Abstract
Description of the basic characteristics of the zoobenthos assemblages at the seafloor of the Boka Kotorska Bay was created by compiling available data from the scientific and gray literature, and they are a result of research on this area during the last 55 years. All data on the benthic fauna of the Boka Kotorska Bay, up until the middle of the last century, are very rare and can be found in publications resulting from the study of the Adriatic Sea by international scientists. With the establishment of the Institute for Marine Biology in Kotor, more intensive research of marine biodiversity of this particular area has started.
Available data indicate the presence of a large number of species of seabed fauna which has adapted to specific environmental conditions. Among the identified taxonomy, there are many species protected by national and international regulations. Most of these species are builders of coralligenous biocoenosis that makes this space unicum. These communities are particularly distributed in the inner part of the Boka Kotorska Bay (the Bay of Kotor and Risan) where they are distributed to the relatively shallow depths (12–30 m).
Since the area of the Boka Kotorska Bay is abundant with underground springs, many species have adapted to life in the brackish environment with reduced salinity. From the species that inhabit the sea bottom of this area, there is a large number of endemic species of the Mediterranean, especially from the group of molluscs and echinoderms.
The area of the Boka Kotorska Bay as well as whole Mediterranean zone is under strong human influence, so the presence of invasive species has been reported, some of which become domesticated. This number is certainly not final and has the tendency to increase.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
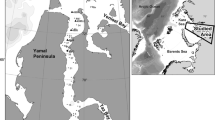
The area of the Boka Kotorska Bay is a complex semi-enclosed basin, consisting of four smaller bays, which conditionally make up the inner part of the Bay (Bay of Kotor and the Bay of Risan) and outer part (Bay of Tivat and the Bay of Herceg Novi) (Fig. 1). Since communication with the open sea is limited, it is noticeable that it is significantly different from the open part of the coast, when it comes to climate, geomorphological, and physical-chemical characteristics. Boka Kotorska Bay enters deep into the mainland, so the individual Bays differ with one another according to values of abiotic factors and the characteristics of the seabed. The bottom of the Bay is mostly covered by thick layers of fine mud. In the Bay of Kotor and Risan, as well as in the Verige Channel, the seabed is covered with clay, with sandy clay directly in front of Risan [1]. In the Bay of Tivat, clay is predominant, and to a lesser extent clay-loamy sand and clay loam. In the Bay of Herceg Novi, aside from the clay, the seafloor is covered with clay loam, clay sand, and sand.
Relief of the sea bottom is very complex and is not always symmetrical. Depth in Bays increases toward the center, except in the Bay of Kotor, where the maximum depth is near the northern coast of the Bay (Perast). The average depth of the entire Bay is 27.6 m and maximum one – 64 m (Bay of Kotor) [2]. Boka Kotorska Bay is surrounded by mountain ranges, which often cause significant cooling in winter months and the appearance of ice on the sea surface. This is an area of intense rainfall, which causes large amounts of fresh water to flow into the Bay in the period from November to April, particularly in its inner part. Such a combination of environmental conditions has caused for the development of specific benthic assemblages [3].
Historical data said that relatively small area of the Boka Kotorska Bay is characterized by high biodiversity of sessile or slow motion organisms [4]. More intensive studies of the Boka Kotorska Bay fauna began in the mid-1970s of the twentieth century, with the establishment of the Institute for Marine Biology in Kotor. Data on the zoobenthos diversity of this area can be found in the papers of the world's scientists as part of the benthic fauna of the Adriatic Sea [5–8]. Depending on the objective of the research project, as well as the available experts, we could say that during the last century, the area of the Bay was very well examined for macromolluscs [9], especially group of cephalopods [10, 11], than polychaetes [12] and anthozoans [13]. The study of echinoderms started during the 1980s [14], just to be intensified during the last 10 years [15]. Data on other zoobenthos organisms that are contained in the papers deal with research of the present benthic biocoenosis [3, 16, 17].
In this chapter, we describe zoobenthos species composition and present benthic assemblages in the area of the Boka Kotorska Bay, based on literature data during the last 55 years, zoobenthos species distribution in relation to the four bays, and checklist of invasive species for the given area.
2 Benthic Biocoenosis in the Boka Kotorska Bay
As it was already pointed out that the Boka Kotorska Bay was characterized by specific values of physical-chemical parameters of the environment, the presence of benthic biocoenosis was specific in relation to the open coast. Occurrence and depth of spread of benthic communities are what makes this area specific. Analysis of the benthic organisms in the area of the Boka Kotorska Bay showed the presence of the biocoenosis of coastal terrigenous muds and elements of other biocoenosis on movable and solid substrate [3]. This biocoenosis occupies more than 87% of the sea bottom in the area of the Bay of Kotor and Risan [17]. Biocoenosis of coastal terrigenous muds is developed along the entire eastern coast of the southern Adriatic, especially in the areas of quiet and weakened bottom currents [18]. In Boka Kotorska Bay, this biocoenosis occupies the largest, mostly central part of the Bay and is only modified in areas where an inflow of fresh water exists (springs, streams, and small rivers which flow into the sea) [18, 19]. Different groups of ascidians are characteristic for this biocoenosis: Diazona violacea, Ascidia virginea, Ascidia mentula, Phallusia mammillata, and others. This biocoenosis is also characterized by soft coral Alcyonium adriaticum; cephalopods Sepia officinalis, Sepia elegans, Sepia orbignyana, Loligo vulgaris, Eledone moschata, Eledone cirrhosa, Alloteuthis media, Octopus vulgaris, Sepiola rondeleti, and Sepieta oweniana; and sea cucumber Eostichopus regalis [18, 19].
The biocoenosis of coastal detritic bottom was recorded at the border between infralittoral and circalittoral steps. Nature of detritic elements is very different and depends on the composition of the shore or from a nearby seabed and surrounding biocoenosis; these are fragments of rock, shards of shells and other skeletal elements, parts of bryozoans, calcareous algae, etc. Within this biocoenosis, there are representatives of sponges Bubaris vermiculata; numerous Polychaeta; snails Turritella tricarinata f. communis and Turritella triplicata; shells Pteria hirundo, Pecten jacobaeus, Pandora pinna, Acanthocardia deshayesii, Moerella donacina, Venus casina, etc.; and echinoderms Labidoplax digitata, Leptopentacta elongata, L. tergestina, Eostichopus regalis, Anseropoda placenta, Psammechinus microtuberculatus, etc. The rest of the sea bottom belongs to the biocoenosis of muddy sands [3].
