Abstract
Background
Increased prevalence of hepatocellular carcinoma (HCC) remains a global health challenge. HCC chemoresistance is a clinical obstacle for its management. Aberrant miRNA expression is a hallmark for both cancer progression and drug resistance. However, it is unclear which miRNAs are involved in HCC chemoresistance.
Methods
MicroRNA microarray analysis revealed a differential expression profile of microRNAs between the hepatocellular carcinoma HA22T cell line and the HDACi-R cell line, which was validated by quantitative real-time PCR (qRT-PCR). To determine the biological function of miR-342-5p and the mechanism of the microRNA-342-5p/CFL1 axis in hepatocellular carcinoma HDACi resistance, loss- and gain-of-function studies were conducted in vitro.
Results
Here we demonstrated the molecular mechanism of histone deacetylase inhibitor (HDACi) resistance in HCC. Differential miRNA expression analysis showed significant down regulation of miR-342-5p in HDACi-R cells than in parental HA22T cells. Mimics of miR-342-5p enhanced apoptosis through upregulation of Bax, cyto-C, cleaved-caspase-3 expressions with concomitant decline in anti-apoptotic protein (Bcl-2) in HDACi-R cells. Although HDACi did not increase cell viability of HDACi-R, overexpression of miR-342-5p decreased cofilin-1 expression, upregulated reactive oxygen species (ROS) mediated apoptosis, and sensitized HDACi-R to HDACi in a dose-dependent manner.
Conclusion
Our findings demonstrated the critical role of miR-342-5p in HDACi resistance of HCC and that this mechanism might be attributed to miR-342-5p/cofilin-1 regulation.
Similar content being viewed by others
Background
The reason behind the second most frequently occurring cancer-related deaths across Taiwan is attributed to hepatocellular carcinoma (HCC) [1]. Globally, it is the cause for the fourth most frequently occurring cancer-related deaths, with approximately 850,000 new cases each year [2, 3]. Most people with HCC are persistently infected by hepatitis C and B viruses (HCV and HBV), with other risk factors being smoking, excessive use of alcohol, diabetes, and obesity [3, 4].
Chemotherapeutic drug resistance is a clinical hurdle for cancer management [5]. Among the several drug resistance mechanisms, diminished apoptosis in cancer cells is one of the main challenges for cancer treatment [6]. The non-coding MicroRNAs (miRNAs) in translational research have garnered attention, since they are important mediators in cell signaling [7]. miRNAs are small RNA with approximately 23 nucleotides [8]. These have specific binding ability for the 3’- untranslated region (UTR) of the target protein and through their complement/mis-pair binding, prevent mRNA translation and downregulate the target protein expression [9]. Thus, altered miRNA expressions determine the activation of several regulatory signaling cascades and remodify the cancer type from drug sensitive to resistant [10]. The miRNAs have been identified as prospective biomarkers in various cancers [11, 12]. Despite several studies demonstrating the crucial role of miRNAs in liver cancer, its involvement in chemotherapy resistance remains unclear. Thus, it is important to analyze the pivotal miRNAs involved in liver cancer drug resistance for enhancing miRNA-based cancer therapy.
The actin-depolymerising factors (ADF) family of proteins including cofilin-1 (CFL1) exhibit comparative biological activities [13]. CFL1 engages actin and induces cytoskeleton remodeling to facilitate cell migration after activation by phosphatases [14]. Activated CFL1 translocates to the mitochondria promoting cell death [15,16,17]. Liver cancer cells treated with reactive oxygen species (ROS) inducers causes mitochondrial dysfunction via cofilin-1[18]. Activated CFL1 reacts with BAX and subsequently gets translocated to the mitochondria inducing neuronal cell apoptosis [15]. Notably, anticancer drug induced apoptosis is linked to their interactions with BAX and accumulation of ROS. CFL1 phosphorylation is particularly critical for the development of chemoresistance in HCC [19].
We previously demonstrated a possible mechanism of HCC chemoresistance [18]. Here, we aimed to determine the functional miRNAs in drug-resistant and parental cells to identify possible therapeutic strategies for HCC. The present study reported the novel role of miR-342-5p in HCC drug resistance. Further, miR-342-5p overexpression might be involved in chemosensitization and cell death in HDAC inhibitor resistant HCC through CFL-1/ROS regulation.
Materials and methods
Cell culture
The human HA22T cell line were procured from Bioresource Collection and Research Center (BCRC), Hsinchu, Taiwan. The cells were grown in Dulbecco's minimal essential medium (DMEM; Sigma, St. Louis, Missouri, USA). HDAC inhibitor (apicidin and suberoylanilide hydroxamic acid (SAHA)) resistant cells (HDACi-R cells) were developed using HA22T parental cells [20]. The HDAC inhibitor cells were incubated with SAHA (2 µM) in this investigation.
Total protein extraction
The protein from the cells were extracted using RIPA Lysis buffer (Thermo Scientific, USA) with phosphatase inhibitors and proteinase K [20, 21]. After lysis, the cells were centrifuged at 15,000 rpm for 30 min at 4 °C. The isolated supernatant was preserved at −20 °C till further analysis.
MTT assay
The cells were seeded in a 96-well plate (1 × 104 cells per well) for 24 h for attachment [14, 22]. They were treated for 24 h with different does (1-5 µM) of SAHA. After removing the supernatant, 100 µl of MTT solution (0.5 mg/ml) was added to each well and incubated at 37 °C for 4 h. The vitality of cells was evaluated by adding 100 µl DMSO in the dark for 10 min to solubilize formazan. A Multiskan™ GO Microplate Spectrophotometer (Thermo Fisher Scientific Inc., Waltham, MA, USA) was used to measure the absorbance at 570 nm. The percentage of cell viability was calculated by comparing it with the control.
