Abstract
Docetaxel (Taxotere®), a semi-synthetic analog of paclitaxel (Taxol®), is a promoter of microtubule polymerization leading to cell cycle arrest at G2/M, apoptosis and cytotoxicity. Docetaxel has significant activity in breast, non-small-cell lung, ovarian and head and neck cancers.
Docetaxel has undergone phase I study in a number of schedules, including different infusion durations and various treatment cycles. Doses studied in adults have ranged from 5 to 145 mg/m2 and those in children from 55 to 235 mg/m2. The most frequently used regimen in adults is 100 mg/m2 every 3 weeks.
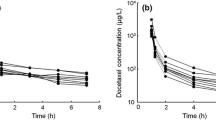
A 1-hour infusion every 3 weeks has been favoured in phase II and III studies, and the disposition of docetaxel after such treatment is best described by a 3 compartment model with α, β and γ half-lives of 4.5 minutes, 38.3 minutes and 12.2 hours, respectively. The disposition of docetaxel appears to be linear, the area under the plasma concentration-time curve (AUC) increasing proportionately with dose.
Docetaxel is widely distributed in tissues with a mean volume of distribution of 74 L/m2 after 100 mg/m2, every 3 weeks. The mean total body clearance after this schedule is approximately 22 L/h/m2, principally because of hepatic metabolism by the cytochrome P450 (CYP)3A4 system and biliary excretion into the faeces. Renal excretion is minimal (<5%). Docetaxel is >90% bound in plasma.
Population pharmacokinetic studies of docetaxel have demonstrated that clearance is significantly decreased with age, decreased body surface area, increased concentrations of α1-acid glycoproteinand albumin. Importantly, patients with elevated plasma levels of bilirubin and/or transaminases have a 12 to 27% decrease in docetaxel clearance and should receive reduced doses.
Although docetaxel is metabolised by CYP3A4, phase I combination studies have not shown major evidence of significant interaction between docetaxel and other drugs metabolised by the same pathway. Nevertheless, care should be taken with the use of known CYP3A4 inhibitors such as erythromycin, ketoconazole and cyclosporin. Conversely, increased doses may be required for patients receiving therapy known to induce this cytochrome (e.g. anticonvulsants).
Preliminary data suggest the erythromycin breath test, an indicator of CYP3A4 function, is a predictor of toxicity after treatment with docetaxel. Such methodologies may eventually enable clinicians to individualise doses of docetaxel for patients with cancer.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Cortes JE, PazdurR. Docetaxel. J Clin Oncol 1995; 13: 2643–55.
Wani MC, Taylor HL, Wall ME, et al. Plant antitumor agents. VI: the isolation and structure of taxol, a novel antileukemic and antitumor agent from Taxus brevifolia. J Am Chem Soc 1971; 93: 2325–7.
Schiff PB, Fant J, Horwitz SB. Promotion of microtubule assembly in vitro by taxol. Nature 1979; 277: 665–7.
Bissery MC. Preclinical pharmacology of docetaxel. Eur J Cancer 1995; 4: S1–6.
Lavelle F, Bissery MC, Combeau C, et al. Preclinical evaluation of docetaxel (Taxotere). Semin Oncol 1995; 22: 3–16.
Ringel I, Horwitz SB. Studies with RP 56976 (taxotere): a semisynthetic analogue of taxol. J Natl Cancer Inst 1991; 83: 288–91.
Commercon A, Bourzat J, Bouchard H, et al. Synthesis and biological properties of docetaxel analogs modified on the side chain [abstract]. Proc Am Assoc Cancer Res 1994; 35: 2383.
Extra JM, Rousseau F, Bruno R, et al. Phase I and pharmacokinetic study of Taxotere (RP 56976; NSC 628503) given as a short intravenous infusion. Cancer Res 1993; 53: 1037–42.
Aapro M, Bruno R, Docetaxel Investigators Group. Early clinical studies with docetaxel. Eur J Cancer 1995; 4: S7–10.