Coralligenous biocoenoses are the best developed in the area of the Bay of Kotor and Risan on a solid substrate in circalittoral layer, in more or less darken conditions, at the shaded area below the underwater meadows of sea grass, as well as in significantly shallower parts of the seabed [17]. Facies Savalia savaglia and facies Leptogorgia sarmentosa can be distinguished within this biocoenosis. The dominant ones are incrusted algae, corals, and species from the group of Bryozoa, Polychaeta, and Echinodermata. It is the most important biodiversity area of the Bay of Kotor and Risan. On the movable sandy-muddy infralittoral substrate, there have developed the biocoenosis of seagrass meadows (Posidonia, Zostera, and Cymodocea). The leaves of Posidonia are often populated by various species of Bryozoa, Hydrozoa, Polychaeta, crabs, and snails. Inside of this biocoenosis as vary important live component are echinoderms (species from genus Holothuria, Echinaster sepositus, Paracentrotus lividus, Sphaerechinus granularis). This biocoenosis is suitable for fish eggs; there are cephalopods and other animals, because in the meadows, the young are protected from predators [3].
Biocoenoses of photophilic algae develop on a solid surface in the upper infralittoral zone, where the light penetration is the strongest and where the variation of temperature and salinity is the most prominent in few-meters-wide zone. This biocoenosis occurs in the form of several facies, but the characteristic species of animals are crabs Acanthonyx lunulatus and Clibanarius erythropus; molluscs Patella pellucida and Cerithium vulgatum; echinoderms Paracentrotus lividus, Arbacia lixula, and Echinaster sepositus; and others [20]. Big haul of fish often swim above algae.
Biocoenoses of the muddy bottom at the Bay are characterized by a huge number of irregular sea urchin Brissopsis lyrifera, so it can be considered as a particular form of the biocoenosis of coastal terrigenous muds. At the same time, coral Veretillum cynomorium is part of this community, otherwise rare in the Adriatic Sea [16].
Recent studies [21] confirm the benthic communities in the infralittoral part of the Bay of Kotor and Risan as unique, due to large presence of communities on moving substrate (soft substrate) and the presence of coralligenous biocoenosis at Dražin Vrt at a depth of 12–30 m.
Area of the Bay of Kotor and Risan is characterized by a very steep rocky coast, and movable bottom has been very close to the coastline, at a depth of 15–25 m, and covers a considerable part of the Bay; below 30–40 m of depth, the seabed has a slight slope and slightly deeper areas that are characterized by the presence of depressions [17].
3 The Diversity of Zoobenthos Assemblages
The inflow of large amounts of fresh water, the presence of underground springs, specific combination of temperature, salinity, and amount of light had a decisive role in the diversity and distribution of benthic fauna. Collected data show the presence of all zoobenthos groups [4] at the seafloor of the Boka Kotorska Bay.
Generally, the inner part of the Boka Kotorska Bay is much better explored than the outer part. Recent studies [17] show that the inner part of the Bay, i.e., the Bay of Kotor and Risan is characterized by a wealth of animal life. This primarily refers to a solid substrate with 124 species recorded, while soft substrates are inhabited by 77 species. Mollusca is the most abundant phylum with a total of 46 taxa, followed by Annelida (38 taxa) and Porifera (37 taxa). Porifera, Bryozoa, and Tunicata are exclusively found on the hard bottom, while Annelida dominates on the soft bottom. Comparing the obtained data with historical records relating to the entire Boka Kotorska Bay (Appendix 1), it is notable that the number of recorded molluscs is significantly lower than in the entire Boka Kotorska Bay, while the number of species of Porifera, Cnidaria, and Annelida is higher than the total number of species recorded during previous research in the area of the entire Boka Kotorska Bay (Fig. 2). This points to the fact that the older studies included only certain groups of organisms and only certain areas so that the least data were collected from two outward parts of the Bay (Bay of Tivat and the Bay of Herceg Novi).
More recently results of the study performed in the inner part of the Boka Kotorska Bay confirmed that the benthic assemblages of the infralittoral represent a unicum, because of the large percentage of soft bottom assemblages and the presence of coralligenous assemblages [21]. In particular, at Dražin Vrt, a coralligenous assemblage was found between 12 and 30 m of depth. Large colonies of Cladocora caespitosa reefs were present and were associated with a rich assemblage of large-sized sponges and cnidarians, notably massive colonies of the false black coral, Savalia savaglia, gorgonian leptogorgia cfr. sarmentosa, and yellow cluster anemone Parazoanthus axinellae [21].
Seabed of the Bay of Tivat is inhabited by different groups of animals, such as Porifera, Cnidaria, Annelida, Crustacea, Mollusca, and Echinodermata [22]. As common species were recorded Axinella brondstedi, Aplysina aerophoba, and Suberites domuncula from phylum Porifera. The most present coral is Cladocora caespitosa. From the group of worms, Spirobranchus triqueter dominated on the soft bottom, while Protula sp. populated on the hard bottom, presented by a large stone or a solid waste. The seafloor of the Tivat Bay is populated by echinoderms Brissopsis lyrifera, Amphiura chiajei, Marthasterias glacialis, Ophiothrix fragilis, Holothuria tubulosa, Holothuria polii, Holothuria mammata, Mesothuria intestinalis, Antedon mediterranea, Echinaster sepositus, Ocnus planci, and Sphaerechinus granularis [23]. Economically important species of marine organisms are presented by cephalopods Sepia officinalis and Loligo vulgaris and then molluscs Nucula nucleus, Mytilus galloprovincialis, Lithophaga lithophaga, Luria lurida, Venus verrucosa, Tonna galea, Pecten jacobaeus, and Ostrea edulis [24].
