Abstract
Modern economic development of the Boka Kotorska Bay, known for its outstanding natural values, in terms of dynamics and size, is often not in line with the principles of environmental protection. As a consequence, over the recent decades, the Bay has been exposed to anthropogenic pollution. Marine sediment acts at the same time as a depositor and also the secondary source of pollutants, thus the analysis of pollutants in sediment is of vital importance for the estimation of its quality. Based on the results obtained over the past two decades, this paper considers characterization of sediment of the Boka Kotorska Bay in terms of heavy metal content and an assessment of heavy metal contamination in sediment and biota (Mytilus galloprovincialis and Posidonia oceanica), by applying environmental quality indexes: Enrichment Factor (EF), Metal Pollution Index (MPI), and Geo-accumulation Index (I geo).
Since 2009, the Institute of Marine Biology Kotor has been conducting Biomonitoring in the framework of the Environmental Monitoring Programme for Montenegro – Program monitoring of the coastal sea ecosystem status of Montenegro, under the MED POL program. The main activities are aimed at determining the environmental status of the Boka Kotorska Bay marine ecosystem by analyzing the parameters and biomarkers described by MED POL program. Beside the recommended biomarkers (metallothioneins, acetilholinesterase, catalase test, glutathione S-transferase, micronuclei test) a new approach of seawater quality biomonitoring was established and it is based on physiological biomarkers of benthic invertebrates. Analysis of all biomarkers showed the pollution trends in Boka Kotorska Bay.
The original version of this chapter was revised: Reference list has been updated. The erratum to this chapter is available at DOI: 10.1007/698_2016_90.
An erratum to this chapter can be found at http://dx.doi.org/10.1007/698_2016_90
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
In recent decades anthropogenic activities (agricultural, urban, and industrial) have led to increased pollution of marine ecosystems, especially in the bays. As a consequence of these actions, pollutants get into the water and often cause irreversible changes in marine ecosystem. Beside these pollutants, heavy metals are of the major concern due to their persistence and bio-accumulative nature [1]. Heavy metals can be introduced into the aquatic environment and accumulate in sediments by disposal of liquid effluents, chemical leachates, and runoff originating from domestic, industrial, and agricultural activities, as well as atmospheric deposition [2, 3].
In Europe, coastal waters are protected by the Water Framework Directive (WFD) and the seas are protected by the Marine Strategy Framework Directive (MSFD). Member States are required to achieve or maintain “good environmental statuses” within their marine waters by 2020 under the MSFD [4]. In order to do that Regional Conventions started to recommend specific sub-lethal biological responses (contaminant-related biomarkers) to be measured in marine organisms [5, 6]. MSFD (Directive 2008/56/EC) has put emphasis on the importance of assessing key biological responses to evaluate the health of organisms and to link any observed responses to contaminant exposure [7]. Marine pollution is defined by GESAMP (The Joint Group of Experts on the Scientific Aspects of Marine Environmental Protection) as introduction by man, directly or indirectly, of substances or energy into the marine environment resulting in such deleterious effects as harm to living resources, hazards to human health, hindrance to marine activities including fishing, impairment of quality for use of sea water, and reduction of amenities. The focus is therefore on humans, rather than natural inputs to the sea and damaging effects of the wastes. Bioavailability is an important aspect of environmental pollution studying. Current scientific knowledge regarding bioavailability, particularly of the microelements, with ecological risk assessment is relatively limited [8].
To assess the ecosystem health, fundamental approach is usage of biomarkers as indicators of pollution. Biomarkers are defined as “a xenobiotically induced variation in cellular or biochemical components, and in processes, structures, or functions, that are measurable in a biological system or sample” [9]. These biomarkers have been incorporated in several regional programs of biomonitoring carried out under international bodies (UNEP and OSPAR) [10]. Biomonitoring is a scientific technique for assessing the environment including human exposures to natural and synthetic chemicals, based on sampling and analysis of an individual organism’s (biomarkers/bioindicators) tissues and fluids [11, 12].
Biomarkers allow the detection of early biological changes that can lead to long-term physiological disorders and can be used as early warning signals of environmental disturbance [12, 13]. Unlike the chemical monitoring that assesses only the presence of pollutants in the cells and tissues using chemical analysis, biomonitoring is not in use to assess the presence exclusively; organisms’ response and impact of pollutants on molecules, cells, tissues/organs, and body of the animals are more important [12]. Biomonitoring cannot replace chemical monitoring but it integrates them and provides unique contribution in toxicity determination of pollutants, even in cases where they are present in low, sub-lethal concentrations.
EU Water Framework Directive (WFD, Directive 2000/60/EC) integrated chemical, biological, physicochemical, hydro-morphological parameters and ecological status in defining water quality [14]. WFD recommends monitoring programs that are necessary for the assessment to achieve good chemical and ecological status of water bodies and stresses the importance of biological monitoring to determine the water quality. Biomarkers, although not incorporated in the WFD, are among the emerging biological monitoring tools considered for use in monitoring programs necessary for the implementation of the WFD [15, 16].
2 Metal Pollution in Sediments in the Boka Kotorska Bay
Biogeochemical cycles of metals in the sea derive from equilibrium processes of chemical substances on the border between the atmosphere, sediment, and seawater. Trace metals enter the sea by different paths such as atmospheric deposition (rain, dust particles, and other precipitation), land erosion, or by rivers loaded with atmospheric and waste waters. Their bioavailability from sediments is affected by the complex relation of the factors: physicochemical features of microelements and sediments, and biological strategies of the organism involved. Considering biological systems complexity, simple relationship between the microelements concentration in the environment and their bioavailability to an organism cannot be expected.
However, biogeochemical cycle of certain natural constituents of seawater is exposed to negative anthropogenic impact. The additional metal quantities in the seawater are derived from industrial waste waters. Accordingly, coastal and industrialized areas are the most vulnerable regarding high metal content in the sea relative to natural content [17, 18]. The purpose and nature of sediment and biota monitoring differ from water monitoring. Sediment monitoring and its quality assessment are mainly conducted in order to determine to what extent sediment acts as a reservoir and secondary source of contaminants in surface waters.
These studies can be conducted in order to determine sediment quality status and its effect on the environment and human health by studying different interactions of the sediment-water system or for regulatory implications such as dredging and final disposal of dredged material.
Sediment quality assessment is mainly limited to chemical characterization. In general, determination of pollutants and their concentration cannot provide enough information in order to adequately determine antagonistic effects or interactions between pollutants or time-dependent availability of pollutants to aquatic organisms.
