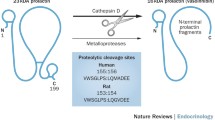
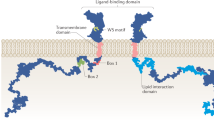
Abstract
Prolactin (PRL), synthesized by the anterior pituitary and to a lesser extent by numerous extrapituitary tissues, affects more physiological processes than all other pituitary hormones combined. This hormone is involved in >300 separate effects in various vertebrate species where its role has been well documented. The initial step in its action is the binding to a specific membrane receptor which belongs to the superfamily of class1cytokine receptors. The function of this receptor is mediated, at least in part, by two families of signaling molecules: Janus kinases and signal transducers and activators of transcription. PRL-binding sites have been identified in a number of cells and tissues of adult animals. Disruption of the gene for the PRL receptor has provided a new animal model with which to better understand the actions of PRL on mammary morphogenesis and mammary gland gene expression. The recent availability of genetic mouse models provides new insights into mammary developmental biology and how the action of a hormone at specific stages of development can have effects later in life on processes such as mammary development and breast cancer initiation and progression.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Neville, M.C. and Daniel, C.W., 1987, The Mammary Gland: Development regulation and function (New York: Plenum Press), pp 383–438.
Li, C.H., Dixon, J.S., Lo, T.B., Pankov, Y.M., and Schmidt, K.D., 1969, Amino acid sequence of ovine lactogenic hormone. Nature 224: 695–696.
Nicoll, C.S., Mayer, G.L., and Russell, S.M., 1986, Structural features of prolactins and growth hormones that can be related to their biological properties. Endocr. Rev. 7: 169–203.
Ben-Jonathan, N., Mershon, J.L., Allen, D.L., and Steinmetz, R.W., 1996, Extrapituitary prolactin: distribution, regulation, functions, and clinical aspects. Endocr. Rev. 17: 639–669.
Kelly, M.A., Rubinstein, M., Asa, S.L., Zhang, G., Saez, C., Bunzow, J.R., Allen, R.G., Hnasko, R., Ben-Jonathan, N., Grandy, D.K., and Low, M.J., 1997, Pituitary lactotroph hyperplasia and chronic hyperprolactinemia in dopamine D2 receptor-deficient mice. Neuron 19: 103–113.
Niall, H.D., Hogan, M.L., Sauer, R., Rosenblum, I.Y., and Greenwood, F.C., 1971, Sequences of pituitary and placental lactogenic and growth hormones: evolution from a primordial peptide by gene duplication. Proc. Natl. Acad. Sci. USA 68: 866–870.
Miller, W.L. and Eberhardt, N.L., 1983, Structure and evolution of the growth hormone gene family. Endocr. Rev. 4: 97–130.
Goffn, V., Shiverick, K.T., Kelly, P.A., and Martial, J.A., 1996, Sequence-function relationships within the expanding family of prolactin, growth hormone, placental lactogen and related proteins in mammals. Endocr. Rev. 17: 385–410.
Kacsoh, B., Veress, Z., Toth, B.E., Avery, L.M., and Grosvenor, C.E., 1993, Bioactive and immunoreactive variants of prolactin in milk and serum of lactating rats and their pups. J. Endocrinol. 138: 243–257.
Nagy, E. and Berczi, I., 1991, Hypophysectomized rats depend on residual prolactin for survival. Endocrinology 128: 2776–2784.
Imagawa, W., Yang, J., Guzman, R., and Nandi, S., 1994, Control of mammary development. In The Physiology of Reproduction. E. Knobil, J.D. Neil, L.L. Ewing, G.S. Greenwald, C.L. Markert, and D.W. Pfaff, eds. (New York: Raven Press), pp. 1033–1063.
Horseman, N.D., Zhao, W., Montecino-Rodriguez, E., Tanaka, M., Nakashima, K., Engle, S.J., Smith, F., Markoff, E., and Dorshkind, K., 1997, Defective mammopoiesis, but normal hematopoiesis, in mice with a targeted disruption of the prolactin gene. EMBO J. 16: 6926–6935.
