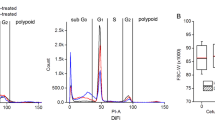
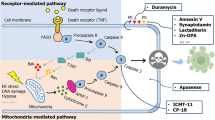
Abstract
The induction of apoptosis plays a key role in non-surgical cancer treatment — whether this occurs by irradiation, chemotherapy, or hormones. Recent advancements in imaging science and metabolic studies by nuclear magnetic resonance have painted an intriguing picture of the metabolic and biophysical processes involved with the progression of apoptosis in situ. It is now possible for us to detect and visualize previously inaccessible and even unrecognized biological phenomena in living cells and tissues undergoing therapeutically induced apoptosis. These new imaging techniques will have an increasing impact in the preclinical design of new anticancer agents and novel treatment protocols to enhance apoptotic cascades and to combat drug resistance. With the advent of molecular medicine and patient-tailored therapeutic options and drug molecules, rapid and accurate visualization of apoptotic response in the clinical settings can be of significant diagnostic and prognostic worth.
Access provided by Autonomous University of Puebla. Download to read the full chapter text
Chapter PDF
Similar content being viewed by others
References
J. Savill and V. Fadok, Corpse clearance defines the meaning of cell death, Nature 407, 784–788 (2000).
R.A. Kinloch et al, The pharmacology of apoptosis, Trends Pharmacol. Sci. 20, 35–42 (1999).
G.I. Evan and K.H. Vousden, Proliferation, cell cycle and apoptosis in cancer, Nature 411, 342–348 (2001).
B. Dubray et al., In vitro radiation-induced apoptosis and early response to low-dose radiotherapy in non-Hodgkin’s lymphomas, Radiother. Oncol. 46, 185–191 (1998).
B. Jansen et al., Chemosensitisation of malignant melanoma by BCL2 antisense therapy, Lancet 356, 1728–1733 (2000).
W.F. Symmans et al., Paclitaxel-induced apoptosis and mitotic arrest assessed by serial fine-needle aspiration: implications for early prediction of breast cancer response to neoadjuvant treatment, Clin. Cancer Res. 6, 4610–4617 (2000).
R.E. Meyn et al., Apoptosis in murine tumors treated with chemotherapy agents, Anti-Cancer Drugs 6, 443–450 (1995).
P.A. Ellis et al., Preoperative chemotherapy induces apoptosis in early breast cancer, Lancet 349, 849 (1997).
J.M. Hakumäki J.M. and K.M. Brindle, Techniques: visualizing apoptosis by nuclear magnetic resonance, Trends Pharmacol. Sci. 24, 146–149 (2003).
C. Bremer et al., Optical-based molecular imaging: contrast agents and potential medical applications, Eur Radiol. 13, 231–243 (2003).
F.G. Blankenberg, K. Ohtsuki, H.W. Strauss, Dying a thousand deaths. Radionuclide imaging of apoptosis, Q. J. Nucl. Med. 43, 170–176 (1999).
C.M. Lahorte et al., Apoptosis-detecting radioligands: current state of the art and future perspectives, Eur. J. Nucl. Med Mol. Imaging 31, 887–919 (2004).
C. Stadelmann and H. Lassmann, Detection of apoptosis in tissue sections, Cell Tissue Res. 301, 19–31 (2000).
S.J. Martin et al., Early redistribution of plasma-membrane phosphatidylserine is a general feature of apoptosis regardless of the initiating stimulus — Inhibition by overexpression of Bcl-2 and Abl, J. Exp. Med. 182, 1545–1556 (1995).
J.T. Kuikka et al., Future developments in nuclear medicine instrumentation: a review., Nucl. Med. Commun. 19, 3–12 (1998).
A. F. Chatziioannou, Molecular imaging of small animals with dedicated PET tomographs, Eur. J. Nucl. Med Mol. Imaging 29, 98–114 (2002).
Blankenberg, F. G. et al., In vivo detection and imaging of phosphatidylserine expression during programmed cell death, Proc. Natl. Acad. Sci. USA 95, 6349–6354 (1998).
T.J. Sweeney et al., Visualizing the kinetics of tumor-cell clearance in living animals, Proc. Natl. Acad Sci. USA 96, 12044–12049 (1999).
J.S. Reynolds et al., Imaging of spontaneous canine mammary tumors using fluorescent contrast agents, Photochem Photobiol. 70, 87–94 (1999).
A. Petrovsky et al., Near-infrared fluorescent imaging of tumor apoptosis, Cancer Res. 63, 1936–42 (2003).
S.J. Mandl et al., Multi-modality imaging identifies key times for annexin V imaging as an early predictor of therapeutic outcome, Mol Imaging 3, 1–8 (2004).
G.J. Czarnota et al., Ultrasonic biomicroscopy of viable, dead and apoptotic cells, Ultrasound Med. Biol. 23, 961–965 (1997).
G. J. Czarnota et al., Ultrasound imaging of apoptosis: high-resolution non-invasive monitoring of programmed cell death in vitro, in situ and in vivo, Br. J. Cancer 81, 520–527 (1999).
D.G. Gadian, NMR and its applications to living systems (Oxford Science, Oxford, 1995).
F. Adebodun and J.F.M. Post, 31P NMR characterization of cellular metabolism during dexamethasone-induced apoptosis in human leukemic cell lines, J. Cell. Physiol. 158, 180–186 (1994).
A.V.W. Nunn et al., Characterisation of secondary metabolites associated with neutrophil apoptosis, FEBS Lett. 392, 295–298 (1996).
S.N.O. Williams et al., Induction of apoptosis in two mammalian cell lines results in increased levels of fructose-1,6-bisphosphate and CDP-choline as determined by 31P MRS, Magn. Reson. Med 40, 411–420 (1998).
