The investigation of pregenual cingulate cortex did detect, in highly anhedonic depressed subjects, decreased glutamine, a metabolic product of glutamate (Walter et al. 2009) and the investigation reporting greater glutamate in the occipital cortex found the greatest levels in melancholia (Hasler et al. 2007; Sanacora et al. 2004; Walter et al. 2009). Ideally, future studies should aim to sample the regions previously investigated concurrently so as to optimally address the regional specificity of findings, and preferentially sample melancholic and anhedonic subjects so as to replicate the findings by Sanacora et al. (2004) and Walter et al.(2009). While there is evidence for disturbances in glutamate levels during major depressive episodes, the mechanism of the relationship to specific mood symptoms requires further study.
6 Conclusions
While neuroimaging is limited by the range in biomarkers available, its ability for in vivo measurement is bridging markers of neurochemistry and neuroplasticity to pathophysiological mechanism of symptom onset for major depressive disorder. For example, indices of greater monoamine oxidase A (MAO-A) level, particularly in the prefrontal and anterior cingulate cortex, are associated with depressed mood states, and high-risk states for onset of major depressive episodes. MAO-A metabolises monoamines, and greater metabolism of monoamines occurs when MAO-A is elevated in brain. Hence, greater levels of MAO-A may be viewed as a key monoamine lowering process during major depressive episodes.
Evidence to date suggests that ongoing deficiency of specific monoamines in specific regions is implicated in symptoms of disease. Lower extracellular serotonin is associated with greater pessimism in humans and chronic serotonin deficiency is associated with upregulation of 5-HT2A (serotonin2A) receptors in cortex. During major depressive episodes, when pessimism is more severe, greater 5-HT2A binding occurs in prefrontal and anterior cingulate cortex. These results argue for a mechanism of lowering extracellular serotonin in the prefrontal and anterior cingulate cortex, consequent to elevated MAO-A level. The relationship between elevated 5-HTT binding greater pessimism during major depressive episodes suggests that greater 5-HTT density in the context of elevated MAO-A level further contributes to serotonin deficiency in these brain regions (see Fig. 9). A similar mechanism may explain the association between neuroimaging indices of greater dorsal striatal D2 density, DAT density and symptoms of motor retardation: Greater MAO-A level and relatively greater DAT density lower extracellular dopamine in the dorsal striatum, leading to motor retardation (also see Fig. 9).
Modern Model of Excessive Extracellular Monoamine Loss During Major Depressive Disorder (a) Monoamine release in a synapse in health. (b) During a major depressive episode, monoamine oxidase A (MAO-A) density is elevated resulting in greater metabolism of monoamines. Outcomes range from (c) to (d). (c) If the monoaminge transporter density is low during a major depressive episode, the effect of elevated MAO-A upon reducing extracellular monoamine is attenuated resulting in a moderate loss of monoamine, eventually resulting in a moderate rise in symptoms. (d) If the monoamine transporter density is not low during a major depressive episode, then there is no protection against the effect of elevated MAO-A. The extracellular concentration of the monoamine is severely reduced and rise in symptom s is severe. This model applies to regions whose functions are affected by changes in monoamine levels. Studies of transporter binding related binding to function (such as motor retardation to striatal DAT binding, and pessmism to prefrontal/anterior cingulate cortex 5-HTT binding. MAO-A in cell types not proximal to the release of the individual monoamine may play a role in its metabolism (for example MAO-A in norepinephrine releasing neurons, glia, and astrocytes may also play a role in the metabolism of serotonin)
Specific mechanisms underlying other neuroimaging abnormalities continue to be investigated. Indices of greater 5-HT1A density, particularly in the cingulate cortex, have been associated with major depressive disorder, and well as anxiety disorders, suggesting that this abnormality is mechanistically related to presence of anxiety symptoms. At this point, abnormalities in glutamate level have been most strongly associated with presence of major depressive episodes, with greater levels in occipital cortex, and reduced levels in prefrontal cortex. Future neurochemical imaging investigations will ultimately focus upon detecting the mechanisms that predispose towards onset of MDE so as to create new biologically informed methods of prevention, and superior, more symptom targeted treatments based upon symptom specific underlying pathology.
References
American Psychiatric Association (1994) Diagnostic and statistical manual of mental disorders. IV edn. Washington, DC. Am Psychiatr Assoc 387
Anda RF, Williamson DF, Escobedo LG, Mast EE, Giovino GA, Remington PL (1990) Depression and the dynamics of smoking a national perspective. JAMA 264:1541–1545
Arango V, Ernsberger P, Marzuk PM, Chen JS, Tierney H, Stanley M, Reis DJ, Mann JJ (1990) Autoradiographic demonstration of increased serotonin 5-HT2 and beta- adrenergic receptor binding sites in the brain of suicide victims. Arch Gen Psychiatry 47:1038–1047