Sediment toxicity can be defined as an ecological and biological change caused by sediment contamination [19]. Even though, in the case when the contaminant concentrations can be measured precisely (such as metal ions measurement), chemical analyzes do not provide the information regarding their bioavailability or possible negative effects [20].
Certain pollutants are less bioavailable than others [21, 22]. For example, many metals are bound to sediment or water particles in quantities inert in terms of influence on organisms. Accordingly, particular metals measured in high levels can have minimal influence. On the other hand, some other compounds (chlorophenols) may be present in relatively low concentrations, but with high impact on organisms. Based on all that, it can be concluded that measuring only the total concentration of different chemical substances is not enough for determination of potential pollutions [22, 23].
In order to understand the distribution of contaminants and ecological value of sediments in a particular area, in addition to elemental composition, it is necessary to know the grain size distribution, mineralogical composition, carbonate content, and content of organic matter in sediment. Granulometric composition of the sediment is a very important indicator of sediment properties [24–26]. It provides data on the concentration of particles with different grain sizes in observed sediment, which means the percentage content of the gravel, sand, and clay in sediment [27].
Based on granulometric composition of the sediment, it is possible to assess its physical and chemical properties. Generally, higher concentrations of heavy metals can be found in the mud, muddy sand, silt, and their mixture compared to carbonated and pure sand sediment [24, 28–30].
Hydrodynamics of the Bay is conditioned by the shape and position of the bay, as well as by a large influx of fresh water from the land in certain periods of the year. The Bay is rich in submerged springs of fresh water, the so-called vrulje that contribute to sediment re-suspension and transport. These conditions in the Bay affect changes in temperature, salinity, and density of seawater, and indirectly the deposition and distribution of metals. Data related to the research on the content of heavy metals in sediment, which have been published in recent years, indicate that the sediment of the Boka Kotorska Bay is loaded with heavy metals and that certain locations, such as ports and marinas, are characterized by a high degree of sediment contamination by heavy metals [3]. Deposition of heavy metals in sediment of the Buy was the result of many years of inadequate treatment of industrial water and municipal water. Environmental risk is greater because the Bay is an almost closed water basin. Although now practically there is no industrial activity in the Bay, the former factory of soap and detergents Riviera, the manufacture and repair of bearings and Jugopetrol warehouses in Kotor, former repair shipyard Arsenal and Avioservis in Tivat, shipyard in Bijela, were in past decades the main sources of heavy metals in the Bay, some of which still have a negative impact on its aquatorium. The intensive marine transport in the Bay, large sailing cruisers, the presence of marinas and small ports, roads that are located along the coast, also contribute to the intake of metals in the waters of the Bay, especially during the tourist season. Due to the natural attractiveness and intense socioeconomic development, the Bay of Boka Kotorska is in recent years exposed to the pressure of rapid urbanization and increasing population, which makes its ecosystem extremely vulnerable to pollution.
Within the framework of monitoring the state of coastal ecosystem of Montenegro which has been implemented in accordance with international standards since 2008, the state of sediment quality is monitored in the Boka Kotorska Bay, especially on the so-called hot spots [3]. This monitoring includes also the analysis of sediment on the content of heavy metals: Cd, Hg, Cu, Ni, Fe, Mn, Pb, Zn, Cr, As. In addition, sediment is the subject of analysis within the national and international projects, the studies on the assessment of environmental impact, or after the contamination caused by accidents.
According to the results of particles’ distribution relative to their size in sediment samples taken from nine locations in the Boka Kotorska Bay in 2014 (Fig. 1), it can be concluded that majority of the sediment samples have muddy consistency, consisted of grain size particles >0.063 mm that belong to clay and silt fractions. Exceptions are sediments in the locations in front of the former repair shipyard “Arsenal” (TV Porto MNE) with 22.35% mud and near the Lustica Peninsula, at the exit from the Bay of Kotor (with 7.10% mud), which mainly consist of different fractions of sand. Mud concentration (clay and silt) on other locations in the Boka Kotorska Bay was in the range 68.12–99.05% [31].
The content of the heavy metals in sediment was investigated at the same locations. Their concentrations in the sediment, expressed in mg/kg dw, were within the following ranges: Cd (0.11–0.36), Hg (0.05–4.40), Cu (12.5–121.5), Ni (68.3–161.7), Fe (12,200–36,000), Mn (341.0–1545.0), Pb (9.0–84.3), Zn (34.5–241.0), Cr (44.9–95.8), As (11.0– 39.0). The presence of the investigated elements in the sediment, expressed on the basis of the average concentration, decreases in the series:
In terms of the content of investigated heavy metals, the lowest mass fraction of Cd, Hg, Cu, Fe, Mn, Pb, Zn, Ni, Cr, As was measured at the reference location in the Lustica Peninsula. This location is close to the open sea where the dynamics of water is more intensive, away from direct sources of anthropogenic pollution, and with the low content of the mud in the composition of its sediment, which support the obtained results. On the other hand, higher concentrations deviations from the average values for specific metals and locations, such as Cu in sediment near the shipyard Bijela (TV B. Bijela), Hg, Pb, and As near former repair shipyard (TV Porto MNE), then Zn, Pb, and Cd in the port of Kotor (KO port), indicate a significant presence of anthropogenic sources of pollution in the aquatorium of the Tivat and Kotor Bays. The sources of these elements in the Bay might be activities in shipyards, discharges of untreated municipal and waste water, surrounding soil erosion, and nautical tourism. Cu, Zn, As, and Pb are constituents in antifouling paints and other marine coatings (corrosion, wood preservation, etc.), solid waste and waste water of different origin.
Most of the heavy metals, regardless the natural or anthropogenic origin, have higher affinity to fine particle material of sediment relative to coarse fraction.
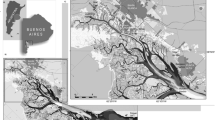
The seabed the Boka Kotorska Bay is mostly covered with layers of fine mud, which further contributes to the accumulation of pollutants in their sediments [32] (Fig. 2). The affinity of heavy metals to the sediment is enhanced by ingredients such as organic matter and clay minerals [33].
Bathymetric contour map of the Montenegro/Northern Albania Continental Margin with indicated main morphologic features [32]
Also, carbonates content is the indicator of heavy metal content in surface sediment. A smaller content of the organic matter and higher carbonate content indicates less presence of heavy metals in sediment [34].