Within the Tivat aquatorium, there is an especially interesting area of the former Naval-Repair Institute Arsenal. Due to specific environmental conditions, it is considered as a separate entity. Benthic organisms in the aquatorium are under great influence of the grit deposited on the bottom as well as waste water from municipal sewage. Survey conducted in 2007 [22] showed the presence of 38 invertebrate species attached on the walls of the piers (Appendix 1). Dominant species were Mytilus galloprovincialis, Spirobranchus triqueter, Phallusia mammillata, Schizobrachiella sanguinea, Sabella spallanzanii, Amphibalanus eburneus, and Protula tubularia. Fauna of the soft bottom (epi- and endo-biocoenosis of soft sediments) is significantly poor compared to the communities at the piers. Majority of these species are organisms of solid substrates that have found a favorable habitat in various solid wastes on the seafloor. Generally, the substrate is muddy covered with a layer of detritus, while black anaerobic mud is observed in some sites, but it is mostly the case of biocoenosis of detritic and terrigenous mud. As specific species are recorded Antedon mediterranea, Myxicola infundibulum, Pecten jacobaeus, Ostrea edulis, Upogebia pusilla, Aplysina aerophoba, Spirobranchus triqueter, and Protula sp. which were frequent on boulders and other types of solid surfaces, which actually represents solid waste (car, tires, etc.) [22]. More recently research conducted in the same area identified 53 species inhabiting the piers (Appendix 1). Classified by groups, the following were identified: 5 species of Porifera, 5 species of Cnidaria, 20 species of Mollusca, 5 species of Annelida, 3 species of Arthropoda, 4 species of Bryozoa, 4 species of Echinodermata, and 7 species of Tunicata. The most numerous were molluscs Mytilus galloprovincialis and Ostrea edulis and tunicates Clavelina lepadiformis, Phallusia fumigata, and Phallusia mammillata [25].
Analyses of benthic fauna in the Bay of Herceg Novi show the presence of all major groups of macrozoobenthos [3]. The largest part of recorded species belongs to the phylum Mollusca. In this Bay, just as in the entire BKB number of settlements Pinna nobilis is increased. Recorded species are relatively small in size, which points to the fact that settlements are relatively young. Presence of Cladocora caespitosa and Spongia officinalis is very significant. Some sites are characterized by abundant populations of this species, as well as Dysidea avara. Also genus Ircinia and Chondrilla nucula are very common. On the locality in the Bay of Herceg Novi, the snail Tylodina perversa has been registered.
During the last decade, special attention is paid to the study of echinoderms [23]. In the area of the Boka Kotorska Bay, the presence of 42 species was identified [15]. Among the identified species, there are six Mediterranean endemics (Antedon mediterranea, Holothuria (Holothuria) mammata, Astropecten spinulosus, Leptopentacta tergestina, Ocnus syracusanus, Astropecten irregularis pentacanthus) and two Mediterranean subendemics (Schizaster canaliferus, Echinocardium fenauxi).
The study of the distribution of species from phylum Porifera shows presence of genus Axinella (A. damicornis, A. verrucosa and A. cannabina) in the BKB [26].
Analysis of the zoobenthos species composition on the basis of the total number recorded species, with the exception of phylum Mollusca, showed that the Bay of Kotor is the richest in species from the group Annelida, Crustacea, Echinodermata, and Tunicata, followed by the Bay of Risan (Fig. 3). In the area of the Bay of Herceg Novi, echinoderms stood out as the most frequent group. These results do not necessarily represent the actual situation, but point to the fact that inner bays are better explored, compared to the Bay of Tivat and the Bay of Herceg Novi.
In terms of distribution of molluscs through bays, we can conclude that most species collected from the Bay of Herceg Novi (104), the Bay of Kotor (85), and the Bay of Risan (84) are almost equal, while the least species have been recorded in the area of the Bay of Risan (72) (Fig. 4, Appendix 2).
4 Non-indigenous (Alien or Nonnative) Species
The biggest threat to marine ecosystems comes from four main negative anthropogenic factors, such as pollution from land (and sea), overfishing, destruction of habitat (especially coastal ones), and introduction of alien species [27]. Introduction of species in a habitat outside their natural distribution (allochthonous species) is a growing problem, primarily because it is almost impossible to predict the behavior of introduced species and its impact on autochthonous species and communities.
Introduction of new species is difficult to prevent especially when you take into account the attractiveness of Boka Kotorska Bay as a tourist destination, which is being visited by a large number of cruise ships and yachts during the year, which are an important vector in the transmission of introduced species [28]. In order to make a good assessment of the impact and risk of the introduced species to a particular ecosystem, it is necessary to have basic knowledge of the origin of the species, its biology and ecology, as well as the manner and place of entry. Timely detection of these species is one of the prerequisites of a successful response in order to protect the environment.
Conducted study showed the presence of five alien species for the area (Table 1). For the area of Boka Kotorska Bay, the presence of introduced species dates back to the mid-1980s of the last century, when the Crassostrea gigas (oyster) was intentionally introduced for mariculture purposes [29]. Except in this area, it has been introduced for the same reasons in many other parts of the Mediterranean Sea, where it is now fully domesticated.
Nonnative species snail Bursatella leachii was recorded at several locations in the inner part of the Bay, so it can be expected that this type becomes domesticated and significantly more numerous than it is now known [30].
For other introduced species listed in Table 1, single appearance is known, and they are probably not present in huge numbers. However, experience from the region indicates to rapid growth of populations of some of the introduced species [31], meaning with more intensive research, it would be possible to register a more number of individuals. This applies particularly to the blue crab (Callinectes sapidus), which was noted in the area of Port Milena in 2006, Jaz in 2009, Oblatno in 2011 (out of the Boka Kotorska Bay), and in 2013 in the Bay of Tivat [32].
Species Farfantepenaeus aztecus was recorded for the first time in of the Bay of Tivat [33].
5 Threats to Zoobenthic Diversity and Protection
Boka Kotorska Bay has limited communication with the open sea and therefore is a very sensitive system. The Bay is surrounded by towns and cities, which represents a major threat to sea biodiversity, direct or indirect.
Even during the 1970s of the last century, it was noted that the declining number of Mollusca, especially in the coastal zone of Boka Kotorska Bay, may be due to the large influx of oil and its products into the sea, as well as a variety of toxic chemical compounds from various warehouses, warships, some factories, and increasing amounts of waste water from urban sewage flowing into the sea [9].
Tendency of the Bay to develop into a well-known and popular tourist destination leads to the great anthropogenic pressure on the coastal strip. The fact that within the Bay of Kotor there is a port for mega cruise ships, in the Bay of Tivat a marina for mega yachts, and a large number of small marinas scattered around the Boka Kotorska Bay shows the presence of the impact of nautical tourism on marine biodiversity. Analyses of seabed in the Bay of Kotor and Risan showed clear traces of anchoring on the seafloor and disturbance of benthic biocoenosis in these places [17]. Based on the number and composition of phytoplankton, we can speak of eutrophication of certain areas [34–36].