Arango V, Underwood M, Mann J (1992) Alterations in monoamine receptors in the brain of suicide victims. J Clin Psychopharm 12:8S–12S
Arango V, Underwood MD, Boldrini M, Tamir H, Kassir SA, Hsiung S, Chen JJ, Mann JJ (2001) Serotonin 1A receptors, serotonin transporter binding and serotonin transporter mRNA expression in the brainstem of depressed suicide victims. Neuro psychopharmacol 25:892–903
Arora RC, Meltzer HY (1989) Serotonergic measures in the brains of suicide victims: 5-HT2 binding sites in the frontal cortex of suicide victims and control subjects. Am J Psychiatry 146:730–736
Attar-Levy D, Martinot J-L, Blin J, Dao-Castellana M-H, Crouzel C, Mazoyer B, Poirer M-F, Bourdel M-C, Aymard N, Syrota A, Feline A (1999) The cortical serotonin2 receptors studied with positron emission tomography and [18F]-setoperone during depressive illness and antidepressant treatment with clomipramine. Biol Psychiatry 45:180–186
Austin MC, Whitehead RE, Edgar CL, Janosky JE, Lewis DA (2002) Localized decrease in serotonin transporter-immunoreactive axons in the prefrontal cortex of depressed subjects committing suicide. Neuroscience 114:807–815
Bacher I, Houle S, Xu X, Zawertailo L, Soliman A, Wilson AA, Selby P, George TP, Sacher J, Miler L, Kish SJ, Rusjan P, Meyer JH (2011) Monoamine oxidase a binding in the prefrontal and anterior cingulate cortices during acute withdrawal from heavy cigarette smoking. Arch Gen Psychiatry 68:817–826
Barbee JG, Jamhour NJ (2002) Lamotrigine as an augmentation agent in treatment-resistant depression. J Clin Psychiatry 63:737–741
Barbee JG, Thompson TR, Jamhour NJ, Stewart JW, Conrad EJ, Reimherr FW, Thompson PM, Shelton RC (2011) A double-blind placebo-controlled trial of lamotrigine as an antidepressant augmentation agent in treatment-refractory unipolar depression. J Clin Psychiatry 72:1405–1412
Barraclough B, Bunch J, Nelson B, Sainsbury P (1974) A hundred cases of suicide: clinical aspects. Br J Psychiatry 125:355–373
Barton DA, Esler MD, Dawood T, Lambert EA, Haikerwal D, Brenchley C, Socratous F, Hastings J, Guo L, Wiesner G, Kaye DM, Bayles R, Schlaich MP, Lambert GW (2008) Elevated brain serotonin turnover in patients with depression: effect of genotype and therapy. Arch Gen Psychiatry 65:38–46
Beck A, Steer R, Kovacs M, Garrison B (1985) Hopelessness and eventual suicide: a 10-year prospective study of patients hospitalized with suicidal ideation. Am J Psychiatry 142(5):559–563
Beck AT, Brown G, Steer RA (1989) Prediction of eventual suicide in psychiatric inpatients by clinical ratings of hopelessness. J Consult Clin Psychol 57:309–310
Benmansour S, Cecchi M, Morilak D, Gerhardt G, Javors M, Gould G, Frazer A (1999) Effects of chronic antidepressant treatments on serotonin transporter function. Density mRNA Level J Neurosci 19(23):10494–10501
Bhagwagar Z, Rabiner EA, Sargent PA, Grasby PM, Cowen PJ (2004) Persistent reduction in brain serotonin1A receptor binding in recovered depressed men measured by positron emission tomography with [11C]WAY-100635. Mol Psychiatry 9:386–392
Bhagwagar Z, Hinz R, Taylor M, Fancy S, Cowen P, Grasby P (2006) Increased 5-HT(2A) receptor binding in euthymic, medication-free patients recovered from depression: a positron emission study with [(11)C]MDL 100,907. Am J Psychiatry 163:1580–1587
Biver F, Wikler D, Lotstra F, Damhaut P, Goldman S, Mendlewicz J (1997) Serotonin 5-HT2 receptor imaging in major depression: focal changes in orbito-insular cortex. Br J Psychiatry 171:444–448
Blier P, Galzin AM, Langer SZ (1989) Diurnal variation in the function of serotonin terminals in the rat hypothalamus. J Neurochem 52:453–459
Bligh-Glover W, Kolli TN, Shapiro-Kulnane L, Dilley GE, Friedman L, Balraj E, Rajkowska G, Stockmeier CA (2000) The serotonin transporter in the midbrain of suicide victims with major depression. Biol Psychiatry 47:1015–1024
Bouvard M, Charles S, Guerin J, Aimard G, Cottraux J (1992) Study of beck’s hopelessness scale. validation and factor analysis. Encephale 18:237–240
Breslau N, Peterson EL, Schultz LR, Chilcoat HD, Andreski P (1998) Major depression and stages of smoking a longitudinal investigation. Arch Gen Psychiatry 55:161–166
Brockington IF, Cernik KF, Schofield EM, Downing AR, Francis AF, Keelan C (1981) Puerperal psychosis phenomena and diagnosis. Arch Gen Psychiatry 38:829–833
Brucke T, Kornhuber J, Angelberger P, Asenbaum S, Frassine H, Podreka I (1993) SPECT imaging of dopamine and serotonin transporters with [123I]beta-CIT. binding kinetics in the human brain. J Neural Transm Gen Sect 94:137–146
Brunswick DJ, Amsterdam JD, Mozley PD, Newberg A (2003) Greater availability of brain dopamine transporters in major depression shown by [99m Tc]TRODAT-1 SPECT imaging. Am J Psychiatry 160:1836–1841
Buchert R, Schulze O, Wilke F, Berding G, Thomasius R, Petersen K, Brenner W, Clausen M (2006) Is correction for age necessary in SPECT or PET of the central serotonin transporter in young, healthy adults? J Nucl Med 47:38–42
Buck A, Gucker PM, Schonbachler RD, Arigoni M, Kneifel S, Vollenweider FX, Ametamey SM, Burger C (2000) Evaluation of serotonergic transporters using PET and [11C](+)McN-5652: assessment of methods. J Cereb Blood Flow Metab 20:253–262
