Abstract
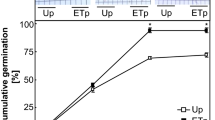
Changes in epigenetic polymorphism degree within wheat seedlings’ population under radiation stimulating exposure of germination were investigated. Variations in seeds sample allocations by germination rate in both control and chronically exposed variants were estimated. Changes in DNA methylation patterns of seedlings from seeds with different germination rate were studied. Variations in epigenetic polymorphism–“distance” between DNA methylation patterns of “fast” and “slow” seedlings of different wheat varieties under radiation exposure were assessed. Indicated, that increased germination rate of seeds sample was associated with decreasing degree of epigenetic polymorphism. Issue concerning both role of epigenetic polymorphism in plant population stability and its decreasing degree that could initiate less effectiveness of production process was discussed.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Prime-A-Plant Group, Conrath, U., Beckers, G.J., Flors, V., García-Agustin, P., Jakab, G., Mauch, F., Newman, M.A., Pieterse, C.M., Poinssot, B., Pozo, M.J., Pugin, A., Schaffrath, U., Ton, J., Wendehenne, D., Zimmerli, L., and Mauch-Mani, B., Priming: getting ready for battle, Mol. Plant-Microbe Interact., 2006, vol. 19, no. 10, pp. 1062–1071.
Ton, J., van der Ent, S., van Hulten, M.H.A., Pozo, M., van Oosten, V., van Loon, L.C., Mauch-Mani, B., Turlings, T.C.J., and Pieterse, C.M.J., Priming as a mechanism behind induced resistance against pathogens, insects and abiotic stress, IOBC wprs Bull., 2009, vol. 44, pp. 3–13.
Kuzin, A.M., Theories of pre-sowing seeds radiation exposure method, in Pre-Sowing Radiation Exposure of Crop Seeds, Moscow, 1963, 311 p.
Kaushanskiy, D.A., and Berezina, N.M., The Effectiveness of Pre-Sowing Seeds’ Radiation Exposure, Moscow, 1975, 91 p.
Lysikov, V.N., Application of Ionizing Exposure in Crop, Kishinev, 1975, 241 p.
Jakab, G., Ton, J., Flors, V., Zimmerli, L., Metra-ux, J.-P., and Mauch-Mani, B., Enhancing Arabidopsis salt and drought stress tolerance by chemical priming for its abscisic acid responses, Plant Physiol., 2005, vol. 139, no. 1, pp. 267–274.
Ton, J., Jakab, G., Toquin, V., Flors, V., Iavicoli, A., Maeder, M.N., Métraux, J.P., and Mauch-Mani, B., Dissecting the beta-aminobutyric acid-induced priming phenomenon in Arabidopsis, Plant Cell., 2005, vol. 17, no. 3, pp. 987–999.
Job, D., Capron, I., Job, C., Dacher, F., Corbineau, F., and Côme, D., Identification of germination-specific protein markers and their use in seed priming technology, in Seed Biology: Advances and Applications, Proc. 6th Intern. Workshop on Seeds, Wallingford, CAB Intern., 2000, pp. 449–459.
Gallardo, K., Job, C., Groot, S.P.C., Puype, M., Demol, H., Vandekerckhove, J., and Job, D., Proteomic analysis of Arabidopsis seed germination and priming, Plant Physiol., 2001, vol. 126, no. 2, pp. 835–848.
Bruce, J.A.T., Matthes, M.C., Napier, J.A., and Pickett, J.A., Stressful “memories” of plants: Evidence and possible mechanisms, Plant Sci., 2007, vol. 173, no. 6, pp. 603–608.
Sokolova, D.A., Vengzhen, G.S., and Kravets, A.P., The effect of DNA modification polymorphism of corn seeds on their germination rate, seedling resistance and adaptive capacity under UV-C exposure, Amer. J. Plant Biol., 2014, vol. 1, no. 1, pp. 1–14.
Sokolova, D.A., Vengzhen, G.S., and Kravets, A.P., Effect of epigenetic polymorphism of corn seeds on their germination rate and resistance of seedlings under UV-C exposure, Cytol. Genet., 2014, vol. 48, no. 4, pp. 227–232.
Sokolova, G.S., Vengzhen, D.A., and Kravets, A.P., Investigation of connection between epigenetic polymorphism of corn seeds and formation of seedlings adaptation to UV-C exposure, Plant Physiol. Genet., 2014, vol. 46, no. 3, pp. 221–229.
