Abstract
This article provides an update of the current trends in the management of acute thromboembolic disease, aimed at emphasizing new options applicable in daily practice. CT is no longer exclusively dedicated to the diagnosis of PE but also participates in the prognostic approach of this disease focusing on the detection of CT features of right ventricular dysfunction. The availability of perfusion CT allows evaluation of the extent of perfusion impairment, thus introducing a more reliable prognostic parameter than the estimation of the clot burden. Radiologists can provide clinicians with relevant information regarding the early risk stratification that is worth describing in daily practice. Optimized management of young patients, pregnant women, and evolution toward chronic pulmonary embolism will also be covered in this chapter.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Multidetector-row computed tomographic angiography (CTA) has become the first-line modality for imaging the pulmonary vasculature in patients with suspected acute pulmonary embolism (PE) (Remy-Jardin et al. 2007). This disease is the third most common acute cardiovascular disease after myocardial infarction and stroke in the Unites States and results in many deaths each year. This data fully justifies a regular update of the radiologists’ role in the management of patients suspected of acute pulmonary embolism. Some aspects are currently well established such as the CT features of endovascular clots whose description has not changed over time. However, their detection has greatly benefited from the improvement of cross-sectional imaging that has almost completely suppressed technically related artifacts. Some clinically relevant aspects have to be considered by radiologists in their CT reports owing to their impact in early risk stratification. The purpose of this chapter is to provide an insight into new options applicable in the daily practice in the clinical context of acute pulmonary embolism.
2 New Options in the Diagnostic Approach
2.1 Clinical Decision Support
Over the last decade, numerous articles have underlined the increase in CT examinations indicated for clinical suspicion of PE with a parallel description of a high rate of negative examinations (Mamlouk et al. 2010). Mamlouk et al. showed that a CT angiogram positive for pulmonary embolism was extremely unlikely (0.95% chance) if patients had none of the studied thromboembolic risk factors, raising questions on the appropriate indication of the CT examination. In order to improve patient selection, clinicians can utilize risk factor assessment. Special attention should be directed to the patient’s age and immobilization, both risk factors of most concern for a CT angiogram positive for PE. Another option has been proposed, based on a computerized clinical decision support (CDS). At each stage of the proposed decision tree, clinicians could either cancel the imaging or ignore the advice. The implementation of evidence-based CDS in the emergency department was found to be associated with a significant decrease in the use and significant increase in the yield of CT pulmonary angiography for the evaluation of acute PE during a 2-year period (Raja et al. 2012). These conclusions are shared by Dunne et al. who have recently reported that implementation of evidence-based clinical decision support was associated with a 12.3% immediate and sustained decrease in use of CT pulmonary angiographic (CTPA) examinations in the evaluation of inpatients for acute PE (Dunne et al. 2015). However, one should underline that a negative chest CTPA for acute PE can help recognize alternative diagnoses for the patient’s symptoms in 25–67% of cases. These cases usually include congestive heart failure, pneumonia, pleural effusion, or atelectasis. Green et al. have recently proposed a structured approach for recognition of alternative explanations approachable on CT images (Green et al. 2015).
2.2 Optimization of the Radiation Dose
In parallel to the abovementioned efforts to decrease the number of unnecessary examinations, the radiological community is directly involved in the optimization of the radiation dose delivered to patients. As recently underlined in a panel discussion (Araoz et al. 2012), the radiation risk from pulmonary CT angiography (CTA) is strongly dependent on age, sex, and pulmonary CTA acquisition parameters (Woo et al. 2012). The radiological community demonstrated the usefulness of several practical methods, the most frequently employed relying on individual adjustment of milliamperage to patient weight, alone or in association with automated tube current modulation systems. This approach achieves an average dose reduction of 20% (Kubo et al. 2008; Christner et al. 2010). However, lowering the kilovoltage has a greater effect on patient dose than reducing the tube current, and the current trend is to perform chest CTAs with a kilovoltage adapted to the patient’s body weight (Matsuoka et al. 2009) (Tables 1a and 1b) (Fig. 1). This approach was investigated in several studies in adult populations, from which several conclusions were drawn (Sigal-Cinqualbre et al. 2004; Schueller-Weidekamm et al. 2006; Szucs-Farkas et al. 2008; Gorgos et al. 2009). First, substantial dose reduction can be achieved, with an average dose reduction of 40% when lowering the setting from 120 to 80 kVp. Second, we improve vascular enhancement, as the attenuation of iodinated contrast media increases at low tube voltage. This was found to improve the analyzability of central and peripheral pulmonary arteries, even when reducing the volume of contrast material (Szucs-Farkas et al. 2011). However, radiologists may be reluctant to apply low kVp protocols in daily clinical routine because of the lack of standardized guidelines not only for the tube potential selection but also for the adjustment of the tube current to avoid grainy images. This difficulty can be overcome by the use of automated systems which can determine the most appropriate kV settings in relation to the patient’s attenuation (Niemann et al. 2013). Lastly, the availability of iterative reconstructions offers a unique means of combining dose reduction (up to 50%, averaged DLP less than 80 mGy.cm), excellent vascular enhancement, and better image quality than that obtained at standard dose (Pontana et al. 2013; Kaul et al. 2014; McLaughlin et al. 2015). With such possibilities of dose reduction, it is no longer necessary to try to save dose by reducing the scan length. Important additional or alternative diagnosis may be excluded from the limited imaging volume and therefore go undetected.