Drugs & antibodies
The anti-c-caspase-3 antibody (#9664) was supplied by Cell Signaling Technology Danvers, USA. The antibodies for Bcl-2 (sc7382), cofilin-1 (sc-53934), GAPDH (sc-47778), anti-Cytochrome c (anti-cyto-C) (sc-13560), and anti-Bax (sc-7480) were supplied by Santa Cruz CA, USA. All secondary antibodies coupled with horseradish peroxidase, were supplied by Santa Cruz. All other reagents were obtained from Sigma-Aldrich.
Western blot
Protein concentration were determined using Bradford protein test dye (Bio-Rad, USA). The proteins (30 μg) were separated using SDS-PAGE [21, 23], and the separated proteins were transferred onto PVDF membranes (Millipore, Belford, Massachusetts). These PVDF membranes were then incubated at RT for 1 h with skimmed milk. After TBST wash, the membranes were treated overnight at 4 °C with specified primary antibodies. The membranes were washed in TBST and incubated with secondary antibody for 1 h at RT. The membranes were detected using chemiluminescence solution (Millipore, Billerica, MA, USA) and the images were acquired using GE Digital Imaging Commerce, CA, USA.
Micro array analysis
The Total RNA isolated from HA22T or HDACi-Resistance cell types were analyzed for differential miRNA analysis using microrna Profiler miRNA Microarray Services (Human miRNA OneArray®). The log-twofold change miRNAs expression of HDACi-R and parental HA22T were calculated [24].
Prediction of mRNA targets
miRNA target predicting website miRTarBase, miRDB, and TargetScanHuman were used to predict the potential targets of miR-342-5p.
RNA preparation & qRT- PCR
The total RNA from HA22T or HDACi-R cells were isolated using RNA Isolation System Quick-RNATM Mini Prep kit (Zymo Research, Irvine, CA, USA) [25]. The Mir-X miRNA First-Strand Synthesis Kit (TAKARA, Ohtsu, Japan) and iScript™ cDNA synthesis kit was used to synthesize cDNA. The miRNA expression pattern was analysed using RT-PCR (CFX96™ Quantitative Real-Time system, Bio-Rad). All the reactions were carried out in triplicates.
Flow cytometry
Apoptosis was analyzed using Annexin V low cytometric analysis. The HA22T and HDACi-R cells were treated with miR-342-5p inhibitor/mimic. Following the manufacturer's instructions, the cells were harvested and stained with Annexin V-FITC apoptotic detecting kit (BD, Biosciences, San Jose, CA, USA) [25] and analyzed using FACS CantoTM equipment (BD, Biosciences). The cells in each quadrant of FITC-A vs PI plot were gated to produce cell singlets. Around 10,000 of the 50,000 visible cells were counted. (1 – (Q3Drug/Q3Control)) resulted in 100 percent cell death.
Analysis of mitochondrial ROS production
MitoSOX Red fluorescence probe was used to determine mitochondrial ROS generation. The live-cell permeable MitoSOX Red dye preferentially target mitochondria and emits red fluorescence in the presence of superoxide. Briefly, 1 × 105 cell suspension per well were added on four-chamber slide. Following treatment, cells were PBS washed and incubated using MitoSOX Red (2.5 µM) for 30 min at 37 0C in 5% carbon dioxide incubator. The images were captured and examined in fluorescence microscopy [20, 26,27,28,29].
MicroRNA and siRNA transfection studies
Depending on the siRNA and miRNA analysis, cell suspension was seeded either in chamber plate or 10 cm petri dish, respectively. The miRNA inhibitor or mimic and scramble inhibitor, and the mimic were obtained from RiboBio Guangzhou, China. siRNA control and siCFL1 were obtained from USA, Life Technologies. The siRNA was predesigned at Ambion®, Carlsbad, CA. Transfection was carried out using Polyplus-transfection (Brant, France) and JetPRIME® reagents in accordance with the manufacturer's instructions [25].
TUNEL assay
Fluorescein-dUTP was used to stain and evaluate the breakage of DNA strand as per manufacturer’s instructions (Roche Molecular Biochemical, Mannheim, Germany). TUNEL positive cells were identified using fluorescence microscopy [30].
Luciferase reporter assay
The Luciferase reporter assays was conducted according to previously described [22,23,24,25]. CFL1 3’UTR wild and mutant type luciferase plasmids were constructed using pmirGLO empty vector (Promega, Madison, WI, USA). According to the JetPRIME® reagents manufactures’s guidelines miR-342-5r mimic, and with pmirGLO-3’UTR or CFLl promoter plasmid were co- transfected after HDACi-R cells. The dual-luciferase assay kit (Promega, Madison,WI, USA) was used to detect the luciferase activity.
Statistical analysis
Statistical analyses were conducted using one-way analysis of variance (ANOVA) and Student's t-test (Prism GraphPad 5). Statistical significance was set at *p < 0.05, ** p < 0.01 and *** < p 0.001. Images were quantified using ImageJ software (NIH, Bethesda, MD, USA).
Results
Assessment of HDACi-R cell properties
We previously generated HDACi-resistance cell line using HDAC inhibitor (SAHA and apicidin) from the parental HA22T cells [19]. To confirm the chemoresistance in HA22T and HDACi-R, the cells were exposed to different doses of SAHA for 24 h and the cell survival was evaluated. HDACi-R cells significantly resisted SAHA-mediated cell death, while the viability was found to decrease dose dependently in HA22T parental cells (Fig. 1A, Fig. S1a-1c). Next, we analyzed the apoptotic-related functional protein expressions from three distinct passages of both cell types. HDACi-R cells showed increased expression of anti-apoptotic protein (Bcl-2) levels than that of HA22T parental cells. In addition, the expression of cleaved-capase-3, Bax, and Cyto-C were down regulated in the HDACi-R cells than in parental cells (HA22T) (Fig. 1B, C). Flow cytometric analysis revealed that, SAHA significantly increased the percentage of apoptotic cells in HA22T parental cells. However, there was a no significant change in apoptotic cell (%) in the HDACi-R cells after SAHA treatment (Fig. 1D, E). These findings confirmed that, HDACi-R cells were resistant to SAHA with increased cell viability than parental HA22T cells.
