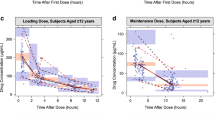
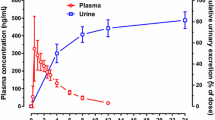
Abstract
Hydroxyurea is used in the treatment of various forms of cancer, sickle-cell anaemia and HIV infection. Oral absorption of the drug is virtually complete, the volume of distribution is equivalent to total body water and elimination is through both renal and nonrenal mechanisms. Nonrenal elimination of hydroxyurea is characterised by Michaelis-Menten kinetics.
Further studies are necessary to clarify several aspects of the pharmacokinetics and pharmacodynamics of hydroxyurea: the effect of age and disease state, concentration-effect relationship, the role of therapeutic drug monitoring, and the mechanisms of renal and nonrenal elimination. The recent development of improved assays for hydroxyurea should have benefits for future pharmacokinetic studies.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Dresler WFC, Stein R. Uber den hydroxhylarnstoff. Justus Liebigs Ann Chem Pharm. 1869; 150: 242–52.
Rosenthal F, Wislicki L, Kollek L. Uber die Beziehungen von schwersten Blutgiften zu Abbauprodukten des Eiweisses: ein Betrag zum Entstehungsmechanismus der perniziosen Anamie. Klin Wochenschr. 1928; 7: 972–7.
Stearns B, Losee KA, Bernstein J. Hydroxyurea: a new type of potential antitumor agent. J Med Chem. 1963; 6: 201.
Yarbro JW, Kennedy BJ, Barnum CP. Hydroxyurea inhibition of DNA synthesis in ascites tumor. Proc Natl Acad Sci U S A. 1965; 53: 1033–5.
Yarbro JW. Mechanism of action of hydroxyurea. Semin Oncol 1992; 19 (3 Suppl. 9): 1–10.
Krakoff IH, Brown NC, Reichard P. Inhibition of ribonucleoside diphosphate reductase by hydroxyurea. Cancer Res. 1968; 28: 1559–65.
Wright JA, Chan AK, Choy BK, et al. Regulation and drug resistance mechanisms of mammalian ribonucleotide reductase, and the significance to DNA sysnthesis. Biochem Cell Biol. 1990; 68(12): 1364–71.
Stubbe J. Ribonucleotide reductases. Adv Enzymol Relat Areas Mol Biol. 1990; 63: 3494–19.
Lassmann G, Liermann B. ESR studies of structure and kinetics of radicals from hydroxyurea: an antitumor drug directed against ribonucleotide reductase. Free Radic Biol Med. 1989; 6(3): 241–4.
Graslund A, Ehrenberg A, Thelander L. Characterization of the free radical of mammalian ribonucleotide reductase. J Biol Chem. 1982; 257(10): 5711–5.
Elford HL, Wampler GL, vant Riet B. New ribonucleotide reductase inhibitors with antineoplastic activity. Cancer Res. 1979; 39: 844–51.
Davidson JD, Winter TS. A method of analyzing for hydroxyurea in biological fluids. Cancer Chemother Rep. 1963; 27: 97–110.
Philips FS, Sternberg SS, Schwartz HS, et al. Hydroxyurea I. Acute cell death in proliferating tissues in rats. Cancer Res. 1967; 27: 61–75.
Fabricius E, Rajewsky MF. Determination of hydroxyurea in mammalian tissues and blood. Rev Eur Etud Clin Biol. 1971; 16(7): 679–83.
Tracewell WG, Trump DL, Vaughan WP, et al. Population pharmacokinetics of hydroxyurea in cancer patients. Cancer Chemother Pharmacol. 1995; 35(5): 417–22.
Veale D, Cantwell BMJ, Kerr N, et al. Phase I study of hydroxyurea in lung cancer. Cancer Chemother Pharmacol. 1988; 21(1): 53–6.
Havard J, Grygiel J, Sampson D. Determination by high-performance liquid chromatography of hydroxyurea in human plasma. J Chromatogr. 1992; 584: 27–4.
Villani P, Maserati R, Regazzi MB, et al. Pharmacokinetics of hydroxyurea in patients infected with human immunodeficiency virus type 1. J Clin Pharmacol. 1996; 36: 117–21.
