Abstract
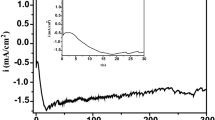
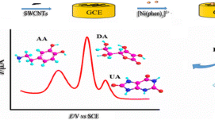
A glassy carbon electrode (GCE) modified by copper-doped titanium dioxide nanoneedles has been fabricated and used for the electrochemical detection of ascorbic acid (AA) in KCl solution. Two pairs of peak currents on quasi-reversible electrochemical cyclic voltammogram peaks (cvps) are located at +0.16 V,–0.03 V (cvp 1 and cvp 2) and +0.01 V,–0.44 V (cvp 1′ and cvp 2′), respectively. The relationship between the peak current and AA concentration is linear in the concentration range from 0.0005 to 2 mM. There is also a linear relationship between the peak current and the scan rate. The detection limit is 0.37 μM and 0.25 μM for cvp 1 and cvp 2, respectively, at a signal-to-noise ratio of 3. A GCE modified by copper-doped titanium dioxide nanoneedles exhibts good stability and has promising characteristics for the detection of AA.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Sun, H.W., Zang, C.M., and Lian, K.Q., Asian J. Pharm. Sci., 2009, vol. 4, pp. 200–206.
Jiang, L. and Jin, W.R., Chin. Chem. Lett., 2002, vol. 13, pp. 1087–1089.
Ardelean, M., Manea, F., Vaszilcsin, N., and Pode, R., Anal. Methods, 2014, vol. 6, pp. 4775–4782.
Jena, B.K. and Raj, C.R., Talanta, 2008, vol. 76, pp. 161–165.
Pandey, P.C., Upadhyay, B.C., and Upadhyay, A.K., Anal. Chim. Acta, 2004, vol. 523, no. 2, pp. 219–223.
Florou, A.B., Prodromidis, M.I., Karayannis, M.I., and Tzouwara-Karayanni, S.M., Anal. Chim. Acta, 2000, vol. 409, pp. 113–121.
Ozcan, L., Sahin, M., and Sahin, Y., Sensors, 2008, vol. 8, pp. 5792–5805.
Zhang, J., Deng, P.H., Feng, Y.L., Kuang, Y.F., et al., Microchim. Acta, 2004, vol. 147, pp. 279–282.
Xia, C. and Ning, W., Analyst, 2011, vol. 136, pp. 288–292.
Keeley, G.P., O’Neill, A., McEvoy, N., Peltekis, N., and Coleman, J.N., J. Mater. Chem., 2010, vol. 20, pp. 7864–7869.
Sun, D., Zhang, Y., Wang, F.R., Wu, K.B., et al., Sens. Actuators B, 2009, vol. 141, pp. 641–645.
Zhao, Y.Q., Bai, J.Y., Wang, L., Xu Hong, E., Huang, P.F., Wang, H.J., and Zhang, L.Y., Int. J. Electrochem. Sci., 2006, vol. 1, no. 7, pp. 363–371.
Pei, L.Z., Xie, Y.K., Cai, Z.Y., Yang, Y., Pei, Y.Q., Fan, C.G., and Fu, D.G., J. Electrochem. Soc., 2012, vol. 159, pp. K55–K60.
Pei, L.Z., Lin, N., Wei, T., Liu, H.D., and Yu, H.Y., J. Mater. Chem. A, 2015, vol. 3, no. 6, pp. 2690–2700.
Li, Y. and Zhan, S.H., J. Dispersion Sci. Technol., 2008, vol. 29, pp. 1421–1425.
Hager, G. and Brolo, A.G., J. Electroanal. Chem., 2009, vol. 625, pp. 109–116.
Davis, J., Moorcroft, M., Wilkins, S.J., Compton, R.G., et al., Analyst, 2000, vol. 125, pp. 737–742.
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
About this article
Cite this article
Pei, L.Z., Wei, T., Lin, N. et al. Analysis of ascorbic acid by electrochemical detection. Surf. Engin. Appl.Electrochem. 52, 398–402 (2016). https://doi.org/10.3103/S1068375516040104
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.3103/S1068375516040104