The main human activities and threats to the coastal and marine environment are fishing [21]. We should particularly note the dangers coming from overfishing; illegal fishing, which refers to the collection of protected species; and the use of illegal means. Another possible threat to marine environment in the area is the dumping of soil from road construction or improvement. The last 20 years have been characterized by a building boom, especially in the narrow coastal zone. During the construction of a large number of apartments and catering facilities, reckless builders discharge large amounts of waste material directly into the sea, leading to a direct threat to communities of photophilic algae that inhabit the shallow narrow zone. The obtained data showed that coralligenous communities in the Bay of Kotor and Risan are located on the small depths which poses a great danger to their preservation. Considering that these communities are one of the main characteristics of the Boka Kotorska Bay, a major threat is global warming as well, human pressure like pollution and coastal development, as well as the presence of introduced species [37].
The invasive red alga Womersleyella setacea could be another potential threat to the Boka Kotorska Bay’s biodiversity. Studies confirm that the presence of this invasive species changes the assemblage structure and reduces species richness in coralligenous communities, particularly decreasing the diversity and abundance of other turf algae [38].
In order to reduce the negative impact on marine biodiversity, it should be approached with the following:
-
Limit and control construction activities and control tourism development.
-
Develop regulations addressed to protect the sensitive species and habitats identified in the Bay.
-
Avoid uncontrolled sewage.
-
Regulate and control soil dumping in the Bay from road construction and waste materials from construction sites.
-
Regulate recreational fishery and the use of fishing gear.
6 Conclusion
Area of the Boka Kotorska Bay is a complex semi-enclosed basin with limited communication with the open sea. Large indentation of the Bay into the mainland, the influx of large amounts of fresh water, and the presence of underwater springs make this aquatorium a specific habitat for plant and animal marine life. Within these specific environmental conditions, benthic biocoenosis had developed, and their composition and distribution make this area particular. The presence of biocoenosis of photophilic algae is reduced to a narrow upper part of the infralittoral and is quite reduced in areas where the coast of the Bay descends steeply into the depths. Biocoenosis of marine phanerogams has been in retreat in the last years, compared to historical data, as a result of anthropogenic impact. The largest part of the Bay, i.e., its central part, is occupied by the biocoenosis of coastal terrigenous mud. Coralligenous biocoenosis stands out as extremely important in terms of biodiversity and vulnerability. They are characterized by the presence of a large number of species, mostly sponges and corals. Their presence at a relatively shallow depth (12–30 m) in the Bay of Kotor and Risan represents unicum and imposes the obligation of their protection.
Preview of the state of zoobenthos species composition in the area of the Boka Kotorska Bay included the compilation of all available literature data for the last 55 years, both from the scientific and gray literature. Obtained results show that certain areas examined in more detail in relation to the rest of the Bay. This primarily refers to the inner part, i.e., the Bay of Kotor and Risan, which was much more the subject of the study, both of plant and animal diversity. Another conclusion to be reached by processing available data is that certain groups of animals have been much better studied, such as molluscs and echinoderms, which may be due to the presence of certain scientists at the Institute of Marine Biology.
Available data on zoobenthos species populated in the territory of the Boka Kotorska Bay show the richness of animal life that occupies the specific and heterogeneous habitats in this relatively small area. Among the identified species, we find a large number of protected species (Pinna nobilis, Lithophaga lithophaga, Savalia savaglia, Aplysina aerophoba, Axinella cannabina, Spongia officinalis, Maya squinado) and Mediterranean endemics (Antedon mediterranea, Holothuria (Holothuria) mammata, Astropecten spinulosus, Leptopentacta tergestina, Ocnus syracusanus, Astropecten irregularis pentacanthus, Schizaster canaliferus, Echinocardium fenauxi), species that have adapted to life in brackish water and low salinity.
The presence of introduced species indicates that this area was not spared from the trend of development of modern society. The presence of five species has been recorded so far (Bursatella leachii, Aplysia dactylomela, Crassostrea gigas, Callinectes sapidus, and Farfantepenaeus aztecus), some of which are already common. This number is certainly not definitive, and each subsequent research may indicate the presence of some new alien species.
References
Lepetić V (1965) Sastav i sezonska dinamika ihtiobentosa i jestivih avertebrata u Bokokotorskom zalivu i mogućnosti njihove eksploatacije (Composition and seasonal dynamics of ihtiobentosa and edible avertebrata in the Bay of Kotor and the possibility of their exploitation). Studia Marina 1:1–164