Pujari MP, Barrientos A, Muggia FM, et al. Determination of hydroxyurea in plasma and peritoneal fluid by high-performance liquid chromatography using electrochemical detection. J Chromatogr B Biomed Sci Appl. 1997; 694(1): 185–91.
El-Yazigi A, Al-Rawithi S. Analysis of hydroxyurea in capsules and aqueous solution and stability study with capillary gas chromatography and therminonic (N-P) detection. Pharm Res. 1992; 9(1): 115–8.
Evered DF, Selhi HS. Transport characteristics of two carcinostatic compounds, hydroxyurea and hadacidin, with rat small intestine [abstract]. Biochem J. 1972; 126: 26P.
Adamson RH, Ague SL, Hess SM, et al. The distribution, excretion, and metabolism of hydroxyurea-14C. J Pharmacol Exp Ther. 1965; 150(2): 322–4.
Tracewell WG, Vaughan WP, Gwilt PR. Pharmacokinetics of hydroxyurea in the rat [abstract]. Pharm Res 1992; 9 Suppl.: S260.
Belt RJ, Haas CD, Kennedy J, et al. Studies of hydroxyurea administered by continuous infusion: toxicity, pharmacokinetics and cell synchronization. Cancer. 1980; 46(3): 455–62.
Morgan JS, Creasey DC, Wright JA. Evidence that the antitumor agent hydroxyurea enters mammalian cells by a diffusion mechanism. Biochem Biophys Res Commun. 1986; 134(3): 1254–9.
Blasberg RG, Patlack C, Fenstermacher JD. Intrathecal chemotherapy: brain tissue profiles after ventriculocisternal perfusion. J Pharmacol Exp Ther. 1975; 195: 73–83.
Beckloff GL, Lerner HJ, Frost D, et al. Hydroxyurea (NSC-32065) in biologic fluids: dose-concentration relationship. Cancer Chemother Rep. 1965; 48: 57–8.
Colvin M, Bono VH. The enzymatic reduction of hydroxyurea to urea by mouse liver. Cancer Res. 1970; 30(5): 1516–9.
Andrae U. Evidence for the involvement of cytochrome P-450 dependent monooxygenase (S) in the formation of genotoxic metabolites from N-hydroxyurea. Biochem Biophys Res Commun. 1984; 118(2): 409–15.
Fishbein WN, Carbone PP. Hydroxyurea: mechanism of action. Science. 1963; 142: 1069–70.
Van Den Berg CL, McGill JR, Kuhn JG, et al. Pharmacokinetics of hydroxyurea in nude mice. Anticancer Drugs. 1994; 5(5): 573–8.
Creasey WA, Capizzi RL, DeConti RC. Clinical and biochemical studies of high-dose intermittent therapy of solid tumors with hydroxyurea (NSC-32065). Cancer Chemother Rep. 1970; 54(3): 191–4.
Newman EM, Carroll M, Akman SA, et al. Pharmacokinetics and toxicity of 120-hour continuous-infusion hydroxyurea in patients with advanced solid tumors. Cancer Chemother Pharmacol. 1997; 39: 254–8.
Charache S, Dover GJ, Moore RD, et al. Hydroxyurea: effects on hemoglobin F production in patients with sickle cell anemia. Blood. 1992; 79(10): 2555–65.
Collins JM. Pharmacological rationales for regional drug delivery. J Clin Oncol. 1984; 2: 498–504.
Guyton AC. Textbook of medical physiology. 8th ed. Philadelphia: WB Saunders, 1991: 186.
Schilsky RL, Ratain JM, Vokes EE, et al. Laboratory and clinical studies of biochemical modulation by hydroxyurea. Semin Oncol 1992; 19 (3 Suppl. 9): 84–9.
Rauscher F, Cadman E. Biochemical and cytokinetic modulation of L1210 and HL-60 cells by hydroxyurea and effect on 1-β-D-arabinofuranosylcytosine metabolism and cytotoxicity. Cancer Res. 1983; 43: 2688–93.
Kubota M, Takimoto T, Tanizawa A, et al. Differential modulation of 1-beta-D-arabinofuranosylcytosine metabolism by hydroxyurea in human leukemic cell lines. Biochem Pharmacol. 1988; 37(9): 1745–9.