Cane D, Olinger L, Gotlib I, Kuiper N (1986) Factor structure of the dysfunctional attitude scale in a student population. J Clin Psychol 42:307–309
Cannon B, Mulroy R, Otto MW, Rosenbaum JF, Fava M, Nierenberg AA (1999) Dysfunctional attitudes and poor problem solving skills predict hopelessness in major depression. J Affect Disord 55:45–49
Cannon DM, Ichise M, Rollis D, Klaver JM, Gandhi SK, Charney DS, Manji HK, Drevets WC (2007) Elevated serotonin transporter binding in major depressive disorder assessed using positron emission tomography and [(11)C]DASB; comparison with bipolar disorder. Biol Psychiatry 62:870–877
Carey MP, Kalra DL, Carey KB, Halperin S, Richards CS (1993) Stress and unaided smoking cessation: a prospective investigation. J Consult Clin Psychol 61:831–838
Carlsson A, Svennerholm L, Winblad B (1980) Seasonal and circadian monoamine variations in human brains examined post mortem. Acta Psychiatr Scand Suppl 280:75–85
Carroll FI, Kotian P, Dehghani A, Gray JL, Kuzemko MA, Parham KA, Abraham P, Lewin AH, Boja JW, Kuhar MJ (1995) Cocaine and 3 beta-(4′-substituted phenyl)tropane-2 beta-carboxylic acid ester and amide analogues. New high-affinity and selective compounds for the dopamine transporter. J Med Chem 38:379–388
Ciliax BJ, Drash GW, Staley JK, Haber S, Mobley CJ, Miller GW, Mufson EJ, Mash DC, Levey AI (1999) Immunocytochemical localization of the dopamine transporter in human brain. J Comp Neurol 409:38–56
Crow TJ, Cross AJ, Cooper SJ, Deakin JF, Ferrier IN, Johnson JA, Joseph MH, Owen F, Poulter M, Lofthouse R et al (1984) Neurotransmitter receptors and monoamine metabolites in the brains of patients with Alzheimer-type dementia and depression, and suicides. Neuropharmacology 23:1561–1569
DeRubeis RJ, Evans MD, Hollon SD, Garvey MJ, Grove WM, Tuason VB (1990) How does cognitive therapy work? Cognitive change and symptom change in cognitive therapy and pharmacotherapy for depression. J Consult Clin Psychol 58:862–869
Deschwanden A, Karolewicz B, Feyissa AM, Treyer V, Ametamey SM, Johayem A, Burger C, Auberson YP, Sovago J, Stockmeier CA, Buck A, Hasler G (2011) Reduced metabotropic glutamate receptor 5 density in major depression determined by [11C]ABP688 PET and postmortem study. Am J Psychiatry 168:727–734
Dewar KM, Grondin L, Carli M, Lima L, Reader TA (1992) [3H]paroxetine binding and serotonin content of rat cortical areas, hippocampus, neostriatum, ventral mesencephalic tegmentum, and midbrain raphe nuclei region following p-chlorophenylalanine and p-chloroamphetamine treatment. J Neurochem 58:250–257
D-haenen H, Bossuyt A, Mertens J, Bossuyt-Piron C, Gijesmans M, Kaufman L (1992) SPECT imaging of serotonin2 receptors in depression. Psychiatry Res Neuroimaging 45:227–237
Drevets WC, Frank E, Price JC, Kupfer DJ, Holt D, Greer PJ, Huang Y, Gautier C, Mathis C (1999) PET imaging of serotonin 1A receptor binding in depression. Biol Psychiatry 46:1375–1387
Elliott R, Rubinsztein JS, Sahakian BJ, Dolan RJ (2002) The neural basis of mood-congruent processing biases in depression. Arch Gen Psychiatry 59:597–604
Fava M, Bless E, Otto M, Pava J, Rosenbaum J (1994) Dysfunctional attitudes in major depression changes with pharmacotherapy. J Nerv Ment Dis 182(1):45–49
Fowler JS, Volkow ND, Wang GJ, Pappas N, Logan J, Shea C, Alexoff D, MacGregor RR, Schlyer DJ, Zezulkova I, Wolf AP (1996) Brain monoamine oxidase A inhibition in cigarette smokers. Proc Natl Acad Sci U S A 93:14065–14069
Freis ED (1954) Mental depression in hypertensive patients treated for long periods with large doses of reserpine. N Engl J Med 251:1006–1008
Frokjaer VG, Mortensen EL, Nielsen FA, Haugbol S, Pinborg LH, Adams KH, Svarer C, Hasselbalch SG, Holm S, Paulson OB, Knudsen GM (2008) Frontolimbic serotonin 2A receptor binding in healthy subjects is associated with personality risk factors for affective disorder. Biol Psychiatry 63:569–576
Garvey AJ, Bliss RE, Hitchcock JL, Heinold JW, Rosner B (1992) Predictors of smoking relapse among self-quitters: a report from the normative aging study. Addict Behav 17:367–377
Ginovart N, Wilson AA, Meyer JH, Hussey D, Houle S (2001) Positron emission tomography quantification of [(11)C]-DASB binding to the human serotonin transporter: modeling strategies. J Cereb Blood Flow Metab 21:1342–1353
Gordon I, Weizman R, Rehavi M (1996) Modulatory effect of agents active in the presynaptic dopaminergic system on the striatal dopamine transporter. Eur J Pharmacol 298:27–30
Graham D, Tahraoui L, Langer SZ (1987) Effect of chronic treatment with selective monoamine oxidase inhibitors and specific 5-hydroxytryptamine uptake inhibitors on [3H]paroxetine binding to cerebral cortical membranes of the rat. Neuropharmacology 26:1087–1092
Han S, Rowell PP, Carr LA (1999) D2 autoreceptors are not involved in the down-regulation of the striatal dopamine transporter caused by alpha-methyl-p-tyrosine. Res Commun Mol Pathol Pharmacol 104:331–338
Hasler G, van der Veen JW, Tumonis T, Meyers N, Shen J, Drevets WC (2007) Reduced prefrontal glutamate/glutamine and gamma-aminobutyric acid levels in major depression determined using proton magnetic resonance spectroscopy. Arch Gen Psychiatry 64:193–200
Hasler G, Fromm S, Carlson PJ, Luckenbaugh DA, Waldeck T, Geraci M, Roiser JP, Neumeister A, Meyers N, Charney DS, Drevets WC (2008) Neural response to catecholamine depletion in unmedicated subjects with major depressive disorder in remission and healthy subjects. Arch Gen Psychiatry 65:521–531