Fumoleau P, Tubiana-Hulin M, Soulie P, et al. A dose finding and pharmacokinetic (PK) study of a new formulation of docetaxel (D) in advanced solid tumors. 10th NCI-EORTC Symposium on New Drugs in Cancer Therapy; 1998 Jun 16–18; Amsterdam.
Rosing H, Lustig V, Koopman FP, et al. Bio-analysis of docetaxel and hydroxylated metabolites in human plasma by high-performance liquid chromatography and automated solid-phase extraction. J Chromatogr B Biomed Sci Appl 1997; 696: 89–98.
Bissery M, Renard A, Montay G, et al. Taxotere: antitumor activity and pharmacokinetics in mice. Proc Am Assoc Cancer Res 1991; 32: 2386.
Vergniol JC, Bruno R, Montay G, et al. Determination of Taxotere in human plasma by a semi-automated high-performance liquid chromatographic method. J Chromatogr 1992; 582: 273–8.
Loos WJ, Verweij J, Nooter K, et al. Sensitive determination of docetaxel in human plasma by liquid-liquid extraction and reversed-phase high-performance liquid chromatography. J Chromatogr B Biomed Appl 1997; 693: 437–41.
Rangel C, Niell H, Miller A, et al. Taxol and taxotere in bladder cancer: in vitro activity and urine stability. Cancer Chemother Pharmacol 1994; 33: 460–4.
Bissery MC, Nohynek G, Sanderink GJ, et al. Docetaxel (Taxotere): a review of preclinical and clinical experience. Pt I: preclinical experience. Anticancer Drugs 1995; 6: 339–55.
Sparreboom A, van Tellingen O, Nooijen WJ, et al. Preclinical pharmacokinetics of paclitaxel and docetaxel. Anticancer Drugs 1998; 9: 1–17.
Gaillard C, Monsarrat B, Vuilhorgne M, et al. Docetaxel (Taxotere) metabolism in the rat in vivo and in vitro. Proc Am Assoc Cancer Res 1994; 35: 428.
Marlard M, Gaillard C, Sanderink G, et al. Kinetics, distribution, metabolism and excretion of radiolabelled taxotere (14C-RP56976) in mice and dogs. Proc Am Assoc Cancer Res 1993; 34: 2343.
Wils P, Phung Ba V, Warnery A, et al. Polarized transport of docetaxel and vinblastine mediated by P-glycoprotein in human intestinal epithelial cell monolayers. Biochem Pharmacol 1994; 48: 1528–30.
Monsarrat B, Alvinerie P, Wright M, et al. Hepatic metabolism and biliary excretion of Taxol in rats and humans. J Natl Cancer Inst Monogr 1993; 15: 39–46.
Commercon A, Bourzat J, Bezard D, et al. Partial synthesis of major human metabolites of docetaxel. Tetrahedron 1994; 50: 10289–98.
Monegier B, Gaillard C, Sable S, et al. Structures of the major human metabolites of docetaxel (RP 56976 — Taxotere®). Tetrahedron Lett 1994; 35: 3715–8.
Royer I, Monsarrat B, Sonnier M, et al. Metabolism of docetaxel by human cytochromes P450: interactions with paclitaxel and other antineoplastic drugs. Cancer Res 1996; 56: 58–65.
Marre F, Sanderink GJ, de Sousa G, et al. Hepatic biotransformation of docetaxel (Taxotere) in vitro: involvement of the CYP3A subfamily in humans. Cancer Res 1996; 56: 1296–302.
Monsarrat B, Royer I, Wright M, et al. Biotransformation of taxoids by human cytochromes P450: structure-activity relationship. Bull Cancer 1997; 84: 125–33.
Sparreboom A, Van Tellingen O, Scherrenburg EJ, et al. Isolation, purification and biological activity of major docetaxel metabolites from human feces. Drug Metab Dispos 1996; 24: 655–8.