Assessment of HDACi-R cell properties A HDAC inhibitor (SAHA) mediated drug resistance were evaluated in HDACi-R and HA22T cells after 24 h treatment. ***p < 0.001 compared to HA22T control, ###p < 0.001compared to HR control. The experiments were conducted in triplicate and the results were expressed as the mean of ± SEM B Effect of SAHA on the protein expression of apoptotic markers (Bcl-2, Bax, Cyto-c, cleaved-caspase-3) in HA22T and HDACi-R cells. C Densitometric analysis of apoptotic markers. The experiments were conducted in triplicate and the results were expressed as the mean of ± SEM **p < 0.01, ***p < 0.001 compared to HA22T control, ##p < 0.01, ###p < 0.001compared to HR control. D Cell death for HA22T and HDACi-R cell types was detected using flow cytometry. E Percentage of apoptotic cells among HA22T and HDACi-R cells. The experiments were conducted in triplicate and the results were expressed as the mean of ± SEM **p < 0.01, ***p < 0.001 compared to HA22T control: and not significant (NS) compared to HDACi-R control
miR-342-5p was the key regulator of cell survival in HDACi-R and HA22T cells
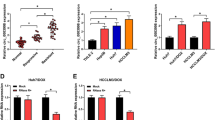
miRNAs are important in tumor development and growth regulation [31]. The impact of miRNAs on HCC drug resistance was examined in the HDACi-R and HA22T cell types and their differential miRNA expressions were determined using micro array. Among the miRNAs, miR-342-5p showed differential expression between HDACi-R and HA22T cells (Fig. 2A). Compared to HA22T cells, the overall level of miR-342-5p expression in HDACi-R cells was lower. The log-2 value of HR/HA hsa-miR-342-5p levels were found to be -1.237847011; p values = 0.000603399 (Fig. 2B). Next, in order to confirm our micro array results we evaluated the expression of miR-342-5p in HDACi-R and HA22T cells by qRT-PCR protocols. Our results revealed that there was a significant downregulation of miR-342-5p expression in HDACi-R cells than in HA22T cells (Fig. 2C). To further examine the involvement of miR-342-5p in chemoresistance, both parental and HDACi-R cells were treated with miR-342-5p mimic and inhibitor Cells without treatment with either mimic or inhibitor showed a significant change in the miR-342-5p expression among the HA22T and HDACi-R cells. There was a significant dose dependent downregulation of miR-342-5p expression in the parental HA22T cells treated with inhibitor, while mimic treatment significantly increased miR-342-5p levels in HDACi-R cells (Fig. 2D). Furthermore, we explored how miR-342-5p stimulated cell death in HCC. Cell viability experiments showed that, HDACi promoted apoptosis in HA22T cells, whereas overexpression of miR-342-5p significantly downregulated cell proliferation in both HDACi-R and HA22T cells (Fig. 2E). Flow cytometry revealed that miR-342-5p mimic significantly enhanced cell death among HDACi-R cells than in HA22T cells (Fig. 2F, G). In contrast to HA22T, the resistant cells showed upregulated Bcl-2 levels with concomitant downregulation in the Bax, cyto-C and cleaved-caspase-3 expressions levels (Fig. 2H, I). However, we found that miR-342-5p overexpression in HDACi-R cells upregulated the apoptotic proteins expressions of Bax, cyto-C, cleaved-caspase-3 with a decline in anti-apoptotic protein expression of Bcl-2 than in HA22T cells (Fig. 2H, I). Cumulatively, these results suggested that, miR-342-5p regulated cell viability and proteins related to apoptosis and pro-survival in HDACi-R cells. These findings suggested that miR-342-5p is vital for chemoresistance, particularly in HDACi-R cells.
miR-342-5p regulates cell survival in HDACi-R and HA22T cells. A Micro array of HDACi-R and HA22T cells showing differential miRNA expression. B Comprehensive micro array assessment showing significant downregulation of hsa-miR-342-5p in HDACi-R cells. C Confirmation of miR-342-5p expression in HDACi-R and HA22T cells through qRT-PCR. HDACi-R cells ***p < 0.001compared to HA22T cells. The experiments were conducted in triplicate and results are expressed as the mean of ± SEM. D Differential expression of miR-342-5p levels in HDACi-R and HA22T cells in the presence of miR-342-5p inhibitor and miR-342-5p mimic. ***p < 0.001 compared to inhibitor negative control, ###p < 0.001 compared to mimic negative control. The experiments were conducted in triplicate and results are expressed as the mean of ± SEM. E Overexpression of miR-342-5p significantly reduced the cell viability of HDACi-R and HA22T cells. ***p < 0.001 compared to HA22T cells, ###p < 0.001 compared to HDACi-R cells. The experiments were conducted in triplicate and results are expressed as the mean of ± SEM. F,G Flow cytometry of apoptosis in HDACi-R and HA22T cells after treatment with miR-342-5p mimic, *p < 0.05 compared to HA22T, ##p < 0.001 compared to HDACi-R control. The experiments were conducted in triplicate and results are expressed as the mean of ± SEM. H Western blotting for analyzing on the effect of apoptotic markers (Bcl-2, Bax, cyto-C, and cleaved-Caspase-3) after the HA22T and HDACi-R cells being treated with miR-342-5p inhibitor and mimic. I Densitometric analysis of apoptotic markers. The experiments were conducted in triplicate and results are expressed as the mean of ± SEM *p < 0.05 compared to HA22T control; and not significant (NS) # < p0.05, ###p < 0.01compared to HR control
miR-342-5p functional targeted CFL1 in hepatocellular carcinoma
We confirmed that miR-342-5p is critical for HDACi-resistance mechanism in HCC in-vitro. Next, we sought to determine target of miR-342-5p. To identify the possible miR-342-5p targets, we searched three online databases: the miRDB, miRTarBase, and TargetScanHuman (Fig. 3A, Table S1). CFL1 was one of the possible targets that spanned all three databases and had been linked to metastasis and invasion in several studies [32, 33]. To support this, we evaluated the mRNA and protein expression of CFL1 in both cell lines. CFL1 mRNA expression was significantly greater in HDACi-R cells than in the parental cells (Fig. 3B). However, we extracted whole-cell protein from three distinct passages through western blotting. Phosphorylated CFL1 expression was upregulated in resistant cells than in HA22T cells (Fig. 3C, D). To determine whether miR-342-5p regulated CFL1 expression, the cells were analyzed for their mRNA and protein expressions after miR-342-5p inhibitor and mimic treatment. CFL1 expression was downregulated in HDACi-R cells followed by miR342-5p overexpression, while it was significantly upregulated after miR-342-5p inhibitor treatment in HA22T cells (Fig. 3E–G). These findings showed that miR-342-5p targeted CFL1 and was a crucial regulator of drug resistance in HDACi-R cells.