Chest CT angiography obtained in a 50-year-old male patient referred to the emergency department with a clinical suspicion of acute pulmonary embolism (182 cm, 69 kg). The examination was obtained at 100 kVp and 90 ref mAs (35% iodinated contrast agent, flow rate: 4 mL/s). The dose-length product was 128 mGy.cm. Sharp delineation of numerous endoluminal clots identified on both sides (a–d). (e) Transverse CT section obtained at the level of cardiac cavities showing a RV/LV ratio greater than 1, suggestive of right ventricular dysfunction
2.3 Optimization of the Iodine Load
2.3.1 Low Contrast Medium Volume
Although iodinated contrast medium is relatively safe, some adverse reactions such as contrast-induced nephropathy may occur. The risk of developing contrast-induced nephrotoxicity is increased with higher doses of iodinated contrast medium. Subsequently, one can decrease the risks by reducing the volume of highly concentrated contrast agents administered during a chest CT angiographic examination. This approach can be facilitated when combining low tube voltage and high-pitch techniques together with iterative reconstruction (Sodickson and Weiss 2012; Lu et al. 2014). The volumes administered varied between 50 and 20 mL of contrast material.
2.3.2 Low-Concentrated Contrast Agents
With the advent of fast CT scanning modes, administration of contrast media with high iodine concentration (i.e., 300–370 mg of iodine per milliliter) has become routine clinical practice to maximize the arterial enhancement of systemic and pulmonary arterial circulation. However, the inflow of highly concentrated contrast material to systemic veins can generate streak artifacts between the concentrated agent and the surrounding structures that may obscure mediastinal and pathological hilar and right upper lobe pulmonary arterial abnormalities. Moreover, the application of CTPA is limited in patients with impaired renal function because of the risk of developing contrast-medium induced nephropathy. Many patients at risk of developing pulmonary embolism are elderly and have comorbid conditions that increase the risk for renal injury. Therefore, it could be interesting to perform chest CT angiographic examinations with reduced iodine load. To date, this approach has not been found to be clinically acceptable owing to the poor level of arterial enhancement on images acquired at high kilovoltages. This limitation has recently been overcome by acquiring data sets with dual-energy CT. This new scanning mode offers the possibility of generating virtual monochromatic spectral (VMS) imaging with a wide range of energy levels accessible from a single data set. Using single-source, dual-energy CT, Yuan et al. were the first authors to demonstrate that these acquisitions facilitated iodine load reduction at CT pulmonary angiography (Yuan et al. 2012). Delesalle et al. investigated the energy levels providing optimal imaging of the thoracic circulation at dual-source, dual-energy CT angiography with reduced iodine load (Delesalle et al. 2013). These authors found that the use of low-concentration contrast media enabled suppression of streak artifacts around systemic veins on high-energy images while providing satisfactory central arterial enhancement on low-energy images. The additional advantage of this scanning mode was the considerable reduction in the amount of iodine administered to patients (Fig. 2). These preliminary studies confirm that dual-energy CT has the potential to represent a new option for greater use of low-concentration contrast agents for chest CT examinations in routine clinical practice.
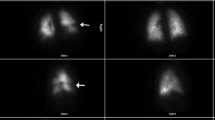
Chest CT angiography obtained in a 52-year-old female patient (164 cm, 60 kg). The examination was acquired with dual source, dual energy after administration of a low-concentrated contrast agent (i.e., 125 mg I/mL). (a) CT section obtained at the level of the right bronchus intermedius illustrating the excellent quality of attenuation within all thoracic circulations on this image generated from both tubes. (b) CT section obtained at the level of the lower lung zones illustrating the additional excellent opacification of the cardiac cavities
2.4 Current Role of CAD Systems
Whereas the diagnosis of acute PE remains based on the visual depiction of endoluminal filling defects, identification of peripheral clots remains a difficult task and more than 30% can be missed on initial review (Ritchie et al. 2007). This limitation can be solved by the use of computer-aided diagnostic (CAD) systems which have been developed to aid radiologists with the depiction of endovascular clots that requires careful analysis of hundreds of pulmonary vascular branches. Used as a second reader, these systems can help detect small clots initially missed (Kligerman et al. 2014), increasing reader sensitivity for the detection of peripheral emboli (Dewailly et al. 2010; Wittenberg and Berger 2012). In addition, the high negative predictive value of these tools is helpful to reassure inexperienced readers (Blackmon et al. 2013). However, these results are obtained at the expense of an increased reading time due to the presence of numerous false-negative and false-positive findings, as recently demonstrated by Wittenberg et al. (Wittenberg and Berger 2012). These authors also demonstrated a strong association between CT image quality and the number of false-positive findings indicated by the CAD system which is a current limitation of CAD in clinical practice.
An alternative to CAD for the detection of small-sized clots can theoretically be found with dual-energy CT which can provide perfusion imaging in addition to cross-sectional imaging of the pulmonary circulation (Fig. 3). Abnormal pulmonary blood distribution shown at dual-source CT improves detection of acute PE, particularly by emphasizing the presence of subsegmental pulmonary iodine mapping defects. However, acute PE cannot be assessed on the sole finding of perfusion defects, even if observed as triangular-shaped defects known to be suggestive of acute PE. Small-airways disease can lead to similar filling defects, depicted in 30% of COPD patients (Pontana et al. 2012). Moreover, the presence of an underlying lung disease altering lung perfusion makes it more difficult to detect PE-related filling defects. Therefore, CT detection of small-sized clots remains a difficult task. In daily practice, this does not represent a major clinical limitation except for the subset of patients with isolated subsegmental PE in whom cross-sectional imaging may fail to depict them. The role of perfusion imaging in the clinical context of acute PE is more generally considered as a helpful tool for identifying the presence or absence of PE (Cai et al. 2015).
