Abstract
The presence and ecological risks of microplastics (MPs) are increasingly reported, whereas the impacts of MPs on human health remain largely unknown. Recent studies have confirmed the MP contamination in food items, including seafood, table salt, drinking water, etc. Dietary exposure is one of the inevitable exposure pathways of MPs, which causes concern about the potential human health risks. Whether we assess health risks or try to reduce food MP contamination, the prerequisites are to figure out the contamination pathways of MPs and their actual level in food items. At present, territorial system is facing serious environmental problems, with soil, freshwater, and air suffering from MP pollution. This leads to diversity and complexity of MP sources in food items. Therefore, we should not be confined to the food itself when considering MPs in food, but should take all pollution possibilities into account. In this chapter, we reviewed the literature concerning MPs in seafood, table salt, drinking water, and other food items. The potential MP sources of food items during the whole process from food acquisition to human ingestion were analyzed, with related human intake of MPs estimated. We also discussed possible translocation and accumulation pathways of MPs within human body. Given the lifetime inevitable exposure to MPs through multiple pathways, we urgently call for a better understanding of the potential MP sources in food items and a comprehensive assessment of human intake.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Since the concept of “microplastic” was introduced in 2004 [1], microplastics (MPs) have been found in various environmental media and organisms globally [2,3,4,5,6]. More recently, the threats of MPs to human health have gained increasing public interests and media attention due to the wide detection of MPs in human-related food.
As the marine environment attracted much attention, seafood has become the most studied food [7,8,9]. More than 690 marine species have been reported to be contaminated by MPs, including edible and nonedible ones [10, 11]. Since inedible species do not normally enter human body, their contribution to MP intake by human is negligible. Until now, MPs have been found in 202 edible species, including 201 marine species and 1 terrestrial species [12]. Subsequently, MPs are also observed in table salt and other food such as honey, sugar, beer, and drinking water [13,14,15,16]. Consumption of these food products will undoubtedly cause MP exposure through human digestive tract. Numerous experiments have demonstrated toxic effects of MPs, such as growth inhibition, oxidative damage, and immune stress [17, 18]. A recent study shows that MPs of high concentration may have caused evolutionary adaptations of some species (e.g., D. magna and G. pulex) [19]. Mammal experiments have proved that polystyrene microplastic can induce gut microbiota dysbiosis and hepatic lipid metabolism disorder in mice [20]. MP particles can also accumulate in marine organisms and transfer through the food chain to higher trophic levels including humans [10]. Despite that ample evidence suggests the MP contamination of human-related food and the related toxicological effects on animals and cells, there is, however, large unknown fields.
Food is indispensable energy supplier for human survival. Therefore, it is necessary to survey our current knowledge on direct human exposure concentration to MPs via food consumption. Related topics have been discussed previously. For example, seafood, as a carrier of MPs, should be treated cautiously considering the influence on human health through biological accumulation and biological amplification [21, 22]. At the same time, some believe that the risk to higher trophic levels is negligible due to the rapid depuration of MPs [23]. In fact, the issue of MPs in food items is no longer about single investigation of MPs in food itself due to inevitable MP contamination in water and air for human consumption in terrestrial environment. During the whole process from food acquisition, production, packaging, and transportation to food intake, extra MPs may be introduced in any link [16]. This is a complex problem that needs to be dissected in depth. When all factors are taken into consideration, we will have more realistic data for risk assessment. Only in this way can we put forward more effective measures to control the main links of food contamination.
2 Microplastics in Seafood
Various foods are summarized in terms of contamination levels of MPs. Since these data have been reported in detail in previous studies [12, 24,25,26], partial data are listed in Table 1.
MPs have been found in fish from many countries and regions, ranging from 0 to 48 items/individual [40,41,42]. The reason that “items/individual” is used instead of “MPs/individual” is that MPs in seafood are usually characterized by their sizes, shapes, and colors, whereas compositions are not universally identified. Among these studies that have completed particle identification, relatively high concentrations occurred in China (13.54–22.21 items/individual) [43], Turkey (9.4 items/individual) [44], and Malaysia (14 items/individual) [45]. However, different methods among studies lead to poor comparability of the results. Therefore, direct comparisons and accurate conclusion cannot be made, and such situation occurs in shellfish and all the other food items.
MP abundance in shellfish (0–57.2 items/individual, 0–75 items/g) is generally higher than that in fish, with blue mussels being the most studied species [46, 47]. The largest numbers occurred in mussels from Canada (34–75 items/g) [29], followed by China (0.9–4.6 items/g) [48] and Equatorial mid-Atlantic area (2 items/g) [49]. In addition to wild mussels, some ones from fishery farms, as well as supermarket, have also been confirmed to be contaminated by MPs. Li et al. have investigated commercial bivalves from fishery farms and supermarket and found that all mussels were contaminated by MPs [27, 28].