Hendricksen M, Thomas AJ, Ferrier IN, Ince P, O’Brien JT (2004) Neuropathological study of the dorsal raphe nuclei in late-life depression and Alzheimer’s disease with and without depression. Am J Psychiatry 161:1096–1102
Herold N, Uebelhack K, Franke L, Amthauer H, Luedemann L, Bruhn H, Felix R, Uebelhack R, Plotkin M (2006) Imaging of serotonin transporters and its blockade by citalopram in patients with major depression using a novel SPECT ligand [(123)I]-ADAM. J Neural Transm 113:659–670
Houle S, Ginovart N, Hussey D, Meyer J, Wilson A (2000) Imaging the serotonin transporter with positron emission tomography: initial human studies with [11C]DAPP and [11C]DASB. Eur J Nucl Med 27:1719–1722
Hoyer D, Pazos A, Probst A, Palacios JM (1986) Serotonin receptors in the human brain. II. characterization and autoradiographic localization of 5-HT1C and 5-HT2 recognition sites. Brain Res 376:97–107
Hrdina P, Vu T (1993) Chronic fluoxetine treatment upregulates 5-HT uptake sites and 5-HT2 receptors in rat brain: an autoradiographic study. Synapse 14:324–331
Hrdina P, Foy B, Hepner A, Summers R (1990) Antidepressant binding sites in brain: autoradiographic comparison of [3H]paroxetine and [3H]imipramine localization and relationship to serotonin transporter. J Pharm Exp Ther 252:410–418
Ichimiya T, Suhara T, Sudo Y, Okubo Y, Nakayama K, Nankai M, Inoue M, Yasuno F, Takano A, Maeda J, Shibuya H (2002) Serotonin transporter binding in patients with mood disorders: a PET study with [11C](+)McN5652. Biol Psychiatry 51:715–722
Ichise M, Liow JS, Lu JQ, Takano A, Model K, Toyama H, Suhara T, Suzuki K, Innis RB, Carson RE (2003) Linearized reference tissue parametric imaging methods: application to [11C]DASB positron emission tomography studies of the serotonin transporter in human brain. J Cereb Blood Flow Metab 23:1096–1112
Ikawa K, Watanabe A, Kaneno S, Toru M (1993) Modulation of [3H]mazindol binding sites in rat striatum by dopaminergic agents. Eur J Pharmacol 250:261–266
Ikoma Y, Suhara T, Toyama H, Ichimiya T, Takano A, Sudo Y, Inoue M, Yasuno F, Suzuki K (2002) Quantitative analysis for estimating binding potential of the brain serotonin transporter with [11 C]McN5652. J Cereb Blood Flow Metab 22:490–501
Innis RB, Seibyl JP, Scanley BE, Laruelle M, Abi-Dargham A, Wallace E, Baldwin RM, Zea-Ponce Y, Zoghbi S, Wang S et al (1993) Single photon emission computed tomographic imaging demonstrates loss of striatal dopamine transporters in Parkinson disease. Proc Natl Acad Sci U S A 90:11965–11969
Joensuu M, Tolmunen T, Saarinen PI, Tiihonen J, Kuikka J, Ahola P, Vanninen R, Lehtonen J (2007) Reduced midbrain serotonin transporter availability in drug-naive patients with depression measured by SERT-specific [(123)I] nor-beta-CIT SPECT imaging. Psychiatry Res 154:125–131
Johnson S, Stockmeier CA, Meyer JH, Austin MC, Albert PR, Wang J, May WL, Rajkowska G, Overholser JC, Jurjus G, Dieter L, Johnson C, Sittman DB, Ou XM (2011) The reduction of R1, a novel repressor protein for monoamine oxidase A, in major depressive disorder. Neuropsychopharmacology 36(10):2139–2148
Kasper S, Wehr TA, Bartko JJ, Gaist PA, Rosenthal NE (1989) Epidemiological findings of seasonal changes in mood and behavior. A telephone survey of Montgomery county. Md Arch Gen Psychiatry 46:823–833
Kendell RE, Chalmers JC, Platz C (1987) Epidemiology of puerperal psychoses. Br J Psychiatry 150:662–673
Kenford SL, Smith SS, Wetter DW, Jorenby DE, Fiore MC, Baker TB (2002) Predicting relapse back to smoking: contrasting affective and physical models of dependence. J Consult Clin Psychol 70:216–227
Kent JM, Coplan JD, Lombardo I, Hwang DR, Huang Y, Mawlawi O, Van Heertum RL, Slifstein M, Abi-Dargham A, Gorman JM, Laruelle M (2002) Occupancy of brain serotonin transporters during treatment with paroxetine in patients with social phobia: a positron emission tomography study with 11C McN 5652. Psychopharmacology (Berl) 164:341–348
Kilbourn MR, Sherman PS, Pisani T (1992) Repeated reserpine administration reduces in vivo [18F]GBR 13119 binding to the dopamine uptake site. Eur J Pharmacol 216:109–112
Kim YJ, Ichise M, Ballinger JR, Vines D, Erami SS, Tatschida T, Lang AE (2002) Combination of dopamine transporter and D2 receptor SPECT in the diagnostic evaluation of PD, MSA, and PSP. Mov Disord 17:303–312
Kish SJ, Shannak K, Hornykiewicz O (1988) Uneven pattern of dopamine loss in the striatum of patients with idiopathic Parkinson’s disease. Pathophysiologic and clinical implications. N Engl J Med 318:876–880
Klimek V, Roberson G, Stockmeier CA, Ordway GA (2003) Serotonin transporter and MAO-B levels in monoamine nuclei of the human brainstem are normal in major depression. J Psychiatr Res 37:387–397
Konradi C, Svoma E, Jellinger K, Riederer P, Denney R, Thibault J (1988) Topographic immunocytochemical mapping of monoamine oxidase-A, monoamine oxidase-B and tyrosine hydroxylase in human post mortem brain stem. Neuroscience 26:791–802
Konradi C, Kornhuber J, Froelich L, Fritze J, Heinsen H, Beckmann H, Schulz E, Riederer P (1989) Demonstration of monoamine oxidase-A and -B in the human brainstem by a histochemical technique. Neuroscience 33:383–400