The recent study [35] in the Boka Kotorska Bay has shown that the surface sediment is mostly composed of lithogenous material, impacted by biogenic and anthropogenic materials. The source of terrigenous fraction in sediments are rock weathering by rain and river erosion, while biological processes like secretion (growth) of skeletal materials by marine organisms feed up biological content in sediment. Sediment in the Boka Kotorska Bay is characterized by high content of organic matter and carbonates [35]. The organic matter contents ranged from 2.84 to 12.93% and carbonates from 10.94 to 37.29%. The source of organic matter are terrigenous materials and decomposition of marine organisms, while carbonate phase probably originates from biogenic precipitation of aquatic organisms, precipitation of the CaCO3 from calcium-rich waters during photosynthesis with increase of pH and partly from natural carbonates [35]. The average content of investigated oxides in the same study of surface sediment in the Boka Kotorska Bay followed the order:
The results indicated positive relationship between heavy metals (Cu, Ni, Co, V, Mo, Rb, Zr, U, and Th) and majority of investigated oxides (K2O, SiO2, Al2O3, Fe2O3, MgO, and TiO2). Accordingly, they have the same source and association with clayey sediments. Also, the results indicated association of elements Ni, Co, Cu, V, Mo, Rb, Zr, U, and Th with aluminosilicates minerals in the sediment. It was concluded that CaO mainly derives from marine biogenic carbonate in surface sediment of the Bay. This was based on the negative correlation between CaO and remaining investigated oxides and elements (Co, Ni, Cu, Rb, Zr, Mo, V, W, U, and Th) [35].
3 Assessment of Metal Contamination
To evaluate degree of contamination in the sediments and biota, we used three parameters: Enrichment Factor (EF), Metal Pollution Index (MPI), and Geo-accumulation Index (I geo).
3.1 Enrichment Factor (EF)
The enrichment factor (EF) of metals is a useful indicator reflecting the status and degree of environmental contamination [36]. The EF calculations compare each value with a given background level, either from the local site, using older deposits formed under similar conditions, but without anthropogenic impact, or from a regional or global average composition [37, 38]. The EF was calculated using the method proposed by [39] as follows:
where (Me/Fe) sample is metal to Fe ratio in the sample of interest; (Me/Fe) background is the natural background value of metal to Fe ratio. In the present study, average shale [40] was used as background or undisturbed value for those metals because no such data was available for the study area.
Elements can be divided into three major groups: elements without enrichment, EF < 10; elements with medium-level enrichment, 10 < EF < 100; and finally highly enriched elements, EF > 100 [41].
In general, EF average values of sediment from autumn 2005 to spring 2007 in the Boka Kotorska indicate that Mn, Zn, Ni, Pb, Cu, Co, As, and Hg have enrichment factors less than 10, and they are in the group of elements without enrichment in surface sediment of the study area. The EF values suggest that metals originate entirely from crustal or natural weathering processes in the surface sediments, except Cd that was with medium-level enrichment in all investigated locations, except Kukuljina, where EF value was less than 10 [42].
Analyzing enrichments of investigated elements in seagrass Posidonia oceanica, the same equation was used to determine EF values as it was used for the sediment. EF average values in seagrass from autumn 2005 to spring 2007 show that Fe, Cu, and Mn in P. oceanica were derived from lithosphere mainly (lithogenic source), considering that EF values were lower than 10. The values were higher in other elements tested, Co and As were low or slightly elevated, Zn, Ni, Pb were high (medium-level enrichment). Cd and Hg were very high (more than 100) [42, 43]. Based on EF value, P. oceanica from location Mamula is enriched by majority of elements, particularly by Hg and Cd, except for As with the lowest value in Mamula.
According to an USEPA regulation [44], sediments with Hg contents below 0.3 mg kg–1 dw were considered as not polluted. This was the case for the analyzed sediments. Besides that, EF values for Hg indicated that Hg was not enriched in the sediments [42].
Accordingly, it is obvious that Hg found in the P. oceanica samples was not adsorbed from surface sediment. On the other hand, EF values for Cd were generally high both in sediment and seagrass samples. However, it was evidenced that sea weed accumulate Cd from water [45, 46].
3.2 Metal Pollution Index (MPI)
To compare the total content of metals at the different sampling stations, the Metal Pollution Index (MPI) was used. The MPI was obtained with the equation:
where: Cf1 = concentration value of the first metal; Cf2 = concentration value of the second metal; n is the number of metals; and CF is the contamination factor. The contamination can be calculated from: Contamination factor (CF) = metal concentration in sediments/Background values of the metal. The MPI > 1 is polluted whereas MPI < 1 indicates no pollution [47]. Based on the values obtained for MPI in sediment, it is obvious that pollution was present on all locations tested in the Boka Kotorska Bay in autumn 2005 and 2006 (Table 1). If we compare the seasons 2005 and 2006, MPI is higher in all location in 2005 (1.84–2.48) relative to 2006 (1.46–2.32). The location with the highest values of MPI in both examined seasons is Kukuljina (the Bay of Tivat), that corresponds with previous statements, because this location has the highest content of microelements in sediment. Although MPI values indicate the presence of pollution, in comparison with Mediterranean countries [47–49], Montenegrin coast is classified in areas with the lowest content of microelements in sediment.
Mapping the spatial distribution of contaminants in soils is the basis of pollution evaluation and risk control. Spatial distribution and concentrations of heavy metals in the bottom sediments are effected by both natural environment factors and anthropogenic factors. Spatial distribution of MPI in surface sediments from the Boka Kotorska Bay was showed to using Ocean DataView 4 (Fig. 3a, b).
If we compare MPI values obtained for sediment and seagrass P. oceanica for autumn 2005 to spring 2007, we can say that Mamula was the least polluted location in the Boka Kotorska Bay. In general, MPI values for P. oceanica were higher than MPI values for sediment on the same locations, the highest MPI average values for this seagrass were on the locations Sveti Stasije and Kukuljina [42, 43].
It can be said that the locations in the Bay of Tivat, particularly Tivat center, are the locations with the highest pollution index, based on MPI values for mussels (Mytilus galloprovincialis) in autumn 2007 to autumn 2009 (5.50–18.50). This is not surprising taking into account that this is the bay with the highest number of anthropogenic sources of pollution (airport, military port, shipyard, agricultural activities). On the other hand, Sveti Stasije in the Kotor Bay is the location with the lowest MPI values in all seasons (3.7–10.00). The highest MPI values were obtained during winter and autumn, while the lowest values were observed in spring for both years tested. On the basis of the MPI values determined for marine seagrass for the same period (autumn 2007 to autumn 2009), it can be said that there is no location with maximum or minimum values. MPI values vary in relation to the investigated locations, seasons, and years. Obviously, in the case of P. oceanica, metals pollution index is the indicator of pollution of both marine water and sediment, because here the physical-chemical parameters of water and sediment [50] play an important role in the bioavailability and accumulation of heavy metals.