miR-342-5p functional targeted CFL1 in HCC. A Bioinformatics analysis of miR-342-5p targets using miRDB, Target Scan, and miRTarbase databases. B mRNA expression of CFL1 was analyzed in HA22T and HDACi-R cells using qRT-PCR. ***p < 0.001 compared to HA22T cells. The experiments were conducted in triplicate and results are expressed as the mean of ± SEM. C Effects of SAHA on CFL1 and p-CFL1 expressions in HA22T and HDACi-R cells. D Densitometric analysis of CFL l. **p < 0.01 compared to HA22T control, and not significant (NS), ##p < 0.01 compared to HR control. The experiments were conducted in triplicate and results are expressed as the mean of ± SEM. E CFL1 expression in HA22T and HDACi resistance cells in the presence of miR-342-5p inhibitor and mimic. *p < 0.05, ***p < 0.001 compared to HA22T control; ##p < 0.01 compared to HDACi-R control the experiments were conducted in triplicate and results are expressed as the mean of ± SEM. F Effects of miR-342-5p mimic/inhibitor on CFL1 and p-CFL1 expression in HA22T and HDACi-R cells. G Densitometric analysis of Fig. F, not significant (NS) compared to HA22T control, #p < 0.05, ###p < 0.001compared to HR control. The experiments were conducted in triplicate and results are expressed as the mean of ± SEM
miR-342-5p regulated HCC chemosensitivity in-vitro
Downregulated miR-342-5p levels in resistant cells inhibited the production of apoptotic proteins. However, miR-342-5p overexpression in HDACi-R cells promoted cell death than in HA22T cells. Here we analyzed the functional importance of miR-342-5p on the chemosensitivity of parental and resistant cells. HDACi suppressed cell growth, whereas miR-342-5p inhibitor reversed the cell death induced by HDACi in HA22T cells (Fig. 4A). HDACi did not cause cell death in resistant cell type. However, overexpression of miR-342-5p increased sensitivity to HDACi in HDACi-R cells (Fig. 4B). Next, to confirm our MTT results we evaluated the impact of apoptosis in HA22T and HDACi-R cells treated with SAHA alone and in combination with miR-342-5p mimic/inhibitor transfection by flow cytometry and western blotting. Cumulatively, the results shown in Fig. 4C–F indicate that, miR-342-5p inhibitor prevented cell death induced by HDACi. Furthermore, miR-342-5p overexpression enhanced the susceptibility of HDACi-R cells to HDACi. Thus, miR-342-5p is an important regulator of chemosensitivity in HCC cells.
miR-342-5p regulated HCC chemosensitivity. A miR-342-5p knockdown inhibited HDACi-induced cell death of HA22T type. **p < 0.01, ***p < 0.001 compared to HA22T control. The experiments were conducted in triplicate and results are expressed as the mean of ± SEM. B miR-342-5p mimic induced chemosensitivity of resistant cells. ###p < 0.001compared to HR control. The experiments were conducted in triplicate and results are expressed as the mean of ± SEM.C Flow cytometry of the combinational treatment of SAHA and miR-342-5p mimic/inhibitor. D Effect of SAHA and miR-342-5p mimic/inhibitor on the percentage of apoptotic cells. ***p < 0.001 compared to parental control, and not significant (NS), ##p < 0.01, ###p < 0.001 control compared to resistant control. The experiments were conducted in triplicate and results are expressed as the mean of ± SEM. E Western blotting analyze the apoptotic proteins in the presence of SAHA and miR-342-5p mimic/inhibitor. F Densitometric analysis of apoptotic markers. The experiments were conducted in triplicate and results are expressed as the mean of ± SEM **p < 0.01, ***p < 0.001 compared to HA22T control, ##p < 0.01, ###p < 0.001compared to HR control
miR-342-5p promoted ROS accumulation and cell death in HCC cells via cofilin-1
Cofilin-1 induces chemoresistance in HCC cells by regulating ROS accumulation [19]. Additional investigation has shown that miR-342 targets cofilin-1 directly in breast cancer [34]. Therefore, we evaluated whether miR-342-5p promoted apoptosis through its regulation of ROS accumulation and cofilin-1 expression. Figure 5A shows the miR-342-5p targeting region on the 3’UTR of cofilin-1 [34]. To investigate the impact of miR-342-5p on target gene suppression, we used luciferase reporter assays with CFL 1 wild-type and mutants 3'UTR. A significant reduction in luciferase activity was observed in HDACi-R cells following co-transfection with miR-342-5p mimic and cofilin-1 3' UTR wild type reporter vectors (Fig. 5B). However, mutant type reporter target sequences did not induce miR-342-5p mimic. Following this report, we investigated whether cell death induced by miR-342-5p was associated with the production of ROS. We found that ROS accumulation was greater in the HA22T control than in HDACi-R cells. Overexpression of miR-342-5p induced ROS production in HDACi-R cell type (Fig. 5C). Further, we assess the potential role of ROS accumulation and apoptosis after co-transfected with miR-342-5p mimic and siCFL1 in HDACi-R and HA22T cells by western blotting and Tunel assay. ROS increased the expression of cleaved-caspase-3 in parental cells than in resistant type. Additionally, miR-342-5p overexpression downregulated cofilin-1 expression and promoted ROS-induced apoptosis (Fig. 5D, E). A similar effect was observed in the siRNA-mediated knockdown of cofilin-1, which also increased the ROS sensitivity of HDACi-R (Fig. 5D, E). Notably, in resistant cells, upregulation of miR-342-5p resulted in increased ROS generation. These findings revealed that, miR-342-5p regulated cell survival in HCC cells via CFL-1 mediated accumulation of ROS.