Kontur PJ, al-Tikriti M, Innis RB, Roth RH (1994) Postmortem stability of monoamines, their metabolites, and receptor binding in rat brain regions. J Neurochem 62:282–290
Korf J, Praag HMV (1971) Retarded depression and the dopamine metabolism. Psychopharmacologia 19:199–203
Koskela A, Kauppinen T, Keski-Rahkonen A, Sihvola E, Kaprio J, Rissanen A, Ahonen A (2008) Brain serotonin transporter binding of [123I]ADAM: within-subject variation between summer and winter data. Chronobiol Int 25:657–665
Kuikka JT, Bergstrom KA, Vanninen E, Laulumaa V, Hartikainen P, Lansimies E (1993) Initial experience with single-photon emission tomography using iodine-123-labelled 2 beta-carbomethoxy-3 beta-(4-iodophenyl) tropane in human brain. Eur J Nucl Med 20:783–786
Lambert GW, Reid C, Kaye DM, Jennings GL, Esler MD (2002) Effect of sunlight and season on serotonin turnover in the brain. Lancet 360:1840–1842
Lanzenberger RR, Mitterhauser M, Spindelegger C, Wadsak W, Klein N, Mien LK, Holik A, Attarbaschi T, Mossaheb N, Sacher J, Geiss-Granadia T, Kletter K, Kasper S, Tauscher J (2007) Reduced serotonin-1A receptor binding in social anxiety disorder. Biol Psychiatry 61:1081–1089
Laruelle M (2000) Imaging synaptic neurotransmission with in vivo binding competition techniques: a critical review. J Cereb Blood Flow Metab 20:423–451
Laruelle M, Giddings SS, Zea-Ponce Y, Charney DS, Neumeyer JL, Baldwin RM, Innis RB (1994) Methyl 3 beta-(4-[125I]iodophenyl)tropane-2 beta-carboxylate in vitro binding to dopamine and serotonin transporters under “physiological” conditions. J Neurochem 62:978–986
Law M, Tang JL (1995) An analysis of the effectiveness of interventions intended to help people stop smoking. Arch Intern Med 155:1933–1941
Lawrence KM, De Paermentier F, Cheetham SC, Crompton MR, Katona CL, Horton RW (1990) Brain 5-HT uptake sites, labelled with [3H]paroxetine, in antidepressant-free depressed suicides. Brain Res 526:17–22
Leake A, Fairbairn AF, McKeith IG, Ferrier IN (1991) Studies on the serotonin uptake binding site in major depressive disorder and control post-mortem brain: neurochemical and clinical correlates. Psychiatry Res 39:155–165
Linnet K, Koed K, Wiborg O, Gregersen N (1995) Serotonin depletion decreases serotonin transporter mRNA levels in rat brain. Brain Res 697:251–253
Liotti M, Mayberg HS, McGinnis S, Brannan SL, Jerabek P (2002) Unmasking disease-specific cerebral blood flow abnormalities: mood challenge in patients with remitted unipolar depression. Am J Psychiatry 159:1830–1840
Little KY, McLauglin DP, Ranc J, Gilmore J, Lopez JF, Watson SJ, Carroll FI, Butts JD (1997) Serotonin transporter binding sites and mRNA levels in depressed persons committing suicide. Biol Psychiatry 41:1156–1164
Luque JM, Kwan SW, Abell CW, Da Prada M, Richards JG (1995) Cellular expression of mRNAs encoding monoamine oxidases A and B in the rat central nervous system. J Comp Neurol 363:665–680
Magnusson A (2000) An overview of epidemiological studies on seasonal affective disorder. Acta Psychiatr Scand 101:176–184
Malison RT, Price LH, Berman R, van Dyck CH, Pelton GH, Carpenter L, Sanacora G, Owens MJ, Nemeroff CB, Rajeevan N, Baldwin RM, Seibyl JP, Innis RB, Charney DS (1998) Reduced brain serotonin transporter availability in major depression as measured by [123I]-2 beta-carbomethoxy-3 beta-(4-iodophenyl)tropane and single photon emission computed tomography [see comments]. Biol Psychiatry 44:1090–1098
Malone KM, Oquendo MA, Haas GL, Ellis SP, Li S, Mann JJ (2000) Protective factors against suicidal acts in major depression: reasons for living. Am J Psychiatry 157:1084–1088
Mann JJ, Stanley M, McBride PA, McEwen BS (1986) Increased serotonin2 and beta-adrenergic receptor binding in the frontal cortices of suicide victims. Arch Gen Psychiatry 43:954–959
Mann J, Underwood M, Arango V (1996) Postmortem studies of suicide victims biology of schizophrenia and affective disorders. American Psychiatric Press, Washington, pp 197–221
Mann JJ, Huang YY, Underwood MD, Kassir SA, Oppenheim S, Kelly TM, Dwork AJ, Arango V (2000) A serotonin transporter gene promoter polymorphism (5-HTTLPR) and prefrontal cortical binding in major depression and suicide [see comments]. Arch Gen Psychiatry 57:729–738
Marazziti D, Rossi A, Giannaccini G, Zavaglia KM, Dell’Osso L, Lucacchini A, Cassano GB (1999) Distribution and characterization of [3H]mesulergine binding in human brain postmortem. Eur Neuropsychopharmacol 10:21–26
Meltzer C, Price J, Mathis C, Greer P, Cantwell M, Houck P, Mulsant B, Ben-Eliezer D, Lopresti B, DeKosky S, Reynolds C (1999) PET imaging of serotonin type 2A receptors in late-life neuropsychiatric disorders. Am J Psych 156(12):1871–1878
Messa C, Colombo C, Moresco RM, Gobbo C, Galli L, Lucignani G, Gilardi MC, Rizzo G, Smeraldi E, Zanardi R, Artigas F, Fazio F (2003) 5-HT(2A) receptor binding is reduced in drug-naive and unchanged in SSRI-responder depressed patients compared to healthy controls: a PET study. Psychopharmacology (Berl) 167:72–78
Meyer JH (2007) Imaging the serotonin transporter during major depressive disorder and antidepressant treatment. J Psychiatry Neurosci 32:86–102
Meyer JH (2008) Applying neuroimaging ligands to study major depressive disorder. Semin Nucl Med 38:287–304
Meyer J, Kapur S, Houle S, DaSilva J, Owczarek B, Brown G, Wilson A, Kennedy S (1999) Prefrontal cortex 5-HT2 receptors in depression: a [18F] setoperone PET imaging study. Am J Psychiatry 156:1029–1034