S. Ronen et al., Magnetic resonance detects metabolic changes associated with chemotherapy-induced apoptosis, Br. J. Cancer 80, 1035–1041 (1999).
M.L. Anthony et al., Inhibition of phosphatidylcholine biosynthesis following induction of apoptosis in HL-60 cells, J. Biol. Chem. 274, 19686–19692 (1999).
F.G. Blankenberg et al., Quantitative analysis of apoptotic cell death using proton nuclear magnetic resonance spectroscopy, Blood 89, 3778–3785 (1997).
C.E. Mountford CE and L.C. Wright, Organization of lipids in the plasma membranes of malignant and stimulated cells: a new model, Trends Biochem Sci. 13, 172–7 (1988).
J.M. Hakumiki and R.A. Kauppinen, 1H NMR visible lipids in the life and death of cells, Trends Biochem. Sci. 25, 357–362 (2000).
J.M. Hakumiki et al., 1H MRS detects polyunsaturated fatty acid accumulation during gene therapy of glioma: implications for the in vivo detection of apoptosis, Nature Med. 5, 1323–1327 (1999).
J.L. Griffin et al., Assignment of 1H nuclear magnetic resonance visible polyunsaturated fatty acids in BT4C gliomas undergoing ganciclovir-thymidine kinase gene therapy-induced programmed cell death, Cancer Res. 63, 3195–3201 (2003).
M. Di Vito et al., 1H NMR-visible mobile lipid domains correlate with cytoplasmic lipid bodies in apoptotic T-lymphoblastoid cells, Biochim. Biophys. Acta. 1530, 47–66 (2001).
N.M. Al-Saffar et al, Apoptosis is associated with triacylglycerol accumulation in Jurkat T-cells, Br. J Cancer 86, 963–970 (2002).
D. De Valck et al., Differential activation of phospholipases during necrosis or apoptosis: a comparative study using tumor necrosis factor and anti-Fas antibodies, J Cell Biochem. 71, 392–399 (1998).
T. Liimatainen et al., 1H magnetic resonance spectroscopic imaging of phospholipase activity in rat gliomas in vivo, Proc. 12th Ann. Sci. Meeting ISMRM, Kyoto, Japan (2004).
L. Le Moyec et al., Proton nuclear magnetic resonance spectroscopy reveals cellular lipids involved in resistance to adriamycin and taxol by the K562 leukemia cell line, Cancer Res. 56, 3461–3467 (1996).
D.S. Ucker et al., Genome digestion is a dispensable consequence of physiological cell-death mediated by cytotoxic T-lymphocytes, Mol. Cell. Biol. 12, 3060–3069 (1992).
C.E. Scott and F. Adebodun, 13C-NMR investigation of protein synthesis during apoptosis in human leukemic cell lines, J Cell Physiol. 181, 147–152 (1999).
M.E. Moseley et al., Diffusion-weighted MR imaging of anisotropic water diffusion in cat central nervous system, Radiology 176, 439–445 (1990).
J.M. Hakumïki et al., Quantitative 1H nuclear magnetic resonance diffusion spectroscopy of BT4C rat glioma during thymidine kinase-mediated gene therapy in vivo: identification of apoptotic response. Cancer Res. 58, 3791–3799 (1998).
S. Hortelano et al., Intracellular water motion decreases in apoptotic macrophages after caspase activation. Cell Death Differ. 8, 1022–1028 (2001).
J.M. Hakumäki et al., Early gene therapy-induced apoptotic response in BT4C gliomas by magnetic resonance relaxation contrast TI in the rotating frame, Cancer Gene Ther. 9, 338–345 (2002).
O.H.J. Gröhn et al., Novel magnetic resonance imaging contrasts for monitoring response to gene therapy in rat glioma, Cancer Res. 63, 7571–7574 (2003).
P.K. Valonen et al., Water diffusion in a rat glioma during ganciclovir-thymidine kinase gene therapy-induced programmed cell death in vivo: correlation with cell density, J Magn. Reson. Imaging 19, 389–396 (2004).
R. Weissleder et al., Superparamagnetic iron oxide: pharmacokinetics and toxicity, AJR Am JRoentgenol. 152, 167–173 (1989).
R. Weissleder et al., In vivo magnetic resonance imaging of transgene expression, Nature Med. 6, 351–355 (2000).
D. Artemov et al., Magnetic resonance molecular imaging of the HER-2/neu receptor, Cancer Res. 63, 2723–2727 (2003).
M. Zhao et al., Non-invasive detection of apoptosis using magnetic resonance imaging and a targeted contrast agent, Nature Med 7, 1241–1244 (2001).
E.A. Schellenberger et al., Annexin V-CLIO: a nanoparticle for detecting apoptosis by MRI, Mol Imaging 1, 102–107 (2002).
A. Flotats and I. Carrio, Non-invasive in vivo imaging of myocardial apoptosis and necrosis. Eur. J. Nucl. Med. Mol. Imaging 30, 615–30 (2003).
M. Doubrovin et al., Imaging transcriptional regulation of p53-dependent genes with positron emission tomography in vivo, Proc. Natl. Acad. Sci. USA. 98, 9300–9305 (2001).
Author information
Authors and Affiliations
Rights and permissions
Copyright information
© 2005 Springer
About this chapter
Cite this chapter
Hakumäki, J. (2005). Imaging of Apoptotic Cells in vivo. In: Application of Apoptosis to Cancer Treatment. Springer, Dordrecht. https://doi.org/10.1007/1-4020-3302-8_14
Download citation
DOI: https://doi.org/10.1007/1-4020-3302-8_14
Publisher Name: Springer, Dordrecht
Print ISBN: 978-1-4020-3303-2
Online ISBN: 978-1-4020-3302-5
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)