Stjepčević J (1974) Ekologija dagnje (Mytilus galloprovincialis LAMK.) i kamenice (Ostrea edulis L.) u gajilištima Bokokotoroskog zaliva (Ecology of mussels (Mytilus galloprovincialis LAMK.) and oysters (Ostrea edulis L.) in farms of Boka Kotorska Bay). Studia Marina 7:5–164
Karaman G, Gamulin-Brida H (1970) Contribution aux recherches des bicenoses benthiques du Golfe de Boka Kotorska. Studia Marina 4:3–42
Petović S, Bataković M (2014) Marine biodiversity of Boka Kotorska bay pilot project on testing Ecosystem Approach (EcAp) application in Boka Kotorska bay: Executive summary. Ed. RAC/SPA, Tunis. 46 pp + Appendices
Olivi G (1792) Zoologia Adriatica, ossia catalogo regionato degli animali del Golfo e delle lagune di Venezia. Bassano
Heller C (1863) Untersuchungen uber die Litoralfauna des Adriatischen Meeres. Sber K Akad Wiss Wien 46:415–448
Heller C (1868) Zoophyten und Echinodermen des Adriatischen Meeres. Druck Von Carl Ueberreuter, Wien, pp 47–86
Kolosváry G v (1938) Echinodermata iz Boke Kotorske (Echinoderms from the Boka Kotorska Bay). Godišnjak Oceanografskog Instituta Kraljevine Jugoslavije 1:121–125
Stjepčević J (1967) Biologija i tehnološki proces uzgoja jadranske kamenice (Ostrea edulis L.) (Biology and technological process of growing oysters (Ostrea edulis L.). Poljoprivreda i šumarstvo XIII 4:33–48
Stjepčević J (1970) Kvalitativno-kvantitativni sastav i distribucija Cephalopoda Bokokotorskog zaliva u jednogodišnjem sezonskom aspektu (Qualitative and quantitative composition and distribution of Cephalopoda in the Bay of Kotor in the one- seasonal aspect). Studia Marina 4(1):43–71
Mandić S (1984) Cephalopoda južnog Jadrana (Cephalopoda of the south Adriatic). Studia Marina 15–16:3–77
Stječević J, Parenzan P, Mandić S, Stjepčević B (1984) Kvalitativno-kvantitativna istraživanja Polychaeta unutrašnjeg dijela Bokokotorskog zaliva (Qualitative and quantitative research of Polychaeta in inner part of the Bay of Kotor). Studia Marina 15–16:79–95
Stjepčević J, Gašić M, Kljajić Z, Stjepčević B, Dogović M, Muller EGW, Zahn R (1986) Prilog proučavanju faune Anthosoa unutrašnjeg dijela Bokokotorskog zaliva (Contribution to the study of fauna Anthosoa in inner part of the Bay of Kotor). Studia Marina 17–18:21–37
Milojević S (1979) Biocenotički pregled faune Echinodermata u Bokokotorskom zalivu (Biocenotic review of fauna of Echinodermata in the Kotor Bay). Paper presented at the 2nd Congress of ecologists of Yugoslavia, Zagreb, pp 1859–1868
Petović S (2011) Contribution to the knowledge of echinodermata of the Boka Kotorska Bay. Studia Marina 25(1):137–158
Gamulin-Brida H (1983) Crnogorsko primorje – specifični dio Jadrana s gledišta bentoskih biocenoza i njihove zaštite (Coast of Montenegro - a specific part of the Adriatic Sea from the viewpoint of the benthic biocenosis and their protection. Studia Marina 13–14:205–214
UNEP - MAP - RAC/SPA (2013) Ecological study to strengthen the creation processes and the management of marine protected areas in Montenegro-Report No. 12508420857
Gamulin-Brida H (1974) Biocoenoses benthiques de la mer Adriatique – Bentoske biocenoze Jadranskog mora. Acta Adriat 15(9):1–102
Gamulin-Brida H (1962) Biocenoze dubljeg litorala u kanalima srednjeg Jadrana = Biocoenoses du littoral plus profond (Circalittoral) dans les canaux de l'Adriatique moyenne. Acta Adriat 9:1–196
Pérès JM, Gamulin-Brida H (1973) Biološka oceanografija (Biological oceanography). Školska knjiga, Zagreb
Badalamenti F, Treviño-Otón J (2012) Development of Marine and Coastal Protected Areas (MPAs) in the Republic of Montenegro. Ed. RAC-SPA
Mandić S, Joksimović A, Joksimović D, Mačić V, Kašćelan S, Đurović M (2007) Report 1. Estimate of environmental impact - marine ecology. CUW-UK
Kašćelan S, Mandić S, Radović I, Krpo-Ćetković J (2009) An annotated checklist of Echinodermata of Montenegro (the south Adriatic Sea). Zootaxa 2275:21–40
Local Biodiversity Action Plan for the Municipality Tivat for the period from 2013 to 2018. http://www.opstinativat.com/cg/images/stories/prostornourbanistickiplan/lbap%20opstina%20tivat1.pdf
University of Montenegro-Institute of Marine biology (2016) Porto Montenegro-monitoring report. Kotor
Mačić V, Petović S, Backović S (2015) Contribution to the knowledge of protected Axinella (Porifera, Demospongiae) species along the Montenegrin coast. Studia Marina 28(1):9–20
Galil BS (2007) Loss or gain? Invasive aliens and biodiversity in the Mediterranean Sea. Mar Pollut Bull 55(7–9):314–322
Statistical yearbook of Montenegro 2015. http://www.monstat.org/userfiles/file/publikacije/godisnjak%202015/GODISNJAK%202015-%20REDAKCIJA,%20RADA%20SISEVIC,%2028.%20DEC.pdf
Stjepčević J, Mandić S, Dragović R (1977) Mogućnost industrijskog uzgoja jestivih školjkaša u Bokokotorskom zalivu i uvonenja novih vrsta u proces uzogja (Possibility of industrial cultivation of edible shellfish in the Bay of Kotor and the introduction of new species in the process). Ichthyologia 9(1):107–120
Mačić V (2013) Contribution to the knowledge of Bursatella leachii (de Blainville, 1817) distribution and reproduction in the Boka Kotorska Bay (Montenegro). Studia Marina 26(1):119–128