Meyer JH, Kruger S, Wilson AA, Christensen BK, Goulding VS, Schaffer A, Minifie C, Houle S, Hussey D, Kennedy SH (2001a) Lower dopamine transporter binding potential in striatum during depression. Neuroreport 12:4121–4125
Meyer JH, Wilson AA, Ginovart N, Goulding V, Hussey D, Hood K, Houle S (2001b) Occupancy of serotonin transporters by paroxetine and citalopram during treatment of depression: a [(11)C]DASB PET imaging study. Am J Psychiatry 158:1843–1849
Meyer JH, McMain S, Kennedy SH, Korman L, Brown GM, DaSilva JN, Wilson AA, Blak T, Eynan-Harvey R, Goulding VS, Houle S, Links P (2003) Dysfunctional attitudes and 5-HT(2) receptors during depression and self-harm. Am J Psychiatry 160:90–99
Meyer JH, Houle S, Sagrati S, Carella A, Hussey DF, Ginovart N, Goulding V, Kennedy J, Wilson AA (2004a) Brain serotonin transporter binding potential measured with carbon 11-labeled DASB positron emission tomography: effects of major depressive episodes and severity of dysfunctional attitudes. Arch Gen Psychiatry 61:1271–1279
Meyer JH, Wilson AA, Sagrati S, Hussey D, Carella A, Potter WZ, Ginovart N, Spencer EP, Cheok A, Houle S (2004b) Serotonin transporter occupancy of five selective serotonin reuptake inhibitors at different doses: an [11C]DASB positron emission tomography study. Am J Psychiatry 161:826–835
Meyer JH, Ginovart N, Boovariwala A, Sagrati S, Hussey D, Garcia A, Young T, Praschak-Rieder N, Wilson AA, Houle S (2006a) Elevated monoamine oxidase a levels in the brain: an explanation for the monoamine imbalance of major depression. Arch Gen Psychiatry 63:1209–1216
Meyer JH, McNeely HE, Sagrati S, Boovariwala A, Martin K, Verhoeff NP, Wilson AA, Houle S (2006b) Elevated putamen D(2) receptor binding potential in major depression with motor retardation: an [11C]raclopride positron emission tomography study. Am J Psychiatry 163:1594–1602
Meyer JH, Wilson AA, Sagrati S, Miler L, Rusjan P, Bloomfield PM, Clark M, Sacher J, Voineskos AN, Houle S (2009) Brain monoamine oxidase A binding in major depressive disorder: relationship to selective serotonin reuptake inhibitor treatment, recovery, and recurrence. Arch Gen Psychiatry 66:1304–1312
Mintun MA, Sheline YI, Moerlein SM, Vlassenko AG, Huang Y, Snyder AZ (2004) Decreased hippocampal 5-HT2A receptor binding in major depressive disorder: in vivo measurement with [18F]altanserin positron emission tomography. Biol Psychiatry 55:217–224
Moll G, Moll R, Riederer P, Gsell W, Heinsen H, Denney RM (1990) Immunofluorescence cytochemistry on thin frozen sections of human substantia nigra for staining of monoamine oxidase A and monoamine oxidase B: a pilot study. J Neural Transm Suppl 32:67–77
Nash JR, Sargent PA, Rabiner EA, Hood SD, Argyropoulos SV, Potokar JP, Grasby PM, Nutt DJ (2008) Serotonin 5-HT1A receptor binding in people with panic disorder: positron emission tomography study. Br J Psychiatry 193:229–234
Neumeister A, Pirker W, Willeit M, Praschak-Rieder N, Asenbaum S, Brucke T, Kasper S (2000) Seasonal variation of availability of serotonin transporter binding sites in healthy female subjects as measured by [123I]-2 beta-carbomethoxy-3 beta-(4-iodophenyl)tropane and single photon emission computed tomography. Biol Psychiatry 47:158–160
Neumeister A, Willeit M, Praschak-Rieder N, Asenbaum S, Stastny J, Hilger E, Pirker W, Konstantinidis A, Kasper S (2001) Dopamine transporter availability in symptomatic depressed patients with seasonal affective disorder and healthy controls. Psychol Med 31:1467–1473
Neumeister A, Bain E, Nugent AC, Carson RE, Bonne O, Luckenbaugh DA, Eckelman W, Herscovitch P, Charney DS, Drevets WC (2004a) Reduced serotonin type 1A receptor binding in panic disorder. J Neurosci 24:589–591
Neumeister A, Nugent AC, Waldeck T, Geraci M, Schwarz M, Bonne O, Bain EE, Luckenbaugh DA, Herscovitch P, Charney DS, Drevets WC (2004b) Neural and behavioral responses to tryptophan depletion in unmedicated patients with remitted major depressive disorder and controls. Arch Gen Psychiatry 61:765–773
Newberg AB, Amsterdam JD, Wintering N, Ploessl K, Swanson RL, Shults J, Alavi A (2005) 123I-ADAM binding to serotonin transporters in patients with major depression and healthy controls: a preliminary study. J Nucl Med 46:973–977
Norman W, Miller I, Dow M (1988) Characteristics of depressed patients with elevated levels of dysfunctional cognitions. Cog Ther Res 12:39–51
O’Hara MW, Swain A (1996) Rates and risk of postpartum depression-a meta analysis. Int Rev Psychiatry 8:37–54
O’Hara MW, Schlechte JA, Lewis DA, Wright EJ (1991) Prospective study of postpartum blues. Biologic and psychosocial factors. Arch Gen Psychiatry 48:801–806
Oliver J, Baumgart E (1985) The dysfunctional attitude scale: psychometric properties and relation to depression in an unselected adult population. Cog Ther Res 9:161–167
O’Regan D, Kwok RP, Yu PH, Bailey BA, Greenshaw AJ, Boulton AA (1987) A behavioural and neurochemical analysis of chronic and selective monoamine oxidase inhibition. Psychopharmacology 92:42–47
Ou XM, Chen K, Shih JC (2006) Monoamine oxidase A and repressor R1 are involved in apoptotic signaling pathway. Proc Natl Acad Sci U S A 103:10923–10928
Pandey GN, Dwivedi Y, Rizavi HS, Ren X, Pandey SC, Pesold C, Roberts RC, Conley RR, Tamminga CA (2002) Higher expression of serotonin 5-HT(2A) receptors in the postmortem brains of teenage suicide victims. Am J Psychiatry 159:419–429