miR-342-5p promoted ROS accumulation and cell death in HCC via cofilin-1. A miR-342-5p targeting region in the 3' UTR of CFL1. B Luciferase reporter assay demonstrates the wild-type and mutant CFL 1 3'UTR targeted by miR-342-5p mimic. ##p < 0.01, ###p < 0.001 compared to resistant control. The experiments were conducted in triplicate and results expressed as the mean of ± SEM. C Effects of miR-342-5p mimic on ROS generation (MitoSOX) in HA22T and HDACi-R cells. D Expression of Cleaved-caspase-3 in the presence of miR-342-5p mimic and si-CFL-1 E Effect of miR-342-5p mimic and siCFL1 on apoptosis analyzed through TUNEL assay. Red apoptotic cells, blue indicates the Nucleus (DAPI counter stain)
The schematic representation of the current investigation. MiR-342-5p overexpression inhibits cofilin-1 phosphorylation, enhances ROS generation, releases cytochrome C, and induces apoptosis in HDACi-R cancer cells.
Discussion
Drug resistance and recurrent metastasis is a significant problem and causes therapeutic failure in cancer prognosis. Here, we sought to uncover key miRNA and the mechanism involved in HCC drug resistance (Fig. 6). CFL-1 is associated with cell metastasis and invasion [32, 33], cytoskeleton reorganization [35], cell differentiation [36]. These molecular pathways suggested that cofilin-1 might be a critical protein for the regulation of cancer cell metastasis. Our previous findings showed that Cofilin-1 may be a potential gene related to chemoresistance in HCC [19]. Our current in vitro study suggested that anti-cancer medication increased ROS generation as well as cell mortality through cofilin-1 translocation into the mitochondria (Fig. 6). Our previous research identified that the drug resistant strategy of cofilin-1 was mediated by decreased mitochondrial translocation after ERK phosphorylation with subsequent decline in ROS levels and cell death [18]. Thus, we hypothesized that cofilin-1 might be a potential therapeutic target for attenuating HCC drug resistance.
miRNAs which are abnormally expressed have been linked in epigenetic regulation of genes involved in cancer [37, 38]. Alteration in miRNA expressions have also been implicated in the pathogenesis of pancreatic, breast [39], liver and lung cancers [40]. The miRNAs influence cancer cell initiation and metastasis [41]. Our findings for the first time provided evidence that miR-342-5p might/may be an important regulator in HCC drug resistance. miR-342-5p has been reported as potential tumor suppressor in neuroblastoma [42,43,44]. miR-342-5p mediated targeting of NAA10 suppresses colon cancer growth and induces apoptosis [45]. miR-342-5p suppresses cell invasion, migration and cell proliferation in osteosarcomas and restores doxorubicin sensitivity by targeting WnT7b [46,47,48]. Accordingly, in this in-vitro study, we reported that miR-342-5p, play an important role in the molecular mechanisms of HCC chemoresistance. Overall, through this investigation, we showed clear evidence that miR-342-5p is associated with anti-drug resistance in HCC in-vitro.
After developing HDACi-R cells, we confirmed the chemoresistance of HDACi-R cells resistant to HDACi SAHA with different concentrations in-vitro (Fig. 1). miR-342-5p expression was significantly reduced than in HA22T cells (Fig. 2). Thus, miR-342-5p overexpression promoted cell death in both parental and resistant cells. However, the effect was considerably higher in HDACi-R cells (Fig. 2). An increased expression level of CFL 1(inactive form), one of the target genes, was observed in HDACi-R cells than in parental cells (Fig. 3). miR-342-5p downregulation promotes chemoresistance and cell survival, suggesting that the miR-342-5p target gene, CFL 1, may be crucial for HCC survival and chemoresistance (Fig. 3). The over expression of miR-342-5p showed higher sensitivity to HDACi-R cells with combinational treatment with SAHA and mimic in-vitro (Fig. 4). Additionally, miR-342-5p mediated CFL-1 targeting induces ROS levels, apoptosis and enhances chemosensitivity, whereas similar observations found after knockdown CFL1 with either siCFL1 alone or siCFL1 and mimic (Fig. 5).
Therefore, we hypothesized that over expression of miR-342-5p suppresses the phosphorylation of cofilin-1, increases the accumulation of reactive oxygen species (ROS), triggers the release of cytochrome C, and promotes death in HDACi-R cancer cells. Notably, this suggests that miR-342-5p might be a potent tumor suppressor in HCC chemosensitivity in-vitro. However, further studies are needed to clarify our hypothesis, since in-vitro settings do not replicate the extensive physiological interactions observed in living organisms [49, 50]. Likewise, the nature and integrity of the cellular and extracellular architecture is missing in in-vitro models. Therefore, hinders its ability to accurately represent the actual biological processes that occur in vivo studies [51]. It also should be noted that the dynamic nature of the living organisms could also affect the HDACi efficacy in vivo studies. It is crucial to acknowledge the limitations inherent in our experimental strategy. Our investigations do not include in-vivo validation, solely focused on in vitro experiments. this study provides significant insights into the association of miR-342-5p with HCC chemoresistance and the possible therapeutic targeting of CFL-1. Future investigations of in-vivo experiments and clinical applications should enhance the significance of targeting miR-342-5p and CFL-1 in HCC chemosensitivity of our in-vitro findings.
