Abstract
Ion exchange constants for various cations adsorbed on clays were determined using compacted-clay samples as membrane electrodes and theory for the membrane response of glass electrodes. The electrochemical cell used is conventionally written

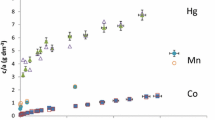
A static electrical potential is developed between the reference electrodes. The potential is a function of the cation species and of the cation activities in the two solutions and is given by an adaptation of the Nernst equation. From measurements of the electrical potential, the free energy of reactions or exchange constants for a series of reactions between various cation species were obtained. These determinations indicate that compacted clays prefer monovalent over divalent cations.
Some exchange constants were determined for the same clays in a non-compacted state. Not only the magnitude but also the order of cation selectivity changed for the same clay when determinations were made in the dispersed state and compared with those obtained by electrochemical means using compacted clay. Publication authorized by the Director, U.S. Geological Survey.
Similar content being viewed by others
References
Eisenman, G. (1962) Cation selective glass electrodes and their mode of operation Biophysical Jour., v.2, part 2, supplement, pp.259–322.
Eisenman, G., Rudin, D. O., and Casby, J. U. (1957) Glass electrodes for measuring sodium ion: Science, v.126, pp.831–834.
Garrels, R. M., Sato, M., Thompson, M. E., and Truesdell, A. H. (1962) Glass electrodes sensitive to divalent cations: Science, v.135, pp.1045–1048.
Govindan, K. P., and Krishnaswamy (1958) Studies on the cation-exchange resin from cashew nut shell liquid: Jour. Sci. Industrial Res., v.17B, pp.416–419.
Hanshaw, B. B. (1962) Membrane properties of compacted clays: Ph.D. Thesis, Harvard University.
Marshall, C. E. (1939) The use of zeolitic membrane electrodes: Jour. Phys. Chem., v.43, pp.1155–1164.
Marshall, C. E. (1948) Theory of selective membrane behavior: Jour. Phys. Chem., v.52, pp.1284–1295.
Marshall, C. E., and Bergman, W. E. (1941) The electrochemical properties of mineral membranes, I. The estimation of K+ activities: Jour. Am. Chem. Soc., v.63, pp.1911–1916.
Marshall, C. E., and Bergman, W. E. (1942a) The electrochemical properties of mineral membranes, II. Measurements of K+ activities in colloidal clays: Jour. Phys. Chem., v.46, pp.52–61.
Marshall, C. E., and Bergman, W. E. (1942b) The electrochemical properties of mineral membranes, III, IV. The estimation of NH4 activities: Jour. Phys. Chem., v.46, pp.325–334.
Marshall, C. E., and Krinball, C. A. (1942c) The electrochemical properties of minera membranes, V. Beidellite membranes and the determination of sodium: Jour. Am. Chem. Soc., v.64, pp.1814–1819.
Marshall, C. E., and Krinball, C. A. (1942d) The clays as colloidal electrolytes: Jour. Phys. Chem., v.46, pp.1077–1090.
Nikolskii, B. P. (1937) Theory of the glass electrode, I: Acta Physicochim. U.R.S.S., v.7, pp.597.
Walton, H. F. (1949) Ion exchange equilibra: In Ion Exchange, F. C. Nachod, Edt., Academic Press, New York, pp.3–28.
Wright, M. P. (1959) pH measurements with the glass electrode: in Symposium on pH and Blood Gas Measurements: Little, Brown, Boston, pp.5–15.
Wyllie, M. R. J. (1948) Some electrochemical properties of shales: Science, v.108, pp.684–685.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hanshaw, B.B. Cation-Exchange Constants for Clays from Electrochemical Measurements. Clays Clay Miner. 12, 397–421 (1963). https://doi.org/10.1346/CCMN.1963.0120136
Published:
Issue Date:
DOI: https://doi.org/10.1346/CCMN.1963.0120136