Abstract
A vermiculite regolith sample from Llano, County, Texas, was divided into 16–32, 8–16, 4–8, 2–4, 1–2, ½–1, ¼–½, 1 10–¼, and 5 100–1 10 micron equivalent spherical diameter size-classes. A well-crystallized vermiculite found in association with the regolith was ground to −400 mesh.
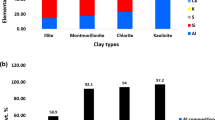
The samples were analyzed by X-ray diffractometry and differential thermal analysis, and cation exchange capacity measurements were made for selected size fractions. Chemical analyses by X-ray fluorescence revealed a systematic increase in silicon with decreasing particle size. An increase in the iron content and a decrease in magnesium were also directly correlated to a decrease in the crystallite size. Aluminum content was constant throughout the range of particle sizes.
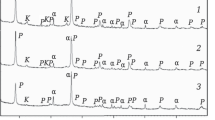
X-ray analyses showed that the coarser particles were trioctahedral, whereas the smaller crystallites were dioctahedral. These data were supported by recast formulae based on the chemical analyses. The recast formulae were substantiated by DTA and cation exchange capacity data as being reasonable approximations of actual formulae.
These observations are interpreted as an expression of chemical alteration that clay minerals may undergo in an aqueous environment. The crystallographic, cation exchange capacity, DTA, and swelling properties are a function of the chemical composition of the crystallites. Chemical composition of the crystallites is shown to be directly correlated to particle size.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.References
Barnes, V. E., and Clabaugh, S. E. (1961) Vermieulite deposits near Llano, Texas: Texas Univ., Guidebook 3, 45–53.
Clabaugh, S. E., and Barnes, V. E. (1959) Vermieulite in central Texas: Texas Univ., Rep. of Invest. 40, 32 pp.
Fyfe, W. S. (1951) Isomorphism and bond type: Amer. Min. 36, 538–42.
Jonas, E. C. (1960) Mineralogy of the micaceous clay minerals: Proa. Internat. Comm. Study Clays, Copenhagen, 1–16.
Jonas, E. C. (1963) Ion exchange at edge and interlayer in montmorillonites differing in size: Science 140, 75–6.
Jonas, E. C., and Roberson, H. E. (1960) Particle size as a factor influencing expansion of the three-layer clay minerals: Amer. Min. 45, 828–38.
Marshall, C. E. (1949) The Colloid Chemistry of the Silicate Minerals: Academic Press, New York, 195 pp.
Paige, S. (1912) Description of the Llano and Burnet quadrangles: U.S. Geol. Surv. Geol. Atlas, Llano-Burnet Folio (No. 183), 16 pp.
Pauling, L. (1948) The Nature of the Chemical Bond: Cornell University Press, Ithaca, New York.
Roberson, H. E., and Jonas, E. C. (1965) Clay minerals intermediate between illite and montmorillonite: Amer. Min. 50, 766–70.
Walker, G. F. (1961) Vermieulite minerals, Chap. VII in X-ray Identification and Crystal Structures of Clay Minerals: Mineralogical Society, London.
Weaver, C. E. (1958) The effects and geologic significance of potassium “fixation” by expanding clay minerals derived from muscovite, biotite, chlorite, and volcanic material: Amer. Min. 43, 839–61.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kerns, R.L., Mankin, C.J. Compositional Variation of a Vermiculite as Related to Particle Size. Clays Clay Miner. 15, 163–177 (1967). https://doi.org/10.1346/CCMN.1967.0150116
Published:
Issue Date:
DOI: https://doi.org/10.1346/CCMN.1967.0150116