Abstract
Asthmatic patients are suspected of having a higher risk of mold sensitization. Thus, precise diagnosis of fungal sensitization is important. Mold allergen extracts are difficult to standardize, but component-resolved allergy diagnosis may be an alternative to replace extract-based tests. In this research, asthmatic and non-asthmatic subjects were studied for their sensitization to Aspergillus fumigatus (Asp f), Cladosporium herbarum (Cla h), Penicillium chrysogenum (Pen ch), Alternaria alternata (Alt a), and Aspergillus versicolor (Asp v). Extract-based tests were applied using the skin prick test (SPT) and allergen-specific immunoglobulin E (sIgE). Subjects with extract-based sensitization to Asp f or Alt a were further investigated for sIgE response to recombinant (r) single mold allergens. At least one mold sensitization was found in about 50% of asthmatic and non-asthmatics with the most frequent sensitization to Alt a, followed by Pen ch, Asp f, Cla h, and Asp v. Interestingly, sensitization rate to individual mold species was always higher in asthmatics and was only significant for Pen ch. The component-resolved diagnosis with the sum of rAsp f 1 - rAsp f 4 plus rAsp f 6 matched the extract-based results (SPT and/or sIgE) in 50% of asthmatics and 46% of non-asthmatics, whereas, rAlt a 1 covered 59% of asthmatics and 50% non-asthmatics of extract-based Alt a sensitization. In conclusion, individual fungal sensitization rate was higher in asthmatics compared to non-asthmatics. Extract-based tests, especially SPTs, were most sensitive, but component-based tests covered 80% of extract-based serological sensitization to Alternaria and Aspergillus.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
- Allergen
- Alternaria alternata
- Aspergillus fumigatus
- Asthma
- Component-based diagnosis
- Extract-based diagnosis
- Mold
- Skin prick test
- Specific IgE
1 Introduction
Health risks induced by mold exposure include allergen-specific immunoglobulin E (sIgE)-mediated sensitization accompanied with allergic symptoms. Even though mold exposure is ubiquitous in environmental airborne samples, sensitization rates to primarily indoor (e.g., Aspergillus sp., Penicillium sp.) and outdoor molds (e.g., Cladosporium sp., Alternaria sp.) were found to be lower (<10%) compared to other environmental allergen sources, such as pollen, mites, and animal dander (20–30%) (Haftenberger et al. 2013). In both general population and atopic subjects, the most frequent fungal sensitization is seen to Alternaria with 2–13%, depending on the test method and patient group (children versus adults). This is followed by Aspergillus with 2–10%, Penicillium with 5–8%, and Cladosporium with 8% (Kespohl and Raulf 2017). In asthmatic patients, the rates of fungal sensitization are clearly increased, with over 20% for Alternaria, Cladosporium, and Penicillium, and are up to 45% for Aspergillus (O’Driscoll et al. 2009). A potentially close relationship between asthma and fungal sensitization, known as allergic fungal airway disease (Rick et al. 2016; Denning et al. 2014) or severe asthma and fungal sensitization, has been described in numerous publications (Knutsen et al. 2012; Agarwal and Gupta 2011; Knutsen and Slavin 2011; Denning et al. 2009; O’Driscoll et al. 2009) and was also the subject of the EAACI Task force (Denning et al. 2014). Therefore, diagnosis of fungal sensitization is very important. However, it is known that fungal test extracts used for the diagnostics are extremely variable, due mainly to the heterogeneity of the fungal raw material used for the production of allergen extracts.
Different strains of mold species, as well as different culture conditions are responsible for the heterogenic protein/allergen mixtures (Esch and Codina 2017). A former study comparing mold skin prick test (SPT) has demonstrated that fungal SPT solutions with identical labeling, provided by different suppliers, are highly variable in protein, antigen, and allergen content (Kespohl et al. 2013). A comparison of SPTs with in vitro measurements of sIgE indicated that positive test results are obtained more often with SPTs than sIgE measurement. Test concordance of SPT and sIgE was best with SPT solutions containing a high fungal allergen content (Kespohl et al. 2016). Thus, to optimize and standardize fungal test extracts, the option of purified or recombinantly produced allergens may eliminate the variability of naturally produced fungal extracts (Esch and Codina 2017). Importantly, there has been a reduction in commercially available fungal allergen test extracts (Klimek et al. 2015), creating a diagnostic gap which may be filled by single allergen components.