Based on continuous monitoring of MPI values of mussels (Mytilus galloprovincialis) growing in the sea grass meadows vicinity, pollution level of selected location can be determined and adequate actions can be undertaken in the case of elevated MPI values. Accordingly, it is possible to decrease destruction and withdrawal of these unique and very useful marine species P. oceanica [50, 51].
3.3 Geo-Accumulation Index (I geo)
A common criterion to evaluate the heavy metal pollution in sediments is the geo-accumulation index (I geo), which was originally defined by Müller [52] to determine metals contamination in sediments, by comparing current concentrations with pre-industrial levels and can be calculated by the following equation [52]:
where C n is the concentration of element n and B n is the geochemical background value. In this study, consider B n = world surface rock average given by Martin and Meybeck [53]. The factor 1.5 is incorporated in the relationship to account for possible variation in background data due to lithogenic effect. Muller has classified I geo in relation to contamination levels into seven classes: unpolluted (Class 0, I geo < 0), unpolluted to moderately polluted (Class 1, 0 < I geo < 1), moderately polluted (Class 2, 1 < I geo < 2), moderately to highly polluted (Class3, 2 < I geo < 3), highly polluted (Class 4, 3 < I geo < 4), highly to extremely polluted (Class 5, 4 < I geo < 5), and extremely polluted (Class 6, I geo > 5), the highest grade reflecting a 100-fold enrichment above baseline values.
According to the categorization based on the obtained I geo values, locations Žanjice and Herceg Novi are categorized as unpolluted to moderately polluted locations by manganese (0.14 respectively 0.77), while the same can be said for nickel (0.42 respectively 1.11) in the case of Herceg Novi and Kukuljina. Kukuljina (Tivat Bay) is location with the highest geoaccumulation index value for Cd, with I geo value for Cd 3.79 in autumn 2006 that represents highly polluted area (Table 1). High contamination of this area by Cd is a consequence of the anthropogenic influence from the land, including waste waters, port activities, and usage of anticorrosive paints for ships and boats [54]. Interestingly, the toxic elements such as mercury, lead and arsenic, as well as the essential elements iron, copper and zinc have such I geo values that all investigated locations are classified practically as non-polluted areas.
Comparing the results of geo-accumulation index to other authors [54], we can conclude that obtained values are significantly lower than those from the data from reference works and that they appear in locations with elevated concentrations of metals.
4 Biomonitoring and Biomarkers in Boka Kotorska Bay
In the framework of the MED POL Program since 2009 in Montenegro is implementing a National Biomonitoring of the coastal waters in Boka Kotorska Bay in order to fulfill the obligations set out in the Convention for the Protection of the Marine Environment and the Coastal Region of the Mediterranean (Barcelona Convention, entered into force 2007). Following the Manual on the biomarkers recommended for the MED POL Biomonitoring programme [5] the Institute of Marine Biology conducted in the period from 2009 to 2015 a research of various biomarkers in order to assess the level and to determine the types of pollution in Boka Kotorska Bay.
The research included biomarkers of effect and biomarkers of exposure. For the detection and assessment of pollution levels in Boka Kotorska Bay the following biomarkers were determined: (a) the content of metallothioneins (MT) in response to exposure to heavy metals, (b) activity of Acetilholinesterase (AChE) in response to exposure to organophosphate and carbamate pesticides, (c) glutathione S-transferase (GST) in response to exposure to organic pollutants (PCB, PAHs), (d) evaluation of the oxidative stress using the Catalase test (CAT), and (e) presence of micronuclei (MN) as a marker of effects of mutagenic and clastogenic substances from the environment on organisms. Also to define the well-being of the animals the Condition (CI) and Gonadosomic (GSI) Indexes were determined.
As a sentinel species the mussel Mytillus galloprovincialis L. was used for monitoring. The mussels are desirable as bioindicators since they have the ability to accumulate water contaminants, have a broad geographical distribution, and can be easily grown in cages. With the parallel use of wild and mussels from the cage we can get information about the long-term and short-term effects of pollution in the areas where they are naturally present.
Biomarker responses were measured in mussels collected in their natural habitat at the location Kotor and the mussels taken from the mussel farm, the location Bijela. Both locations have different levels of pollution and ecological characteristics and are under great anthropogenic pressures due to the outflow of waste water, small shipyard, maritime transport, tourism, etc.
One of the first researches on biomarkers was done in 2008. Biological effects were investigated analyzing both generic and specific biomarkers at cellular level (metallothionein content, lysosomal membrane stability, lipofuscin and neutral lipid accumulation, lysosomal structural changes) [55].
Comparing the data from 2009 to 2015 for CI and GSI shows the decrease in the conditional index in both locations, which may be an indication that some adverse effect on mussels exists. CI values ranged from 10.78 g/cm3 in the location Kotor in 2015 up to 19.95 g/cm3 also in the location Kotor in 2013 [31].
The location Kotor has a higher CI than the location Bijela. The reason for this is the fact that the location Bijela sustains a larger anthropogenic impact (load of industrial pollution, shipbuilding, port activities), while the location Kotor is affected mainly by the waste water from households. Reduced nutritional status of mussels in both locations can lead to increased stress in mussels.
The content of metallothionein is determined in response to exposure to heavy metals. Metallothionein are a group of proteins of low molecular weight with a high content of thiol groups and metal atoms embedded in a relatively stable molecule tetrathionate cluster configuration type. The content of MT which is found in a particular tissue or cell is a direct reflection of the state of equilibrium that is established between the MT synthesis process and its degradation. Violation of this balance was due to the slow decomposition or due to increased synthesis leading to increased concentrations of MT in the cell. Although a variety of substances can cause the synthesis of MT, only some metals (Zn, Cd, Cu) and glucocorticoids are considered as primary inducers. Metallothionein concentration was highest in 2014 in the location Bijela (165.15 mg/g wet weight), and the lowest also was recorded in 2014 in the location Kotor (33.74 mg/g wet weight) [31]. In comparison to the Southern Adriatic (Slovenian coastal waters) the MT levels are lower in Southern Adriatic [56]. The essential difference between the heavy metal pollutions is the concentration of some metals. The location Bijela has higher levels of arsenic and lead with low capacity of binding to MT. On the other hand, it was found that the concentration of total Hg was ten times higher in the northern Adriatic than in other parts. A higher level of MT in the northern locations may be due to the high binding affinity for Hg MT, despite its low induction ability [57].