Schilsky RL, Williams SF, Ultmann JE, et al. Sequential hydroxyurea-cytarabine chemotherapy for refractory non-Hodgkins lymphoma. J Clin Oncol. 1987; 5(3): 419–25.
DeGramont A, Louvet C, Varette C, et al. Reversal of resistance to high-dose folinic acid (LV) and 5-fluorouracil (5-FU) in metastatic colorectal cancer by hydroxyurea [abstract]. Proc Am Soc Clin Oncol. 1992; 11: 178.
Heidelberger C, Danenberg PV, Moran RG. Fluorinated pyrimidines and their nucleosides. Adv Enzymol Relat Areas Mol Biol. 1983; 54: 58–119.
Li JC, Kaminskas E. Progressive formulation of DNA lesions in cultured Ehrlich ascites tumor cells treated with hydroxyurea. Cancer Res. 1987; 47(11): 2755–8.
Minford J, Kerrigan D, Nichols M, et al. Enhancement of the DNA breakage and cytotoxic effects of intercalating agents by treatment with sublethal doses of 1-β-D-arabinofuranosylcytosine or hydroxyurea in L1210 cells. Cancer Res 1984; 44 (12 Pt 1): 5583–93.
Ratain MJ, Schilsky RL, Wojack BR, et al. Hydroxyurea and etoposide: in vitro synergy and phase I clinical trial. J Natl Cancer Inst. 1988; 80(17): 1412–6.
Swinnen LJ, Barnes DM, Fisher SG, et al. 1-β-D-arabinofuranosylcytosine and hydroxyurea production of cytotoxic synergy with cis-diamminedichloroplatinum (II) and modification of platinum-induced DNA inter Strand cross-linking. Cancer Res. 1989; 49(6): 1383–9.
Christen RD, Shalinsky DR, Howell SB. Enhancement of the loss of multiple drug resistance by hydroxyurea. Semin Oncol 1992; 19 (3 Suppl. 9): 94–100.
Snapka RM, Varshavsky A. Loss of unstably amplified dihydrofolate reductase genes from mouse cells is greatly accelerated by hydroxyurea. Proc Natl Acad Sci U S A. 1983; 80(24): 7533–7.
Von Hoff DD, Waddelow T, Forseth B, et al. Hydroxyurea accelerates loss of extrachromosomally amplified genes from tumor cells. Cancer Res 1991; 51 (23 Pt 1): 6273–9.
Kennedy BJ. Hydroxyurea therapy in chronic myelogenous leukemia. Cancer. 1972; 29(4): 1052–6.
Sharon R, Tatrsky I, Ben-Arieh Y. Treatment of polycythemia vera with hydroxyurea. Cancer. 1986; 57(4): 718–20.
Ariel IM. Therapeutic effects of hydroxyurea: an experience with 118 patients with inoperable solid tumors. Cancer. 1970; 25(3): 705–14.
Carter SK, Wasserman TH. The chemotherapy of urologic cancer. Cancer. 1975; 36: 729–47.
Mundy AR. A pilot study of hydroxyurea in hormone ‘escaped’ metastatic carcinoma of the prostate. Br J Urol. 1982; 54: 20–5.
Hussey DH, Abrams JR. Combined therapy in advanced head and neck cancer: hydroxyurea and radiotherapy. Prog Clin Cancer. 1975; 6: 79–86.
Hreshchyshyn MM, Aron BS, Boronow RC, et al. Hydroxyurea or placebo combined with radiation to treat stages IIIB and IV cervical cancer confined to the pelvis. Int J Radiat Oncol Biol Phys. 1979; 5: 317–22.
Piver MS, Vongtama V, Emrich LJ. Hydroxyurea plus pelvic radiation versus placebo plus pelvic radiation in surgically staged state IIIB cervical cancer. J Surg Oncol. 1987; 35(2): 129–34.
Layton AM, Sheehan-Dare RA, Goodfile MJ, et al. Hydroxyurea in the management of therapy resistant psoriasis. Br J Dermatol. 1989; 121(5): 647–53.
Lori F, Malykh A, Cara A, et al. Hydroxyurea as an inhibitor of human immunodeficiency virus-type 1 replication. Science. 1994; 266: 801–5.