Dual-energy chest CT angiography obtained in a 30-year-old male suspected of acute pulmonary embolism (185 cm, 73 kg). The examination was obtained with dual-source, dual-energy CT (tube A: 80 kV; tube B: 140 kVp; 35% iodinated contrast agent; flow rate: 4 mL/s). The dose-length product was 260 mGy.cm. (a) The presence of a small-sized, peripheral clot in a subsegmental pulmonary artery of the anterior segment of the left upper lobe (arrow). (b) Whereas the small clot is difficult to visualize, the corresponding perfusion defect (arrowheads) is easily depicted
2.5 Is There Always a High Level of Confidence in Diagnosing Acute PE?
From the radiologist perspective, the confidence in identifying endovascular clots has dramatically increased with CT technological improvement over years. In a recent study, Bedayat et al. have evaluated the clinical characteristics associated with low confidence in diagnosis of acute PE as expressed in CTPA reports and evaluated the effect of confidence level in PE diagnosis on patient clinical outcomes (Bedayat et al. 2015). They observed that roughly 10% of positive CTPA reports had uncertainties in PE findings; patients with reports categorized as low confidence had smaller emboli and more comorbidities. Although the low-confidence group was less likely to receive PE-related therapies, patients in this group were not associated with higher probability of short-term mortality.
3 New Options in the Prognostic Assessment
3.1 Clot Burden
The degree of vascular obstruction on CT images can be estimated with semiquantitative scores (Quanadli et al. 2001; Mastora et al. 2003) with recent introduction of quantitative estimates of blood clot volume (Furlan et al. 2011; Hariharan et al. 2016). There is a controversy on the association between mortality and clot burden indexes, mainly related to the lack of correlation between these results and the presence of a pre-existing pulmonary disease. Whereas an isolated subsegmental clot may have no clinical consequence in an otherwise healthy patient, the same clot may lead to respiratory failure in a patient with poor respiratory condition due to previously altered pulmonary perfusion. Therefore, it appears interesting to switch from clot burden estimates to the analysis of the extent of perfusion impairment on dual-energy CT perfusion images (Chae et al. 2010; Bauer and Frellesen 2011; Thieme et al. 2012). New scores have been defined, grading the degree of perfusion defect per lobe (Chae et al. 2010; Thieme et al. 2012), estimating the volume of perfusion defects relatively to the total lung volume (Bauer and Frellesen 2011). Good correlations were found between perfusion impairment and CT features of right ventricular dysfunction, suggesting that perfusion defects could be a predictor of patient outcome. To our knowledge, a single study used a model adjusted for age, gender, and prior history of COPD and heart failure (Thieme et al. 2012). Good correlations were found between the proposed dual-energy based PE score and a number of parameters of PE severity. These authors concluded that this approach was easier and faster to perform than the traditional CT scoring methods for vascular obstruction with better prognostic implications.
3.2 Right Ventricular Dysfunction
As previously underlined, the presence of right ventricular dysfunction in hemodynamically stable patients is the major determinant of patient’s outcome. Recent updates in the literature should help radiologists gather accurate information. Regarding the CT features of right ventricular dysfunction, the description published by Reid and Murchison in 1998 remains of major clinical usefulness (Reid and Murchison 1998) (Table 2). In the list of CT features of right ventricular dysfunction, the RV/LV diameter ratio is the most important parameter to consider, as recently confirmed by Becattini et al. in a meta-analysis (Becattini et al. 2014).
Several practical approaches have been proposed to determine the RV/LV diameter ratio, including measurements on transverse CT sections, short-axis images and four-chamber views of the cardiac cavities. Kamel et al. were the first authors to suggest that measurements of ventricular diameters on transverse CT sections were accurate enough to estimate the RV/LV ratio (Kamel et al. 2008). This has been recently confirmed by Lu et al. (2012) who have reported that the axial RV/LV diameter ratio is no less accurate than the reformatted four-chamber RV/LV diameter ratio for predicting 30-day mortality after PE (Fig. 1). A step further in the simplification of the estimation of the RV/LV diameter ratio has been proposed by Kumamaru et al. (2012). These authors have recently demonstrated that complex measurements of RV/LV diameter ratios can be replaced by subjective determination of right ventricular enlargement. When the right ventricle appeared larger than the left ventricle, it provided prognostic information that did not significantly differ from that of more traditional, quantitative RV/LV diameter ratios. The authors concluded that a right ventricle that appears larger than the left ventricle should be reported by the radiologist and interpolated into clinical risk stratification. This information can be reinforced when a prior CT examination negative for PE indications is available. In such circumstances, Lu et al. have shown that the interval increase in four-chamber RV/LV diameter ratio is more accurate than the diameter ratio of the CT examination with positive findings for PE alone for mortality prediction after acute PE (Lu et al. 2012). Kim et al. have recently completed the list of conventional chest CT-derived hemodynamic findings with the left-bulging atrial septum, an abnormal sign indicating hemodynamic overloading of the right heart (Kim et al. 2014).