3 Microplastics in Table Salt
MPs have been widely identified in table salt of more than 100 brands all over the world [16, 33, 36, 50], with their concentrations varying widely. The highest concentration was reported in Croatia (1.35 × 104–1.98 × 104 MPs/kg salt), followed by Indonesia (1.36 × 104 MPs/kg), Italy (1.57 × 103–8.23 × 103 MPs/kg) [32, 33], the USA (0.47 × 102–8.1 × 102 MPs/kg), and China (5.5 × 102–6.8 × 102 MPs/kg) [16, 36]. A recent study compared MP concentrations in table salts collected from different regions, using sea salt as a seawater MP pollution indicator, which indicated a significant higher MP concentration in Asia than in other continents [38]. The lowest concentrations of MPs were reported by Karami et al. [51]. This was probably due to its filters with larger pores (149 μm), which allowed smaller-sized MPs to escape in the filtration process and thus underestimated the MP abundance.
4 Microplastics in Drinking Water
Compared to seafood and table salt, relatively few studies reported MP contamination in drinking water. The available data cover raw and treated water from drinking water treatment plants (DWTP), tap water, and bottled water [37, 38, 52]. According to the MP abundance in raw and treated water, particles larger than 50 μm can be removed from water at the treatment plants. The removal rate of MPs by traditional drinking water treatment processes varied from 25 to 90%, depending on treatment technologies [35]. For tap water samples, the lowest abundance was observed in Italy and Denmark (0 MPs/L), while the highest abundance (9.24 MPs/L) was found in the tap water of the USA [36]. The abundance of MPs in bottled water varied from 0 to 5.4 × 107 MPs/L [37, 38, 52]. Water in returnable-used plastic containers had significantly more MPs compared with that in single-used bottles [37].
The difference of detection limits among studies in the drinking water field is obvious. MPs in tap water were often analyzed by micro-Fourier transform infrared spectroscopy (μ-FTIR), capturing MPs > 20 μm [45]. All bottled water and the water from Czech DWTP were analyzed using non-FTIR or μ-FTIR combined with other method [43, 47]. These methods included μ-Raman, dyeing method combined with μ-FTIR, and “method for the extraction and determination of MPs in organic and inorganic matrix samples,” making “small-sized MPs (< 10 μm)” detectable. The MP concentration in tap water samples may be underestimated due to the non-detectable smaller MPs using μ-FTIR, which leads to the biased result that the higher concentrations of MPs were detected in bottled water than in tap water. The identification of “small-sized MPs (<10 μm)” is in an urgent need for accurate recognition of MPs and the relative risk assessment, regarding not only drinking water but other food items [37].
5 Microplastics in Other Food Items
MP contamination also occurred in other food products according to the literature. These rarely reported food types include beer, sugar, honey, chicken, tea, as well as canned sardines and sprats [13,14,15, 39, 53,54,55]. Besides, we have found MPs in edible seaweed (unpublished data). However, the data of these food items are so limited that more investigations are needed on a broader range, covering more regions and food types.
In recent years, MP pollution has been widely recognized in soil, where the crops and edible vegetables we eat grow. This undoubtedly poses a risk to land plants. Although there is no evidence from field investigations, laboratory studies have shown that polystyrene microsphere (0.2 μm) can be absorbed by lettuce roots and then migrate to shoots and accumulate in edible stems and leaves [56]. Although larger MPs are difficult to enter root cortex, they may adhere to plant surface and be ingested by human [57].
6 Source, Human Burden, and Potential Health Risks
6.1 Source Diagnostics
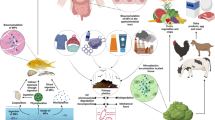
To date, MPs are ubiquitous in terrestrial environment. Together with the whole process from food acquisition to ingestion, the sources of MPs in food become diverse and complex. Figure 1 shows the potential MP sources during the whole process of seafood consumption, and other food items experience similar processes to seafood.
In the beginning, the main factor affecting food contamination is the pollution degree of the surrounding environment. Typical examples are table salt and seafood. MPs in the surrounding environment can fuse into, adhere to, or be ingested by marine animals [58, 59]. Several studies found that the abundance of MPs in sea salt was higher than that in rock salt or lake salt, which could be explained by the high level of MP pollution in coastal zones [16]. Besides, both mussel and sea salt are proposed as indicators of MP pollution in marine environment [6, 33]. There are also some plastic appliances and ropes acting as pollution sources of MPs in cultured seafood. Another MP source of farmed seafood is the feeding materials produced from MP-contaminated fish or other animals [21].
After acquisition, food items commonly experience food processing. The presence of MPs in table salt suggests that MPs might be introduced during collection, transportation to surface water, concentration, drying, crystallization, or refinement processes [16]. Some food items are processed with additional substances such as salt or water. If these additives have been already contaminated, MPs will be introduced. Moreover, food packaging materials are often made of plastics, causing the possibility of food contamination. According to the latest research, a single plastic tea bag can release approximately 11.6 billion MPs and 3.1 billion nanoplastics into a single cup of the beverage [55]. The staggering data reminds us to pay more attention to food packaging and all external factors that may have similar effects. For instance, takeout food industry is booming in today’s quick living pace, resulting in more plastic packaging for food. Such situation makes food more likely to be contaminated.
