Summary
Teicoplanin is a glycopeptide antibiotic whose activity is selectively oriented against Gram-positive aerobic and anaerobic bacteria, including Staphylococcus aureus, coagulase-negative staphylococci, Clostridium difficile, Peptostreptococcus spp. and Corynebacterium jeikeium; such activity is affected by neither methicillin resistance nor β-lactamase production.
Teicoplanin is not significantly absorbed from the gastrointestinal tract; consequently, it has to be administered intravenously (either by infusion or by rapid injection) or intramuscularly. Its long half-life allows regimens based upon once daily administration.
The adverse effects most frequently associated with teicoplanin treatment are local and hypersensitivity reactions, such as itching and drug fever; anaphylactoid reactions (the ‘red man syndrome’) are seldom observed. Teicoplanin also has less potential than vancomycin to cause nephrotoxicity, especially when administered in combination with an aminoglycoside.
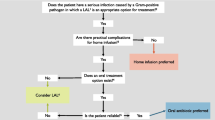
Teicoplanin has been proven to be effective in the treatment of microbiologically documented Gram-positive infections, including ‘difficult to treat infections’ such as endocarditis and prosthetic infections. Furthermore, recent trials in patients with haematological malignancies or other cancers have clearly demonstrated that teicoplanin is at least as efficacious as vancomycin in the empirical initial antibiotic regimen for febrile neutropenic patients, and is associated with fewer adverse effects. Finally, owing to its good tolerability profile and the advantage of once daily administration by both intravenous and intramuscular routes, teicoplanin has proven to be very useful for the outpatient treatment of serious Gram-positive infections.
In conclusion, teicoplanin is potentially an effective alternative to vancomycin both in immunocompetent and immunocompromised patients, with the advantage over vancomycin of single daily dose administration and lower toxicity. Further comparative studies with vancomycin are, however, required to better define the therapeutic role of teicoplanin for particular infections (i.e. infective endocarditis).
Similar content being viewed by others
References
Parenti F. Structure and mechanism of action of teicoplanin. J Hosp Infect 1986; 7 Suppl. A: 79–83
Brogden RN, Peters DH. Teicoplanin. A reappraisal of its antimicrobial activity, pharmacokinetic properties and therapeutic efficacy. Drugs 1994; 47: 823–54
Reynolds PE. Inhibitors of bacterial wall synthesis. In: Greenwood D, O’Grady F, editors. The scientific basis of antimicrobial chemotherapy. Cambridge: Cambridge University Press, 1985: 13–40
National Committee for Laboratory Standards. 1992; 12: No. 20; Villanova, PA, USA
Dutka-malen S, Leclercq R, Coutant V, et al. Phenotypic and genotypic heterogenicity of glycopeptide resistance determinants in Gram-positive bacteria. Antimicrob Agents Chemother 1990; 34: 1875–9
Verbist L, Tjandramaga B, Hendrickx B, et al. In vitro activity and human pharmacokinetics of teicoplanin. Antimicrob Agents Chemother 1984; 26: 881–6
Wise R, Donovan IA, McNulty CAM, et al. Teicoplanin, its pharmacokinetics, blister and peritoneal fluid penetration. J Hosp Infect 1986; 7: Suppl. A: 47–55
Buniva G, Del Favero A, Bernareggi A, et al. Pharmacokinetics of 14C-teicoplanin in healthy volunteers. J Antimicrob Chemother 1988; 21: Suppl. A: 23–8
Del Favero A, Patoia L, Rosina R, et al. Pharmacokinetics and tolerability of teicoplanin in healthy volunteers after single increasing doses. Antimicrob Agents Chemother 1991; 35: 2551–7
Thompson GA, Smithers JA, Kenny MT, et al. Pharmacokinetics of teicoplanin upon multiple dose intravenous administration to normal healthy male volunteers. Biopharm Drug Dispos 1992; 13: 213–20