Improvements in the molecular allergy diagnostics have been recently reviewed (Kleine-Tebbe and Jakob 2017), with examples that included increasing test sensitivity by spiking natural allergen extracts with single recombinant allergens and test selectivity by the application of recombinant marker allergens (Huss-Marp et al. 2015). Regarding fungal sensitization, 111 single allergens (including molds, yeast, dermatophytes, and mushrooms) are confirmed and listed in the Allergen Nomenclature (2018) database. Of these, six are commercially available as singleplex and two as components of a multiplex system (Kespohl and Raulf 2017). Specifically, recombinant Aspergillus fumigatus allergens are helpful tools for the differentiation of asthmatic patients with allergic bronchopulmonary aspergillosis (ABPA) from Aspergillus fumigatus-sensitized asthmatics without ABPA (Kurup et al. 2000; Crameri 1998).
In the current study, sIgE-mediated fungal sensitization rates were investigated in asthmatic versus non-asthmatic subjects with suspected mold sensitization by applying different extract-based test systems (SPT versus sIgE). Furthermore, component-based sIgE tests were compared to extract-based sIgE test results in both groups.
2 Methods
2.1 Study Group and Data Collection
From a former multicenter study (Kespohl et al. 2016), an asthma group and a non-asthma group were reinvestigated. In brief, patients with suspected mold allergy and/or mold exposure were recruited from 13 allergy practices and clinics (12 German and 1 Polish). The inclusion criteria were the anamnestic, self-reported suspicion, or diagnosed mold allergy or mold exposure and/or mold-induced allergic symptoms. Mold-induced respiratory symptoms could have occurred occupationally, at home, or both. The study consisted of questionnaires, SPTs, and sIgE measurements. Mold exposure was documented by self-reported patient questionnaire asking for visual mold formation (bigger/smaller DIN A4) in living areas, at workplaces, or during recreation.
The group of asthmatics (n = 81) included participants with asthma symptoms in the self-reported patient questionnaire, answering Yes to the question “Have you ever been diagnosed with asthma?” and reporting additional asthma medication by answering Yes to the question “Are you currently taking regular medicines for respiratory problems?” The group of non-asthmatics (n = 56) was composed of participants without asthmatic symptoms, answering No to the question “Have you ever been diagnosed with asthma?”
Sensitization to Aspergillus fumigatus (Asp f), Cladosporium herbarum (Cla h), Penicillium chrysogenum (Pen ch), Alternaria alternata (Alt a), and Aspergillus versicolor (Asp v) was tested using extract-based SPTs and sIgE (Kespohl et al. 2016). Component-based allergy diagnostics were exclusively conducted with serological measurement using singleplex recombinant allergen components, one for Alt a: rAlt a 1 and five for Asp f: rAsp f 1-4 plus rAsp f 6.
2.2 Determination of SPT
Mold SPT solutions of Asp f, Cla h, Pen ch, and Alt a were purchased from four different manufacturers: (1) Allergopharma GmbH & Co. KG (Reinbek, Germany), (2) ALK (Hørsholm, Denmark), (3) HAL Allergy (Leiden, Netherlands), and (4) Lofarma (Milano MI, Italy), and the extracts for Asp v were prepared in-house as described earlier (Kespohl et al. 2013). SPT solutions were pricked in duplicate, and test results were calculated as the mean value of both SPT determinations. SPT cut-point was evaluated by the Youden index, taking sIgE measurements as the “positive standard.” A mean wheal size ≥1.5 mm (right/left arm double testing) was considered positive as described in detail in Kespohl et al. (2016).