There is also an important source that needs special emphasis, which is airborne MPs. Air contact exists almost throughout the entire process of food consumption, from food acquisition to human ingestion. To date, atmospheric MPs have been discovered in many countries and regions, both indoor and outdoor environments [60, 61]. Airborne MPs may have greater contribution to food MP pollution than other sources. The risk of plastic exposure caused by mussel has been confirmed to be minimal compared to fiber ingestion through air fallout during a meal [62].
6.2 Human Body Burden
Contaminated food items are undoubtedly sources of gastrointestinal exposure for human. A preliminary estimate on the body burden of MPs was made based on detected MP concentrations in table salt, seafood, and drinking water. MP intake through other food items cannot be estimated due to scarce data. The abundance of MPs in table salt ranges widely from 0 to 2.0 × 104 MPs/kg. Considering the global mean intake of table salt of 10.06 g/day [63], the intake of MPs ranges from 0 to 198 MPs per day, equivalent to 0 to 7.3 × 104 MPs per year. The highest value is calculated according to the data of salt from Croatia [32]. The actual MP exposure through salt intake depends on the types and brands of table salts, as well as the study regions.
The presence of MPs in seafood has been widely recognized [27, 28, 48]. In 2014, van Cauwenberghe and Janssen first estimated the potential MP intake through seafood consumption. It showed that in Europe, where shellfish consumption was high, an adult may ingest up to 11,000 MPs per year [22]. In countries with low shellfish consumption, consumers ingest an average of 1800 MPs per year, which is still a considerable exposure. The annual intake of MPs through seafood consumption worldwide has been estimated by Hantoro et al., ranging from 11 to 3.5 × 104 particles per person [64]. Since MPs are mostly detected in gastrointestinal tracts (GITs), which are normally removed before seafood consumption, species that we eat whole pose greater threats than gutted ones. However, MPs in the eviscerated flesh of two commonly consumed dried fish species were significantly more than in excised organs, evidencing that the evisceration does not necessarily eliminate the risk of MP intake [45]. Moreover, MPs were also detected in the muscle of commercial fish [30]. These findings raise concerns about possible implications for human consumers.
With around 1.4 L water intake per day [65], the annual MP ingestion through drinking water, including tap water and bottled water, could be in the range of 0–2.8 × 1010 MPs. However, the MPs detected in bottled water have different size fractions (<10 μm) from those detected in tap water, seafood, and table salt. The abundance, as well as the potential human risk of particles with different size ranges, is not comparable. Therefore, when estimating the MPs in the three media, we only calculated human MP intake through drinking tap water (0–4.7 × 103 MPs per year), without considering the data of bottled water.
Despite that we have estimated the intake of MPs by human through food consumption, these data cannot represent the real situation. When calculating it, we mostly take MP level of food itself into consideration but ignore other contamination possibilities (from food processing, air, package, etc.). Thus, more attention needs to be paid to these areas. When all factors are taken into consideration, the total amount of MPs ingested by human through food is likely to rise by orders of magnitude. However, excessive panic is uncalled-for before there is sufficient toxicological evidence related to human body. Our body is in a process of dynamic metabolism, and the unabsorbed MPs will be discharged with feces. Therefore, the absorbed MPs and the amount of pollutants (organic pollutants and heavy metals) released during MP metabolism are needed to make clear.
6.3 Translocation and Accumulation in Human Body and Health Risks
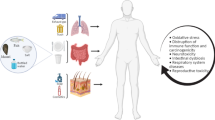
After ingestion, MPs are capable of translocating and accumulating in different organs and tissues. MPs have been found to be internalized in the gastrointestinal tract, and the unabsorbed portion is excreted with human feces [66]. The studies on other nano-sized particles provided evidence of penetration in the blood-brain barrier and placenta and even crossing the cell membrane [67, 68]. However, there is still no direct evidence showing the exact distribution and accumulation of MPs in human organs such as the liver and kidney or in human blood.