Acetylcholinesterase (AChE) is an enzyme involved in the transmission of nerve impulses, and its inhibition leads to neurotoxicity that occurs due to exposure of the body to organophosphorus and carbamate pesticides. Activity of acetylcholinesterase is inhibited by many pesticides (organophosphorus and carbamate pesticides) and metals such as cadmium (Cd) and chromium (Cr). Lately, there has been increasing evidence that different pollutants inhibit AChE and is therefore considered that the AChE biomarker of general stress. Activity of AChE was measured in whole mussel tissue and it ranged from 1.093 in the location Kotor (2013) to 12.63 nmol/min/mg in the location Bijela (2009) [31]. These concentrations were in the same range at the northern and southern stations [57].
Glutathione S-transferase (GST) is a detoxification enzyme which catalyzes the reaction of detoxification of numerous toxic compounds using glutathione. In this way, it prevents any interaction between the reactive products and cellular proteins or nucleic acids. If the organism is an animal under the influence of high oxidative stress, that would result in high activity of the enzyme glutathione S-transferase. The average specific activity of GST was the highest in the location Bijela in 2013 (8.179 nmol/mg protein/min) while the lowest value was in the location Kotor (4.390 nmol/mg protein/min) [31]. The activity of GST at the location Bijela was also higher than in Eastern Mediterranean [58].
Catalase (CAT) is an enzyme that is mainly found in the digestive tract (hepatopancreas) of animals and is a part of the antioxidant protection of the organism. Catalase breaks down toxic hydrogen peroxide to oxygen and water molecules. If there is no decomposition of hydrogen peroxide, then the oxidative stress induced by hydrogen peroxide leads to the cell death. If the animal is exposed to oxidative stress, hydrogen peroxide concentration value is increased resulting in higher activity of the enzyme catalase. Average maximum and minimum values are obtained in the location Kotor and ranged from 0.952 μmol/min/mg of protein in 2013 to 4.185 μmol/min/mg in 2015 [31]. There were no difference in the activity of the enzyme between the south of Adriatic and Eastern Mediterranean [58].
The application of genotoxicity biomarkers in sentinel organisms allows for the assessment of mutagenic hazards and/or for the identification of the sources and fate of the contaminants. Micronucleus (MN) test as an index of accumulated genetic damage during the lifespan of the cells is one of the most appropriate techniques for identification of the integrated response to the complex mixture of contaminants [59]. MN are formed in the process of cell division and their expression can occur at different times after the DNA damage event, depending on the cell cycle kinetics and the mechanism of induction [59]. During 2009 and 2010 MN test was done for the two sampling sites in the Boka Kotorska Bay. The MN frequencies were detected in the stations Kotor (3 ± 1) and Bijela (4 ± 1). Comparing the obtained results with the results from other sampling locations indicates that the test results do not differ and that there is an increased frequency of micronuclei. In July 2011, the Comet test was performed and it confirmed the results of the micronucleus test. The comet assay measured the levels of DNA damage molecules in the gills, hemolymph, and hepatopancreas caused by mutagenic and genotoxic substances. The level of DNA damage was significantly higher at the site Bijela [60].
In the cooperation framework between the University of Montenegro with Environmental Protection Agency of Montenegro and Saint-Petersburg Scientific Research Centre for Ecological Safety, Russian Academy of Sciences from Russia, at Institute of Marine Biology (IMB) Kotor, Montenegro established a new approach of seawater quality biomonitoring based on physiological biomarkers of benthic invertebrates. There are two monitoring types in use: active biomonitoring has been conducted since 2012 while passive biomonitoring has been implemented since 2016. The collaboration is aimed to improve the existing methodology of sea water quality assessment in the Boka Kotorska Bay by the integration of procedure already validated in Russia [61].
Due to specific seasonal variations in salinity and temperature of seawater surface in the BokaKotorska Bay [62], to overcome these environmental conditions and survive, intertidal animals attached to one location almost their entire life cycle are forced to activate protective physiological mechanisms. It was suggested that disruption of any physiological process of an organism will surely decrease its ability to survive, if the problem would occur on population level; it is not difficult to conclude that physiological biomarkers have direct relevance to entire population [63]. The physiological biomarkers of Mytilid mussels such as heart rate, clearance rate, condition indices, scope for metabolic arrest, scope for growth, shell growth, and byssus thread production were reviewed in the study of [64].
Recently, preliminary study of seawater quality assessment by active biomonitoring based on cardiac activity of the Mediterranean mussel Mytilus galloprovincialis L. as physiological biomarker has been conducted in a part of the Boka Kotorska Bay [65]. Since the heart rate (Hr) is a reliable indicator of general health condition of an organism, Hr recovery time and the coefficient of variation (CV) within a group of M. galloprovincialis L. are calculated after the stress, induced by the addition of distilled water in seawater aquarium. The method was developed by S. V. Kholodkevich and it is based on non-invasive recording of cardiac activity of invertebrates with hard skeletons by means of photoplethismograph technology with infrared laser fiber-optic sensors [66]. Experimental unit is presented in Fig. 4. Photoplethismographs are widely used as medical devices in optical detection of blood volume changes derived from contractile activity of the heart [67]. The usage of optical sensors in cardiac activity recording of aquatic invertebrates is innovative and reliable, also animal stress during handling is minimized. To avoid drilling a hole in shell valves to obtain Hr signal, small sensors are attached by waterproof glue above the heart area.
Beside the active biomonitoring of seawater quality including the addition of distilled water as functional loading in static system, passive biomonitoring of sea water quality based on Hr of M. galloprovincialis L. in the Boka Kotorska Bay is conducted. Experimental procedure was described in [61], whereby crayfish Pontascus leptodactylus was used as a model organism. Briefly, procedure requires permanent circulation of water directly from the sea through aquaria with mussels connected to a System for Industrial Biological Water Quality Monitoring (SIBWQM). For example, if potentially polluted seawater induced significant Hr variation within a group of mussels, the system would rapidly activate the alarm signal and seawater that caused the reaction would be sampled and used for physical-chemical analyses to reveal the source of pollution. Accordingly, this biomonitoring approach can be considered as reliable early warning system in detection of different pollution types in the aquatic ecosystems. Since the environmental pollution incidents are caused by human factor in most cases, there is an opportunity for implementation of “Polluter Pays Principle” [68], particularly in the inner part of the Boka Kotorska Bay, characterized by low level of water exchange and intensive maritime transport in relatively small area during the summer season. The application of aforementioned early warning system for biomonitoring enables detection of pollution on the initial phase and prevents further contamination of aquatic ecosystem.