2.3 Determination of Serological Parameters
Serum of patients was collected, and mold sensitization was measured with extract-based ImmunoCAPs (ThermoScientific; Uppsala, Sweden) for the following mold species: Asp f (m3), Cla h (m2), Pen ch (m-1), Alt a (m6), and Asp v (Gm25). Rates of sensitization were given in the percentage values. All patients with extract-based sensitization (at least to one of the four SPTs and/or sIgE) to Alt a and/or Asp f were additionally analyzed for component-based sensitization with recombinant (r) allergens as singleplex ImmunoCAPs. In case of SPT and/or sIgE sensitization to Alt a, recombinant major allergen rAlt a 1 (m229) was tested. Asp f-sensitized subjects (SPT and/or sIgE) were tested with five recombinant allergens: rAsp f 1 (m218), rAsp f 2 (m219), rAsp f 3 (m220), rAsp f 4 (m221), and rAsp f 6 (m222) by ImmunoCAP. Specific IgE values ≥0.35 kU/L were considered positive, with the measuring range starting at 0.01 kU/L.
2.4 Statistical Analysis
Comparison of diagnostic agreement of extract-based serological test versus component-based test sIgE values was assessed with nonparametric Spearman’s correlation coefficient. In case of Asp f, the sum of single Asp f components was correlated with extract-based Asp f values, using GraphPad Prism 7.04. Significance of contingency table analysis regarding fungal sensitization in asthmatic and non-asthmatic subjects was done with two-sided Fisher’s exact test. A p-value <0.05 defined statistically significant differences. A commercial statistical package of GraphPad Prism v7.04 was use for the analysis.
3 Results
3.1 Fungal Sensitization in Asthmatics and Non-asthmatic Subjects
Among the 81 subjects with reported asthmatic symptoms plus asthma medication, 56% were shown to have fungal sensitization to at least one of the five tested mold species using SPT and/or sIgE (Fig. 1). Subjects without asthmatic symptoms (non-asthmatics, n = 56) but with suspected fungal allergy exhibited a positive reaction to at least one mold species in 52% of the cases. The most prominent fungal sensitization was seen to Alt a, with 42% in asthmatics and 32% in non-asthmatics. Sensitization to Pen ch was just as frequent as sensitization to Alt a among asthmatics, amounting to 41%. However, in non-asthmatics, the rate of Pen ch sensitization was significantly lower (23%) compared to asthmatics. Sensitization to Asp f was present in 32% of asthmatic vs. 23% of non-asthmatics, followed by sensitization rates to Cla h (26% asthmatics vs. 20% non-asthmatics) and to Asp v (21% asthmatics vs. 13% non-asthmatics). The rate of individual sensitization was always higher among the asthmatic subjects compared to the non-asthmatics but significant only for Pen ch sensitization.
Differences in the applied extract-based diagnostic tests were investigated for SPT and sIgE in asthmatics and non-asthmatics (Fig. 2). SPT detected mold sensitization more frequently compared to sIgE tests using ImmunoCAP, independent of asthma symptoms. With SPTs, 52% of asthma and non-asthmatic subjects showed positive skin reactions to at least one mold species in at least one of the four applied SPT extracts, whereas 37% of asthmatics and 25% of non-asthmatics were tested positive to at least one mold species using the serological sIgE test with ImmunoCAPs. Considering the single SPT results according to individual manufacturers, it was seen that the rate of mold sensitization was lower using only one SPT compared to the combined. However, discrepancy between mold sensitization measured by SPT and sIgE was still there. For asthmatics, the rate of fungal sensitization to at least one mold species was shown to be between 33% and 47% with different SPTs compared to 37% with sIgE. In non-asthmatics, 38–46% showed positive skin reactions in SPTs and only 25% with sIgE. Nevertheless, the concordance of positive SPT and positive sIgE was always higher in asthmatics than in non-asthmatics (Table 1). The best concordance was seen for Alt a with 71% positive SPT plus sIgE in asthmatics and 56% in non-asthmatics. For all other mold species, the test concordance was below 50% with a range of difference from 2% to 25% in asthmatics compared to non-asthmatics (Table 1). The highest rate of discrepancy in SPT/sIgE concordance (32%) was seen with Pen ch sensitization, with 8% in non-asthmatics compared to 40% in asthmatics. Only this high discrepancy of concordance between asthmatic and non-asthmatics was significant.