3.3 Novel Approaches
In addition to the estimation of the clot burden and features of RV dysfunction, CT angiography can also reveal important ancillary findings in the scanned field, particularly malignancy and abnormal lung parenchymal features (e.g., infarction, interstitial lung disease, or effusions) that affect the patient’s prognosis. Based on these assumptions, Kumamaru et al. have developed a comprehensive risk-scoring system based on CTPA findings for predicting mortality within 30 days after acute PE (Kumamaru et al. 2016). Their model was found to be superior to PESI (PE severity index) in predicting mortality, leading the authors to suggest that the incorporation of this scoring system into image interpretation workflows might help clinicians to select the most appropriate management approach for individual patients.
4 The Radiologist’s Report: Which Information Is Particularly Relevant for Clinicians?
The various mortality rates reported among studies illustrate the heterogeneous clinical and prognostic spectrum in acute PE (Table 3). This situation has raised debates on the most appropriate therapeutic options for the various PE-related risk categories. Regarding the prognostic parameters, it is well established that the hemodynamic status at the time of presentation has the strongest prognostic implication for short-term mortality. Therefore, the highest risk is that of “massive acute PE,” characterized by the presence of PE-associated arterial hypotension or shock. Accounting for 5% of all cases of PE, consensus guidelines recommend treatment with thrombolysis. These patients are not referred to the CT room. In the remaining majority of PE patients who present without hypotension, there is a subgroup of patients with “submassive acute PE,” characterized by the presence of right ventricular dysfunction at the time of diagnosis. These patients are considered as having a higher risk of clinical deterioration than those with preserved systemic arterial blood pressure and normal right ventricular function. They correspond to the “intermediate risk” category. Current debates question the recommendations for thrombolytic therapy in this subset of patients (Todd and Tapson 2009; Piazza and Goldhaber 2010; Jimenez et al. 2013). It was only recently that improved risk assessment strategies permitted advances in the identification of another category, i.e., the “low-risk PE.” The current state of knowledge of this category has been recently summarized by Lankeit and Konstantinides (2012). Patients presenting without hemodynamic instability and without elevated biomarker levels or imaging findings indicating right ventricular dysfunction or myocardial injury may constitute this low-risk group (Torbicki et al. 2008). In this category, selected patients might be considered for early discharge and treatment at home. Lastly, the high quality of chest CT examinations currently achievable enables depiction of small-sized pulmonary embolism, sometimes incidentally diagnosed, which could represent a “no-risk category.” These situations have raised debates in the literature on the clinical significance of such clots, described as “isolated subsegmental PE,” comprising a spectrum ranging from a single subsegmental clot to multiple clots exclusively confined to the subsegmental arterial bed. Eyer et al. were the first authors to report the clinicians’ decision to withhold anticoagulation in this category of patients, without adverse effects of this clinical decision (Eyer et al. 2005). Later, Anderson et al. suggested that some pulmonary emboli detected by CT might be clinically unimportant, the equivalent of deep vein thrombosis isolated to the calf veins that might not require anticoagulant therapy (Anderson et al. 2007). In a recent review article, Stein et al. summarized the conditions which should be fulfilled for such therapeutic decisions (Stein et al. 2012). From this description of current trends in PE-risk stratification (Table 4), one can deduce that the radiologist’s report should provide clinicians with relevant information for early risk stratification. Consequently, particular attention should be directed toward analysis of the amount and location of clots in the pulmonary arterial tree, description of cardiac cavity morphology with special attention to CT features suggestive of right ventricular dysfunction as well as abnormalities suggesting pre-existing pulmonary and/or cardiac disease.
5 Pulmonary Embolism from Pregnancy to Young Adults
5.1 Pulmonary Embolism in Pregnancy
To prepare the mother for the blood losses associated with delivery, a state of hypercoagulability develops during pregnancy that explains the increased risk for venous thromboembolism reported during pregnancy. Because clinical symptoms are nonspecific, reliable diagnostic tests are needed but the most adapted diagnostic strategy remained a matter of discussion until the publication of clinical practice guidelines in 2011 (Leung et al. 2011). A multidisciplinary panel developed evidence-based guidelines using the Grades of Recommendation, Assessment, Development and Evaluation (GRADE) system. Strong recommendations were made for three specific scenarios: (a) performance of chest radiography as the first radiation-associated procedure, (b) use of lung scintigraphy as the preferred test in the setting of a normal chest radiograph, and (c) performance of CT pulmonary angiography rather than digital substraction angiography in a pregnant woman with a nondiagnostic ventilation-perfusion result. In addition to general recommendations for radiation dose savings for the fetus and the maternal breast, the scanning protocol for a chest CT pulmonary angiography in a pregnant patient should be adapted to the hemodynamic effects of pregnancy. These effects combine an increase in cardiac output, heart rate, and plasma volume leading to dilution of the contrast bolus (Ridge et al. 2011). Moreover, suboptimal opacification can also be due to the increased venous return of nonopacified blood to the right atrium during inspiration. Consequently, several technical adjustments have been proposed, including the use of automated bolus triggering, a high contrast medium flow rate, a high concentration of contrast medium and acquisition during quiet or suspended respiration rather than at deep inspiration (Fig. 4). Regarding potential harmful effects of a chest CT examination to the fetus, it is important to be aware that the radiation dose delivered is in the range of that absorbed by the fetus from naturally occurring background radiation during the 9-month gestational period. In a series of 343 neonates exposed to an iodinated contrast agent at various stages of gestation, all had a normal tyroxine level at birth. In the 85 neonates tested for thyroid-stimulating hormone, only one (with comorbid conditions) had a transiently abnormal level that reverted to normal by day 6 (Bourjeily et al. 2010). From this study, it was concluded that a single, high-dose in utero exposure to water-soluble, low-osmolar iodinated intravenous products, such as iohexol, is unlikely to have a clinically important effect on thyroid function at birth.