Experimental unit for cardiac activity recording of aquatic invertebrates is also used for testing the toxicity of different chemical pollutants to cardiac physiology of mussels sampled in the Boka Kotorska Bay. The study [69] indicated high toxicity of cadmium-chloride to Hr of M. galloprovincialis L. In spite of washing out polluted seawater from the system after the exposure to toxicant, cardiac activity was at the low level a few hours later, suggesting possible inhibition of neurophysiological pathways.
In areas with intensive marine traffic, release of oil and oil derivatives in the seawater is inevitable [70]. Recently, in the Boka Kotorska Bay, 32.7 m ship has sunk, consequently, dispersed diesel fuel droplets were detected in the whole water column [71]. Intertidal animals and filter feeders were particularly endangered. The impact of diesel oil, dispersed diesel oil, and oil dispersant (Superdispersant-25) on Hr and level of DNA damage in haemocytes of M. galloprovincialis from the same area was investigated in studies [72, 73]. The results indicated high susceptibility of M. galloprovincialisto oil related pollution. Superdispersant-25 induced impairment of cardiac activity despite low concentrations. In parallel with Hr, level of DNA molecule damage in haemocytes of the same species was analyzed by single-cell gel electrophoresis (SCGE), known as comet assay. In general, genotoxic effect of diesel oil was not significantly expressed while exposure to Superdispersant-25 induced significant increase of tail intensity parameter (TI%) indicating on genotoxic potential of oil dispersant. Accordingly, chemical dispersion of oil should not be applied in partly closed systems such as the Boka Kotorska Bay. Previously, the evaluation of comet assay on three tissues of M. galloprovincials L. sampled on different locations in the Boka Kotorska Bay was conducted [60].
The studies including genotoxicity assessment are implemented in cooperation between the IMB Kotor, Department of Microbiology Faculty of Biology and Institute for Multidisciplinary Research, University of Belgrade, Serbia. The aim of the further collaboration is to integrate environmental toxicology methods and reinforce capacities for future participation in international projects regarding environmental protection.
5 Conclusion
Over the past few decades, the Boka Kotorska Bay with its extraordinary natural and socioeconomic values, distinct both in the historical and contemporary context, has been undergoing a dynamic economic development that implies a number of risks related to the protection of the marine ecosystem. The specific character of this Bay in terms of geological, hydrological, and hydrographic characteristics and limited communication with the open sea increases the pressure of its exposure to the anthropogenic pollution above all. This complicates research and monitoring of the state of environmental quality in the Bay and establishing of a connection between the degree of contamination of the bay on one side and the response and rehabilitation capacity of its ecosystem on the other side. In addition to regular monitoring based on internationally agreed standards, which our country has accepted as binding, there are, depending on the context, also other approaches in assessing the contamination of water, sediment, and biota. Data reported in this paper in terms of the heavy metals content in sediment indicate that the Bay of Tivat and the Bay of Kotor are more exposed to anthropogenic sources of contamination, which is related to more intensive economic activities in these two Bays. The indexes of environmental quality as a powerful tool for assessing the degree of contamination with heavy metals were applied for sediment and biota (P. oceanica and M. galloprovincialis) through the Enrichment Factor (EF), Metal Pollution Index (MPI), and Geo-accumulation Index (I geoE). They pointed out the seasonal and annual variability of assessment results, especially the assessment of the degree of contamination in biota, and indicated the Tivat Bay as the most contaminated. Also, available data and their comparison show that it is necessary to continuously monitor the degree of heavy metals contamination in sediment and biota in the long term, and to determine the trend of contamination the Boka Kotorska Bay in terms of heavy metal content in these environments.
Biomonitoring activities based on the analysis of all mentioned biomarkers and indexes have showed that the location Bijela is under the higher pressure from human activities than the location Kotor (industrial pollution load, shipbuilding, port activities, etc.) while the location Kotor is most affected by the waste water from households. This assertion is supported by the fact that the inhibition of the enzyme was higher in the location Bijela (caused by oxidative stress, pollution with organophosphate pesticide, carbamate pesticides and metals), and also that the maximum load of metals was in the location Bijela.
Active biomonitoring method has major potential in seawater monitoring of the Montenegrin coast; however, it should be further tested and elaborated. Furthermore, since the Town of Kotor in the Boka Kotorska Bay is an important destination for large touristic ships – cruisers – passive biomonitoring of sea water quality will significantly contribute to environmental protection and sustainable development of this part of the Adriatic Sea.
