Abstract
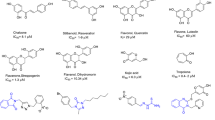
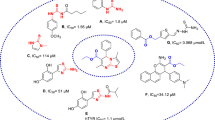
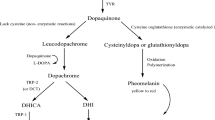
Tyrosinase is a copper-containing bifunctional metalloenzyme, widely distributed around the phylogeny. This enzyme is involved in the production of melanin and some other pigments in humans, animals, etc. Abnormal accumulation of melanin, which is due to the overexpression of the enzyme, is called hyperpigmentation and underexpression is called vitiligo, which is a major skin problem around the world. The inhibitors of this enzyme have been utilized in cosmetics, especially as depigmenting agents in the case of hyperpigmentation. They are also involved in several other disease conditions. In the last few decades a large number of tyrosinase inhibitors have been discovered and reported by several groups including ours. This chapter principally emphasizes the discovery of some interesting inhibitors, mainly of heterocyclic origin, and their impacts on drug discovery; some of the inhibitors might not be heterocyclic but their chemistry is quite interesting in terms of the inhibition.

Access provided by Autonomous University of Puebla. Download to read the full chapter text
Chapter PDF
Similar content being viewed by others
References
Gadd GW (1980) Melanin production and differentiation in batch cultures of the polymorphic fungus. FEMS Microbiol Lett 9:237–240
Bell AA, Wheeler MH (1986) Biosynthesis and functions of fungal melanins. Annu Rev Phytopathol 24:411–451
Zimmerman WC et al. (1995) Melanin and perithecial development in Ophiostoma piliferum. Mycologia 87:857–863
Zarivi O et al. (2003) Truffle thio-flavours reversibly inhibit truffle tyrosinase. FEMS Microbiol Lett 220(1):81–88
Ohguchi K et al. (2003) Effects of hydroxystilbene derivatives on tyrosinase activity. Biochem Biophys Res Commun 307(4):861–863
Xu Y et al. (1997) Tyrosinase mRNA is expressed in human substantia nigra. Mol Brain Res 45(1):159–162
Chen QX, Kubo I (2002) Kinetics of mushroom tyrosinase inhibition by quercetin. J Agric Food Chem 50(14):4108–4112
Rescigno A et al. (2002) Tyrosinase inhibition: general and applied aspects. J Enzyme Inhib Med Chem 17(4):207–218
Seo SY, Sharma VK, Sharma N (2003) Mushroom tyrosinase: recent prospects. J Agric Food Chem 51(10):2837–2853
Martinez MV, Whitaker JR (1995) The biochemistry and control of enzymatic browning. Trends Food Sci Technol 6:195–200
Yaropolov AI et al. (1995) Flow-injection analysis of phenols at a graphite electrode modified with co-immobilized laccase and tyrosinase. Anal Chim Acta 308:137–144
Mayer AM (1987) Polyphenol oxidases in plants: recent progress. Phytochemistry 26:11–20
van Gelder CWG, Flurkey WH, Wichers HJ (1997) Sequence and structural features of plant and fungal tyrosinases. Phytochemistry 45:1309–1323
Sanchez-Ferrer A et al. (1995) Tyrosinase: a comprehensive review of its mechanism. Biochim Biophys Acta 1247:1–11
Raper HS (1928) The anaerobic oxidases. Physiol Rev 8:245–282
Mason HS (1948) The chemistry of melanin. III. Mechanism of the oxidation of trihydroxyphenylalanine by tyrosinase. J Biol Chem 172:83–99
Lerner AB et al. (1949) Mammalian tyrosinases preparation and properties. J Biol Chem 179:185–195
Kobayashi T et al. (1995) Modulation of melanogenic protein expression during the switch from eu- to pheomelanogenesis. J Cell Sci 108(Pt6):2301–2309
Olivares C et al. (2001) The 5,6-dihydroxyindole-2-carboxylic acid (DHICA) oxidase activity of human tyrosinase. Biochem J 354(Pt1):131–139
Liangli YU (2003) Inhibitory effects of (S)- and I-6-hydroxy-2,5,7,8–tetramethylchroman-2-carboxylic acids on tyrosinase activity. J Agric Food Chem 51:2344–2347
Lee SE et al. (2000) Inhibition effects of cinnamomum cassia bark-derived materials on mushroom tyrosinase. Food Sci Biotechnol 9:330–333
Sugumaran M (1988) Molecular mechanism for cuticular sclerotization. Adv Insect Physiol 21:179–231
Barrett FM (1984) Wound-healing phenoloxidase in larval cuticle of Calpodes ethlius (Lepidoptera: Hesperiidae). Can J Zool 62:834–838
Pawelek JM, Korner AM (1982) The biosynthesis of mammalian melanin. J Am Chem Soc 70:136–145