Ormandy, C.J., Camus, A., Barra, J., Damotte, D., Lucas, B.K., Buteau, H., Edery, M., Brousse, N., Babinet, C., Binart, N., and Kelly, P.A., 1997, Null mutation of the prolactin receptor gene produces multiple reproductive defects in the mouse. Genes Dev. 11: 167–178.
Ormandy, C.J., Binart, N., and Kelly, P.A., 1997, Mammary gland development in prolactin receptor knockout mice. J. Mammary Gland. Biol. Neopl. 2: 355–364.
Brisken, C., Kaur, S., Chavarria, T.E., Binart, N., Sutherland, R.L., Weinberg, R.A., Kelly, P.A., and Ormandy, C.J., 1999, Prolactin controls mammary gland development via direct and indirect mechanisms. Dev. Biol. 210: 96–106.
Lydon, J.P., DeMayo, F. J., Funk, C.R., Mani, S.K., Hughes, C.A., Montgomery, C.A., Shyamala, G., Conneely, O.M., and O’Malley, B.W., 1995, Mice lacking progesterone receptor exhibit pleiotropic reproductive abnormalities. Genes Dev. 9: 2266–2278.
Korach, K.S., 1994, Insights from the study of animals lacking functional estrogen receptor. Science 266: 1524–1527.
Liu, X., Robinson, G.W., Wagner, K.U., Garrett, L., Wynshaw-Boris, A., and Hennighausen, L., 1997, Stat5a is mandatory for adult mammary gland development and lactogenesis. Genes Dev. 11: 179–186.
Udy, G.B., Towers, R.P., Snell, R.G., Wilkins, R.J., Park, S.H., Ram, P.A., Waxman, D.J., and Davey, H.W., 1997, Requirement of Stat5b for sexual dimorphism of body growth rates and liver gene expression. Proc. Natl. Acad. Sci. USA 94: 7239–7244.
Toscani, A., Mettus, R.V., Coupland, R., Simpkins, H., Litvin, J., Orth, J., Hatton, K.S., and Reddy, E.P., 1997, Arrest of spermatogenesis and defective breast development in mice lacking A-myb. Nature 386: 713–717.
Fantl, V., Stamp, G., Andrews, A., Rosewell, I., and Dickson, C., 1995, Mice lacking cyclin D 1 are small and show defects in eye and mammary gland development. Genes Dev. 9: 2364–2372.
Jones, F.E., Jerry, D.J., Guarino, B.C., Andrews, G.C., and Stem, D.F., 1996, Heregulin induces in vivo proliferation and differentiation of mammary epithelium into secretory lobuloalveoli. Cell Growth Differ. 7: 1031–1038.
Krane, I.M. and Leder, P., 1996, NDF/heregulin induces persistence of terminal end buds and adenocarcinomas in the mammary glands of transgenic mice. Oncogene 12: 1781–1788.
Yang, Y., Spitzer, E., Meyer, D., Sachs, M., Niemann, C., Hartmann, G., Weidner, K.M., Birchmeier, C., and Birchmeier, W., 1995, Sequential requirement of hepatocyte growth factor and neuregulin in the morphogenesis and differentiation of the mammary gland. J. Cell Biol. 131: 215–226.
Sicinski, P., Donaher, J.L., Parker, S.B., Li, T., Fazeli, A., Gardner, H., Haslam, S.Z., Bronson, R.T., Elledge, S.J., and Weinberg, R.A., 1995, Cyclin D1 provides a link between development and oncogenesis in the retina and breast. Cell 82: 621–630.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2002 Kluwer Academic Publishers
About this chapter
Cite this chapter
Binart, N., Ormandy, C.J., Kelly, P.A. (2002). Mammary Gland Development and the Prolactin Receptor. In: Mol, J.A., Clegg, R.A. (eds) Biology of the Mammary Gland. Advances in Experimental Medicine and Biology, vol 480. Springer, Boston, MA. https://doi.org/10.1007/0-306-46832-8_10
Download citation
DOI: https://doi.org/10.1007/0-306-46832-8_10
Publisher Name: Springer, Boston, MA
Print ISBN: 978-0-306-46414-0
Online ISBN: 978-0-306-46832-2
eBook Packages: Springer Book Archive