Conclusion
Our findings indicate that down regulated miR-342-5p expression is an important contributor for HDACi-R. Further, overexpression of miR-342-5p in HDACi-R cells promoted cell death through ROS/CFL-1 regulation in vitro. In summary, miR-342-5p might act as potent sensitizer for HADACi mediated chemoresistance in HCC.
Availability of data and materials
The data sets used and/or analysed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- HCC:
-
Hepatocellular carcinoma
- CFL1:
-
Cofilin1
- p-CFL1:
-
Phosphorylated Cofilin1
- HDAC:
-
Histone deacetylase
- HDACi:
-
Histone deacetylase inhibitor
- HDACi-R:
-
Histone deacetylase inhibitor resistant
- SAHA:
-
Suberoylanilide hydroxamic acid
- miRNA:
-
MicroRNA
- ROS:
-
Reactive oxygen species
- siCFL1:
-
Small interfering Cofilin1
References
Hsieh CH, Hsu HH, Shibu MA, Day CH, Bau DT, Ho CC, Lin YM, Chen MC, Wang SH, Huang CY. Down-regulation of β-catenin and the associated migration ability by Taiwanin C in arecoline and 4-NQO-induced oral cancer cells via GSK-3β activation. Mol Carcinog. 2017;56(3):1055–67.
Serper M, Taddei TH, Mehta R, D’Addeo K, Dai F, Aytaman A, Baytarian M, Fox R, Hunt K, Goldberg DS. Association of provider specialty and multidisciplinary care with hepatocellular carcinoma treatment and mortality. Gastroenterology. 2017;152(8):1954–64.
Ding X-X, Zhu Q-G, Zhang S-M, Guan L, Li T, Zhang L, Wang S-Y, Ren W-L, Chen X-M, Zhao J. Precision medicine for hepatocellular carcinoma: driver mutations and targeted therapy. Oncotarget. 2017;8(33):55715.
Huang SM, Jeng L-B, Shyu W-C, Chen H-Y. Combination treatment of pembrolizumab with DC-CIK cell therapy for Advanced hepatocellular carcinoma: a case report. Biomedicine. 2023;13(3):57–62.
Geretto M, Pulliero A, Rosano C, Zhabayeva D, Bersimbaev R, Izzotti A. Resistance to cancer chemotherapeutic drugs is determined by pivotal microRNA regulators. Am J Cancer Res. 2017;7(6):1350.
Mansoori B, Mohammadi A, Davudian S, Shirjang S, Baradaran B. The different mechanisms of cancer drug resistance: a brief review. Adv Pharm Bull. 2017;7(3):339.
Virani SS, Alonso A, Benjamin EJ, Bittencourt MS, Callaway CW, Carson AP, Chamberlain AM, Chang AR, Cheng S, Delling FN. Heart disease and stroke statistics—2020 update: a report from the American heart association. Circulation. 2020;141(9):e139–596.
Siomi H, Siomi MC. Posttranscriptional regulation of microRNA biogenesis in animals. Mol Cell. 2010;38(3):323–32.
O’Brien J, Hayder H, Zayed Y, Peng C. Overview of microRNA biogenesis, mechanisms of actions, and circulation. Front Endocrinol. 2018;9:402.
Ali Syeda Z, Langden SSS, Munkhzul C, Lee M, Song SJ. Regulatory mechanism of MicroRNA expression in cancer. Int J Mol Sci. 2020;21(5):1723.
Wery M, Kwapisz M, Morillon A. Noncoding RNAs in gene regulation. Wiley Interdiscip Rev Syst Biol Med. 2011;3(6):728–38.
Chung T, Lau T, Cheung T, Yim S, Lo K, Siu N, Chan L, Yu M, Kwong J, Doran G. Dysregulation of microRNA-204 mediates migration and invasion of endometrial cancer by regulating FOXC1. Int J Cancer. 2012;130(5):1036–45.
Wang W, Goswami S, Lapidus K, Wells AL, Wyckoff JB, Sahai E, Singer RH, Segall JE, Condeelis JS. Identification and testing of a gene expression signature of invasive carcinoma cells within primary mammary tumors. Cancer Res. 2004;64(23):8585–94.
Tang W, Zhang Y, Xu W, Harden TK, Sondek J, Sun L, Li L, Wu D. A PLCβ/PI3Kγ-GSK3 signaling pathway regulates cofilin phosphatase slingshot2 and neutrophil polarization and chemotaxis. Dev Cell. 2011;21(6):1038–50.
Posadas I, Pérez-Martínez FC, Guerra J, Sánchez-Verdú P, Ceña V. Cofilin activation mediates Bax translocation to mitochondria during excitotoxic neuronal death. J Neurochem. 2012;120(4):515–27.
Chua BT, Volbracht C, Tan KO, Li R, Yu VC, Li P. Mitochondrial translocation of cofilin is an early step in apoptosis induction. Nat Cell Biol. 2003;5(12):1083–9.
Klamt F, Zdanov S, Levine RL, Pariser A, Zhang Y, Zhang B, Yu L-R, Veenstra TD, Shacter E. Oxidant-induced apoptosis is mediated by oxidation of the actin-regulatory protein cofilin. Nat Cell Biol. 2009;11(10):1241–6.
Hsieh Y-C, Rao YK, Wu C-C. Huang C-YF, Geethangili M, Hsu S-L, Tzeng Y-M: Methyl antcinate A from Antrodia camphorata induces apoptosis in human liver cancer cells through oxidant-mediated cofilin-and Bax-triggered mitochondrial pathway. Chem Res Toxicol. 2010;23(7):1256–67.
Liao P, Hsu H, Chen T, Chen M, Day C, Tu C, Lin Y, Tsai F, Kuo W, Huang C. Phosphorylation of cofilin-1 by ERK confers HDAC inhibitor resistance in hepatocellular carcinoma cells via decreased ROS-mediated mitochondria injury. Oncogene. 2017;36(14):1978–90.