Parsey RV, Kegeles LS, Hwang DR, Simpson N, Abi-Dargham A, Mawlawi O, Slifstein M, Van Heertum RL, Mann JJ, Laruelle M (2000) In vivo quantification of brain serotonin transporters in humans using [11C]McN 5652. J Nucl Med 41:1465–1477
Parsey RV, Hastings RS, Oquendo MA, Huang YY, Simpson N, Arcement J, Huang Y, Ogden RT, Van Heertum RL, Arango V, Mann JJ (2006a) Lower serotonin transporter binding potential in the human brain during major depressive episodes. Am J Psychiatry 163:52–58
Parsey RV, Oquendo MA, Ogden RT, Olvet DM, Simpson N, Huang YY, Van Heertum RL, Arango V, Mann JJ (2006b) Altered serotonin 1A binding in major depression: a [carbonyl-C-11]WAY100635 positron emission tomography study. Biol Psychiatry 59:106–113
Perry EK, Marshall EF, Blessed G, Tomlinson BE, Perry RH (1983) Decreased imipramine binding in the brains of patients with depressive illness. Br J Psychiatry 142:188–192
Post RM, Kotin J, Goodwin FK, Gordon EK (1973) Psychomotor activity and cerebrospinal fluid amine metabolites in affective illness. Am J Psychiatry 130:67–72
Praag HM, Korf J, Lakke J, Schut T (1975) Dopamine metabolism in depressions, psychoses, and Parkinson’s disease: the problem of the specificity of biological variables in behaviour disorders. Psychol Med 5:138–146
Praschak-Rieder N, Wilson AA, Hussey D, Carella A, Wei C, Ginovart N, Schwarz MJ, Zach J, Houle S, Meyer JH (2005) Effects of tryptophan depletion on the serotonin transporter in healthy humans. Biol Psychiatry 58:825–830
Praschak-Rieder N, Willeit M, Wilson AA, Houle S, Meyer JH (2008) Seasonal variation in human brain serotonin transporter binding. Arch Gen Psychiatry 65:1072–1078
Pratt L, Brody D (2010) Depression and smoking in the U.S. household population aged 20 and over, 2005–2008. NCHS Data Brief 34:1–8
Rajkowska G, Miguel-Hidalgo JJ (2007) Gliogenesis and glial pathology in depression. CNS Neurol Disord Drug Targets 6:219–233
Robins E, Murphy G, Wilkinson R, Gassner S, Kayes J (1959) Some clinical considerations in the prevention of suicide based on a study of 134 successful suicides. Am J Publ Health 49(7):888–899
Rommelspacher H, Meier-Henco M, Smolka M, Kloft C (2002) The levels of norharman are high enough after smoking to affect monoamineoxidase B in platelets. Eur J Pharmacol 441:115–125
Rosen LN, Targum SD, Terman M, Bryant MJ, Hoffman H, Kasper SF, Hamovit JR, Docherty JP, Welch B, Rosenthal NE (1990) Prevalence of seasonal affective disorder at four latitudes. Psychiatry Res 31:131–144
Roth B, McLean S, Zhu X, Chuang D (1987) Characterization of two [3H]ketanserin recognition sites in rat striatum. J Neurochem 49:1833–1838
Rovescalli AC, Brunello N, Riva M, Galimberti R, Racagni G (1989) Effect of different photoperiod exposure on [3H]imipramine binding and serotonin uptake in the rat brain. J Neurochem 52:507–514
Ruhe HG, Mason NS, Schene AH (2007) Mood is indirectly related to serotonin, norepinephrine and dopamine levels in humans: a meta-analysis of monoamine depletion studies. Mol Psychiatry 12:331–359
Ruhe HG, Booij J, Reitsma JB, Schene AH (2009) Serotonin transporter binding with [123I]beta-CIT SPECT in major depressive disorder versus controls: effect of season and gender. Eur J Nucl Med Mol Imaging 36:841–849
Sacher J, Wilson A, Houle S, Hassan S, Rusjan P, Bloomfield P, Stewart D, Meyer J (2010) Elevated brain monoamine oxidase A binding in early postpartum. Arch Gen Psychiatry 67(5):468–474
Sanacora G, Gueorguieva R, Epperson CN, Wu YT, Appel M, Rothman DL, Krystal JH, Mason GF (2004) Subtype-specific alterations of gamma-aminobutyric acid and glutamate in patients with major depression. Arch Gen Psychiatry 61:705–713
Sarchiapone M, Carli V, Camardese G, Cuomo C, Di Giuda D, Calcagni ML, Focacci C, De Risio S (2006) Dopamine transporter binding in depressed patients with anhedonia. Psychiatry Res 147:243–248
Sargent PA, Kjaer KH, Bench CJ, Rabiner EA, Messa C, Meyer J, Gunn RN, Grasby PM, Cowen PJ (2000) Brain serotonin1A receptor binding measured by positron emission tomography with [11C]WAY-100635: effects of depression and antidepressant treatment. Arch Gen Psychiatry 57:174–180
Saura J, Kettler R, Da Prada M, Richards JG (1992) Quantitative enzyme radioautography with 3H-Ro 41-1049 and 3H-Ro 19-6327 in vitro: localization and abundance of MAO-A and MAO-B in rat CNS, peripheral organs, and human brain. J Neurosci 12:1977–1999
Saura J, Bleuel Z, Ulrich J, Mendelowitsch A, Chen K, Shih JC, Malherbe P, Da Prada M, Richards JG (1996) Molecular neuroanatomy of human monoamine oxidases A and B revealed by quantitative enzyme radioautography and in situ hybridization histochemistry. Neuroscience 70:755–774
Schmuck K, Ullmer C, Engels P, Lubbert H (1994) Cloning and functional characterization of the human 5-HT2B serotonin receptor. FEBS Lett 342:85–90
Selvaraj S, Venkatesha Murthy N, Bhagwagar Z, Bose SK, Hinz R, Grasby PM, Cowen PJ (2009) Diminished brain 5-HT transporter binding in major depression: a positron emission tomography study with [(11)C]DASB. Psychopharmacology 213(2-3):555–562
Sharot T, Riccardi AM, Raio CM, Phelps EA (2007) Neural mechanisms mediating optimism bias. Nature 450:102–105
Simons AD, Murphy GE, Levine JL, Wetzel RD (1986) Cognitive therapy and pharmacotherapy for depression. Sustained improvement over one year. Arch Gen Psychiatry 43:43–48