Mosher DB, Pathak MA, Fitzpatric TB (1983) Update: dermatology in general medicine. McGraw Hill, New York, pp 205–225
Maeda K, Fukuda M (1991) In vitro effectiveness of several whitening cosmetic components in human melanocytes. J Soc Cosmet Chem 42:361–368
Palumbo A et al. (1991) Mechanism of inhibition of melanogenesis by hydroquinone. Biochim Biophys Acta 1073(1):85–90
Ohyama Y, Mishima Y (1990) Melanogenesis inhibitory effects of kojic acid and its action mechanism. Fragrance J 6:53
Maeda K, Fukuda M (1991) In vitro effectiveness of several whitening cosmetic components in human melanocytes. J Soc Cosmet Chem 42:361–368
Arung ET, Shimizu K, Kondo R (2006) Inhibitory effect of artocarpanone from Artocarpus heterophyllus on melanin biosynthesis. Biol Pharm Bull 29(9):1966–1969
Boissy RE, Visscher M, DeLong MA (2005) DeoxyArbutin: a novel reversible tyrosinase inhibitor with effective in vivo skin lightening potency. Exp Dermatol 14(8):601–608
Choi H et al. (2005) Inhibition of skin pigmentation by an extract of Lepidium apetalum and its possible implication in IL-6 mediated signaling. Pigment Cell Res 18(6):439–446
Farooqui JZ et al. (1995) Isolation of a unique melanogenic inhibitor from human skin xenografts: initial in vitro and in vivo characterization. J Invest Dermatol 104(5):739–743
Funasaka Y et al. (1999) The depigmenting effect of alpha-tocopheryl ferulate on human melanoma cells. Br J Dermatol 141(1):20–29
Hamed SH et al. (2006) Comparative efficacy and safety of deoxyarbutin, a new tyrosinase-inhibiting agent. J Cosmet Sci 57(4):291–308
Ichihashi M et al. (1999) The inhibitory effect of dl-alpha-tocopheryl ferulate in lecithin on melanogenesis. Anticancer Res 19(5A):3769–3774
Imokawa G et al. (1986) Differential analysis of experimental hypermelanosis induced by UVB, PUVA, and allergic contact dermatitis using a brownish guinea pig model. Arch Dermatol Res 278(5):352–362
Imokawa G et al. (1997) The role of endothelin-1 in epidermal hyperpigmentation and signaling mechanisms of mitogenesis and melanogenesis. Pigment Cell Res 10(4):218–228
Lee JY, Kang WH (2003) Effect of cyclosporin A on melanogenesis in cultured human melanocytes. Pigment Cell Res 16(5):504–508
Okombi S et al. (2006) Discovery of benzylidenebenzofuran-3(2H)-one (aurones) as inhibitors of tyrosinase derived from human melanocytes. J Med Chem 49(1):329–333
Schallreuter KU, Wood JW (1990) A possible mechanism of action for azelaic acid in the human epidermis. Arch Dermatol Res 282(3):168–171
Usuki A et al. (2003) The inhibitory effect of glycolic acid and lactic acid on melanin synthesis in melanoma cells. Exp Dermatol 12(Suppl 2):43–50
Saboury AA, Alijanianzadeh M, Mansoori-Torshizi H (2007) The role of alkyl chain length in the inhibitory effect of n-alkyl xanthates on mushroom tyrosinase activities. Acta Biochim Pol 54(1):183–191
Ha SK et al. (2005) Inhibition of tyrosinase activity by N,N-unsubstituted selenourea derivatives. Biol Pharm Bull 28(5):838–840
Ahn SJ et al. (2006) Regulation of melanin synthesis by selenium-containing carbohydrates. Chem Pharm Bull (Tokyo) 54(3):281–286
Koketsu M et al. (2002) Inhibitory effects of 1,3-selenazol-4-one derivatives on mushroom tyrosinase. Chem Pharm Bull (Tokyo) 50(12):1594–1596
Khan MT et al. (2005) Structure–activity relationships of tyrosinase inhibitory combinatorial library of 2,5-disubstituted-1,3,4-oxadiazole analogues. Bioorg Med Chem 13(10):3385–3395
Shaheen F et al. (2005) Alkaloids of Aconitum laeve and their anti-inflammatory antioxidant and tyrosinase inhibition activities. Phytochemistry 66(8):935–940
Sultankhodzhaev MN et al. (2005) Tyrosinase inhibition studies of diterpenoid alkaloids and their derivatives: structure–activity relationships. Nat Prod Res 19(5):517–522
Casanola-Martin GM et al. (2006) New tyrosinase inhibitors selected by atomic linear indices-based classification models. Bioorg Med Chem Lett 16(2):324–330
Ahmad VU et al. (2004) Tyrosinase inhibitors from Rhododendron collettianum and their structure–activity relationship (SAR) studies. Chem Pharm Bull (Tokyo) 52(12):1458–1461
Azhar UH et al. (2006) Tyrosinase inhibitory lignans from the methanol extract of the roots of Vitex negundo Linn. and their structure–activity relationship. Phytomedicine 13(4):255–260