Hsu W-C, Ramesh S, Shibu MA, Chen M-C, Wang T-F, Day CH, Chen R-J, Padma VV, Li C-C, Tseng Y-C. Platycodin D reverses histone deacetylase inhibitor resistance in hepatocellular carcinoma cells by repressing ERK1/2-mediated cofilin-1 phosphorylation. Phytomedicine. 2021;82: 153442.
Liu Y-S, Chang Y-C, Kuo W-W, Chen M-C, Hsu H-H, Tu C-C, Yeh Y-L, Viswanadha VP, Liao P-H, Huang C-Y. Inhibition of protein phosphatase 1 stimulates noncanonical ER stress eIF2α activation to enhance fisetin-induced chemosensitivity in HDAC inhibitor-resistant hepatocellular carcinoma cells. Cancers. 2019;11(7):918.
Arfianti A, Ulfah U, Hutabarat LS, Budiarti AD, Sahara NS, Saputra NPK. Hipoxia modulates the secretion of growth factors in of human umbilical cord-derived mesenchymal stem cells. Biomedicine. 2023;13(3):49–56.
Chang JHM, Lin CH, Shibu MA, Chou YC, Liu JY, Chou YH, Shen CY, Yeh YL, Viswanadha VP, Huang CY. Cryptotanshinone (Dsh-003) from Salvia miltiorrhiza Bunge inhibits prostaglandin E2-induced survival and invasion effects in HA22T hepatocellular carcinoma cells. Environ Toxicol. 2018;33(12):1254–60.
Hsu H-H, Kuo W-W, Shih H-N, Cheng S-F, Yang C-K, Chen M-C, Tu C-C, Viswanadha VP, Liao P-H, Huang C-Y. FOXC1 regulation of miR-31-5p confers oxaliplatin resistance by targeting LATS2 in colorectal cancer. Cancers. 2019;11(10):1576.
Kannathasan T, Kuo W-W, Chen M-C, Viswanadha VP, Shen C-Y, Tu C-C, Yeh Y-L, Bharath M, Shibu MA, Huang C-Y. Chemoresistance-associated silencing of miR-4454 promotes colorectal cancer aggression through the GNL3L and NF-κB pathway. Cancers. 2020;12(5):1231.
Singuru MMR, Sun S-C, Chuang M-C. Advances in oligonucleotide-based detection coupled with fluorescence resonance energy transfer. TrAC Trends Anal Chem. 2020;123: 115756.
Singuru MMR, Liao W-C, Chuang M-C. Genotoxin-responsive DNA capsules for the detection of styrene oxide. Sens Actuators B Chem. 2021;349: 130776.
Singuru MMR, Liao Y-C, Lin GM-H, Chen W-T, Lin Y-H, To CT, Liao W-C, Hsu C-H, Chuang M-C. Engineered multivalent DNA capsules for multiplexed detection of genotoxicants via versatile controlled release mechanisms. Biosensors Bioelectronics. 2022;216:114608.
Lin Y-H, Singuru MMR, Marpaung DSS, Liao W-C, Chuang M-C. Ethylene glycol-manipulated syntheses of calcium carbonate particles and DNA capsules toward efficient ATP-responsive cargo release. ACS Appl Bio Mater. 2023;6(8):3351–60.
Chang-Lee SN, Hsu H-H, Shibu MA, Ho T-J, Tsai C-H, Chen M-C, Tu C-C, Viswanadha VP, Kuo W-W, Huang C-Y. E 2/ERβ inhibits PPARα to regulate cell-proliferation and enhance apoptosis in Hep3b-hepatocellular carcinoma. Pathol Oncol Res. 2017;23:477–85.
Chen H-A, Li C-C, Lin Y-J, Wang T-F, Chen M-C, Su Y-H, Yeh Y-L, Padma VV, Liao P-H, Huang C-Y. Hsa-miR-107 regulates chemosensitivity and inhibits tumor growth in hepatocellular carcinoma cells. Aging. 2021;13(8):12046.
Zhou Y, Su J, Shi L, Liao Q, Su Q. DADS downregulates the Rac1-ROCK1/PAK1-LIMK1-ADF/cofilin signaling pathway, inhibiting cell migration and invasion. Oncol Rep. 2013;29(2):605–12.
Yamaguchi H, Condeelis J. Regulation of the actin cytoskeleton in cancer cell migration and invasion. Biochimica et Biophysica Acta (BBA) Mol Cell Res. 2007;1773(5):642–52.
Liu C, Xing H, Luo X, Wang Y. MicroRNA-342 targets Cofilin 1 to suppress the growth, migration and invasion of human breast cancer cells. Arch Biochem Biophys. 2020;687: 108385.
Serezani CH, Kane S, Medeiros AI, Cornett AM, Kim S-H, Marques MM, Lee S-P, Lewis C, Bourdonnay E, Ballinger MN. PTEN directly activates the actin depolymerization factor cofilin-1 during PGE2-mediated inhibition of phagocytosis of fungi. Sci Signaling. 2012. https://doi.org/10.1126/scisignal.2002448.
Zhou J, Wang Y, Fei J, Zhang W. Expression of cofilin 1 is positively correlated with the differentiation of human epithelial ovarian cancer. Oncol Lett. 2012;4(6):1187–90.
Mansouri S, Khansarinejad B, Mosayebi G, Eghbali A, Mondanizadeh M. Alteration in expression of miR-32 and FBXW7 tumor suppressor in plasma samples of patients with T-cell acute lymphoblastic leukemia. Cancer Manag Res. 2020;12:1253.
Pinho JD, Silva GEB, Júnior AALT, de Castro Belfort MR, Macedo JM, da Cunha IW, Quintana LG. Calixto JdRR, Nogueira LR, Coelho RWP: MIR-107, MIR-223-3P and MIR-21-5P reveals potential biomarkers in penile cancer. Asian Pac J Cancer Prev. 2020;21(2):391.