References
Kaushik A, Kansal A, Santosh et al (2009) Heavy metal contamination of river Yamuna, Haryana, India: assessment by metal enrichment factor of the sediment. J Hazard Mater 164:265–270
Stanković S, Cantaluppi C, Mandić S, Degetto S (2002) The environmental state of aquatic systems through sediments analysis: radiochemical approach. Stud Mar 23:57–64
Tanaskovski B, Petrovic M, Kljajic Z, Degetto S, Stankovic S (2014) Analysis of major, minor and trace elements in surface sediments by X-ray fluorescence spectrometry for assessment of possible contamination of Boka Kotorska Bay, Montenegro. Maced J Chem Chem Eng 33:139–150
Schettino T, Caricato R, Calisi A et al (2012) Biomarker approach in marine monitoring and assessment: new insights and perspectives. Open Environ Sci 6:20–27
UNEP/RAMOGE: Manual on the biomarkers recommended for the MED POL biomonitoring programme (1999) UNEP, Athens
OSPAR Commission (2008) Coordinated environmental monitoring programme – assessment manual for contaminants in sediment and biota, publication no. 379/2008, p 39
Law R, Hanke G, Angelidis M et al (2010) Marine strategy framework directive – task group 8 report contaminants and pollution effects. Scientific and Technical Research Series, vol 161. EUR 24335 EN – Joint Research Centre Scientific and Technical Reports. Office for Official 622 Publications of the European Communities, Luxembourg
Peijenenburg WJGM, Posthuma L, Eijsacheis HJP et al (1997) A conceptual frame work of implementation of bioavailability of metals for environmental management purposes. Ecotoxicol Environ Saf 37:163–172
Committee on Biological Markers of the National Research Council (1987) Biological markers in environmental health research. Environ Health Perspect 74:3–9
Ramsak A, Scancar J, Horvat M (2012) Evaluation of metallothioneins in blue mussels (Mytilus galloprovincialis) as a biomarker of mercury and cadmium exposure in the Slovenian waters (Gulf of Trieste): a long-term field study. Acta Adriat 53(1):71–86
Zhou Q, Zhang J, Fu J et al (2008) Biomonitoring: an appealing tool for assessment of metal pollution in the aquatic ecosystem. Anal Chim Acta 606:135–150
Stankovic S, Kalaba P, Stankovic RA (2014) Biota as toxic metals indicators. A review. Environ Chem Lett 12:63–84
Walker CH, Hopkin SP, Sibly RM et al (2006) Principles of ecotoxicology. CRC Presss, Taylor & Francis Group, Boca Raton, FL
Vincent C, Heinrich H, Edwards A et al (2002) Guidance on typology, reference conditions and classification systems for transitional and coastal waters. CIS Working Group 2.4 (COAST). Common Implementation Strategy of the Water Framework Directive, European Commision, p 119
Allan IJ, Vrana B, Greenwood R et al (2006) “Toolbox” for biological and chemical monitoring requirements for the European Union’s Water Framework Directive. Talanta 69:302–322
Mills GA, Greenwood R, Gonzalez C (2007) Environmental monitoring within the Water Framework Directive (WFD). Trends Anal Chem 26:450–453
Libes SM (1992) An introduction to marine biogeochemistry. Wiley, New York
Phillips DJH, Rainbow PS (1993) Biomonitoring of trace aquatic contaminants. Elsevier Applied Science, London
Balkis N, Çağatay MN (2001) Factors controlling metal distribution in the surface sediment in the Erdek Bay, Sea of Marmara, Turkey. Environ Int 27:1–13
Chapman PM (2007) Determining when contamination is pollution – weight of evidence determinations for sediments and effluents. Environ Int 33:492–501
Cenci RM, Basset A, Sena F et al (2002) Trace elements in sediment cores of Lake Alimini (Lecce Italy). Fresenius Environ Bull 11:681–685
Ansari TM, Marr L, Tariq N (2004) Heavy metals in marine pollution perspective-A: mini review. J Appl Sci 4:1–20
Papatheodorou G, Hotos G, Gerada M et al (2002) Heavy metal concentrations in sediments of Klisova lagoon (Southeast Mesalonghi-Aetolikon lagoon complex), W. Greece. Fresenius Environ Bull 11:951–956
Zabetoglou K, Voutsa D, Samara C (2002) Toxicity and heavy metal contamination of surficial sediment from the Bay of Thessaloniki (Northwestern Aegean Sea) Greece. Chemosphere 49:17–26
Ünlü S, Topcuoğlu S, Alpar B et al (2008) Heavy metal pollution in surface sediments and mussel samples in the Gulf of Gemlik. Environ Monit Assess 144:169–178
Ujević I, Kljaković-Gašpić Z, Bogner D (2010) Influence of suspended matter on cadmium accumulation in sediment from Kaštela Bay, Adriatic Sea, Croatia. Acta Adriat 51:79–88
Dugalić JG, Gajić AB (2005) Pedologija – praktikum. Agronomski fakultet, Čačak
UNEP/IOC/IAEA (1995). Manual for the geochemical analyses of marine sediments and suspended particulate matter. Reference Methods for Marine Pollution Studies No.63, UNEP
Tam NFY, Wong YS (2000) Spatial variation of heavy metals in surface sediments of Hong Kong mangrove swamps. Environ Pollut 10:195–205
Aloupi M, Angelidis MO, Gabriel A et al (2007) Marine monitoring along the eastern coastal area of the island of Lesvos, Greece during 2004 in the framework of MedPol III. J Global Nest 9:83–97
Joksimovic D (2015) Complex research of the ecosystem of coastal sea of Montenegro. University of Montenegro, Final study of the KOTOR project. Institute of Marine Biology, pp 1–145
Del Bianco F, Gasperini L, Giglio F et al (2014) Seafloor morphology of the Montenegro/N. Albania Continental Margin (Adriatic Sea—Central Mediterranean). Geomorphology 226:202–216
ICES (2009) Report of the Working Group on Marine Sediments in Relation to Pollution (WGMS). ICES CM 2009/MHC:02, p 59
Dolenec T, Faganeli J, Pirc S (1998) Major, minor and trace elements in surficial sediments from the open Adriatic Sea: a regional geochemical study. Geol Croatica 51:59–73. doi:10.4154/GC.1998.08
Tanaskovski B, Jovic M, Milicic L et al (2016) The geochemistry model of the surface sediment determined by using ED-XRF technique: a case study of the Boka Kotorska bay, Adriatic Sea. Environ Sci Pollut Res 1–13. doi: 10.1007/s11356-016-6353-6
Feng H, Han X, Zhang WG et al (2004) A preliminary study of heavy metal contamination in Yangtze River Intertidal Zone Due to urbanization. Mar Pollut Bull 49(11–12):910–915. doi:10.1016/j.marpolbul.2004.06.014
Cato I (1977) Recent sedimentological and geochemical conditions and pollution problems in two marine areas in Southwestern Sweden. Striae 6:1–150
Choi K, Kim S, Hong G et al (2012) Distribution of heavy metals in the sediments of South Korean harbours. Environ Geochem Health 34:71–82. doi:10.1007/s10653-011-9413-3
Sinex S, Helz G (1981) Regional geochemistry of trace elements in Chesapeak Bay sediments. Environ Geol 3(6):315–323. doi:10.1007/BF02473521