Sensitization rate to at least one mold species, diagnosed by skin prick test (SPT) against Alternaria alternata, Aspergillus fumigatus, Cladosporium herbarum, and Penicillium chrysogenum from four different manufacturers (Manuf. 1–4) or diagnosed by allergen-specific immunoglobulin E (sIgE) measured with the corresponding ImmunoCAPs in asthmatic and non-asthmatic subjects. *Manufacturer offered no Claosporium herbarum SPT solution
3.2 Component-Based Versus Extract-Based sIgE Testing
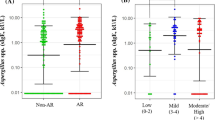
Among the studied subjects, sensitization to Alt a was found to be most common, including 34/81 (42%) asthmatics and 18/56 (32%) non-asthmatics (Fig. 1), as diagnosed with both SPT and sIgE. Serological sensitization was measured in 26 of the 34 Alt a-sensitized asthmatics by means of sIgE concentration ≥ 0.35 kU/L and in 11 of 18 Alt a-sensitized non-asthmatics (Table 1). The concordance of SPT and sIgE was between 71% in asthmatics and 56% in non-asthmatics. A component-based test for Alt a was conducted with the major allergen, rAlt a 1, using singleplex ImmunoCAP in all subjects who exhibited Alternaria sensitization (SPT and/or sIgE) (Fig. 3a and b). With component-based rAlt a 1 measurement, no additional sensitization was detected that was not detected with the extract-based sIgE testing. Specific IgE concentration to component rAlt a 1 ≥ 0.35 kU/L was measured in 20 asthmatics (Fig. 3a), all of whom also had sIgE concentration ≥ 0.35 kU/L to the allergen extract Alt a. Therefore, taking only the serological tests into account, rAlt a 1, when used as a component-based tool, detected 77% (20/26) of extract-based serological sensitization in asthmatics. Nevertheless, eight subjects with Alt a sensitization diagnosed solely by SPT were not detected using the component-based rAlt a 1.
In the group of non-asthmatics, similar results were obtained (Fig. 3b). Here, component-based sensitization to rAlt a 1 was seen in nine non-asthmatic subjects all of whom reacted positively to extract-based Alt a in SPT and/or sIgE. Calculating only serological Alt a sensitization, component-based rAlt a 1 measured positive sIgE values (≥ 0.35 kU/L) in 9 out of the 11 (82%) extract-based positive non-asthmatic subjects. Of note, patients with Alt a sensitization in SPT without sIgE to Alt a were not detected by rAlt a 1. In both groups, single allergen rAlt a 1 met the criteria for a major allergen and was recognized by more than 50% of subjects. Nonparametric Spearman’s correlation was highly significant (p < 0.0001) among both asthmatics (r = 0.871) and non-asthmatics (r = 0.863).
In comparison to Alt a, Asp f sensitization with extract-based tools (SPT and/or sIgE) was seen in 26 asthmatics and 13 non-asthmatics (Table 1). For component-based diagnostic tools, five single allergens rAsp f 1, rAsp f 2, rAsp f 3, rAsp f 4, and rAsp f 6 were tested. There was no additional sIgE sensitization detected by component-based testing compared to extract-based tests, neither in asthmatics nor in non-asthmatics (Fig. 4a and b). Specific IgE to single components was exclusively detected in subjects with sIgE sensitization to Asp f (extract-based ImmunoCAPs). Specific IgE to allergen components rAsp f 1 and rAsp f 3 was most frequently measured in about 50% of both groups – in 6 out of the 13 asthmatics and in 4 (rAsp f 1) and 3 (rAsp f 3) out of the 6 non-asthmatics (Table 2). Aspergillus allergens, rAsp f 2 and rAsp f 4, each tested positively in five subjects and rAsp f 6 in three subjects from both groups. Taken together, the component-based allergen pattern for Aspergillus sensitization was comparable in asthmatics and non-asthmatics. For a comparison of extract-based serological sensitization with component-based tests, the sum of all single components (rAsp f 1–Asp f 6) was taken. In asthmatics, 10 out of the 13 (77%) subjects with extract-based serological Asp f sensitization were also positive in the component-based tests (Fig. 4a). In non-asthmatic subjects, five out of the six (83%) tested positive to the sum of single rAsp f components (Fig. 4b). Correlation of extract-based vs. sum of component-based tests for Asp f sensitization was significant in both groups (p < 0.005), with r = 0.850 for asthmatics and r = 0.770 for non-asthmatics.