Zenetos A, Gofas S, Verlaque M, Çinar ME, García Raso E, Bianchi CN, Morri C, Azzurro E et al (2010) Alien species in the Mediterranean Sea by 2010. A contribution to the application of European Union’s Marine Strategy Framework Directive (MSFD). Part I. Spatial distribution. Mediterr Mar Sci 11(2):381–493
Marković O, Đurović M (2014) First documented record of the American blue crab, Callinectes sapidus Rathbun, 1896 in the Boka Kotorska Bay, Southern Adriatic Sea, Montenegro p. 208–209. In: Kapiris et al., 2014 New Mediterranean marine biodiversity records. Mediterr Mar Sci 15(1):198–212. doi:10.12681/mms.737
Marković O, Gokoglu M, Petović S, Mandić M (2014) First record of the Northern brown shrimp, Farfantepenaeus aztecus (Ives, 1891) (Crustacea: Decapoda: Penaeidae) in the South Adriatic Sea, Montenegro. Mediterr Mar Sci 15(1):165–167
Krivokapic S, Stankovic Z, Vuksanovic N (2009) Seasonal variations of phytoplankton biomass and environmental conditions in the inner Boka Kotorska Bay (eastern Adriatic Sea). Acta Bot Croat 68(1):45–55
Krivokapić S, Pestorić B, Drakulović D (2010) Temporal variability of nutrients and chlorophyll a in Boka Kotorska Bay, Eastern Adriatic Sea. BALWOIS
Drakulović D, Vuksanović N, Joksimović D (2010) Dynamics of phytoplankton in Boka Kotorska Bay. Studia Marina 25(1):1–20
Kruzic P, Benkovic L (2008) Bioconstructional features of the coral Cladocora caespitosa (Anthozoa, Scleractinia) in the Adriatic Sea (Croatia). Mar Ecol 29:125–139
Piazzi L, Balata D, Cinelli F (2007) Invasions of alien macroalgae in Mediterranean coralligenous assemblages. Cryptogam Algol 28(3):289–301
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Appendices
Appendix 1: List of Zoobenthos Species Diversity from the Boka Kotorska Bay According to Literature Data
Species | Herceg Novi Bay | Tivat Bay | Risan Bay | Kotor Bay |
|---|---|---|---|---|
Porifera | ||||
Acanthella acuta | x | x | x | |
Agelas oroides | x | x | ||
Anchinoe fictitious | x | x | ||
Anchinoe tenacior | x | x | ||
Aplysina aerophoba cfr. | x | x | x | x |
Aplysina cavernicola | x | x | ||
Axinella cannabina | x | x | ||
Axinella damicornis | x | x | ||
Axinella verrucosa | x | x | ||
Cacospongia scalaris | x | |||
Calyx niceaensis | x | x | ||
Chondrilla nucula | x | x | x | |
Chondrosia reniformis | x | x | ||
Clathrina cfr. cerebrum | x | x | ||
Clathrina coriacea | x | x | ||
Cliona celata | x | x | ||
Cliona schmidti | x | x | ||
Cliona sp. | x | x | ||
Cliona viridis | x | x | ||
Crambe crambe | x | x | x | |
Dictyonella incisa | x | x | ||
Dysidea fragilis | x | x | ||
Dysidea avara | x | x | x | x |
Geodia cydonium | x | x | ||
Haliclona cratera | x | |||
Haliclona fulva | x | x | ||
Haliclona mucosa | x | x | ||
Hexadella racovitzai | x | x | ||
Ircinia oros | x | x | ||
Ircinia variabilis | x | x | x | |
Mycale massa | x | x | ||
Petrosia ficiformis | x | x | ||
Poecilosclerida spp. | x | x | ||
Raspailia viminalis | x | x | ||
Sarcotragus cfr. foetidus | x | x | ||
Sarcotragus spinosulus | x | x | ||
Spirastrella cunctatrix | x | x | x | x |
Spongia officinalis | x | x | x | |
Suberites carnosus | x | |||
Suberites domuncula | x | x | x | |
Suberitidae n.i. | x | x | ||
Tethya aurantium | x | x | ||
Tethya citrina | x | x | ||
Cnidaria | ||||
Actinia equina | x | x | ||
Aiptasia mutabilis | x | x | x | |
Alcyonium brioniense | x | x | ||
Alcyonium coralloides | x | x | ||
Alcyonium palmatum | x | x | x | x |
Anemonia sulcata | x | x | ||
Balanophyllia europaea | x | x | x | |
Balanophyllia italica | x | x | ||
Bugula aquilirostris | x | |||
Calliactis parasitica | x | x | ||
Caryophyllia cfr. inornata | x | x | ||
Caryophyllia cfr. smithii | x | x | ||
Caryophyllia clavus | x | x | ||
Caryophyllia spp. | x | x | ||
Cerianthus membranaceus | x | x | ||
Cladocora caespitosa | x | x | x | x |
Condylactis aurantiaca | x | x | x | |
Corallium rubrum | x | |||
Epizoanthus cfr. arenaceus | x | x | ||
Epizoanthus mediterraneus | x | x | ||
Epizoanthus sp. | x | x | ||
Eunicella cavolini | x | x | ||
Eunicella stricta | x | x | ||
Gerardia savaglia | x | x | ||
Hoplangia durotrix cfr. | x | x | ||
Hydractinia inermis | x | x | ||
Hydrozoa n.i. | x | x | ||
Leptogorgia sarmentosa | x | x | ||
Madrepora oculata | x | |||
Nemertesia antennina | x | x | ||
Nemertesia ramosa | x | x | x | |
Obelia sp. | x | x | ||
Parazoanthus axinellae | x | x | ||
Pennatula phosphorea | x | |||
Phyllangia mouchezi | x | x | ||
Plumularia setacea | x | x | ||
Pteroeides spinosum | x | |||
Phymanthus pulcher | x | x | ||
Savalia savaglia | x | x | ||
Scleractinia n.i. | x | x | ||
Veretillum cynomorium | x | x | x | |
Annelida | ||||
Amage adspersa | x | |||
Ampharete grubei | x | x | ||
Amphicteis gunneri | x | x | ||
Amphictene auricoma | x | |||
Bispira volutacornis | x | x | ||
Brada villosa | x | x | x | |
Ceratonereis hircinicola | x | x | ||
Chaetopterus variopedatus | x | x | ||
Chaetozone sp. | x | x | ||
Dervillea rubrovittata | x | x | ||
Drilonereis filum | x | x | ||
Eteone siphonodonta | x | x | ||
Eunice torquata | x | x | ||
Eunice vittata | x | x | ||
Eupolymnia nebulosa | x | x | ||
Glycera rouxii | x | x | ||
Hermonia hystris | x | x | ||
Hydroides norvegica | x | x | ||
Jasmineira elegans | x | x | ||
Lagis koreni | x | x | ||
Lanice conchilega | x | x | ||
Leanira yhleni | x | x | ||
Lumbriconereis latreilli | x | x | ||
Lumbrineris cf. tetraurata | x | x | ||
Lumbrineris latreilli | x | x | ||