Turekian KK, Wedepohl KH (1961) Distribution of the elements in some major units of the earth’s crust. Geol Soc Am Bull 72:175–92
Pekey H (2006) The distribution and sources of heavy metals in Izmit Bay surface sediments affected by a polluted stream. Mar Pollut Bull 52:1197–1208
Stanković S, Jović M, Tanaskovski B et al (2015) Can the origin of some metals in the seagrass Posidonia oceanica be determined by the indexes of metals pollutions? Environ Sci Pollut Res 22(11):8253–8263. doi:10.1007/s11356-014-3953-x
Stanković S, Jović M, Mihajlović ML et al (2015) Metal pollution determined by pollution indices for sea grass P. oceanica and surface sediments. Arch Biol Sci 67(1):91–101
Ligero RA, Barrera M, Casas-Ruiz M et al (2002) Dating of marine sediments and time evolution of heavy metal concentrations in the Bay of Cadiz, Spain. Environ Pollut 118:97–108
Lee WY, Wang XW (2001) Metal accumulation in the green macroalgae Ulva fasciata: effect of nitrate, ammonium and phosphate. Sci Total Environ 278:11–22
Joksimović D, Stanković S (2012) Accumulation of trace metals in marine organisms of the southeastern Adriatic coast, Montenegro. J Serb Chem Soc 77:105–117
Usero J, Morillo J, Gracia I (2005) Heavy metal concentrations in molluscs from the Atlantic coast of southern Spain. Chemosphere 59:1175–1181
Rodríguez-Barroso MR, Benhamou Y, El Moumni B (2009) Evaluation of metal contamination in sediments from North of Morocco: geochemical and statistical approaches. Environ Monit Assess 159:169–181
Uluturhan E (2010) Heavy metal concentrations in surface sediments from two regions (Saros and Gökova Gulfs) of the Eastern Aegean Sea. Environ Monit Assess 165:657–684
Stanković S, Jović M, Petrović M et al (2012) Trace elements concentrations in the seagrass Posidonia oceanica and surface sediments samples at the southeastern Adriatic coast. Int Conf Mar Coast Ecos, Tirana, Albania 25–28:89
Jović M, Stanković S (2014) Determination of marine pollution by comparative analysis of metal pollution indices. Arch Biol Sci 66(3):1205–1215
Müller G (1981) Die Schwemetallbelastungder sedimente des Neckars und seiner Nebenflusse: eine Bestandsaufnahme. Chem Ztg 105:157–164
Martin JM, Meybeck M (1979) Elemental mass balance of materials carried by major world rivers. Mar Chem 7:173–206
Buccolieri A, Buccolieri G, Cardellicchio N et al (2006) Heavy metals in marine sediments of Taranto Gulf (Ionian Sea, Southern Italy). Mar Chem 99:227–235
Da Ros L, Moschino V, Macic V et al (2011) An ecotoxicological approach for the Boka Kotorska Bay (south-eastern Adriatic Sea): first evaluation of lysosomal responses and metallothionein induction in mussels. Mar Pollut Bull 63:326–333
Kristan U, Kanduc T, Osterc A et al (2014) Assessment of pollution level using Mytilus galloprovincialis as a bioindicator species: The case of the Gulf of Trieste. Mar Pollut Bull 89(1–2):455–63
Ramsak A, Tkalcic B, Kljajic Z (2010) Evaluation of biomarkers response at the SE Gulf of Trieste (Slovenia) and Boka Kotorska (Montenegro). CIESM - 39th Congress, Venezia, 10-14May 2010
Tsangaris C, Moschino V, Strogyloudi E et al (2016) Biochemical biomarker responses to pollution in selected sentinel organisms across the Eastern Mediterranean and the Black Sea. Environ Sci Pollut Res 23:1789–1804
Bolognesi C, Hayashi M (2011) Micronucleus assay in aquatic animals. Mutagenesis 26(1):205–213
Heberger K, Kolarevic S, Kracun-Kolarevic M et al (2014) Evaluation of single-cell gel electrophoresis data: Combination of variance analysis with sum of ranking differences. Mutat Res 771:15–22
Kholodkevich SV, Ivanov AV, Kurakin AS et al (2008) Real time biomonitoring of surface water toxicity level at water supply stations. Environ Bioindic 3(1):23–34
Bellafiore D, Guarnieri A, Grilli F et al (2011) Study of the hydrodynamical processes in the Boka Kotorska Bay with a finite element model. Dyn Atmos Oceans 52(1):298–321
Depledge MH, Aagaard A, Györkös P (1995) Assessment of trace metal toxicity using molecular, physiological and behavioural biomarkers. Mar Pollut Bull 31(1):19–27
Nicholson S, Lam PKS (2005) Pollution monitoring in Southeast Asia using biomarkers in the mytilid mussel Perna viridis (Mytilidae: Bivalvia). Environ Int 31(1):121–132
Martinović R, Kurakin AS, Kholodkevich SV et al (2013a) Preliminary results of sea water quality assessment based on physiological biomarkers in part of the Boka Kotorska Bay. Water Resour Manag 3(1):31–34
Fedotov VP, Kholodkevich SV, Strochilo AG (2000) Study of contractile activity of the crayfish heart with the aid of a new non-invasive technique. J Evol Biochem Physiol 36(3):288–293
Allen J (2007) Photoplethysmography and its application in clinical physiological measurement. Physiol Meas 28(3):R1
Gaines SE (1991) Polluter-pays principle: from economic equity to environmental ethos. Tex Int'l LJ 26:463
Martinović R, Gacic Z, Kljajic Z (2013) Heart rate changes of the Mediterranean mussel (Mytilus galloprovincialis L.). Stud Mar 26(1):111–118
Etkin DS (2011) Spill occurrences: a world overview. In: Fingas M (ed) Oil spill science and technology: prevention, response and clean up, 1st edn. Gulf Professional Publishing, Burlington, pp 7–48
DeNardis NI, Svetličić V, Pletikapić G et al (2014) Presence of dispersed diesel fuel in the water column of the Boka Kotorska Bay: a case study. Stud Mar 27(1):43–64
Martinovic R, Gacic Z, Kljajic Z (2015a) The influence of oil, dispersed oil and the oil dispersant SD-25 on the heart rate of the mediterranean mussel (M.galloprvincialis L.). In: Stylios C et al (eds) Sustainable development of sea-corridors and coastal waters. Springer, Switzerland, pp 21–27. doi:10.1007/978-3-319-11385-2_2
Martinović R, Kolarević S, Kračun-Kolarević M et al (2015b) Genotoxic potential and heart rate disorders in the Mediterranean mussel Mytilus galloprovincialis exposed to Superdispersant-25 and dispersed diesel oil. Mar Environ Res 108:83–90
Acknowledgments
We would like to thank our colleague Branka Pestorić, PhD and Aleksandar Jovičić, who provided maps.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Joksimović, D. et al. (2016). Metal Pollution and Ecotoxicology of the Boka Kotorska Bay. In: Joksimović, A., Djurović, M., Semenov, A., Zonn, I., Kostianoy, A. (eds) The Boka Kotorska Bay Environment . The Handbook of Environmental Chemistry, vol 54. Springer, Cham. https://doi.org/10.1007/698_2016_40
Download citation
DOI: https://doi.org/10.1007/698_2016_40
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-51613-4
Online ISBN: 978-3-319-51614-1
eBook Packages: Earth and Environmental ScienceEarth and Environmental Science (R0)