4 Discussion
The current study compared mold sensitization in asthmatics and non-asthmatics with extract-based allergy tests applied as SPT and as serological sIgE measurements. The results showed that fungal sensitization rate to at least one of the five tested mold species was comparable – with 56% in asthmatics and 52% in non-asthmatics. A high mold sensitization rate revealed here was consistent with a previous survey in which the mold sensitization rate was 66% in asthmatics (O’Driscoll et al. 2009). But for non-asthmatics, more than 50% fungal sensitization observed in the current study was unexpectedly high. This could be due to the recruiting criteria for patients, which only included subjects with reported mold exposure or suspected mold allergy, who were obviously more sensitized compared to the general population.
For the five individual mold species tested, it was shown that sensitization was always higher in asthmatics compared to non-asthmatics. The most frequent mold sensitization seen in both groups (42% in asthmatics and 32% in non-asthmatics) was to Alternaria. The dominant role of Alternaria as a source of IgE-mediated sensitizer and inducer of respiratory symptoms (asthma, wheeze, or allergy) has been reported in several studies and case reports (Behbod et al. 2015; Salo et al. 2006; Bush and Prochnau 2004; Zureik et al. 2002). Sensitization rates against Aspergillus, Cladosporium and Penicillium in non-asthmatics also were with >20% higher in the current study compared to former studies in the general population and in patients with allergic problems (Haftenberger et al. 2013; Gent et al. 2012; Szewzyk et al. 2011; Heinzerling et al. 2009). Of note, there was a significantly higher sensitization rate to Pen ch in asthmatics (41%) compared to non-asthmatics (23%). Former studies have shown significantly higher odds ratio of asthma in relation to specific IgE to Aspergillus and Cladosporium (Jaakkola et al. 2006) or Alternaria (Zureik and Orehek 2002), but not for Penicillium in adult asthmatics. Nevertheless, there are neonate cohorts or studies in children indicating that Penicillium is significantly associated with respiratory effects among the sensitized children (Caillaud et al. 2018; Gent et al. 2012; Rosenbaum et al. 2010; Bundy et al. 2009). Therefore, Penicillium sensitization should be considered regarding fungal sensitization in asthmatics.
Among the extract-based test systems, SPT revealed mold sensitization with >50% higher to at least one mold species. This was more than what was observed with serological sIgE tests with 25% in non-asthmatics and 37% in asthmatics. These results may be due to the fact that in the current study, positive SPT results were generated from skin tests using four different SPT solutions, from different suppliers, all tested as double values. When calculating the sensitization rate of only one SPT solution with sIgE, it was shown that it was higher in SPT compared to sIgE only if SPT solutions with high antigen/allergen content were used (Kespohl et al. 2016). In contrast, if SPT solution had a low antigen/allergen content, the sIgE-based sensitization rate was higher in comparison to SPT. Discordant fungal sensitivity, as diagnosed by SPT or sIgE, has been investigated previously by O’Driscoll et al. (2009) who report 77% concordance of SPT and sIgE for fungal sensitization to at least one mold species, with slightly more positive sIgE tests. Further, double skin prick testing improves test reproducibility enormously, especially in extracts without major allergens, such as many occupational and mold allergens (van Kampen et al. 2013). In addition, SPT results depend on the arm position used, as wheal size closer to the elbow is slightly larger than that at the wrist (Kespohl et al. 2016).
Component-based allergy diagnosis has been introduced to resolve the issue of standardization in fungal allergen test solutions, thereby increasing test sensitivity and selectivity (Kleine-Tebbe and Jakob 2015). In the current study, rAlt a 1 was used as a singleplex for Alternaria sensitization and was detected in about 80% of Alt a extract-based serological sensitization, independent of asthmatic symptoms. For Alternaria, component-based diagnosis was comparable with extract-based serological diagnosis, since rAlt a 1 was the major allergen available in sufficient amounts in the Alternaria test extracts used. That is in line with the result of a former study by Asturias et al. (2005) showing that Alternaria diagnostics could be substituted for by natural or recombinant Alt a 1 that was recognized in 98% of 42 patients with positive SPT and serology to Alt a. Another study by Vailes et al. (2001) has found sIgE response to rAlt a 1 in 93% of patients with asthma/rhinitis, exhibiting sIgE concentration (> CAP-class 2) to Alternaria extract. Other patients, such as those with atopic dermatitis or cystic fibrosis, with sIgE concentration (> CAP-class 2) to Alternaria, were less frequently sensitized to single allergen rAlt a 1, in 47% and 60%, respectively.