Our current knowledge is very poor about whether MPs will reach human organs and cause adverse health impacts. The available animal experiment results may have some implications for human health effects of MPs. Ingestion of MPs caused inflammatory responses in the digestive system of Mytilus [69]. The immune system of fish was the target of MP attack [70]. Inflammations including chemokine expression and pulmonary hypertension were induced by intrajugular injection of polystyrene (PS) microspheres in rats, probably due to the increased blood coagulability or vascular occlusions [71, 72]. In vivo experiments showed that PS could be internalized in macrophages, erythrocytes, as well as rat alveolar epithelial cells, showing damages to intracellular structures [73, 74]. Moreover, persistent organic pollutants, metals, and pathogenic microorganisms can be adsorbed on MPs, and the leaching of chemical additives can also aggravate the toxic effects of MPs [75,76,77,78]. MPs have been verified to be transport vectors for hydrophobic organic chemicals (HOCs) in aqueous environments [79, 80]. Apparent enrichment coefficients of HOCs on MPs might be up to five or six orders of magnitude higher than the background concentration in the surrounding seawater. MPs may then transport HOCs over long distances and affect the environmental and biological systems [80]. The debate on the harmfulness of MPs to human health remains. Some researchers emphasized the danger posed by food chain transfer, while others claimed no adverse effect caused by MPs or MP additives [78, 81]. The controversies mostly lie in the uncertainty of MP intake estimate, which calls for more precise MP intake measurements or stimulating analysis. More research is urged to quantify the concentrations of MPs in the tissue and to understand the mechanisms of the induced human symptoms [82].
7 Conclusions
Food safety is an important issue closely related to human health. MP-contaminated food poses a global concern, and humans are vulnerable to MP exposure through consumption of these food items. The related information is scarce, and there may be more kinds of food contaminated by MPs. What’s more, broader range of research subjects and the detection technologies of small-sized particles are required in addition to the uniformity of methods when we are assessing food safety. Besides, we should take all pollution sources and possibilities into consideration. On this basis, the human intake we calculate will be closer to the actual value.
References
Thompson RC, Olsen Y, Mitchell RP, Davis A, Rowland SJ, John AWG, McGonigle D, Russell AE (2004) Lost at sea: where is all the plastic? Science 304(5672):838–838. https://doi.org/10.1126/science.1094559
Rezania S, Park J, Din MFM, Taib SM, Talaiekhozani A, Yadav KK, Kamyab H (2018) Microplastics pollution in different aquatic environments and biota: a review of recent studies. Mar Pollut Bull 133:191–208. https://doi.org/10.1016/j.marpolbul.2018.05.022
Wright SL, Thompson RC, Galloway TS (2013) The physical impacts of microplastics on marine organisms: A review. Environ Pollut 178:483–492. https://doi.org/10.1016/j.envpol.2013.02.031
Van Cauwenberghe L, Devriese L, Galgani F, Robbens J, Janssen CR (2015) Microplastics in sediments: a review of techniques, occurrence and effects. Mar Environ Res 111:5–17. https://doi.org/10.1016/j.marenvres.2015.06.007
Van Cauwenberghe L, Claessens M, Vandegehuchte MB, Janssen CR (2015) Microplastics are taken up by mussels (Mytilus edulis) and lugworms (Arenicola marina) living in natural habitats. Environ Pollut 199:10–17. https://doi.org/10.1016/j.envpol.2015.01.008
Li JN, Lusher AL, Rotchell JM, Deudero S, Turra A, Brate ILN, Sun CJ, Hossain MS, Li QP, Kolandhasamy P, Shi HH (2019) Using mussel as a global bioindicator of coastal microplastic pollution. Environ Pollut 244:522–533. https://doi.org/10.1016/j.envpol.2018.10.032
Lusher AL, McHugh M, Thompson RC (2013) Occurrence of microplastics in the gastrointestinal tract of pelagic and demersal fish from the English Channel. Mar Pollut Bull 67(1–2):94–99. https://doi.org/10.1016/j.marpolbul.2012.11.028
Devriese LI, van der Meulen MD, Maes T, Bekaert K, Paul-Pont I, Frere L, Robbens J, Vethaak AD (2015) Microplastic contamination in brown shrimp (Crangon crangon, Linnaeus 1758) from coastal waters of the Southern North Sea and Channel area. Mar Pollut Bull 98(1–2):179–187. https://doi.org/10.1016/j.marpolbul.2015.06.051