Urien S, Barre J, Morin C, et al. Docetaxel serum protein binding with high affinity to alpha 1-acid glycoprotein. Invest New Drugs 1996; 14: 147–51.
de Valeriola D, Brassine C, Gaillard C, et al. Study of excretion balance, metabolism and protein binding of C14 radiolabelled taxotere (TXT) (RP56976, NSC628503) in cancer patients. Proc Am Assoc Cancer Res 1993; 34: 2221.
Burris H, Irvin R, Kuhn J, et al. Phase I clinical trial of taxotere administered as either a 2-hour or 6-hour intravenous infusion. J Clin Oncol 1993; 11: 950–8.
Bissett D, Setanoians A, Cassidy J, et al. Phase I and pharmacokinetic study of taxotere (RP 56976) administered as a 24-hour infusion. Cancer Res 1993; 53: 523–7.
Pazdur R, Newman RA, Newman BM, et al. Phase I trial of Taxotere: five-day schedule. J Natl Cancer Inst 1992; 84: 1781–8.
Tomiak E, Piccart MJ, Kerger J, et al. Phase I study of docetaxel administered as a 1-hour intravenous infusion on a weekly basis. J Clin Oncol 1994; 12: 1458–67.
Hainsworth J, Burris III H, Erland J, et al. Phase I trial of docetaxel administered by weekly infusion in patients with advanced refractory cancer. J Clin Oncol 1998; 16: 2164–8.
Blaney SM, Seibel NL, O’Brien M, et al. Phase I trial of docetaxel administered as a 1-hour infusion in children with refractory solid tumors: a collaborative pediatric branch, National Cancer Institute and Children’s Cancer Group trial. J Clin Oncol 1997; 15: 1538–43.
McLeod H, Kearns C, Kuhn J, et al. Evaluation of the linearity of docetaxel pharmacokinetics. Cancer Chemother Pharmacol 1998; 42: 155–9.
Launay Iliadis MC, Bruno R, Cosson V, et al. Population pharmacokinetics of docetaxel during phase I studies using nonlinear mixed-effect modeling and nonparametric maximum-likelihood estimation. Cancer Chemother Pharmacol 1995; 37: 47–54.
Bruno R, Riva A, Hille D, et al. Pharmacokinetic and pharmacodynamic properties of docetaxel: results of phase I and phase II trials. Am J Health Syst Pharm 1997; 54: S16–9.
Bruno R, Vivier N, Vergniol JC, et al. A population pharmacokinetic model for docetaxel (Taxotere): model building and validation. J Pharmacokinet Biopharm 1996; 24: 153–72.
Bruno R, Hille D, Thomas L, et al. Population pharmacokinetics/pharmacodynamics (PK/PD) of docetaxel (Taxotere) in phase II studies [abstract]. Proc Am Soc Clin Oncol 1995; 14: 457.
Gurney H. Dose calculation of anticancer drugs: a review of the current practice and introduction of an alternative. J Clin Oncol 1996; 14: 2590–611.
Bruno R, Hille D, Riva A, et al. Population pharmacokinetics/pharmacodynamics of docetaxel in phase II studies in patients with cancer. J Clin Oncol 1998; 16: 187–96.
Tanigawara Y, Sasaki Y, Otsu T, et al. Population pharmacokinetics of docetaxel in Japanese patients [abstract no. 1518]. Proc Am Soc Clin Oncol 1996; 15: 479.
Oulid Aissa D, Bruno R, Lebecq A, et al. Taxotere safety in patients with impaired liver function (LF) [abstract no. 1508]. Proc Am Soc Clin Oncol 1996; 15: 476.
Baker S, Ravdin P, Aylesworth C, et al. A phase I and pharmacokinetic (PK) study of docetaxel in cancer patients with liver dysfunction due to malignancies. 10th NCI-EORTC Symposium on New Drugs in Cancer Therapy; 1998 Jun 16–18; Amsterdam.
Hudis CA, Seidman AD, Crown JP, et al. Phase II and pharmacologic study of docetaxel as initial chemotherapy for metastatic breast cancer. J Clin Oncol 1996; 14: 58–65.