Griffith KM. Hydroxyurea (NSC-32065): results of a phase I study. Cancer Chemother Rep. 1964; 40: 33–6.
Vaughan WP, Kris E, Vose J, et al. Phase I/II study incorporating intravenous hydroxyurea into high-dose chemotherapy for patients with primary refractory or relapsed and refractory intermediate-grade and high-grade malignant lymphoma. J Clin Oncol. 1995; 13(5): 1089–95.
Donehower RC. Hydroxyurea. In: Chabner B, editor. Pharmacologic principles of cancer treatment. Philadelphia: WB Saunders, 1982: 269–75.
Wilson JG, Scott WJ, Ritter EJ, et al. Comparative distribution and embryotoxicity of hydroxyurea in pregnant rats and rhesus monkeys. Teratology. 1975; 11(2): 169–78.
Ford SS, Shackney SE. Lethal and sublethal effects of hydroxyurea in relation to drug concentration and duration of drug exposure in sarcoma 180, in vitro. Cancer Res 1977; 37 (8 Pt 1): 2628–37.
Moran RE, Straus MJ. Cytokinetic analysis of L1210 leukemia after continuous infusion of hydroxyurea in vivo. Cancer Res. 1979; 39(5): 1616–22.
Blumenreich MS, Kellihan MJ, Joseph UG, et al. Long-term intravenous hydroxyurea infusions in patients with advanced cancer: a phase I trial. Cancer. 1993; 71(9): 2828–32.
Brown PC, Tlsty TD, Schimke RT. Enhancement of methotrexate resistance and dihydrofolate reductase gene amplification by treatment of mouse 3T6 cells in hydroxyurea. Mol Cell Biol. 1983; 3(6): 1097–1107.
Dover GJ, Humphries RK, Moore JG, et al. Hydroxyurea induction of hemoglobin F production in sickle cell disease: relationship between cytotoxicity and F cell production. Blood. 1986; 67(3): 735–8.
Gao WY, Johns DG, Chokekijchai S, et al. Disparate actions of hydroxyurea in potentiation of purine and pyrimidine 2′,3′-dideoxynucleoside activities against replication of human immunodeficiency virus. Proc Natl Acad Sci U S A. 1995; 92: 8333–7.
Malley SD, Grange JM, Hamedi-Sangsari F, et al. Synergistic anti-human immunodeficiency virus type 1 effect of hydroxamate compounds with 2,3-dideoxyinosine in infected resting human lymphocytes. Proc Natl Acad Sci U S A. 1994; 91: 11017–21.
Giacca M, Zanussi S, Comar M, et al. Treatment of human immunodeficiency virus infection with hydroxyurea: virologic and clinical evaluation. J Infect Dis. 1996; 174: 204–9.
Simonelli C, Nasti G, Vaccher E, et al. Hydroxyurea treatment in HIV-infected patients. J Acquir Immune Defic Syndr Hum Retrovirol. 1996; 13: 462–4.
Biron F, Lucht F, Peyramond D, et al. Anti-HIV activity of the combination of didanosine and hydroxyurea in HIV-1-in-fected individuals. J Acquir Immune Defic Syndr Hum Retrovirol. 1995; 10: 36–40.
Vila J, Biron F, Nugier F, et al. 1-year follow-up of the use of hydroxycarbamide and didanosine in HIV infection. Lancet. 1996; 348(9021): 203–4.
Montaner JSG, Zala C, Conway B, et al. A pilot study of hydroxyurea among patients with advanced human immunodeficiency virus (HIV) disease receiving chronic didanosine therapy: Canadian HIV trials network protocol 080. J Infect Dis. 1997; 175: 801–6.
Malley SD, Grange JM, Hamedi-Sangsari F, et al. Suppression of HIV production in resting lymphocytes by combining didanosine and hydroxamate compounds [abstract]. Lancet. 1994; 343: 1292.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gwilt, P.R., Tracewell, W.G. Pharmacokinetics and Pharmacodynamics of Hydroxyurea. Clin Pharmacokinet 34, 347–358 (1998). https://doi.org/10.2165/00003088-199834050-00002
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003088-199834050-00002