Stanley M, Mann JJ (1983) Increased serotonin-2 binding sites in frontal cortex of suicide victims. Lancet 1:214–216
Stockmeier CA (2003) Involvement of serotonin in depression: evidence from postmortem and imaging studies of serotonin receptors and the serotonin transporter. J Psychiatr Res 37:357–373
Stockmeier CA, Kellar KJ (1986) In vivo regulation of the serotonin-2 receptor in rat brain. Life Sci 38:117–127
Stockmeier CA, Dilley GE, Shapiro LA, Overholser JC, Thompson PA, Meltzer HY (1997) Serotonin receptors in suicide victims with major depression. Neuropsychopharmacology 16:162–173
Stockmeier CA, Howley E, Shi X, Sobanska A, Clarke G, Friedman L, Rajkowska G (2009) Antagonist but not agonist labeling of serotonin-1A receptors is decreased in major depressive disorder. J Psychiatr Res 43:887–894
Todd KG, McManus DJ, Baker GB (1995) Chronic administration of the antidepressants phenelzine, desipramine, clomipramine, or maprotiline decreases binding to 5-hydroxytryptamine2A receptors without affecting benzodiazepine binding sites in rat brain. Cell Mol Neurobiol 15:361–370
Tom SM, Fox CR, Trepel C, Poldrack RA (2007) The neural basis of loss aversion in decision-making under risk. Science 315:515–518
Trivedi MH, Rush AJ, Wisniewski SR, Nierenberg AA, Warden D, Ritz L, Norquist G, Howland RH, Lebowitz B, McGrath PJ, Shores-Wilson K, Biggs MM, Balasubramani GK, Fava M (2006) Evaluation of outcomes with citalopram for depression using measurement-based care in STAR*D: implications for clinical practice. Am J Psychiatry 163:28–40
Turecki G, Briere R, Dewar K, Antonetti T, Lesage AD, Seguin M, Chawky N, Vanier C, Alda M, Joober R, Benkelfat C, Rouleau GA (1999) Prediction of level of serotonin 2A receptor binding by serotonin receptor 2A genetic variation in postmortem brain samples from subjects who did or did not commit suicide. Am J Psychiatry 156:1456–1458
Verhoeff NP, Kapur S, Hussey D, Lee M, Christensen B, Psych C, Papatheodorou G, Zipursky RB (2001) A simple method to measure baseline occupancy of neostriatal dopamine d(2) receptors by dopamine in vivo in healthy subjects. Neuropsychopharmacology 25:213–223
Walter M, Henning A, Grimm S, Schulte RF, Beck J, Dydak U, Schnepf B, Boeker H, Boesiger P, Northoff G (2009) The relationship between aberrant neuronal activation in the pregenual anterior cingulate, altered glutamatergic metabolism, and anhedonia in major depression. Arch Gen Psychiatry 66:478–486
Weissman A (1979) The dysfunctional attitude scale: a validation study. Dissertation Abstr Int 40:1389B–1390B
Willeit M, Ginovart N, Graff A, Rusjan P, Vitcu I, Houle S, Seeman P, Wilson AA, Kapur S (2008) First human evidence of d-amphetamine induced displacement of a D2/3 agonist radioligand: a [11C]-(+)-PHNO positron emission tomography study. Neuropsychopharmacology 33:279–289
Wilson A, Schmidt M, Ginovart N, Meyer J, Houle S (2000) Novel radiotracers for imaging the serotonin transporter by positron emission tomography: synthesis, radiosynthesis, in vitro and ex vivo evaluation of [11C]-labelled 2-(Phenylthio) araalkylamines. J Med Chem 43:3103–3110
Wilson AA, Ginovart N, Hussey D, Meyer J, Houle S (2002) In vitro and in vivo characterisation of [11C]-DASB: a probe for in vivo measurements of the serotonin transporter by positron emission tomography. Nucl Med Biol 29:509–515
World Health Organization (2008) The global burden of disease: 2004 update. Department of Health Statistics and Informatics, Information Evidence and Research Cluster, WHO, Geneva
Xiao Q, Pawlyk A, Tejani-Butt SM (1999) Reserpine modulates serotonin transporter mRNA levels in the rat brain. Life Sci 64:63–68
Yates M, Leake A, Candy JM, Fairbairn AF, McKeith IG, Ferrier IN (1990) 5HT2 receptor changes in major depression. Biol Psychiatry 27:489–496
Yatham LN, Liddle PF, Shiah IS, Scarrow G, Lam RW, Adam MJ, Zis AP, Ruth TJ (2000) Brain serotonin2 receptors in major depression: a positron emission tomography study. Arch Gen Psychiatry 57:850–858
Youdim MB, Edmondson D, Tipton KF (2006) The therapeutic potential of monoamine oxidase inhibitors. Nat Rev Neurosci 7:295–309
Young SN, Smith SE, Pihl RO, Ervin FR (1985) Tryptophan depletion causes a rapid lowering of mood in normal males. Psychopharmacology 87:173–177
Yu A, Yang J, Pawlyk AC, Tejani-Butt SM (1995) Acute depletion of serotonin down-regulates serotonin transporter mRNA in raphe neurons. Brain Res 688:209–212
Zarate CA Jr, Singh JB, Carlson PJ, Brutsche NE, Ameli R, Luckenbaugh DA, Charney DS, Manji HK (2006) A randomized trial of an N-methyl-D-aspartate antagonist in treatment-resistant major depression. Arch Gen Psychiatry 63:856–864
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2012 Springer-Verlag Berlin Heidelberg
About this chapter
Cite this chapter
Meyer, J.H. (2012). Neurochemical Imaging and Depressive Behaviours. In: Cowen, P., Sharp, T., Lau, J. (eds) Behavioral Neurobiology of Depression and Its Treatment. Current Topics in Behavioral Neurosciences, vol 14. Springer, Berlin, Heidelberg. https://doi.org/10.1007/7854_2012_219
Download citation
DOI: https://doi.org/10.1007/7854_2012_219
Published:
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-642-35424-3
Online ISBN: 978-3-642-35425-0
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)