Aspergillus fumigatus component-based diagnosis has been intensively investigated (Crameri 1998), with a specific goal to differentiate between allergic/asthmatics and patients with allergic bronchopulmonary aspergillosis (ABPA) or cystic fibrosis. A combination of sIgE sensitization to rAsp 2 plus rAsp f 4 plus rAsp f 6 appears significant enough to discriminate between asthmatics and patient with ABPA (Kurup et al. 2000). In the current study, single component-based sIgE diagnosis using singleplex rAsp f 1, rAsp f 2, rAsp f 3, rAsp f 4, and rAsp f 6 assessed in 10 out of the 26 Asp f (extract-based)-sensitized asthmatics and in 5 out of the 13 non-asthmatics resulted in a positive response (≥ 0.35 kU/L) to the sum of all five rAsp f components (Table 2). There was no difference in sIgE sensitization profile or frequency to any of the tested single Asp f allergens depending on asthma or non-asthma status. In both groups, rAsp f 1 and rAsp f 3 were the most frequently detected allergens. A previous study by Crameri (1998) also shows rAsp f 1 and rAsp f 3 to be the most prominent allergens in allergic, asthmatic and ABPA patients. A reported frequency of up to 70% sensitization to single rAsp f components was not confirmed in the current study, taking all (SPT and/or sIgE) Asp f-sensitized subjects into account. Here, none of the tested single rAsp f allergens exceeded the 26% sensitization rate among Aspergillus-sensitized subjects. However, calculating the sensitization rate to single rAsp f allergens on the basis of subjects with sIgE to Asp f resulted in up to 53% sensitization to rAsp f 1 and 47% to rAsp f 3.
In conclusion, it was shown that extract-based diagnosis conducted as skin prick test was the most sensitive and was therefore recommended as the first choice. Serological sensitization to Alternaria was measured by rAlt a 1 in 78% of all patients and could substitute for the extract-based serological measurement. For serological Aspergillus sensitization, all five rAsp f single allergens were necessary to account for 80% of the extract-based sIgE sensitization. Further research is necessary to identify clinically relevant allergens, especially marker allergens; for instance, serine proteases may be relevant for diagnostic purposes in fungal allergy.
References
Agarwal R, Gupta D (2011) Severe asthma and fungi: current evidence. Med Mycol 49(Suppl 1):150–157
Allergen Nomenclature (2018) WHO/IUIS allergen nomenclature sub-committee. https://www.allergen.org. Accessed on 20 Dec 2018
Asturias JA, Ibarrola I, Ferrer A, Andreu C, López-Pascual E, Quiralte J, Florido F, Martínez A (2005) Diagnosis of Alternaria alternata sensitization with natural and recombinant Alt a 1 allergens. J Allergy Clin Immunol 115(6):1210–1217
Behbod B, Sordillo JE, Hoffman EB, Datta S, Webb TE, Kwan DL, Kamel JA, Muilenberg ML, Scott JA, Chew GL, Platts-Mills TA, Schwartz J, Coull B, Burge H, Gold DR (2015) Asthma and allergy development: contrasting influences of yeasts and other fungal exposures. Clin Exp Allergy 45(1):154–163
Bundy KW, Gent JF, Beckett W, Bracken MB, Belanger K, Triche E, Leaderer BP (2009) Household airborne Penicillium associated with peak expiratory flow variability in asthmatic children. Ann Allergy Asthma Immunol 103(1):26–30
Bush RK, Prochnau JJ (2004) Alternaria-induced asthma. J Allergy Clin Immunol 113(2):227–234
Caillaud D, Leynaert B, Keirsbulck M, Nadif R, mold ANSES working group (2018) Indoor mold exposure, asthma and rhinitis: findings from systematic reviews and recent longitudinal studies. Eur Respir Rev 27(148). https://doi.org/10.1183/16000617.0137-2017
Crameri R (1998) Recombinant Aspergillus fumigatus allergens: from the nucleotide sequences to clinical applications. Int Arch Allergy Immunol 115(2):99–114