Chest CT angiography obtained in a 29-year-old pregnant patient at 21 weeks gestation (163 cm, 54 kg). The examination was obtained at 100 kVp and 90 ref mAs (30% iodinated contrast agent, 4 mL/s). The dose-length product was 83 mGy.cm. Images were reconstructed with raw-data-based iterative reconstruction. Transverse CT section obtained at the level of the right upper lobe bronchus (a) and right middle lobe bronchus (b) illustrating the good level of vascular enhancement and the possibility to depict large endoluminal clots on both sides
5.2 Pulmonary Embolism in Children
As recently reported by Lee et al. (Lee et al. 2012a), the incidence of PE ranges from 0.73% to 4.2% in the pediatric population with concerns about potential overutilization of CT pulmonary angiography in children suspected of having PE. From their study, it was concluded that risk factor assessment should be a primary tool for guiding when to perform CT pulmonary angiography in this population. With such an approach, CT pulmonary angiography can be targeted more appropriately, with the potential to substantially reduce costs and radiation exposure. Five independent risk factors were found to be significantly associated with a positive CT pulmonary angiography result, namely immobilization, hypercoagulable state, excess estrogen state, indwelling central venous line, and prior PE and/or deep venous thrombosis. Lastly, they observed that the D-dimer test was of little value in screening for PE among children with a high clinical probability of PE. From the same group, it appears that similar conclusions can be drawn in older children and young adults (Lee et al. 2012b). In a recent study, Hennelly et al. have proposed a pediatric PE clinical decision rule that was derived from commonly used adult-based PE algorithms (Hennelly et al. 2016).
6 When Acute PE Evolves Toward Chronic PE
Whereas complete resolution of endoluminal clots represents the most frequent outcome of acute pulmonary embolism, some patients can develop chronic obstruction of the pulmonary circulation. Dependent on the severity of chronic obstruction, pulmonary hypertension can subsequently develop, related to incomplete resolution of clots or recurrent pulmonary embolism (Fig. 5). Chronic thromboembolic pulmonary hypertension (CTEPH) represents one of the leading causes of pulmonary hypertension, with a prevalence occurring in 0.1–4.0% of patients in retrospective studies (Fedullo et al. 2001; Pengo et al. 2004). In a recent prospective study, the incidence of symptomatic CTEPH was found to represent a relatively common complication after an episode of acute pulmonary embolism, reported in 5.4% of the studied population (Guerin et al. 2014). The high rate of complete thrombus resolution in the majority of patients combined with the lack of precise knowledge of the risk factors for CTEPH do not justify the routine use of follow-up CTPA imaging in patients treated for acute PE (Den Exter et al. 2015). The interpretation of residual defects after acute PE is not straightforward. Whereas it may be related to a slow rate of resolution of endoluminal clots, one should integrate the possibility of acute PE in the context of unknown chronic PE. Guerin et al. have recently underlined that a majority of patients with CTEPH had previously unknown pulmonary hypertension at the time of the acute event (Guerin et al. 2014). Six months after diagnosis of first or recurrent PE, residual pulmonary perfusion defects encountered on V/Q-SPECT were found to correspond with hypoperfusion on iodine map dual-energy CT in the majority of patients with chronic thromboembolic disease seen on dual-energy CT (de Broucker et al. 2012).
CT angiography obtained in a 38-year-old female patient evaluated for chronic thromboembolic pulmonary hypertension. The examination was obtained with dual-source, dual-energy CT (tube A: 80 kV; tube B: 140 kVp; 35% iodinated contrast agent; flow rate: 4 mL/s). The dose-length product was 378 mGy.cm. (a) CT section obtained at the level of the right pulmonary artery showing an intraluminal web at the level of the left interlobar pulmonary artery. (b) CT section obtained at the level of the origin of the right middle lobe artery showing a mural defect (arrow) at the level of the right interlobar pulmonary artery. (c) CT section obtained at the level of the carina showing tiny arterial sections in the posterior segment of the right upper lobe and mosaic perfusion on both sides
Conclusion
CT pulmonary angiography is a well-recognized diagnostic tool but is also a unique means of providing prognostic information from the same examination as that used for diagnostic purposes. Radiologists should be aware of current trends in the management of patients with acute PE as this knowledge has direct influence on the content of their daily reports. RV function and information on the likelihood of an underlying cardiopulmonary disease is of the utmost importance for risk stratification. Lastly, CT is a rapidly evolving technology, and radiologists should regularly adapt their single energy scanning protocols and consider new options with dual-energy CT whenever available.