Baker L, Hirth J, Carson E, et al. Cytochrome P4503A activity as a predictor of docetaxel toxicity and response [abstract]. Proc Am Soc Clin Oncol 1998; 17: 718.
Klink-Alakl M, Riva A, Bruno R, et al. Taxotere (T) safety profile in patients (pts) with liver metastases (LM) with or without impaired liver function (ILF) [abstract]. Proc Am Soc Clin Oncol 1997; 16: 220a.
Seibel NI, Blaney SM, O’Brien M, et al. Pediatric phase I trial of docetaxel (D) with G-CSF: a collaborative pediatric branch, NCI and Children’s Cancer Group trial [abstract]. Proc Am Soc Clin Oncol 1997; 16: 220a.
LeBlanc GA, Sundseth SS, Weber GF, et al. Platinum anticancer drugs modulate P-450 mRNA levels and differentially alter hepatic drug and steroid hormone metabolism in male and female rats. Cancer Res 1992; 52: 540–7.
LeBlanc GA, Waxman DJ. Feminization of rat hepatic P-450 expression by cisplatin: evidence for perturbations in the hormonal regulation of steroid-metabolizing enzymes. J Biol Chem 1988; 263: 15732–9.
Rowinsky EK, Gilbert MR, McGuire WP, et al. Sequences of taxol and cisplatin: a phase I and pharmacologic study. J Clin Oncol 1991; 9: 1692–703.
Huizing MT, van Warmerdam LJ, Rosing H, et al. Phase I and pharmacologic study of the combination paclitaxel and carboplatin as first-line chemotherapy in stage III and IV ovarian cancer. J Clin Oncol 1997; 15: 1953–64.
Pronk LC, Schellens JH, Planting AS, et al. Phase I and pharmacologie study of docetaxel and cisplatin in patients with advanced solid tumors. J Clin Oncol 1997; 15: 1071–9.
Millward MJ, Zalcberg J, Bishop JF, et al. Phase I trial of docetaxel and cisplatin in previously untreated patients with advanced non-small-cell lung cancer. J Clin Oncol 1997; 15: 750–8.
de Vos AI, Nooter K, Verweij J, et al. Differential modulation of cisplatin accumulation in leukocytes and tumor cell lines by the paclitaxel vehicle Cremophor EL. Ann Oncol 1997; 8: 1145–50.
Ma J, Verweij J, Planting AS, et al. Docetaxel and paclitaxel inhibit DNA-adduct formation and intracellular accumulation of cisplatin in human leukocytes. Cancer Chemother Pharmacol 1996; 37: 382–4.
Webster LK, Linsenmeyer ME, Rischin D, et al. Plasma concentrations of polysorbate 80 measured in patients following administration of docetaxel or etoposide. Cancer Chemother Pharmacol 1997; 39: 557–60.
Sparreboom A, van Tellingen O, Nooijen WJ, et al. Nonlinear pharmacokinetics of paclitaxel in mice results from the pharmaceutical vehicle Cremophor EL. Cancer Res 1996; 56: 2112–5.
Webster LK, Cosson EJ, Stokes KH, et al. Effect of the paclitaxel vehicle, Cremophor EL, on the pharmacokinetics of doxorubicin and doxorubicinol in mice. Br J Cancer 1996; 73: 522–4.
D’Incalci M, Colombo T, Zucchetti M, et al. Pharmacokinetic (PK) interaction between taxanes and anthracyclines: in-vivo study in mice [abstract]. Proc Am Soc Clin Oncol 1998; 17: 239.
Ellis AG, Crinis NA, Webster LK. Inhibition of etoposide elimination in the isolated perfused rat liver by Cremophor EL and Tween 80. Cancer Chemother Pharmacol 1996; 38: 81–7.