Bessa F, Barria P, Neto JM, Frias JPGL, Otero V, Sobral P, Marques JC (2018) Occurrence of microplastics in commercial fish from a natural estuarine environment. Mar Pollut Bull 128:575–584. https://doi.org/10.1016/j.marpolbul.2018.01.044
Carbery M, O’Connor W, Thavamani P (2018) Trophic transfer of microplastics and mixed contaminants in the marine food web and implications for human health. Environ Int 115:400–409. https://doi.org/10.1016/j.envint.2018.03.007
Seltenrich N (2015) New link in the food chain? Marine plastic pollution and seafood safety. Environ Health Perspect 123(2):A34–A41. https://doi.org/10.1289/ehp.123-A34
Toussaint B, Raffael B, Angers-Loustau A, Gilliland D, Kestens V, Petrillo M, Rio-Echevarria IM, Eede GVD (2019) Review of micro- and nanoplastic contamination in the food chain. Food Addit Contam Part A Chem Anal Control Expo Risk Assess 36:1–35. https://doi.org/10.1080/19440049.2019.1583381
Liebezeit G, Liebezeit E (2013) Non-pollen particulates in honey and sugar. Food Addit Contam Part A Chem Anal Control Expo Risk Assess 30(12):2136–2140. https://doi.org/10.1080/19440049.2013.843025
Liebezeit G, Liebezeit E (2014) Synthetic particles as contaminants in German beers. Food Addit Contam Part A Chem Anal Control Expo Risk Assess 31(9):1574–1578. https://doi.org/10.1080/19440049.2014.945099
Liebezeit G, Liebezeit E (2015) Origin of synthetic particles in honeys. Pol J Food Nutr Sci 65(2):143–147. https://doi.org/10.1515/pjfns-2015-0025
Yang D, Shi H, Li L, Li J, Jabeen K, Kolandhasamy P (2015) Microplastic pollution in table salts from China. Environ Sci Technol 49(22):13622–13627. https://doi.org/10.1021/acs.est.5b03163
Avio CG, Gorbi S, Milan M, Benedetti M, Fattorini D, d’Errico G, Pauletto M, Bargelloni L, Regoli F (2015) Pollutants bioavailability and toxicological risk from microplastics to marine mussels. Environ Pollut 198:211–222. https://doi.org/10.1016/j.envpol.2014.12.021
Lu YF, Zhang Y, Deng YF, Jiang W, Zhao YP, Geng JJ, Ding LL, Ren HQ (2016) Uptake and accumulation of polystyrene microplastics in zebrafish (Danio rerio) and toxic effects in liver. Environ Sci Technol 50(7):4054–4060. https://doi.org/10.1021/acs.est.6b00183
Besseling E, Redondo-Hasselerharm P, Foekema EM, Koelmans AA (2019) Quantifying ecological risks of aquatic micro- and nanoplastic. Crit Rev Env Sci Technol 49(1):32–80. https://doi.org/10.1080/10643389.2018.1531688
Lu L, Wan ZQ, Luo T, Fu ZW, Jin YX (2018) Polystyrene microplastics induce gut microbiota dysbiosis and hepatic lipid metabolism disorder in mice. Sci Total Environ 631-632:449–458. https://doi.org/10.1016/j.scitotenv.2018.03.051
Barboza LGA, Vethaak AD, Lavorante BRBO, Lundebye AK, Guilhermino L (2018) Marine microplastic debris: an emerging issue for food security, food safety and human health. Mar Pollut Bull 133:336–348. https://doi.org/10.1016/j.marpolbul.2018.05.047
Van Cauwenberghe L, Janssen CR (2014) Microplastics in bivalves cultured for human consumption. Environ Pollut 193:65–70. https://doi.org/10.1016/j.envpol.2014.06.010
Santana MFM, Moreira FT, Turra A (2017) Trophic transference of microplastics under a low exposure scenario: insights on the likelihood of particle cascading along marine food-webs. Mar Pollut Bull 121(1–2):154–159. https://doi.org/10.1016/j.marpolbul.2017.05.061
Hantoro I, Lohr AJ, Van Belleghem FGAJ, Widianarko B, Ragas AMJ (2019) Microplastics in coastal areas and seafood: implications for food safety. Food Addit Contam Part A Chem Anal Control Expo Risk Assess 36(5):674–711. https://doi.org/10.1080/19440049.2019.1585581
Peixoto D, Pinheiro C, Amorim J, Oliva-Teles L, Guilhermino L, Vieira MN (2019) Microplastic pollution in commercial salt for human consumption: a review. Estuar Coast Shelf Sci 219:161–168
Koelmans AA, Nor NHM, Hermsen E, Kooi M, Mintenig SM, De France J (2019) Microplastics in freshwaters and drinking water: critical review and assessment of data quality. Water Res 155:410–422. https://doi.org/10.1016/j.watres.2019.02.054
Li JN, Yang DQ, Li L, Jabeen K, Shi HH (2015) Microplastics in commercial bivalves from China. Environ Pollut 207:190–195. https://doi.org/10.1016/j.envpol.2015.09.018