Denning DW, O’Driscoll BR, Powell G, Chew F, Atherton GT, Vyas A, Miles J, Morris J, Niven RM (2009) Randomized controlled trial of oral antifungal treatment for severe asthma with fungal sensitization: The Fungal Asthma Sensitization Trial (FAST) study. Am J Respir Crit Care Med 179(1):11–18
Denning DW, Pashley C, Hartl D, Wardlaw A, Godet C, Del Giacco S, Delhaes L, Sergejeva S (2014) Fungal allergy in asthma-state of the art and research needs. Clin Transl Allergy 4:14
Esch RE, Codina R (2017) Fungal raw materials used to produce allergen extracts. Ann Allergy Asthma Immunol 118(4):399–405
Gent JF, Kezik JM, Hill ME, Tsai E, Li DW, Leaderer BP (2012) Household mold and dust allergens: exposure, sensitization and childhood asthma morbidity. Environ Res 118:86–93
Haftenberger M, Laußmann D, Ellert U, Kalcklösch M, Langen U, Schlaud M, Schmitz R, Thamm M (2013) Prävalenz von Sensibilisierungen gegen Inhalations- und Nahrungsmittelallergene: Ergebnisse der Studie zur Gesundheit Erwachsener in Deutschland (DEGS1). Bundesgesundheitsblatt 56(5–6):687–697
Heinzerling LM, Burbach GJ, Edenharter G, Bachert C, Bindslev-Jensen C, Bonini S et al (2009) GA(2)LEN skin test study I: GA(2)LEN harmonization of skin prick testing: novel sensitization patterns for inhalant allergens in Europe. Allergy 64(10):1498–1506
Huss-Marp J, Raulf M, Jakob T (2015) Spiking with recombinant allergens to improve allergen extracts: benefits and limitations for the use in routine diagnostics. Allergo J Int 24:236–243
Jaakkola MS, Ieromnimon A, Jaakkola JJK (2006) Are atopy and specific IgE to mites and molds important for adult asthma? J Allergy Clin Immunol 117(3):642–648
Kespohl S, Raulf M (2017) Mold allergens and their importance in molecular allergy diagnosis. In: Kleine-Tebbe J, Jakob T (eds) Molecular allergy diagnostics. Springer International Publishing, Switzerland, pp 445–458
Kespohl S, Maryska S, Zahradnik E, Sander I, Bruening T, Raulf-Heimsoth M (2013) Biochemical and immunological analysis of mold skin prick test solution: current status of standardization. Clin Exp Allergy 43(11):1286–1296
Kespohl S, Maryska S, Bünger J, Hagemeyer O, Jakob T, Joest M, Knecht R, Koschel D, Kotschy-Lang N, Merget R, Mülleneisen NK, Rabe U, Röseler S, Sander I, Stollewerk D, Straube H, Ulmer HM, van Kampen V, Walusiak-Skorupa J, Wiszniewska M, Wurpts G, Brüning T, Raulf M (2016) How to diagnose mold allergy? Comparison of skin prick tests with specific IgE results. Clin Exp Allergy 46(7):981–991
Kleine-Tebbe J, Jakob T (2015) Molecular allergy diagnostics using IgE singleplex determinations: methodological and practical considerations for use in clinical routine. Allergo J Int 24:185–197
Kleine-Tebbe J, Jakob T (2017) Molecular allergy diagnostics. Innovation for a better patient management. Springer International Publishing, Switzerland
Klimek L, Werfel T, Vogelberg C, Jung K (2015) Authorised allergen products for intracutaneous testing may no longer be available in Germany: allergy textbooks have to be re-written. Allergo J Int 24(3):84–93
Knutsen AP, Slavin RG (2011) Allergic bronchopulmonary aspergillosis in asthma and cystic fibrosis. Clin Dev Immunol 2011:843763
Knutsen AP, Bush RK, Demain JG, Denning DW, Dixit A, Fairs A, Greenberger PA, Kariuki B, Kita H, Kurup VP, Moss RB, Niven RM, Pashley CH, Slavin RG, Vijay HM, Wardlaw AJ (2012) Fungi and allergic lower respiratory tract diseases. J Allergy Clin Immunol 129(2):280–291
Kurup VP, Banerjee B, Hemmann S, Greenberger PA, Blaser K, Crameri R (2000) Selected recombinant Aspergillus fumigatus allergens bind specifically to IgE in ABPA. Clin Exp Allergy 30(7):988–993