Kravets, O.P., Sokolova, D.O., Berestyana, A.M., Shnurenko, O.R., Bannikova, M.O., Morgun, B.V., Kuchuk, M.V., and Grodzinsky, D.M., Correlation between ecological plasticity of elite winter wheat varieties and DNA methylation pattern polymorphism within variety, Sci. Innov., 2016, vol. 12, no. 2, pp. 50–59.
Lakin, A.B., Biometry, Moscow, 1990, 352 p.
Ausubel, F.M., Current Protocols in Molecular Biology, Biophotometer—Operating Manual, 2004. http://www.eppendorf.com.
Bartlett, J.M.S., and Stirling, D., PCR Protocols. Methods in Molecular Biology, New York, 2003, 545 p.
Nei, A new measure of genetic distance, in Genetic Distance, J.F.Crow et al., Eds, New York, 1974, pp. 63–76.
Zhimulev, I.F., and Belyaeva, E.S., Heterochromatin, position effect and genetic silencing, Russ. J. Genet., 2003, vol. 39, no. 2, pp. 133–146.
Clark, D.J., Nucleosome positioning, nucleosome spacing and the nucleosome code, J. Biomol. Struct. Dyn., 2010, vol. 27, no. 6, pp. 781–793.
Teif, V.B., Shkrabkou, A.V., Egorova, V.P., and Krot, V.I., Nucleosomes in gene regulation: theoretical approach, Mol. Biol., 2010, vol. 46, no. 1, pp. 1–11.
Teif, V.B., Nucleosome positioning: resources and tools online, Brief. Bioinform., 2015, vol. 17, no. 5, pp. 745–757.
Zhang, Y.Y., Fisher, M., Colot, V., and Bossdorf, O., Epigenetic variation creates potential for evolution of plant phenotypic plasticity, New Phytol., 2013, vol. 197, no. 1, pp. 314–322.
Zhang, and Hsien, T.-F., Heritable epigenetic variation and its potential applications for crop improvement, Plant Breed. Biotechnol., 2013, vol. 1, pp. 307–319.
Kravets, O.P., Grodzinskiy, D.M., and Mazur, L.O., Cumulative effects of chronic radiation exposure of plants from Chernobyl exclusion zone, Scientific Papers of the Institute for Nuclear Research, 2005, vol. 7, no. 5, pp. 115–124.
Mirkin, B.M., Types of coenotic plant strategies, Biol. Bull. Rev., 1983, vol. 44, no. 5, pp. 603–613.
Zlobin, Y.A., Plant Population Ecology, Current Research Status, Growth Terms, Sumy, 2009, 263 p.
Becker, C., Hagmann, J., Müller, J., Koenig, D., Stegle, O., Borgwardt, K., and Weigel, D., Spontaneous epigenetic variation in the Arabidopsis thaliana methylome, Nature, 2011, vol. 480, pp. 245–249.
Salmon, A., Clotault, J., Jenczewski, E., Chable, V., and Manzanares-Dauleux, M.J., Brassica oleracea displays a high level of DNA methylation polymorphism, Plant Sci., 2008, vol. 174, no. 1, pp. 61–70.
Landan, G., Cohen, N.M., Mukamel, Z., Bar, A., Molchadsky, A., Brosh, R., Horn-Saban, S., Zalcenstein, D.A., Goldfinger, N., Zundelevich, A., Gal-Yam, E.N., Rotter, V., and Tanay, A., Epigenetic polymorphism and the stochastic formation of differentially methylated regions in normal and cancerous tissues, Nat. Genet., 2012, vol. 44, no. 11, pp. 1207–1214.
Kravets, A.P., Vengzhen, G.S., and Grodzinsky, D.M., Distant interaction between exposed and non-exposed with radiation plants, Radiation biology. Radioecology, 2009, vol. 49, no. 4, pp. 561–570.
Kravets, A.P., Gatilova, G.D., and Grodzinsky, D.M., Dynamics of cytogenetical abnormalities in seedlings’ meristems under chronic radiation exposure of seeds, Radiat. Biol. Radioecol., 2008, vol. 48, no. 3, pp. 208–219.
Tornly, J.G.M., Mathematics in Plant Physiology, Kiev, 1982, 302 p.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © O.P. Kravets, D.O. Sokolova, 2017, published in Tsitologiya i Genetika, 2017, Vol. 51, No. 5, pp. 30–37.
About this article
Cite this article
Kravets, O.P., Sokolova, D.O. Epigenetic mechanisms regulating seed germination rate. Cytol. Genet. 51, 346–351 (2017). https://doi.org/10.3103/S0095452717050048
Received:
Published:
Issue Date:
DOI: https://doi.org/10.3103/S0095452717050048