Li JN, Green C, Reynolds A, Shi HH, Rotchell JM (2018) Microplastics in mussels sampled from coastal waters and supermarkets in the United Kingdom. Environ Pollut 241:35–44. https://doi.org/10.1016/j.envpol.2018.05.038
Rochman CM, Tahir A, Williams SL, Baxa DV, Lam R, Miller JT, Teh FC, Werorilangi S, Teh SJ (2015) Anthropogenic debris in seafood: plastic debris and fibers from textiles in fish and bivalves sold for human consumption. Sci Rep 5:14340. https://doi.org/10.1038/srep14340
Akhbarizadeh R, Moore F, Keshavarzi B (2018) Investigating a probable relationship between microplastics and potentially toxic elements in fish muscles from northeast of Persian Gulf. Environ Pollut 232:154–163. https://doi.org/10.1016/j.envpol.2017.09.028
Bellas J, Martinez-Armental J, Martinez-Camara A, Besada V, Martinez-Gomez C (2016) Ingestion of microplastics by demersal fish from the Spanish Atlantic and Mediterranean coasts. Mar Pollut Bull 109(1):55–60. https://doi.org/10.1016/j.marpolbul.2016.06.026
Renzi M, Blaskovic A (2018) Litter & microplastics features in table salts from marine origin: Italian versus Croatian brands. Mar Pollut Bull 135:62–68. https://doi.org/10.1016/j.marpolbul.2018.06.065
Kim JS, Lee HJ, Kim SK, Kim HJ (2018) Global pattern of microplastics (MPs) in commercial food-grade salts: sea salt as an indicator of seawater MP pollution. Environ Sci Technol 52(21):12819–12828. https://doi.org/10.1021/acs.est.8b04180
Iniguez ME, Conesa JA, Fullana A (2017) Microplastics in Spanish table salt. Sci Rep 7:8620. https://doi.org/10.1038/S41598-017-09128-X
Pivokonsky M, Cermakova L, Novotna K, Peer P, Cajthaml T, Janda V (2018) Occurrence of microplastics in raw and treated drinking water. Sci Total Environ 643:1644–1651. https://doi.org/10.1016/j.scitotenv.2018.08.102
Kosuth M, Mason SA, Wattenberg EV (2018) Anthropogenic contamination of tap water, beer, and sea salt. PLoS One 13(4):e0194970. https://doi.org/10.1371/journal.pone.0194970
Ossmann BE, Sarau G, Holtmannspotter H, Pischetsrieder M, Christiansen SH, Dicke W (2018) Small-sized microplastics and pigmented particles in bottled mineral water. Water Res 141:307–316. https://doi.org/10.1016/j.watres.2018.05.027
Mason SA, Welch VG, Neratko J (2018) Synthetic polymer contamination in bottled water. Front Chem 6:407. https://doi.org/10.3389/Fchem.2018.00407
Karami A, Golieskardi A, Choo CK, Larat V, Karbalaei S, Salamatinia B (2018) Microplastic and mesoplastic contamination in canned sardines and sprats. Sci Total Environ 612:1380–1386. https://doi.org/10.1016/j.scitotenv.2017.09.005
Phillips MB, Bonner TH (2015) Occurrence and amount of microplastic ingested by fishes in watersheds of the Gulf of Mexico. Mar Pollut Bull 100(1):264–269. https://doi.org/10.1016/j.marpolbul.2015.08.041
Nie H, Wang J, Xu K, Huang Y, Yan M (2019) Microplastic pollution in water and fish samples around Nanxun Reef in Nansha Islands, South China Sea. Sci Total Environ 696:134022. https://doi.org/10.1016/j.scitotenv.2019.134022
Pazos RS, Maiztegui T, Colautti DC, Paracampo AH, Gomez N (2017) Microplastics in gut contents of coastal freshwater fish from Rio de la Plata estuary. Mar Pollut Bull 122(1–2):85–90. https://doi.org/10.1016/j.marpolbul.2017.06.007
Feng Z, Zhang T, Li Y, He X, Wang R, Xu J, Gao G (2019) The accumulation of microplastics in fish from an important fish farm and mariculture area, Haizhou Bay, China. Sci Total Environ 696:133948. https://doi.org/10.1016/j.scitotenv.2019.133948
Guven O, Gokdag K, Jovanovic B, Kideys AE (2017) Microplastic litter composition of the Turkish territorial waters of the Mediterranean Sea, and its occurrence in the gastrointestinal tract of fish. Environ Pollut 223:286–294. https://doi.org/10.1016/j.envpol.2017.01.025
Karami A, Golieskardi A, Ho YB, Larat V, Salamatinia B (2017) Microplastics in eviscerated flesh and excised organs of dried fish. Sci Rep 7:5473. https://doi.org/10.1038/s41598-017-05828-6
Thushari GGN, Senevirathna JDM, Yakupitiyage A, Chavanich S (2017) Effects of microplastics on sessile invertebrates in the eastern coast of Thailand: an approach to coastal zone conservation. Mar Pollut Bull 124(1):349–355. https://doi.org/10.1016/j.marpolbul.2017.06.010
Murray F, Cowie PR (2011) Plastic contamination in the decapod crustacean Nephrops norvegicus (Linnaeus, 1758). Mar Pollut Bull 62(6):1207–1217. https://doi.org/10.1016/j.marpolbul.2011.03.032