O’Driscoll BR, Powell G, Chew F, Niven RM, Miles JF, Vyas A, Denning DW (2009) Comparison of skin prick tests with specific serum immunoglobulin E in the diagnosis of fungal sensitization in patients with severe asthma. Clin Exp Allergy 39(11):1677–1683
Rick EM, Woolnough K, Pashley CH, Wardlaw AJ (2016) Allergic fungal airway disease. J Investig Allergol Clin Immunol 26(6):344–354
Rosenbaum PF, Crawford JA, Anagnost SE, Wang CJK, Hunt A, Anbar RD, Hargrave TM, Hall EG, Liu CC, Abraham JL (2010) Indoor airborne fungi and wheeze in the first year of life among a cohort of infants at risk for asthma. J Expo Sci Environ Epidemiol 20(6):503–515
Salo PM, Arbes SJ, Sever M, Jaramillo R, Cohn RD, London SJ, Zeldin DC (2006) Exposure to Alternaria alternata in US homes is associated with asthma symptoms. J Allergy Clin Immunol 118(4):892–898
Szewzyk R, Becker K, Hünken A, Pick-Fuß H, Kolossa-Gehring M (2011) Kinder-Umwelt-Survey (KUS) 2003/06 Sensibilisierung gegenüber Innenraum Schimmelpilzen. In: Schriftreihe Umwelt & Gesundheit im Auftrag des Umweltbundesamt. http://www.uba.de/uba-info-medien/4176.html. Accessed on 20 Dec 2018
Vailes LD, Perzanowski MS, Wheatley LM, Platts-Mills TA, Chapman MD (2001) IgE and IgG antibody responses to recombinant Alt a 1 as a marker of sensitization to Alternaria in asthma and atopic dermatitis. Clin Exp Allergy 31(12):1891–1895
van Kampen V, de Blay F, Folletti I, Kobierski P, Moscato G, Olivieri M, Quirce S, Sastre J, Walusiak-Skorupa J, Kotschy-Lang N, Müsken H, Mahler V, Schliemann S, Ochmann U, Sültz J, Worm M, Sander I, Zahradnik E, Brüning T, Merget R, Raulf-Heimsoth M (2013) Evaluation of commercial skin prick test solutions for selected occupational allergens. Allergy 68(5):651–658
Zureik M, Orehek J (2002) Diagnosis and severity of asthma in the elderly: results of a large survey in 1485 asthmatics recruited by lung specialists. Respiration 69(3):223–228
Zureik M, Neukirch C, Leynaert B, Liard R, Bousquet L, Neukirch F, European Community Respiratory Health Survey (2002) Sensitisation to airborne molds and severity of asthma: cross sectional study from European Community respiratory health survey. BMJ 325(7361):11–414
Acknowledgments
The study was supported by the German Social Accident Insurance (DGUV), Sankt Augustin, Germany, IPA-project 107. Data herein were generated by reinvestigation and further testing of sera from former QuaSchi-patient group with the known skin prick tests and serological data published by Kespohl et al. (2016). We acknowledge the support of the QuaSchi-study group for their collaboration in recruiting and medical examination of patients with potential mold exposure. We also thank Silke Maryska and Ursula Meurer for their excellent technical assistance.
Conflicts of Interest
The authors declare no conflict of interest in relation to this article.
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. The Ethics Committee of the Medical Faculty of the Ruhr-University Bochum in Germany approved the implementation of all necessary examinations (register no. 4104-11).
Informed Consent
Written informed consent was obtained from all individual participants included in the study.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Kespohl, S., Raulf, M. (2019). Mold Sensitization in Asthmatic and Non-asthmatic Subjects Diagnosed with Extract-Based Versus Component-Based Allergens. In: Pokorski, M. (eds) Medical Science and Research. Advances in Experimental Medicine and Biology(), vol 1153. Springer, Cham. https://doi.org/10.1007/5584_2019_342
Download citation
DOI: https://doi.org/10.1007/5584_2019_342
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-19058-3
Online ISBN: 978-3-030-19059-0
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)