Briganti S, Camera E, Picardo M (2003) Chemical and instrumental approaches to treat hyperpigmentation. Pigment Cell Res 16(2):101–110
Fechner GA et al. (1992) Antiproliferative and depigmenting effects of the histamine (H2) agonist dimaprit and its derivatives on human melanoma cells. Biochem Pharmacol 43(10):2083–2090
Imokawa G, Mishima Y (1985) Analysis of tyrosinases as asparagin-linked oligosaccharides by concanavalin A lectin chromatography: appearance of new segment of tyrosinases in melanoma cells following interrupted melanogenesis induced by glycosylation inhibitors. J Invest Dermatol 85(2):165–168
Inoue S et al. (1990) Mechanism of growth inhibition of melanoma cells by 4-S-cysteaminylphenol and its analogues. Biochem Pharmacol 39(6):1077–1083
Mishima Y (1994) Molecular and biological control of melanogenesis through tyrosinase genes and intrinsic and extrinsic regulatory factors. Pigment Cell Res 7(6):376–387
Monji A et al. (2005) Tyrosinase induction and inactivation in normal cultured human melanocytes by endothelin-1. Int J Tissue React 27(2):41–49
Nerya O et al. (2004) Chalcones as potent tyrosinase inhibitors: the effect of hydroxyl positions and numbers. Phytochemistry 65(10):1389–1395
Ohyama Y, Mishima Y (1993) Isolation and characterization of high molecular weight melanogenic inhibitors naturally occurring in melanoma cells. Pigment Cell Res 6(1):7–12
Park YD et al. (2006) TXM13 human melanoma cells: a novel source for the inhibition kinetics of human tyrosinase and for screening whitening agents. Biochem Cell Biol 84(1):112–116
Parvez S et al. (2006) Survey and mechanism of skin depigmenting and lightening agents. Phytother Res 20(11):921–934
Prince S, Illing N, Kidson SH (2001) SV-40 large T antigen reversibly inhibits expression of tyrosinase, TRP-1, TRP-2 and Mitf, but not Pax-3, in conditionally immortalized mouse melanocytes. Cell Biol Int 25(1):91–102
Saha B et al. (2006) Transcriptional activation of tyrosinase gene by human placental sphingolipid. Glycoconj J 23(3–4):259–268
Schallreuter KU et al. (1994) Defective tetrahydrobiopterin and catecholamine biosynthesis in the depigmentation disorder vitiligo. Biochim Biophys Acta 1226(2):181–192
Song KK et al. (2006) Inhibitory effects of cis- and trans-isomers of 3,5-dihydroxystilbene on the activity of mushroom tyrosinase. Biochem Biophys Res Commun 342(4):1147–1151
Townsend E et al. (1992) Reversible depigmentation of human melanoma cells by halistanol trisulphate, a novel marine sterol. Melanoma Res 1(5–6):349–357
Tsuji-Naito K et al. (2007) Modulating effects of a novel skin-lightening agent, alpha-lipoic acid derivative, on melanin production by the formation of DOPA conjugate products. Bioorg Med Chem 15(5):1967–1975
Vogel FS et al. (1977) γ-l-Glutaminyl-4-hydroxybenzene, an inducer of cryptobiosis in Agaricus bisporus and a source of specific metabolic inhibitors for melanogenic cells. Cancer Res 37(4):1133–1136
Yang F, Boissy RE (1999) Effects of 4-tertiary butylphenol on the tyrosinase activity in human melanocytes. Pigment Cell Res 12(4):237–245
Chakraborty DP et al. (1978) Interrelationship of tryptophan pyrrolase with tyrosinase in melanogenesis of Bufo melanostictus. Clin Chim Acta 82(1–2):55–59
Schallreuter KU, Wood JM, Berger J (1991) Low catalase levels in the epidermis of patients with vitiligo. J Invest Dermatol 97(6):1081–1085
Author information
Authors and Affiliations
Corresponding author
Editor information
Rights and permissions
Copyright information
© 2007 Springer-Verlag Berlin Heidelberg
About this chapter
Cite this chapter
Khan, M.T.H. (2007). Heterocyclic Compounds Against the Enzyme Tyrosinase Essential for Melanin Production: Biochemical Features of Inhibition. In: Khan, M.T.H. (eds) Bioactive Heterocycles III. Topics in Heterocyclic Chemistry, vol 9. Springer, Berlin, Heidelberg. https://doi.org/10.1007/7081_2007_077
Download citation
DOI: https://doi.org/10.1007/7081_2007_077
Published:
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-540-73401-7
Online ISBN: 978-3-540-73402-4
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)