Antony KK, Lewis EW, Kenny MT, et al. Pharmacokinetics and bioavailability of a new formulation of teicoplanin following intravenous and intramuscular administration to humans. J Pharm Sci 1991; 80: 605–7
Cruciani M, Navarra A, Di Perri G, et al. Evaluation of intra-ventricular teicoplanin for the treatment of neurological shunt infections. Clin Infect Dis 1992; 15: 285–9
Wilson APR, Gruneberg RN, Neu H. Dosage recommendations for teicoplanin. J Antimicrob Chemother 1993; 32: 792–6
Greenberg RN. Treatment of bone, joint, and vascular-access-associated Gram-positive bacterial infections with teicoplanin. Antimicrob Agents Chemother 1990; 34: 2392–7
Fernandez-Guerrero M, Gobernado M, Ariza J, et al. Spanish experience with teicoplanin. Scand J Infect Dis Suppl. 1990; 72: 38–44
Livornese LL, Gold M, Johnson CC, et al. Clinical evaluation of teicoplanin in the treatment of Gram-positive bacterial intravascular infections. J Antimicrob Chemother 1993; 31: 188–91
Presterl E, Graninger W, Georgopoulos A. The efficacy of teicoplanin in the treatment of endocarditis caused by Gram-positive bacteria. J Antimicrob Chemother 1993; 31: 755–66
Venditti M, Gelfusa V, Serra P, et al. 4-week treatment of streptococcal native valve endocarditis with high-dose teicoplanin. Antimicrob Agents Chemother 1992; 36: 723–6
Davey P, Williams AH. Teicoplanin monotherapy of serious infections caused by Gram-positive bacteria: a re-evaluation of patients with endocarditis or Staphylococcus aureus bacteraemia from a European open trial. J Antimicrob Chemother 1991; 27: Suppl. B: 43–50
Gilbert DN, Wood CA, Kimbrough RC, et al. Failure of treatment with teicoplanin at 6 milligrams/kilogram/day in patients with Staphylococcus aureus intravascular infection. Antimicrob Agents Chemother 1991; 35: 79–87
Van der Auwera P, Aoun M, Meunier F. Randomized study of vancomycin versus teicoplanin for the treatment of Gram-positive bacterial infections in immunocompromised hosts. Antimicrob Agents Chemother 1991; 35: 451–7
Lewis P, Garaud JJ, Parenti F. A multicentre open clinical trial of teicoplanin in infections caused by Gram-positive bacteria. J Antimicrob Chemother 1988; 21: Suppl. A: 61–7
Nathwani D, Reid TMS, Gould IM, et al. An open study of teicoplanin in the treatment of Gram-positive infections. J Chemother 1991; 3: 315–20
Edelstein HE, Oster SE, Karp RA, et al. Intravenous or intramuscular teicoplanin once daily for skin and soft-tissue infections. DICP 1991; 25: 914–8
LeFrock JL, Ristuccia AM, Ristuccia PA, et al. Teicoplanin in the treatment of bone and joint infections. Eur J Surg 1992; Suppl. 567: 10–3
Weinberg WG. Safety and efficacy of teicoplanin for bone and joint infections: results of a community-based trial. South Med J 1993; 86: 891–7
Concia E. Ruolo della teicoplanina nelle infezioni delle basse vie respiratorie. Antibioticoter Prat 1991; 1: 41–7
Amaducci S, Barbieri D, Cogo R, et al. Valutazione comparativa di trattamenti antibiotici empirici in pazienti in insufficienza respiratoria cronica con riacutizzazione infettiva bronchiale. Antibioticoter Prat 1992; 2: 63–70
Pizzo PA. Management of fever in patients with cancer and treatment induced neutropenia. N Engl J Med 1993; 328: 1323–32
Menichetti F, Del Favero A. The role of Gram-positive therapy in the neutropenic patient. J Antimicrob Chemother 1991; 27: Suppl. B: 51–60
Del Favero A, Menichetti F, Guerciolini R, et al. Prospective randomized clinical trial of teicoplanin for empiric combined antibiotic therapy in febrile, granulocytopenic acute leukemia patients. Antimicrob Agents Chemother 1987; 31: 1126–9
EORTC International Antimicrobial Therapy Cooperative Group and the National Cancer Institute of Canada-Clinica Trials Group. Vancomycin added to empirical combination therapy for fever in granulocytopenic cancer patients. J Infect Dis 1991; 163: 951–8
Shenep J, Hughes WT, Roberson PK, et al. Vancomycin, ticarcillin and amikacin compared with ticarcillin-clavulanate and amikacin in the empirical treatment of febrile neutropenic children with cancer. N Engl J Med 1988; 319: 1053–8