References
Anderson DR, Kahn SR, Rodgers MA et al (2007) Computed tomographic pulmonary angiography vs ventilation-perfusion lung scanning in patients with suspected pulmonary embolism. JAMA 298:2743–2753
Araoz PA, Haramati LB, Mayo JR, Mortani Barbosa EJ Jr, Rybicki FJ, Colletti PM (2012) Panel discussion : pulmonary embolism imaging and outcomes. AJR Am J Roentgenol 198:1313–1319
Bauer RW, Frellesen C, Renker M et al (2011) Dual energy CT pulmonary blood volume assessment in acute pulmonary embolism – correlation with D-dimer level, right heart strain and clinical outcome. Eur Radiol 21:1914–1921
Becattini C, Agnelli G, Germini F, Vedovati MC (2014) Computed tomography to assess risk of death in acute pulmonary embolism: a meta-analysis. Eur Respir J 43:1678–1690
Bedayat A, Sewatkar R, Cai T et al (2015) Association between confidence level of acute pulmonary embolism diagnosis on CTPA images and clinical outcome. Acad Radiol 22:1555–1561
Blackmon KN, Florin C, Bogoni L, McCain JW, Koonce JD, Bastarrika G, Thilo C, Costello P, Salganicoff M, Schoepf UJ (2013) Computer-aided detection of pulmonary embolism at CT pulmonary angiography: can it improve performance of inexperienced readers? Eur Radiol 21:1214–1223
Bourjeily G, Chalhoub M, Phornphutkul C, Alleyne TC, Woodfield CA, Chen KK (2010) Neonatal thyroid function: effect of a single exposure to iodinated contrast medium in utero. Radiology 256:744–750
Cai XR, Feng YZ, Qiu L et al (2015) Iodine distribution map in dual-energy computed tomography pulmonary artery imaging with rapid kVp switching for the diagnosis analysis and quantitative evaluation of acute pulmonary embolism. Acad Radiol 22:743–751
Chae EJ, Seo JB, Jang MY et al (2010) Dual-energy CT for assessment of the severity of acute pulmonary embolism: pulmonary perfusion defect score compared with CT angiographic obstruction score and right ventricular/left ventricular diameter ratio. AJR Am J Roentgenol 194:604–610
Christner JA, Zavaletta VA, Eusemann CD, Walz-Flannigan AI, McCollough CH (2010) Dose reduction in helical CT: dynamically adjustable z-axis X-ray beam collimation. AJR Am J Roentgenol 194:W49–W55
de Broucker T, Pontana F, Santangelo T, Faivre JB, Tacelli N, Delannoy-Deken V, Duhamel A, Remy J, Remy-Jardin M (2012) Single- and dual-source chest CT protocols: levels of radiation dose in routine clinical practice. Diagn Interv Imaging 93:852–858
Delesalle MA, Pontana F, Duhamel A, Faivre JB, Flohr T, Tacelli N, Remy J, Remy-Jardin M (2013) Spectral optimization of chest CT angiography with reduced iodine load: experience in 80 patients evaluated with dual-source, dual-energy CT. Radiology 267:256–266
Den Exter PL, Van Es J, LJM K et al (2015) Thromboembolic reoslution assessed by CT pulmonary angiography after treatment of acute pulmonary embolism. Thromb Haemost 114:26–34
Dewailly M, Remy-jardin M, Duhamel A et al (2010) Computer-aided detection of acute pulmonary embolism with 64-slice multidetector row computed tomography: impact of the scanning conditions and overall image quality in the detection of peripheral clots. J Comput Assist Tomogr 34:23–30
Dunne RM, Ip IK, Abbett S et al (2015) Effect of evidence-based clinical decision support on the use and yield of CT pulmonary angiographic imaging in hospitalized patients. Radiology 276:167–174
Eyer BA, Goodman LR, Washington L (2005) Clinicians’ response to radiologists’ reports of isolated subsegmental pulmonary embolism or inconclusive interpretation of pulmonary embolism using MDCT. AJR Am J Roentgenol 184:623–628
Fedullo PF, Auger WR, Kerr KM et al (2001) Chronic thromboembolic pulmonary hypertension. N Engl J Med 345:1465–1472
Furlan A, Patil A, Park B et al (2011) Accuracy and reproducibility of blood clot burden quantification with pulmonary CT angiography. AJR Am J Roentgenol 196:516–523
Gorgos A, Remy-Jardin M, Duhamel A, Faivre JB, Tacelli N, Delannoy V, Remy J (2009) Evaluation of peripheral pulmonary arteries at 80 kV and 140 kV: dual-energy computed tomography assessment in 51 patients. J Comput Assist Tomogr 33:981–986
Green DB, Raptis CA, Garin IAH, Bhalla S (2015) Negative computed tomography for acute pulmonary embolism: important differential diagnosis considerations for acute dyspnea. Radiol Clin North Am 53:789–799
Guerin L, Coutiraud F, Parent F et al (2014) Prevalence of chronic thromboembolic pulmonary hypertension after acute pulmonary embolism. Thromb Haemost 112:598–605
Hariharan P, Dudzinski DM, Rosovosky R et al (2016) Relation among clot burden, right-sided heart strain and adverse events after acute pulmonary embolism. Am J Cardiol 118:1568–1573
Hennelly KE, Baskin MN, Monuteuax MC et al (2016) Detection of pulmonary embolism in high-risk children. J Pediatr 178:214–218