Li JN, Qu XY, Su L, Zhang WW, Yang DQ, Kolandhasamy P, Li DJ, Shi HH (2016) Microplastics in mussels along the coastal waters of China. Environ Pollut 214:177–184. https://doi.org/10.1016/j.envpol.2016.04.012
Taylor ML, Gwinnett C, Robinson LF, Woodall LC (2016) Plastic microfibre ingestion by deep-sea organisms. Sci Rep 6:33997. https://doi.org/10.1038/srep33997
Seth CK, Shriwastav A (2018) Contamination of Indian sea salts with microplastics and a potential prevention strategy. Environ Sci Pollut Res 25:1–10. https://doi.org/10.1007/s11356-018-3028-5
Karami A, Golieskardi A, Choo CK, Larat V, Galloway TS, Salamatinia B (2017) The presence of microplastics in commercial salts from different countries (vol 7, 46173, 2017). Sci Rep 7:46838. https://doi.org/10.1038/Srep46838
Schymanski D, Goldbeck C, Humpf HU, Furst P (2018) Analysis of microplastics in water by micro-Raman spectroscopy: release of plastic particles from different packaging into mineral water. Water Res 129:154–162. https://doi.org/10.1016/j.watres.2017.11.011
Muhlschlegel P, Hauk A, Walter U, Sieber R (2017) Lack of evidence for microplastic contamination in honey. Food Addit Contam Part A Chem Anal Control Expo Risk Assess 34(11):1982–1989. https://doi.org/10.1080/19440049.2017.1347281
Lwanga EH, Vega JM, Quej VK, Chi JD, del Cid LS, Chi C, Segura GE, Gertsen H, Salanki T, van der Ploeg M, Koelmans AA, Geissen V (2017) Field evidence for transfer of plastic debris along a terrestrial food chain. Sci Rep 7:14071. https://doi.org/10.1038/s41598-017-14588-2
Hernandez LM, Xu EG, Larsson HCE, Tahara R, Maisuria VB, Tufenkji N (2019) Plastic teabags release billions of microparticles and nanoparticles into tea. Environ Sci Technol 53(21):12300–12310. https://doi.org/10.1021/acs.est.9b02540
Li LZ, Zhou Q, Yin N et al (2019) Uptake and accumulation of microplastics in an edible plant (in Chinese). Chin Sci Bull 64:928–934. https://doi.org/10.1360/N972018-00845
Ng EL, Lwanga EH, Eldridge SM, Johnston P, Hu HW, Geissen V, Chen DL (2018) An overview of microplastic and nanoplastic pollution in agroecosystems. Sci Total Environ 627:1377–1388. https://doi.org/10.1016/j.scitotenv.2018.01.341
Li Q, Sun C, Wang Y, Cai H, Li L, Li J, Shi H (2019) Fusion of microplastics into the mussel byssus. Environ Pollut 252:420–426. https://doi.org/10.1016/j.envpol.2019.05.093
Su L, Deng H, Li B, Chen Q, Pettigrove V, Wu C, Shi H (2019) The occurrence of microplastic in specific organs in commercially caught fishes from coast and estuary area of east China. J Hazard Mater 365:716–724. https://doi.org/10.1016/j.jhazmat.2018.11.024
Vianello A, Jensen RL, Liu L, Vollertsen J (2019) Simulating human exposure to indoor airborne microplastics using a Breathing Thermal Manikin. Sci Rep 9:8670. https://doi.org/10.1038/s41598-019-45054-w
Dris R, Gasperi J, Mirande C, Mandin C, Guerrouache M, Langlois V, Tassin B (2017) A first overview of textile fibers, including microplastics, in indoor and outdoor environments. Environ Pollut 221:453–458. https://doi.org/10.1016/j.envpol.2016.12.013
Catarino AI, Macchia V, Sanderson WG, Thompson RC, Henry TB (2018) Low levels of microplastics (MP) in wild mussels indicate that MP ingestion by humans is minimal compared to exposure via household fibres fallout during a meal. Environ Pollut 237:675–684. https://doi.org/10.1016/j.envpol.2018.02.069
Powles J, Fahimi S, Micha R, Khatibzadeh S, Shi PL, Ezzati M, Engell RE, Lim SS, Danaei G, Mozaffarian D, Dis GBDNC (2013) Global, regional and national sodium intakes in 1990 and 2010: a systematic analysis of 24 h urinary sodium excretion and dietary surveys worldwide. BMJ Open 3(12):e003733. https://doi.org/10.1136/bmjopen-2013-003733
Hantoro I, Lohr AJ, Van Belleghem F, Widianarko B, Ragas AMJ (2019) Microplastics in coastal areas and seafood: implications for food safety. Food Addit Contam Part A Chem Anal Control Expo Risk Assess 36(5):1–38. https://doi.org/10.1080/19440049.2019.1585581
Kim SK, Kho YL, Shoeib M, Kim KS, Kim KR, Park JE, Shin YS (2011) Occurrence of perfluorooctanoate and perfluorooctanesulfonate in the Korean water system: Implication to water intake exposure. Environ Pollut 159(5):1167–1173. https://doi.org/10.1016/j.envpol.2011.02.004
Liebmann B, Köppel S, Königshofer P, Bucsics T, Reiberger T, Schwabl P (2018) Assessment of microplastic concentrations in human stool – preliminary results of a prospective study. https://doi.org/10.13140/RG.2.2.16638.02884