Menichetti F, Martino P, Bucaneve G, et al. Effects of teicoplanin and those of vancomycin in initial empirical antibiotic regimen for febrile, neutropenic patients with hematological malignancies. Antimicrob Agents Chemother 1994; 38: 2041–6
Rolston KVI, Nguyen H, Amos G, et al. A randomized double-blind trial of vancomycin versus teicoplanin for the treatment of Gram-positive bacteremia in patients with cancer. J Infect Dis 1994; 169: 350–5
Chow AW, Jewesson PJ, Kureishi A, et al. Teicoplanin versus vancomycin in the empirical treatment of febrile neutropenic patients. Eur J Haematol 1993; 51 Suppl. 54: 18–24
Kureishi A, Jewesson PJ, Rubinger M, et al. Double-blind comparison of teicoplanin versus vancomycin in febrile neutropenic patients receiving concomitant tobramycin and piperacillin: effect of ciclosporin A associated nephrotoxicity. Antimicrob Agents Chemother 1991; 35: 2246–52
Nováková IR, Donnelly JP, Verhagen CS, et al. Teicoplanin as modification of initial empirical therapy in febrile granulocytopenic patients. J Antimicrob Chemother 1990; 25: 985–93
Fauser AA, Lang E, Schmid J, et al. Antibiotische Behandlung der Sepsis mit Teicoplanin bei Knochenmarktransplantierten Patienten. Onkologie 1991; 14: 248–55
Periti P, Stringa G, Donati L, et al. Teicoplanin. Its role as systemic therapy of burn infections and as prophylaxis for orthopaedic surgery. Eur J Surg 1991; Suppl. 567: 3–8
de Lalla F, Nicolin R, Rinaldi E, et al. Prospective study of oral teicoplanin versus oral vancomycin for therapy of pseudomembranous colitis and Clostridium difficile-associated diarrhea. Antimicrob Agents Chemother 1992; 36: 2192–6
The Swedish CDAD Study Group. Treatment of Clostridium difficile associated diarrhea and colitis with an oral preparation of teicoplanin; a dose finding study. Scand J Infect Dis 1994; 26: 309–16
Goodman SB, Schurman DJ. Prophylaxis in orthopedic surgery. In: Schlossberg D, editor. Orthopedic infection. New York: Springier-Verlag, 1988: 144–55
Gorbach SL, Conbdon RE, Conte JE, et al. Evaluation of new anti-infective drugs for surgical prophylaxis. Clin Infect Dis 1992; 15: S313–38
Mollan RAB, Haddock M, Webb CH. Teicoplanin vs cephamandole for antimicrobial prophylaxis in prosthetic joint implant surgery (preliminary results). Eur J Surg 1992; Suppl. 567: 19–21
de Lalla F, Novelli A, Pellizzer GP, et al. Regional and systemic prophylaxis with teicoplanin in monolateral and bilateral total knee replacement procedures: study of pharmacokinetics and tissue penetration. Antimicrob Agents Chemother 1993; 37: 2693–8
de Lalla F, Viola R, Pellizzer GP, et al. Efficacy and safety of regional prophylaxis (RP) with teicoplanin (T) in total knee replacement (TKR) [abstract K3]. Abstracts of the 34th Inter-science Conference on Antimicrobial Agents and Chemotherapy: 1994 Oct 4–7; Orlando, Florida
Lim SH, Smith MP, Machin SJ, et al. A prospective randomised study of prophylactic teicoplanin to prevent early Hickman catheter-related sepsis in patients receiving intensive chemotherapy for haematological malignancies. Eur J Haematol 1993; 51 Suppl. 54: 10–3
Pellizzer GP, Nicolin R, D’Emilio A, et al. Teicoplanin (T) prophylaxis in tunnelled central venous catheter (TCVC) [abstract 89]. Abstracts of the 35th Interscience Conference on Antimicrobial Agents and Chemotherapy: 1995 Sep 17–20; San Francisco
Davey PG, Williams AH. A review of the safety profile of teicoplanin. J Antimicrob Chemother 1991; 27: Suppl. B: 69–73
Polk RE. Anaphylactoid reactions to glycopeptide antibiotics. J Antimicrob Chemother 1991; 27: Suppl. B: 17–29
Dubettier S, Boibieux A, Lagable M, et al. Red man syndrome with teicoplanin. Rev Infect Dis 1991; 13: 770–1
Sahai J, Healy DP, Shelton MJ, et al. Comparison of vancomycin- and teicoplanin-induced histamine release and ‘red man syndrome’. Antimicrob Agents Chemother 1990; 34: 765–9