Jimenez D, Billello KL, Murin S (2013) Point/Counterpoint editorials. Should systemic lytic therapy be used for submassive pulmonary embolism? Yes. Chest 143:296–299
Kamel EM, Schmidt S, Doenz F, Adler-Etechami G, Schnyder P, Quanadli SD (2008) Computed tomographic angiography in acute pulmonary embolism: do we need multiplanar reconstructions to evaluate the right ventricular dysfunction? J Comput Assist Tomogr 32:438–443
Kaul D, Grupp U, Kahn J, Ghadjar P, Wiener E, Hamm B, Streitparth F (2014) Reducing radiation dose in the diagnosis of pulmonary embolism using adaptive statistical iterative reconstruction and lower tube potential in computed tomography. Eur Radiol 24:2685–2692
Kim MJ, Jung HO, Jung JI, Kim KJ, Jeon DS, Youn HJ (2014) CT-derived atrial and ventricular septal signs for risk stratification of patients with acute pulmonary embolism: clinical associations of CT-derived signs for prediction of short-term mortality. Int J Cardiovasc Imaging 30:25–32
Kligerman SJ, Lahiji K, Galvin JR et al (2014) Missed pulmonary emboli on CT angiography/ assessment with pulmonary embolism – computer-aided detection. AJR Am J Roentgenol 202:65–73
Kubo T, Lin PJP, Stiller W, Takahashi M, Kauczor HU, Ohno Y, Hatabu H (2008) Radiation dose reduction in chest CT: a review. AJR Am J Roentgenol 190:335–343
Kumamaru KK, Hunsaker AR, Bedayat A, Soga S, Signorelli J, Adams K, Wake N, Lu MT, Rybicki FJ (2012) Subjective assessment of right ventricle enlargement from computed tomography pulmonary angiography images. Int J Cardiovasc Imaging 28:965–973
Kumamaru KK, Saboo SS, Aghayev A et al (2016) CT pulmonary angiography-based scoring system to predict the prognosis of acute pulmonary embolism. J Cardiovasc Comput Tomogr 10(6):473–479
Lankeit M, Konstantinides S (2012) Is it time for home treatment of pulmonary embolism? Eur Respir J 40:742–749
Lee EY, Tse SKS, Zurakowski D, Johnson VM, Lee NJ, Tracy DA, Boiselle PM (2012a) Children suspected of having pulmonary embolism: multidetector CT pulmonary angiography – thromboembolic risk factors and implications for appropriate use. Radiology 262:242–251
Lee EY, Neuman MI, Lee NJ, Johnson VM, Zurakowski D, Tracy DA, Boiselle PM (2012b) Pulmonary embolism detected by pulmonary MDCT angiography in older children and young adults: risk factor assessment. AJR Am J Roentgenol 198:1431–1437
Leung AN, Bull TM, Jaeschke R et al (2011) An official American Thoracic Society/Society of Thoracic radiology clinical practice guideline: evaluation of suspected pulmonary embolism in pregnancy. Am J Respir Crit Care Med 184:1200–1208
Lu MT, Demehri S, Cai T, Parast L, Hunsaker AR, Goldhaber SZ, Rybicki FJ (2012) Axial and reformatted four-chamber right ventricle-to-left ventricle diameter ratios on pulmonary CT angiography as predictors of death after acute pulmonary embolism. AJR Am J Roentgenol 198:1353–1360
Lu GM, Luo S, Meinel FG, McQuiston AD, Zhou CS et al (2014) High-pitch computed tomography pulmonary angiography with iterative reconstruction at 80 kVp and 20 mL contrast agent volume. Eur Radiol 24:3260–3268
Mamlouk MD, vanSonnenberg E, Gosalia R et al (2010) Pulmonary embolism at CT angiography: implications for appropriateness, cost and radiation exposure in 2003 patients. Radiology 256:625–632
Mastora I, Remy-Jardin M, Masson P, Galland E, Delannoy V, Bauchart JJ, Remy J (2003) Severity of acute pulmonary embolism: evaluation of a new spiral CT angiographic score in correlation with echocardiographic data. Eur Radiol 13:29–35
Matsuoka S, Hunsaker AR, Gill RR et al (2009) Vascular enhancement and image quality of MDCT pulmonary angiography in 400 cases: comparison of standard and low kilovoltage settings. AJR Am J Roentgenol 192:1651–1656
McLaughlin PD, Liang T, Homiedan M, Louis LJ, O’Connel TW, Krzymyk K, Nicolaou S, Mayo MR (2015) High pitch, low voltage dual source CT pulmonary angiography: assessment of image quality and diagnostic acceptability with hybrid iterative reconstruction. Emerg Radiol 22:117–123
Niemann T, Simon H, Faivre JB, Yasunaga K, Bendaoud S, Simeone A, Remy J, Duhamel A, Flohr T, Remy-Jardin M (2013) Clinical evaluation of automatic tube voltage selection in chest CT angiography. Eur Radiol 23:2643–2651
Pengo V, Lensing AW, Prins MH et al (2004) Incidence of thromboembolic pulmonary hypertension after pulmonary embolism. N Engl J Med 350:2257–2264
Piazza G, Goldhaber SZ (2010) Management of submassive pulmonary embolism. Circulation 122:1124–1129
Pontana F, Chalayer C, Faivre JB, Murphy C, Remy-Jardin M, Remy J (2012) Pseudo-embolic perfusion defects in COPD: evaluation with dual-energy CT angiography (DECT) in 170 patients. Abstract B 0272, European Congress of Radiology, SS504 – CTTA - Dual energy and dose reduction