Lindholm EM, Leivonen S-K, Undlien E, Nebdal D, Git A, Caldas C, Børresen-Dale A-L, Kleivi K. miR-342-5p as a potential regulator of HER2 breast cancer cell growth. Microrna. 2019;8(2):155–65.
Han Z, Li Y, Zhang J, Guo C, Li Q, Zhang X, Lan Y, Gu W, Xing Z, Liang L. Tumor-derived circulating exosomal miR-342-5p and miR-574-5p as promising diagnostic biomarkers for early-stage Lung Adenocarcinoma. Int J Medical Sci. 2020;17(10):1428.
Xuan Y, Yang H, Zhao L, Lau WB, Lau B, Ren N, Hu Y, Yi T, Zhao X, Zhou S. MicroRNAs in colorectal cancer: small molecules with big functions. Cancer Lett. 2015;360(2):89–105.
Soriano A, Masanas M, Boloix A, Masiá N, París-Coderch L, Piskareva O, Jiménez C, Henrich K-O, Roma J, Westermann F. Functional high-throughput screening reveals miR-323a-5p and miR-342-5p as new tumor-suppressive microRNA for neuroblastoma. Cell Mol Life Sci. 2019;76:2231–43.
Veys C, Jammes M, Rédini F, Poulain L, Denoyelle C, Legendre F, Galera P. Tumor suppressive role of miR-342-5p and miR-491-5p in human osteosarcoma cells. Pharmaceuticals. 2022;15(3):362.
Veys C, Benmoussa A, Contentin R, Duchemin A, Brotin E, Lafont JE, Saintigny Y, Poulain L, Denoyelle C, Demoor M. Tumor suppressive role of miR-342-5p in human chondrosarcoma cells and 3D organoids. Int J Mol Sci. 2021;22(11):5590.
Yang H, Li Q, Niu J, Li B, Jiang D, Wan Z, Yang Q, Jiang F, Wei P, Bai S. microRNA-342-5p and miR-608 inhibit colon cancer tumorigenesis by targeting NAA10. Oncotarget. 2016;7(3):2709.
Liu Q, Wang Z, Zhou X, Tang M, Tan W, Sun T, Deng Y. miR-342-5p inhibits osteosarcoma cell growth, migration, invasion, and sensitivity to Doxorubicin through targeting Wnt7b. Cell Cycle. 2019;18(23):3325–36.
Wu Y-Y, Lai H-F, Huang T-C, Chen Y-G, Ye R-H, Chang P-Y, Lai S-W, Chen Y-C, Lee C-H, Liu W-N. Aberrantly reduced expression of miR-342-5p contributes to CCND1-associated chronic myeloid leukemia progression and imatinib resistance. Cell Death Dis. 2021;12(10):908.
Dou Y, Chen F, Lu Y, Qiu H, Zhang H. Effects of Wnt/β-catenin signal pathway regulated by miR-342-5p targeting CBX2 on proliferation, metastasis and invasion of ovarian cancer cells. Cancer Manag Res. 2020;12:3783.
Corrò C, Novellasdemunt L, Li VS. A brief history of organoids. Am J Physiol Cell Physiol. 2020;319(1):C151–65.
Fedi A, Vitale C, Ponschin G, Ayehunie S, Fato M, Scaglione S. In vitro models replicating the human intestinal epithelium for absorption and metabolism studies: a systematic review. J Cont Release. 2021;335:247–68.
Carannante V, Wiklund M, Önfelt B. In vitro models to study natural killer cell dynamics in the tumor microenvironment. Front Immunol. 2023. https://doi.org/10.3389/fimmu.2023.1135148.
Acknowledgements
This study was supported by the China Medical University, Taichung, Taiwan, and Hualien Tzu Chi Hospital, Buddhist Tzu Chi Medical Foundation, Hualien 970, Taiwan. All the experiments were conducted at China Medical University Chih-Yang Huang's Laboratory Taichung group.
Funding
Thank you Kaohsiung Armed Forces General Hospital, Kaohsiung, Taiwan. Ministry of Science and Technology: 111-2314-B-303-008-MY3, China Medical University and Asia University Taiwan 106-ASIA-16.
Author information
Authors and Affiliations
Contributions
Research Concept: Parvathi Nakka, and Chikondi Jassi.; Experimental Investigations, Original Manuscript writing: Parvathi Nakka.; Review & Editing: Parvathi Nakka, Chikondi Jassi, Yu-Chun Chang, Ming-Cheng Chen, Chi-Cheng Li, and Yi-Sheng Liu and Jer-Yuh Liu, Wei-Wei Kuo, and Chih-Yang Huang.; Data Analysis and Validation: Parvathi Nakka, Yu-Chun Chang, Ming-Cheng Chen, Chung-Min Yeh, Wei-Wei Kuo, and Chih-Yang Huang.; Supervision, Jer-Yuh Liu, Wei-Wei Kuo, and Chih-Yang Huang.; Project Administration, Chi-Cheng Li and Chih-Yang Huang.; Funding Acquisition, Wei-Wen Kuo, Chih-Yang Huang. All authors read and approved teh final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
12935_2024_3450_MOESM1_ESM.docx
Additional file 1 Fig. S.1MTT assay was used to evaluate cell viability in HA22T and HDACi-R cells after they were treated with the appropriate amounts of SAHA for 24 hours, 48 hours, and 72 hours. Expression of stem cell markers are higher in HDAC inhibitor-resistant cells than HA22T cells.
12935_2024_3450_MOESM2_ESM.xlsx
Additional file 2 Table S1, related to Fig.3A, The name of genes identified as potential targets of miR-342-5p as predicted by four algorithms.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Nakka, P., Jassi, C., Chen, MC. et al. Sensitization of hepatocellular carcinoma cells to HDACi is regulated through hsa-miR-342-5p/CFL1. Cancer Cell Int 24, 291 (2024). https://doi.org/10.1186/s12935-024-03450-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12935-024-03450-x