Chang S, Kuhn J, Rizzo J, et al. Phase I study of paclitaxel in patients with recurrent malignant glioma: a North American Brain Tumor Consortium report. J Clin Oncol 1998; 16: 2188–94.
Adis International. Taxotere (docetaxel) international product monograph. Auckland: Adis, 1996.
Watanabe K, Hayashi I, Hiraki S, et al. Phase I/I dose escalation study of docetaxel and cisplatin for stage IV non-small cell lung cancer [abstract]. Proc Am Soc Clin Oncol 1997; 16: 254.
Llombart-Cussac A, Pivot X, Rixe O, et al. Docetaxel plus cisplatin: an active regimen in patients with anthracycline-resistant breast cancer (ARBC): definitive results of a phase I/II study [abstract]. Proc Am Soc Clin Oncol 1998; 17: 154.
Baille P, Vernillet L, Vergniol O, et al. Docetaxel (D) pharmacokinetics (PK): influence of coadministered drugs in phase I combination studies [abstract]. Proc Am Soc Clin Oncol 1998; 17: 189.
de Valeriola D, Brassinne C, Kerger J, et al. Clinical and pharmacokinetic phase I study of docetaxel (TXT) combined to continuous infusion (CI) of 5-fluorouracil (FU) in advanced cancer patients [abstract]. Proc Am Soc Clin Oncol 1997; 16: 221.
Sano M, Adachi I, Watanabe T, et al. Aphase I/II study of docetaxel (TXT) in combination with continuous infusion of fluorouracil (5-FU) in patients with advanced or recurrent breast cancer [abstract]. Proc Am Soc Clin Oncol 1997; 16: 189.
Itoh K, Fujiii H, Minami H, et al. Phase I and pharmacological study of docetaxel combined with doxorubicin for advanced breast cancer [abstract]. Proc Am Soc Clin Oncol 1997; 16: 174.
Schuller J, Czejka M, Kletzl H, et al. Doxorubicin (DOX) and Taxotere (TXT): a pharmacokinetic (PK) study of the combination in advanced breast cancer [abstract]. Proc Am Soc Clin Oncol 1998; 17: 205.
Malik U, Sparano J, Wolffe A. Phase I trial of liposomal doxorubicin (Doxil) and docetaxel (taxotere) in patients (pts) with advanced breast cancer (ABC) [abstract]. Proc Am Soc Clin Oncol 1998; 17: 175.
Vasey P, Atkinson R, Coleman R, et al. Preliminary report of a dose-finding study of a docetaxel-carboplatin combination in untreated advanced epithelial ovarian cancer (EOC) [abstract]. Proc Am Soc Clin Oncol 1998; 17: 349.
Belani C, Hadeed V, Ramanathan R, et al. Docetaxel and carboplatin: a phase I and pharmacokinetic trial for advanced non-haematological malignancies [abstract]. Proc Am Soc Clin Oncol 1997; 16: 220.
Langer C, McAleer C, Millenson M, et al. Phase I evaluation of docetaxel and carboplatin in advanced malignancy: stratification of patient cohorts based on prior treatment (tx) status yields useful results [abstract]. Proc Am Soc Clin Oncol 1998; 17: 204.
Giannakakis T, Papadouris S, Ziras N, et al. Phase I/II study of docetaxel and carboplatin as first line chemotherapy in advanced non-small cell lung cancer: preliminary results [abstract]. Proc Am Soc Clin Oncol 1998; 17: 486.
Pronk L, Vasey A, Sparreboom A, et al. Amatrix-designed phase I dose-finding and pharmacokinetic study of the combination of Xeloda plus taxotere [abstract]. Proc Am Soc Clin Oncol 1998; 17: 212.
Tkaczuk K, Brooks S, Fedenko K, et al. Phase I trial of docetaxel (Dotax) and topotecan hydrochloride (Topo) in patients with advanced solid tumours [abstract]. Proc Am Soc Clin Oncol 1998; 17: 254.