Pontana F, Pagniez J, Duhamel A et al (2013) Reduced-dose low-voltage chest CT angiography with sinogram-affirmed iterative reconstruction versus standard-dose filtered back projection. Radiology 267:609–618
Quanadli SD, EI Hajjam M, Vieillard-Baron A, Joseph T, Mesurolle B, Oliva VL, Barré O, Bruckert F, Dubourg O, Lacombe P (2001) New CT index to quantify arterial obstruction in pulmonary embolism: comparison with angiographic index and echocardiography. AJR Am J Roentgenol 176:1415–1420
Raja AS, Ip IK, Prevedello LM et al (2012) Effect of computerized clinical decision support on the use and yield of CT pulmonary angiography in the emergency department. Radiology 262:468–474
Reid JH, Murchison JT (1998) Acute right ventricular dilatation: a new helical CT sign of massive pulmonary embolism. Clin Radiol 53:694–698
Remy-Jardin M, Pistolesi M, Goodman LR, Gefter WB, Gottschalk A, Mayo JR, Sostman HD (2007) Management of suspected acute pulmonary embolism in the era of CT angiography: a statement from the Fleischner Society. Radiology 245:315–329
Ridge CA, Mhuircheartaigh JN, Dodd JD, Skehan SJ (2011) Pulmonary CT angiography protocol adapted to the hemodynamic effects of pregnancy. AJR Am J Roentgenol 197:1058–1063
Ritchie G, McGurk S, Mc Creath C, Graham C, Murchison JT (2007) Prospective evaluation of unsuspected pulmonary embolism on contrast multidetector CT (MDCT) scanning. Thorax 62:536–540
Schueller-Weidekamm C, Schaefer-Prokop CM, Weber M, Herold CJ, Prokop M (2006) CT angiography of pulmonary arteries to detect pulmonary embolism: improvement of vascular enhancement with low kilovoltage settings. Radiology 241:899–907
Sigal-Cinqualbre AB, Hennequin R, Abada HT, Chen X, Paul JF (2004) Low-kilovoltage multi-detector row chest CT in adults: feasibility and effect on image quality and iodine dose. Radiology 231:169–174
Sodickson A, Weiss M (2012) Effect of patient size on radiation dose reduction and image quality in low-kVp CT pulmonary angiography performed with reduced IV contrast dose. Emerg Radiol 19:437–445
Stein PD, Goodman LR, Hull RD et al (2012) Diagnosis and management of isolated subsegmental pulmonary embolism: review and assessment of the options. Clin Appl Thromb Hemost 18:20–26
Szucs-Farkas Z, Kurmann L, Strautz T, Patak MA, Vock P, Schindera ST (2008) Patient exposure and image quality of low-dose pulmonary computed tomography angiography: comparison of 100- and 80-kVp protocols. Invest Radiol 43:871–876
Szucs-Farkas Z, Schibler F, Cullmann J et al (2011) Diagnostic accuracy of pulmonary CT angiography at low tube voltage: intraindividual comparison of a normal-dose protocol at 120 kVp and a low-dose protocol at 80 kVp using reduced amount of contrast medium in a simulation study. AJR Am J Roentgenol 197:W852–W859
Thieme SF, Ashoori N, Bamberg F et al (2012) Severity assessment of pulmonary embolism using dual energy CT – correlation of a perfusion defect score with clinical and morphological parameters of blood oxygenation and right ventricular failure. Eur Radiol 22:269–278
Todd JL, Tapson VF (2009) Thrombolytic therapy for acute pulmonary embolism: a critical appraisal. Chest 135:1321–1329
Torbicki A, Perrier A, Konstantinides SV et al (2008) Guidelines on the diagnosis and management of acute pulmonary embolism: the task force for the diagnosis and management of acute pulmonary embolism of the European Society of Cardiology (ESC). Eur Heart J 29:2276–2315
Wittenberg R, Berger FH, Peters JF et al (2012) Acute pulmonary embolism: effect of a computer-assisted detection prototype on diagnosis – an observer study. Radiology 262:305–313
Woo JKH, Chiu RYW, Thakur Y, Mayo JR (2012) Risk-benefit analysis of pulmonary CT angiography in patients with suspected pulmonary embolus. AJR Am J Roentgenol 198:1332–1339
Yuan R, Shuman WP, Earls JP, Hague CJ, Mumtaz HA, Scott-Moncrieff A, Ellis JD, Mayo JR, Leipsic JA (2012) Reduced iodine load at CT pulmonary angiography with dual-energy monochromatic imaging: comparison with standard CT pulmonary angiography – a prospective randomized trial. Radiology 262:290–297
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer International Publishing AG
About this chapter
Cite this chapter
Hutt, A., Felloni, P., Remy, J., Remy-Jardin, M. (2017). CT of Pulmonary Embolism: Imaging Update. In: Nikolaou, K., Bamberg, F., Laghi, A., Rubin, G.D. (eds) Multislice CT. Medical Radiology(). Springer, Cham. https://doi.org/10.1007/174_2017_19
Download citation
DOI: https://doi.org/10.1007/174_2017_19
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-42585-6
Online ISBN: 978-3-319-42586-3
eBook Packages: MedicineMedicine (R0)