Lysidice ninetta | x | x | ||
Maldane globifex | x | x | ||
Marphysa bellii | x | x | ||
Melinna palmata | x | x | ||
Nematonereis unicornis | x | x | ||
Nephthys hystricis | x | x | ||
Nereis irrorata | x | x | ||
Notomastus latericeus | x | x | ||
Onuphis conchylega | x | x | ||
Pontobdella muricata | x | x | x | |
Pomatoceros triqueter | x | x | x | x |
Praxillella gracilis | x | x | ||
Protula sp. | x | x | x | x |
Sabella spallanzani | x | x | x | x |
Serpula vermicularis | x | x | x | x |
Spirographis | x | x | x | |
Spirorbis sp. | x | |||
Sternaspis scutata | x | x | x | x |
Sthenolepis sp. | x | x | ||
Syllidae 1 | x | x | ||
Terebellida n.i. | x | x | ||
Crustacea | ||||
Alpheus cf. glaber | x | x | ||
Alpheus dentipes | x | x | ||
Anapagurus bicorniger | x | x | ||
Anapagurus breviaculeatus | x | x | ||
Callianassa minor | x | x | ||
Carcinus mediterraneus | x | |||
Diogenes pugilator | x | |||
Dorippe lanata | x | |||
Ebalia granulosa | x | |||
Eriphia spinifrons | x | x | x | |
Ethusa mascarone | x | |||
Eurynome aspera | x | x | ||
Galathea nexa | x | x | x | |
Galathea squamifera | x | x | x | |
Gonoplax angulata | x | |||
Galathea intermedia | x | x | ||
Ilia nucleus | x | |||
Inachus dorsettensis | x | |||
Inachus leptocheirus | x | |||
Inachus thoracicus | x | |||
Macropipus arcuatus | x | x | ||
Macropipus pusillus | x | x | x | |
Macropodia longirostris | x | x | ||
Macropodia rostrata | x | x | ||
Maia squinado | x | x | x | |
Mysidacea sp. | x | x | ||
Paguristes oculatus | x | x | ||
Pagurus alatus | x | |||
Pagurus cuanensis | x | x | ||
Palaemon adspersus | x | |||
Palaemon serratus | x | |||
Parthenope massena | x | |||
Penaeus trisulcatus | x | x | x | |
Periclimenes amethysteus | x | x | ||
Periclimenes scriptus cfr. | x | x | ||
Pilumnus hirtellus | x | x | x | |
Pisidia bluteli | x | |||
Pisidia longimana | x | x | ||
Processa canaliculata | x | |||
Squilla mantis | x | x | x | x |
Sicyonia carinata | x | |||
Tanaidacea unid. | x | x | ||
Upogebia deltaura | x | |||
Upogebia litoralis | x | x | x | x |
Upogebia typica | x | |||
Xantho poressa | x | |||
Bryozoa | ||||
Beania magellanica | x | x | ||
Bugula aquilirostris | x | x | ||
Bugula sp. | x | x | x | |
Cellaria fistulosa | x | x | ||
Celleporina caminata | x | |||
Cribilaria radiata | x | |||
Crisia sp. | x | |||
Disporella hispida | x | |||
Frondipora reticulata | x | x | x | |
Frondipora verrucosa | x | x | ||
Hippothoa flagellum | x | |||
Idmonea sp. | x | |||
Lichenopora radiata | x | x | ||
Margareta cereoides | x | |||
Microporella marsupiata | x | |||
Myriapora truncata | x | x | x | |
Myriozoum truncatum | x | |||
Pherusella tubulosa | x | |||
Phoronis sp. | x | |||
Porella cervicornis | x | x | ||
Porella compressa cfr. | x | x | ||
Retepora beaniana | x | x | x | |
Reteporella cfr. grimaldi | x | x | ||
Schizobrachiella sanguinea | x | x | x | x |
Schizomavella mamillata | x | x | ||
Sertella sp. | x | |||
Schizoporella magnifica | x | |||
Schizoporella sanguine | x | x | ||
Echinodermata | ||||
Amphiura chiajei | x | x | x | x |
Amphiura filiformis | x | x | ||
Amphiuridae juv. unid. | x | x | ||
Anseropoda placenta | x | x | x | |
Antedon mediterranea | x | x | x | x |
Arbacia lixula | x | |||
Astropecten auranciacus | x | x | x | |
Astropecten irregularis pentacanthus | x | x | ||
Astropecten spinulosus | x | x | ||
Brissus unicolor | x | x | ||
Brissopsis lyrifera | x | x | x | x |
Cidaris cidaris | x | |||
Coscinasterias tenuispina | x | |||
Echinaster sepositus | x | x | x | x |
Echinocardium cordatum | x | |||
Echinocardium fenauxi | x | |||
Echinocyamus pusillus | x | x | ||
Eostichopus regalis | x | x | ||
Hacelia attenuata | x | |||
Holothuria (Panningothuria) forskali | x | |||
Holothuria mammata | x | |||
Holothuria polii | x | x | x | |
Holothuria tubulosa | x | x | x | |
Labidoplax digitata | x | x | ||
Lepidoplx digitata | x | x | ||
Leptopentacta elongata | x | x | x | x |
Leptopentacta tergestina | x | x | x | x |
Marthasterias glacialis | x | x | x | x |
Mesothuria intestinalis | x | |||
Ocnus planci | x | x | x | |
Ocnus syracusana | x | |||
Ophidiaster ophidianus | x | |||
Ophioderma longicauda | x | |||
Ophiomyxa pentagona | x | x | x | |
Ophiothrix fragilis | x | x | x | |
Ophiura alba | x | x | x | |
Ophiura ophiura | x | |||
Paracentrotus lividus | x | |||
Psammechinus microtuberculatus | x | |||
Schizaster canaliferus | x | |||
Spatangus purpureus | x | |||
Sphaerechinus granularis | x | x | x | |
Tunicata | ||||
Amaroucium proliferum | x | |||
Ascidia mentula | x | x | x | |
Ascidiella aspersa | x | x | ||
Ascidiella scabra | x | |||
Ciona intestinalis | x | x | ||
Didemnum candidum | x | |||
Didemnum maculosum | x | |||
Diplosoma spongiforme | x | x | ||
Eugira arenosa | x | |||
Halocynthia papillosa | x | x | x | |
Microcosmus sp. | x | x | x | |
Molgula appendiculata | x | x | ||
Phallusia fumigata | x | x | x | |
Phallusia mammillata | x | x | x | x |
Polycarpa gracilis | x | |||
Styela plicata | x | x | ||
Appendix 2: List of Macromollusca from the Boka Kotorska Bay (Source: Stjepčević, 1967)
Vrsta | Herceg Novi Bay | Tivat Bay | Risan Bay | Kotor Bay |
|---|---|---|---|---|
Aloidis gibba Olivi | x | x | x | x |
Anomia ephippium L. | x | x | x | x |
Aplysia dactylomela | x | |||
Aporrhais pespelecani L. | x | x | x | x |