Jani P, Halbert GW, Langridge J, Florence AT (2011) Nanoparticle uptake by the rat gastrointestinal mucosa: quantitation and particle size dependency. J Pharm Pharmacol 42(12):821–826. https://doi.org/10.1111/j.2042-7158.1990.tb07033.x
Schirinzi GF, Perez-Pomeda I, Sanchis J, Rossini C, Farre M, Barcelo D (2017) Cytotoxic effects of commonly used nanomaterials and microplastics on cerebral and epithelial human cells. Environ Res 159:579–587. https://doi.org/10.1016/j.envres.2017.08.043
von Moos N, Burkhardt-Holm P, Kohler A (2012) Uptake and effects of microplastics on cells and tissue of the blue mussel Mytilus edulis L. after an experimental exposure. Environ Sci Technol 46(20):11327–11335. https://doi.org/10.1021/es302332w
Greven AC, Merk T, Karagoz F, Mohr K, Klapper M, Jovanovic B, Palic D (2016) Polycarbonate and polystyrene nanoplastic particles act as stressors to the innate immune system of fathead minnow (Pimephales promelas). Environ Toxicol Chem 35(12):3093–3100. https://doi.org/10.1002/etc.3501
Jones AE, Watts JA, Debelak JP, Thornton LR, Younger JG, Kline JA (2003) Inhibition of prostaglandin synthesis during polystyrene microsphere-induced pulmonary embolism in the rat. Am J Physiol Lung Cell Mol Physiol 284(6):1072–1081. https://doi.org/10.1152/ajplung.00283.2002
Churg A, Brauer M (2000) Ambient atmospheric particles in the airways of human lungs. Ultrastruct Pathol 24(6):353–361. https://doi.org/10.1080/019131200750060014
Geiser M, Rothen-Rutishauser B, Kapp N, Schurch S, Kreyling W, Schulz H, Semmler M, Hof VI, Heyder J, Gehr P (2005) Ultrafine particles cross cellular membranes by nonphagocytic mechanisms in lungs and in cultured cells. Environ Health Perspect 113(11):1555–1560. https://doi.org/10.1289/ehp.8006
Yacobi NR, DeMaio L, Xie JS, Hamm-Alvarez SF, Borok Z, Kim KJ, Crandall ED (2008) Polystyrene nanoparticle trafficking across alveolar epithelium. Nanomedicine 4(2):139–145. https://doi.org/10.1016/j.nano.2008.02.002
Oliveira M, Ribeiro A, Hylland K, Guilhermino L (2013) Single and combined effects of microplastics and pyrene on juveniles (0+ group) of the common goby Pomatoschistus microps (Teleostei, Gobiidae). Ecol Indic 34(11):641–647. https://doi.org/10.1016/j.ecolind.2013.06.019
Oberbeckmann S, Loder MGJ, Labrenz M (2015) Marine microplastic-associated biofilms – a review. Environ Chem 12(5):551–562. https://doi.org/10.1071/EN15069
Iniguez ME, Conesa JA, Fullana A (2017) Pollutant content in marine debris and characterization by thermal decomposition. Mar Pollut Bull 117(1–2):359–365. https://doi.org/10.1016/j.marpolbul.2017.02.022
Bouwmeester H, Hollman PCH, Peters RJB (2015) Potential health impact of environmentally released micro- and nanoplastics in the human food production chain: experiences from nanotoxicology. Environ Sci Technol 49(15):8932–8947. https://doi.org/10.1021/acs.est.5b01090
Liu X, Shi H, Xie B, Dionysiou DD, Zhao Y (2019) Microplastics as both a sink and a source of bisphenol A in the marine environment. Environ Sci Technol 53(17):10188–10196. https://doi.org/10.1021/acs.est.9b02834
Mato Y, Isobe T, Takada H, Kanehiro H, Ohtake C, Kaminuma T (2001) Plastic resin pellets as a transport medium for toxic chemicals in the marine environment. Environ Sci Technol 35(2):318–324. https://doi.org/10.1021/es0010498
Hussain N, Jaitley V, Florence AT (2001) Recent advances in the understanding of uptake of microparticulates across the gastrointestinal lymphatics. Adv Drug Deliv Rev 50(1–2):107–142. https://doi.org/10.1016/S0169-409x(01)00152-1
Prata JC (2018) Airborne microplastics: consequences to human health? Environ Pollut 234:115–126. https://doi.org/10.1016/j.envpol.2017.11.043
Acknowledgments
The authors gratefully acknowledge the financial support by the grants from the National Key Research and Development Program (2016YFC1402204).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Zhang, Q., Zhao, Y., Li, J., Shi, H. (2020). Microplastics in Food: Health Risks. In: He, D., Luo, Y. (eds) Microplastics in Terrestrial Environments. The Handbook of Environmental Chemistry, vol 95. Springer, Cham. https://doi.org/10.1007/698_2020_453
Download citation
DOI: https://doi.org/10.1007/698_2020_453
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-56270-0
Online ISBN: 978-3-030-56271-7
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)