Davenport A. Allergic cross-reactivity to teicoplanin and vancomycin. Nephron 1993; 63: 482
Grek V, Andrien F, Collignon J, et al. Allergic cross-reaction of teicoplanin and vancomycin. J Antimicrob Chemother 1991; 28: 476–7
Knudsen JD, Pedersen M. IgE-mediated reaction to vancomycin and teicoplanin after treatment with vancomycin. Scand J Infect Dis 1992; 24: 395–6
Lewis EW, Gibson CB, Heilman CJ, et al. Teicoplanin administration in patients with vancomycin hypersensitivity [abstract no. 1672]. Program and Abstracts of the 32nd Interscience Conference on Antimicrobial Agents and Chemotherapy: 1992 Oct 11–14; Anaheim, California
Del Favero A, Patoia L, Bucaneve G, et al. Leukopenia with neutropenia associated with teicoplanin therapy. DICP 1989; 23: 45–7
Novakòvà I, Donnelly JP, De Pauw B. Ceftazidime as monotherapy or combined with teicoplanin for initial empiric treatment of presumed bacteremia in febrile granulocytopenic patients. J Antimicrob Chemother 1991; 35: 672–8
Lang E, Schafer V, Schaaf B, et al. Comparison of efficacy and safety of teicoplanin in Gram-positive infections: a multi-centre study. Scand J Infect Dis 1990; 72: Suppl.: 54–60
Guaglianone MH, Bachini V, Mosconi G, et al. Teicoplanin for empiric combined antibiotic therapy in febrile granulocytopenic patients. Multicenter open trial: preliminary analysis of results in 614 treated patients. J Chemother 1993; 5: Suppl. 1:173–4
Frye RF, Job ML, Dretler RH, et al. Teicoplanin nephrotoxicity: first case report. Pharmacotherapy 1992; 12: 240–2
Wilson APR, Gruneberg RN, Neu H. A critical review of the dosage of teicoplanin in Europe and the USA. Int J Antimicrob Agents 1994; 4: Suppl. 1: S1–30
Shanson DC, Shehata A, Tadayon M, et al. Comparison of intravenous teicoplanin with intramuscular amoxycillin for the prophylaxis of streptococcal bacteremia in dental patients. J Antimicrob Chemother 1987; 20: 85–93
Wilson APR, Treasure T, Gruneberg RN, et al. Antibiotic prophylaxis in cardiac surgery: a prospective comparison of two dosage regimens of teicoplanin with a combination of flucloxacillin and tobramycin. J Antimicrob Chemother 1988; 21: 213–23
Webster A, Wilson APR, Williams AH, et al. The use of a new glycopeptide antibiotic, teicoplanin, in the treatment of bacterial endocarditis. Postgrad Med J 1987; 63: 621–4
Maher ER, Hollman A, Gruneberg RN. Teicoplanin-induced ototoxicity in Down’s syndrome. Lancet 1986; 1: 613
Dagan R, Einhorn M, Howard CB, et al. Infections due to Gram-positive organisms in children: possible role for teicoplanin. J Antimicrob Chemother 1991; 27: Suppl. B: 37–41
Peller P, Aichholzer B, Fell J, et al. Safety and efficacy of teicoplanin in the treatment of Gram-positive infections in pediatric patients in Germany. Pediatr Infect Dis J 1993; 12: S7–9
Kacet N, Dubos JP, Roussel-Delvallez M, et al. Teicoplanin and amikacin in neonates with staphylococcal infection. Pediatr Infect Dis J 1993; 12: S10–12
Wilson AP, Gruneberg RN. Use of teicoplanin in community medicine. Eur J Clin Microbiol Infect Dis 1994; 13: 701–10
Davey PG, Rowley DR, Phillips GA. Teicoplanin: home therapy for prosthetic joint infections. Eur J Surg 1992; Suppl. 567: 23–5
Ball LM, Siddal S, Van-Saenen H. Teicoplanin in home therapy of the terminally ill child. Eur J Haematol 1993; 54 Suppl.: 14–7
Preis S, Jurgens H, Friedland C, et al. Ceftriaxone alone or in combination with teicoplanin in the management of febrile episodes in neutropenic children and adolescents with cancer on an outpatient base. Klin Padiatr 1993; 205: 295–9
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
de Lalla, F., Tramarin, A. A Risk-Benefit Assessment of Teicoplanin in the Treatment of Infections. Drug-Safety 13, 317–328 (1995). https://doi.org/10.2165/00002018-199513050-00005
Published:
Issue Date:
DOI: https://doi.org/10.2165/00002018-199513050-00005