Arca barbata L. | x | x | x | x |
Arca diluvii Lamk. | x | x | x | |
Arca lactea L. | x | x | x | x |
Arca noae L. | x | x | x | x |
Arca tetragona Poli | x | |||
Astraea rugosa L. | x | x | x | |
Avicula tarentina Lamk. | x | x | ||
Buccinulum corneum L. | x | |||
Bursatella leachii | x | x | x | |
Calliostoma conulus L. | x | x | x | x |
Calliostoma laugieri Payr | x | x | ||
Calliostoma zizyphinum L. | x | x | x | x |
Calyptraea chinensis L. | x | x | x | x |
Cantharidus striatus L. | x | |||
Capulus hungaricus L. | x | |||
Cardium echinatum L. | x | x | ||
Cardium edule L. | x | x | x | x |
Cardium exiguum Gmel. | x | x | x | x |
Cardium paucicostatum Sowerby | x | x | x | x |
Cardium tuberculatum L. | x | x | x | x |
Cassidaria echinophora L. | x | x | ||
Cerithium rupestre Risso | x | x | x | x |
Cerithium vulgatum Brug. | x | x | x | x |
Chama gryphina Lamk. | x | |||
Chama lamellosa Lamk. | x | |||
Chiton olivaceus Speng. | x | x | x | x |
Chlamys glabra L. | x | |||
Chlamys opercularis L. | x | x | ||
Chlamys varia L. | x | x | x | x |
Clanculus corallinus Gm. | x | x | ||
Columbella rustica L. | x | x | x | x |
Conus mediterraneus Brug. | x | x | x | x |
Crepidula moulinsii Mich. | x | x | x | x |
Cuspidaria (Neaera) cuspidata Olivi | x | x | ||
Cypraea lurida L. | x | |||
Cypraea pyrum Gm | x | |||
Cypraea spurca L. | x | |||
Dentalium (Antalis) dentale L. | x | x | x | x |
Dentalium (Antalis) vulgare da Costa | x | x | ||
Diodora gibberula Lamk. | x | x | x | x |
Diodora graeca L. | x | x | x | x |
Divaricella divaricata L. | x | |||
Dolium galea L. | x | x | ||
Donacilla cornea Poli | x | |||
Dosinia lupina L. | x | x | ||
Eledone moschata Leach. | x | x | x | x |
Emerginula fissura L. | x | |||
Fucus pulchellus Phil. | x | |||
Fusinus rostratus Olivi | x | x | x | x |
Fusinus syracusanus L. | x | |||
Gibbula divaricata L. | x | x | x | |
Gibbula magus L. | x | x | x | x |
Gibbula obliquata Gm. | x | |||
Gibbula umbilicalis L. | x | |||
Glycymeris glycymeris L. | x | |||
Glycymeris pilosa L. | x | x | ||
Glycymeris violacescenns Lamk. | x | |||
Haliotis lamellosa Lamk. | x | |||
Hiatella arctica L. | x | x | ||
Hiatella rugosa L. | x | x | x | x |
Irus irus L. | x | |||
Isocardia cor L. | x | x | x | |
Laevicardium oblongum Gmel. | x | |||
Leda fragilis Shem. | x | x | ||
Leda pella L. | x | x | x | x |
Leptotyra sanguinea L. | x | |||
Lima hians Gmel. | x | |||
Lima lima L. | x | x | ||
Lithophaga lithophaga L. | x | x | x | x |
Littorina neritoides L. | x | x | x | x |
Loligo vulgaris Lamk. | x | x | x | x |
Loripes lacteus L. | x | x | ||
Mactra corallina L. | x | x | ||
Mitra ebenina Lamk. | x | |||
Modiolus barbatus L. | x | x | x | x |
Monodonta turbinata Born | x | x | x | x |
Murex brandaris L. | x | x | x | x |
Murex trunculus L. | x | x | x | x |
Mytilus galloprovincialis Lamk. | x | x | x | x |
Nassa costulata Renijer | x | |||
Nassa neritea L. | x | |||
Nassarius mutabilis L. | x | |||
Nassarius pygmaeus Lamk. | x | x | x | x |
Nassarius reticulatus L. | x | x | x | x |
Natica hebraea Mart | x | x | x | x |
Natica josephina Risso | x | |||
Natica millepunctata Lamk. | x | x | ||
Nucula nucleus L. | x | x | x | x |
Octopus vulgaris Lamk. | x | x | x | x |
Ostrea edulis L. | x | x | x | |
Patella caerulea L. | x | x | x | x |
Patella lusitanica Gmel. | x | x | x | x |
Patella vulgata L. | x | x | x | x |
Pecten jacobaeus L. | x | x | ||
Pharus legumen L. | x | x | x | |
Philine aperta | x | |||
Pinna nobilis L. | x | x | x | x |
Pinna pectinata L. | x | x | x | |
Pisania maculosa Lamk. | x | x | x | x |
Pitar rudis Poli | x | |||
Polynices (Lunatia) alderi Forbes | x | |||
Primovula adriatica Sow | x | |||
Psammobia depressa Pennant | x | x | ||
Pteria hirundo L. | x | x | ||
Scala communis lamk. | x | x | ||
Scrobicularia plana da Costa | x | x | x | |
Sepia elegans D’Orb. | x | x | x | x |
Sepia officinalis L. | x | x | x | x |
Sepiola oweniana D’Orb. | x | x | ||
Sepiola petersii Ststrp. | x | x | ||
Sepiola rondeletii Leach. | x | x | x | x |
Solen vagina L. | x | x | x | x |
Solenocurtus pelucidus L. | x | x | ||
Spondylus gaederopus L. | x | x | x | x |
Strombiformis subulata Don. | x | |||
Tellina distorta Poli | x | |||
Tellina pulchella Lamk. | x | |||
Teredo navalis L. | x | x | x | x |
Thracia combulordea de Bl. | x | |||
Tritonalia erinacea L. | x | |||
Trivia adriatica Monten | x | |||
Turritella tricarinata Risso | x | x | x | x |
Venerupis aureus Gmel. | x | |||
Venerupis decussata L. | x | x | x | x |
Venus fasciata Donov. | x | |||
Venus gallina L. | x | x | x | x |
Venus verrucosa L. | x | x | x | x |
Vermetus (Petaloconchus) subcancellatus Biv. | x | |||
Vermetus (Serpulorbis) arenarius L. | x | x | x |
Rights and permissions
Copyright information
© 2016 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Petović, S., Marković, O. (2016). Characteristics of the Zoobenthos in Boka Kotorska Bay. In: Joksimović, A., Djurović, M., Semenov, A., Zonn, I., Kostianoy, A. (eds) The Boka Kotorska Bay Environment . The Handbook of Environmental Chemistry, vol 54. Springer, Cham. https://doi.org/10.1007/698_2016_23
Download citation
DOI: https://doi.org/10.1007/698_2016_23
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-51613-4
Online ISBN: 978-3-319-51614-1
eBook Packages: Earth and Environmental ScienceEarth and Environmental Science (R0)