Spiridonidis C, Laufman L, Jones J, et al. Weekly gemcitabine (GEM) combined with monthly docetaxel (Doc) in patients (pts) with advanced malignancies: a phase I trial [abstract]. Proc Am Soc Clin Oncol 1998; 17: 206.
Schlosser N, Richel D, van Zandwijk N, et al. Phase I study of docetaxel and gemcitabine combination chemotherapy in chemotherapy naive patients with advanced or metastatic non small cell lung cancer [abstract]. Proc Am Soc Clin Oncol 1998; 17: 499.
Davis T, Rigas J, Maurer L, et al. Unique two week schedule of docetaxel and short infusion gemcitabine for advanced solid tumors: preliminary results of a phase I trial [abstract]. Proc Am Soc Clin Oncol 1998; 17: 239.
Eckardt J, Schmidt A, Needles B, et al. A phase I study of the combination of docetaxel (D) and gemcitabine (G) [abstract]. Proc Am Soc Clin Oncol 1998; 17: 240.
Shirazi F, Bahrami G, Fanaee G, et al. Pharmacokinetic interactions of docetaxel and etoposide [abstract]. Proc Am Soc Clin Oncol 1998; 17: 252.
Valero V. Docetaxel and cyclophosphamide in patients with advanced solid tumors. Oncology Huntingt 1997; 11: 21–3.
Pronk LC, Schrijvers D, Schellens JH, et al. Phase I study on docetaxel and ifosfamide in patients with advanced solid tumours. Br J Cancer 1998; 77: 153–8.
Taub R, Keohan M, Fine R, et al. Phase I/II study of combined estramustine/taxotere in advanced sarcomas [abstract]. Proc Am Soc Clin Oncol 1998; 17: 524.
Kudoh S, Negoro S, Masuda N, et al. Phase I/II study of docetaxel (Doc) and Irinotecan (CPT-11) for previously untreated advanced non-small cell lung cancer [abstract]. Proc Am Soc Clin Oncol 1998; 17: 491.
Couteau C, Lokiec F, Vernillet L, et al. Phase I dose-finding and pharmacokinetic (PK) study of docetaxel (D) in combination with irinotecan (I) in advanced solid tumours [abstract]. Proc Am Soc Clin Oncol 1997; 16: 708.
Alexopoulos A, Kouroussis C, Kakolyris S, et al. A phase I/II study of docetaxel (D) and mitoxantrone (M) in metastatic breast cancer (MBC) [abstract]. Proc Am Soc Clin Oncol 1998; 17: 126.
Fumoleau P, Fety R, Delecroix V, et al. Docetaxel combined with vinorelbine: phase I results and new study designs. Oncology (Huntingt) 1997; 11 (6 Suppl. 6): 29–31.
Miller VA. Docetaxel (Taxotere) and vinorelbine in the treatment of advanced non-small cell lung cancer: preliminary results of a phase I/II trial. Semin Oncol 1997; 24 (4 Suppl. 14): S14/15–S14/17.
Trudeau M, Crump M, Latreille J, et al. Escalating doses of docetaxel and epirubicin as first line therapy for metastatic breast cancer: a phase I study of the National Cancer Institute of Canada Clinical Trials Group [abstract]. Proc Am Soc Clin Oncol 1998; 17: 178.
Bruno R, Olivares R, Berille J, et al. Alpha-1-acid glycoprotein is an independent predictor of efficacy and survival in NSCLC patients treated with docetaxel (taxotere) [abstract]. Proc Am Soc Clin Oncol 1998; 18: 1812.
Seidman AD, Hochhauser D, Gollub M, et al. Ninety-six-hour paclitaxel infusion after progression during short taxane exposure: a phase II pharmacokinetic and pharmacodynamic study in metastatic breast cancer. J Clin Oncol 1996; 14: 1877–84.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Clarke, S.J., Rivory, L.P. Clinical Pharmacokinetics of Docetaxel. Clin Pharmacokinet 36, 99–114 (1999). https://doi.org/10.2165/00003088-199936020-00002
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003088-199936020-00002